These Highlights Do Not Include All The Information Needed To Use Zycubo Safely And Effectively. See Full Prescribing Information For Zycubo.
e3aeefc2-f44d-4337-9c2e-3dba44a89e48
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZYCUBO is indicated for the treatment of Menkes disease in pediatric patients.
Indications and Usage
ZYCUBO is indicated for the treatment of Menkes disease in pediatric patients.
Dosage and Administration
The recommended dosage of ZYCUBO in pediatric patients: Less than 1 year of age is 1.45 mg administered subcutaneously twice daily (8-12 hours between injections). 1 year of age to less than 17 years of age is 1.45 mg administered subcutaneously once daily.
Warnings and Precautions
Copper Accumulation and Risk of Toxicity : Exogenous administration of copper with ZYCUBO may lead to further copper accumulation and has the potential to result in drug-induced kidney injury, liver dysfunction, and hematological abnormalities. Monitor patients during ZYCUBO treatment. Adjust dosage if necessary. ( 2.2 , 5.1 , 6.1 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Copper Accumulation and Risk of Toxicity: Drug-Induced Kidney Injury, Liver Dysfunction, Hematological Abnormalities [see Warnings and Precautions ( 5.1 )]
Medication Information
Warnings and Precautions
Copper Accumulation and Risk of Toxicity : Exogenous administration of copper with ZYCUBO may lead to further copper accumulation and has the potential to result in drug-induced kidney injury, liver dysfunction, and hematological abnormalities. Monitor patients during ZYCUBO treatment. Adjust dosage if necessary. ( 2.2 , 5.1 , 6.1 )
Indications and Usage
ZYCUBO is indicated for the treatment of Menkes disease in pediatric patients.
Dosage and Administration
The recommended dosage of ZYCUBO in pediatric patients: Less than 1 year of age is 1.45 mg administered subcutaneously twice daily (8-12 hours between injections). 1 year of age to less than 17 years of age is 1.45 mg administered subcutaneously once daily.
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Copper Accumulation and Risk of Toxicity: Drug-Induced Kidney Injury, Liver Dysfunction, Hematological Abnormalities [see Warnings and Precautions ( 5.1 )]
Description
ZYCUBO is indicated for the treatment of Menkes disease in pediatric patients.
Section 42229-5
Limitations of Use
ZYCUBO is not indicated for the treatment of Occipital Horn Syndrome.
Section 51945-4
Principal Display Panel - 2.9 mg Carton Label
NDC 42358-329-01
Zycubo
®
(copper histidinate) for injection
2.9 mg/vial
For subcutaneous injection
One Single-Dose vial
Discard unused portion after each single use.
Do not administer more than one dose from the vial.
Rx only
Section 59845-8
INSTRUCTIONS FOR USE
ZYCUBO ® [zye kyoo boe]
(copper histidinate)
for injection, for subcutaneous use
This Instructions for Use contains information on how to prepare and inject ZYCUBO. Read this Instructions for Use before you prepare and inject a dose of ZYCUBO for the first time and each time you get a ZYCUBO refill. There may be new information. This information does not take the place of talking to your healthcare provider about your child's medical condition or their treatment.
ZYCUBO is for subcutaneous injection only (inject directly under the skin). Always follow the specific instructions given by your healthcare provider.
- If you have questions about preparing or injecting ZYCUBO, call SentynlCares | ZYCUBO Patient Support Services at 1-888-251-2800.
Important information you need to know before preparing and injecting ZYCUBO:
- Your healthcare provider should show you the right way to prepare and inject your child's prescribed dose of ZYCUBO before you do this for the first time.
- Your healthcare provider will prescribe the amount of ZYCUBO needed for each dose for your child. Confirm the amount of ZYCUBO needed at each visit with your child's healthcare provider.
- ZYCUBO comes as a powder or cake in a vial. Each vial of ZYCUBO must be mixed with 0.9% sodium chloride to mix (dissolve) the powder or cake before use.
- Do not mix ZYCUBO with anything other than 0.9% sodium chloride.
- Vials of ZYCUBO are for 1 time use only. Throw the vial away after use, even if there is medicine left in the vial. Do not save for later use. Throw away used vials in your household trash.
- If your child misses a dose of ZYCUBO, inject the dose as soon as possible. Inject the next scheduled dose at least 6 hours after you finish injecting the missed dose.
- Do not expose ZYCUBO to any heat source, such as a microwave or hot water.
- Do not share needles and syringes. See Step 13: “Throw away (dispose of) used needles and syringes.”
Storing ZYCUBO and other supplies:
Vials of ZYCUBO before mixing:
- Store ZYCUBO in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep ZYCUBO vials in the original carton until you are ready to use it.
Vials of ZYCUBO after mixing:
- If you do not use the ZYCUBO solution right away after mixing, store the mixed ZYCUBO vial:
- in the refrigerator between 36°F to 46°F (2°C to 8°C) and use within 24 hours. Throw away (discard) the mixed ZYCUBO vial if not used within 24 hours.
- at room temperature between 68°F to 77°F (20°C to 25°C) and use within 4 hours. Throw away (discard) the mixed ZYCUBO vial if not used within 4 hours.
- Write the date and time you mixed ZYCUBO with 0.9% sodium chloride on the carton.
- Do not shake ZYCUBO after it has been mixed.
Other supplies:
- Store other supplies according to the manufacturer instructions (see Step 2: “Gather and check other supplies” for a list of supplies needed).
Keep ZYCUBO and all medicines out of the reach of children.
Preparing and injecting ZYCUBO
Step 1: Gather and check ZYCUBO vial for damage and expiration date
|
|
Step 2: Gather and check other supplies
|
|
|
Step 3: Wash your hands
If you have been told to wear gloves to prepare and inject ZYCUBO, put them on now. |
|
Step 4: Prepare the vials
Note: If you touch the rubber stopper, clean it again with a new alcohol wipe. |
|
Step 5: Prepare the syringe used for withdrawing sodium chloride
|
|
Step 6: Withdraw 1 mL of sodium chloride into syringe
|
|
Step 7: Mix ZYCUBO until dissolved
|
|
|
Step 8: Check solution
Check that the ZYCUBO solution is a blue color.
|
|
|
Step 9: Determine the injection time
After mixing, inject the ZYCUBO solution right away or inject the ZYCUBO solution:
|
|
Step 10: Prepare a syringe with the prescribed dose of ZYCUBO
|
|
|
Step 11: Prepare the injection site
ZYCUBO is injected under the skin (subcutaneously).
|
|
Step 12: Inject ZYCUBO
|
|
Step 13: Throw away (dispose of) used needles and syringes
|
|
| If you have questions about preparing or injecting ZYCUBO, call SentynlCares | ZYCUBO Patient Support Services at 1-888-251-2800. Manufactured by: Zydus Lifesciences Ltd., Vadodara – 391510, India Manufactured for: Sentynl Therapeutics, Inc., Solana Beach, CA 92075 This Instructions for Use has been approved by the U.S. Food and Drug Administration. Issued: 1/2026 |
11 Description
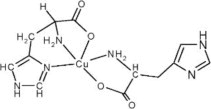
ZYCUBO (copper histidinate) for injection is a copper replacement product. The chemical name is copper, (L-histidinato-ϰN,ϰN3,ϰO)(L-histidinato-ϰN,ϰO)-, (SP-5-14-C)-. The molecular formula is C12H16CuN6O4, and the molecular weight is 371.84 g/mol. Copper histidinate is soluble in water.
The chemical structure is:
ZYCUBO is a sterile, preservative-free, blue lyophilized powder or cake for subcutaneous injection after reconstitution with 1 mL sterile 0.9% Sodium Chloride Injection, USP. Each single-dose vial contains 2.9 mg of copper histidinate (equivalent to 0.5 mg elemental copper). The resultant solution has a concentration of 2.9 mg/mL and a pH of 7.4.
2.7 Missed Dose
If a ZYCUBO dose is missed, administer the missed dose as soon as possible. Administer the next scheduled dose at least 6 hours after the administration of the missed dose.
8.4 Pediatric Use
The safety and effectiveness of ZYCUBO for the treatment of Menkes disease have been established in pediatric patients, and the information on this use is discussed throughout the labeling.
8.5 Geriatric Use
Menkes disease is a disease of pediatric patients. Clinical trials of ZYCUBO did not include patients 65 years of age and older.
14 Clinical Studies
The efficacy of ZYCUBO was evaluated in pediatric patients with Menkes disease (age at treatment initiation ranges 0.1 to 31.4 months) receiving 3 years of copper histidinate treatment in two open-label, single-arm clinical trials (Trial 1, NCT00001262 and Trial 2, NCT00811785). Data from ZYCUBO-treated patients in these two trials were compared to data from an untreated contemporaneous external control cohort as collected under a protocol amendment of Trial 2. In both trials, pediatric patients:
- Less than 1 year of age received 1.45 mg of ZYCUBO administered subcutaneously twice daily until 1 year of age.
- Equal to or greater than 1 year of age received 1.45 mg of ZYCUBO subcutaneously once daily for up to 3 years.
Overall survival was evaluated in a subset of the pooled population from Trial 1 and Trial 2, referred to as the pooled efficacy population. This efficacy population included only patients with Menkes disease who carried a severe pathogenic variant of the ATP7A gene (duplication/deletion, nonsense, or a canonical splice junction variant) and were born after 1999. There were 83 pediatric patients (66 ZYCUBO; 17 external control) in this pooled efficacy population: 21 patients (21 ZYCUBO) from Trial 1 and 62 patients (45 ZYCUBO; 17 external control) from Trial 2.
Patients in the pooled efficacy population were assigned to 1 of 4 cohorts as described in Table 2.
|
All values are in median (range) |
||||
|
1 Within 4 weeks of birth or within 4 weeks of birth corrected for prematurity (i.e., < 40 weeks' gestation) |
||||
|
2 After 4 weeks of birth or after 4 weeks of birth corrected for prematurity (i.e., < 40 weeks' gestation) |
||||
| Treated Cohorts | Untreated Cohorts | |||
|
ZYCUBO- Early Treatment
(ZYCUBO-ET) n=31 |
ZYCUBO- Late Treatment
(ZYCUBO-LT) n=35 |
External Control-Early Treatment
(EC-ET) n=17 |
External Control-Late Treatment
(EC-LT) n=16 |
|
| Eligibility | Started ZYCUBO treatment within 4 weeks of birth [1] | Started ZYCUBO treatment after 4 weeks of birth [2] |
|
|
| Age at diagnosis (months) | 0.1 (-4.5 – 1.9) | 4.8 (0.4 – 29.4) | 4.7 (2.1 – 22.2) | 5.6 (2.1 – 22.2) |
| Age at treatment initiation (months) | 0.4 (0.1 – 1.9) | 7.1 (1.3 – 31.4) | NA | NA |
| Treatment duration (months) | 34.1 (1.1 – 36) | 20 (1.3 – 36) | NA | NA |
In the 4 cohorts, 81 patients were male (98%) except for 2 female (2%) patients in ZYCUBO-LT. The pooled efficacy population included patients with the following race and ethnicity: 52 White (63%), 11 Hispanic (13%), 8 Black or African American (10%), 6 Unknown (7%), 4 Other (5%), and 2 Asian or Pacific Islander (2%). The majority of patients in all 4 cohorts were born prematurely: ZYCUBO-ET = 77%, ZYCUBO-LT = 66%, EC-ET = 82%, and EC-LT = 81%.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Copper Accumulation and Risk of Toxicity: Drug-Induced Kidney Injury, Liver Dysfunction, Hematological Abnormalities [see Warnings and Precautions (5.1)]
12.2 Pharmacodynamics
Patients with Menkes disease have low serum concentrations of copper and ceruloplasmin. Treatment with ZYCUBO increases serum copper and ceruloplasmin concentrations in patients with Menkes disease.
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of ZYCUBO have not been fully characterized.
12.3 Pharmacokinetics
The geometric mean (CV%) maximum serum concentration (Cmax) of copper histidinate was 67 (36%) ng/mL, the geometric mean (CV%) area under the concentration-time curve from time 0 to 24 hours (AUC0-24hr) was 186 (21%) ng•hr/mL, and the geometric mean (CV%) area under the concentration-time curve from time 0 to infinity (AUC0-inf) was 296 (15%) ng•hr/mL following a single subcutaneous dose of 3 mg copper histidinate (approximately twice the approved recommended dose for Menkes disease patients 1 year old or older) in healthy adult subjects.
At the recommended dosage, the mean (SD) serum copper concentration increased from a baseline concentration of 30 (25) mcg/dL to 114 (38) mcg/dL at 12 months, and gradually decreased over the 36-month treatment period, with a mean (SD) serum copper concentration of 63 (31) mcg/dL at 36 months. The mean (SD) serum ceruloplasmin concentration was 12 (12) mg/dL at baseline, 33 (11) mg/dL at 12 months, and 20 (8) mg/dL at 36 months [see Clinical Studies (14)].
1 Indications and Usage
ZYCUBO is indicated for the treatment of Menkes disease in pediatric patients.
12.1 Mechanism of Action
Menkes disease is an X-linked recessive disorder caused by pathogenic variants in the copper transport ATPase encoded by ATP7A. Patients with Menkes disease have impaired absorption of copper from their diet, impaired transport of copper across the blood-brain barrier, and dysregulation of many copper-dependent enzymes. ZYCUBO is a bioavailable copper replacement therapy that is administered as a subcutaneous injection to bypass the impaired gastrointestinal absorption observed in patients with Menkes disease.
5 Warnings and Precautions
Copper Accumulation and Risk of Toxicity: Exogenous administration of copper with ZYCUBO may lead to further copper accumulation and has the potential to result in drug-induced kidney injury, liver dysfunction, and hematological abnormalities. Monitor patients during ZYCUBO treatment. Adjust dosage if necessary. (2.2, 5.1, 6.1)
2 Dosage and Administration
- Before initiating ZYCUBO, obtain baseline serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count. (2.1)
- The recommended dosage of ZYCUBO in pediatric patients:
- Monitor serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC). (2.3)
- Reconstitute ZYCUBO and administer subcutaneously. (2.4, 2.6)
- See Full Prescribing Information for additional preparation, storage, and administration instructions. (2.4, 2.5, 2.6)
3 Dosage Forms and Strengths
For Injection: 2.9 mg of copper histidinate (equivalent to 0.5 mg elemental copper) as a blue lyophilized powder or cake in a single-dose vial for reconstitution.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety analysis from 2 open-label, single-arm clinical trials included a total of 129 ZYCUBO-treated patients with an age range from 0 to 48 months. Patients less than 1 year of age received ZYCUBO 1.45 mg twice daily, and patients 1 year of age and older received ZYCUBO 1.45 mg once daily. The median exposure duration was 24 months (range: 1 to 39 months) [see Clinical Studies (14)].
2.6 Administration Instructions
A caregiver may administer ZYCUBO to patients after proper training in subcutaneous injection technique if a healthcare provider determines that it is appropriate (see Instructions for Use ). Administer ZYCUBO using a sterile disposable 1 mL syringe and 1/2 inch injection needle (between 23 to 27 gauge).
Slowly withdraw 0.5 mL of reconstituted ZYCUBO solution from the vial and inject subcutaneously.
Administer ZYCUBO by subcutaneous injection at separate sites in the abdominal area (2 inches from the navel), buttocks, and the outer lateral aspect of the upper arm or thigh. Rotate injection sites with each injection to reduce the risk of lipodystrophy. Do not give injections into areas where the skin is scarred, tender, bruised, red, or hard.
Discard unused portion after each single use. Do not administer more than one dose from the vial.
17 Patient Counseling Information
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Instructions for Use).
2.5 Storage of Reconstituted Solution
If the reconstituted ZYCUBO vial is not used immediately, store the vial refrigerated at 2°C to 8°C (36° to 46°F) for up to 24 hours or at controlled room temperature at 20°C to 25°C (68°F to 77°F) for up to 4 hours.
Discard the reconstituted ZYCUBO vial if not used within 24 hours of refrigeration or within 4 hours at room temperature.
2.2 Recommended Dosage and Administration
The recommended dosage of ZYCUBO in pediatric patients:
- Less than 1 year of age is 1.45 mg administered subcutaneously twice daily (8-12 hours between injections).
- 1 year of age to less than 17 years of age is 1.45 mg administered subcutaneously once daily.
5.1 Copper Accumulation and Risk of Toxicity
Impaired copper transport in patients with Menkes disease can lead to copper accumulation and organ impairment in the kidneys, liver, and hematopoietic system. Treatment with ZYCUBO may lead to further copper accumulation and related toxicity, especially in the first two years of life given renal and hepatic immaturity.
Obtain baseline serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC). After initiating ZYCUBO, monitor laboratory values every 6 weeks for the first 6 months, then every 3 months for 18 months, and then every 6 months thereafter during ZYCUBO treatment. If laboratory abnormalities are detected, consider reducing the frequency of ZYCUBO administration or temporarily withholding or permanently discontinuing ZYCUBO. Return to increased frequency of laboratory monitoring when resuming a dosage as clinically indicated.
2.1 Recommended Testing Before Initiating Zycubo
Before initiating ZYCUBO, obtain baseline serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC) [see Warnings and Precautions (5.1)].
2.3 Dosage and Administration Modifications and Monitoring
Monitor serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC) every 6 weeks for the first 6 months, then every 3 months for 18 months, and then every 6 months thereafter during ZYCUBO treatment. If laboratory abnormalities are detected, consider reducing the frequency of ZYCUBO administration or temporarily withholding or permanently discontinuing ZYCUBO. Return to increased frequency of laboratory evaluation when resuming a dosage as clinically indicated [see Warnings and Precautions (5.1)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
ZYCUBO is not indicated for the treatment of Occipital Horn Syndrome.
Section 51945-4 (51945-4)
Principal Display Panel - 2.9 mg Carton Label
NDC 42358-329-01
Zycubo
®
(copper histidinate) for injection
2.9 mg/vial
For subcutaneous injection
One Single-Dose vial
Discard unused portion after each single use.
Do not administer more than one dose from the vial.
Rx only
Section 59845-8 (59845-8)
INSTRUCTIONS FOR USE
ZYCUBO ® [zye kyoo boe]
(copper histidinate)
for injection, for subcutaneous use
This Instructions for Use contains information on how to prepare and inject ZYCUBO. Read this Instructions for Use before you prepare and inject a dose of ZYCUBO for the first time and each time you get a ZYCUBO refill. There may be new information. This information does not take the place of talking to your healthcare provider about your child's medical condition or their treatment.
ZYCUBO is for subcutaneous injection only (inject directly under the skin). Always follow the specific instructions given by your healthcare provider.
- If you have questions about preparing or injecting ZYCUBO, call SentynlCares | ZYCUBO Patient Support Services at 1-888-251-2800.
Important information you need to know before preparing and injecting ZYCUBO:
- Your healthcare provider should show you the right way to prepare and inject your child's prescribed dose of ZYCUBO before you do this for the first time.
- Your healthcare provider will prescribe the amount of ZYCUBO needed for each dose for your child. Confirm the amount of ZYCUBO needed at each visit with your child's healthcare provider.
- ZYCUBO comes as a powder or cake in a vial. Each vial of ZYCUBO must be mixed with 0.9% sodium chloride to mix (dissolve) the powder or cake before use.
- Do not mix ZYCUBO with anything other than 0.9% sodium chloride.
- Vials of ZYCUBO are for 1 time use only. Throw the vial away after use, even if there is medicine left in the vial. Do not save for later use. Throw away used vials in your household trash.
- If your child misses a dose of ZYCUBO, inject the dose as soon as possible. Inject the next scheduled dose at least 6 hours after you finish injecting the missed dose.
- Do not expose ZYCUBO to any heat source, such as a microwave or hot water.
- Do not share needles and syringes. See Step 13: “Throw away (dispose of) used needles and syringes.”
Storing ZYCUBO and other supplies:
Vials of ZYCUBO before mixing:
- Store ZYCUBO in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep ZYCUBO vials in the original carton until you are ready to use it.
Vials of ZYCUBO after mixing:
- If you do not use the ZYCUBO solution right away after mixing, store the mixed ZYCUBO vial:
- in the refrigerator between 36°F to 46°F (2°C to 8°C) and use within 24 hours. Throw away (discard) the mixed ZYCUBO vial if not used within 24 hours.
- at room temperature between 68°F to 77°F (20°C to 25°C) and use within 4 hours. Throw away (discard) the mixed ZYCUBO vial if not used within 4 hours.
- Write the date and time you mixed ZYCUBO with 0.9% sodium chloride on the carton.
- Do not shake ZYCUBO after it has been mixed.
Other supplies:
- Store other supplies according to the manufacturer instructions (see Step 2: “Gather and check other supplies” for a list of supplies needed).
Keep ZYCUBO and all medicines out of the reach of children.
Preparing and injecting ZYCUBO
Step 1: Gather and check ZYCUBO vial for damage and expiration date
|
|
Step 2: Gather and check other supplies
|
|
|
Step 3: Wash your hands
If you have been told to wear gloves to prepare and inject ZYCUBO, put them on now. |
|
Step 4: Prepare the vials
Note: If you touch the rubber stopper, clean it again with a new alcohol wipe. |
|
Step 5: Prepare the syringe used for withdrawing sodium chloride
|
|
Step 6: Withdraw 1 mL of sodium chloride into syringe
|
|
Step 7: Mix ZYCUBO until dissolved
|
|
|
Step 8: Check solution
Check that the ZYCUBO solution is a blue color.
|
|
|
Step 9: Determine the injection time
After mixing, inject the ZYCUBO solution right away or inject the ZYCUBO solution:
|
|
Step 10: Prepare a syringe with the prescribed dose of ZYCUBO
|
|
|
Step 11: Prepare the injection site
ZYCUBO is injected under the skin (subcutaneously).
|
|
Step 12: Inject ZYCUBO
|
|
Step 13: Throw away (dispose of) used needles and syringes
|
|
| If you have questions about preparing or injecting ZYCUBO, call SentynlCares | ZYCUBO Patient Support Services at 1-888-251-2800. Manufactured by: Zydus Lifesciences Ltd., Vadodara – 391510, India Manufactured for: Sentynl Therapeutics, Inc., Solana Beach, CA 92075 This Instructions for Use has been approved by the U.S. Food and Drug Administration. Issued: 1/2026 |
11 Description (11 DESCRIPTION)
ZYCUBO (copper histidinate) for injection is a copper replacement product. The chemical name is copper, (L-histidinato-ϰN,ϰN3,ϰO)(L-histidinato-ϰN,ϰO)-, (SP-5-14-C)-. The molecular formula is C12H16CuN6O4, and the molecular weight is 371.84 g/mol. Copper histidinate is soluble in water.
The chemical structure is:
ZYCUBO is a sterile, preservative-free, blue lyophilized powder or cake for subcutaneous injection after reconstitution with 1 mL sterile 0.9% Sodium Chloride Injection, USP. Each single-dose vial contains 2.9 mg of copper histidinate (equivalent to 0.5 mg elemental copper). The resultant solution has a concentration of 2.9 mg/mL and a pH of 7.4.
2.7 Missed Dose
If a ZYCUBO dose is missed, administer the missed dose as soon as possible. Administer the next scheduled dose at least 6 hours after the administration of the missed dose.
8.4 Pediatric Use
The safety and effectiveness of ZYCUBO for the treatment of Menkes disease have been established in pediatric patients, and the information on this use is discussed throughout the labeling.
8.5 Geriatric Use
Menkes disease is a disease of pediatric patients. Clinical trials of ZYCUBO did not include patients 65 years of age and older.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of ZYCUBO was evaluated in pediatric patients with Menkes disease (age at treatment initiation ranges 0.1 to 31.4 months) receiving 3 years of copper histidinate treatment in two open-label, single-arm clinical trials (Trial 1, NCT00001262 and Trial 2, NCT00811785). Data from ZYCUBO-treated patients in these two trials were compared to data from an untreated contemporaneous external control cohort as collected under a protocol amendment of Trial 2. In both trials, pediatric patients:
- Less than 1 year of age received 1.45 mg of ZYCUBO administered subcutaneously twice daily until 1 year of age.
- Equal to or greater than 1 year of age received 1.45 mg of ZYCUBO subcutaneously once daily for up to 3 years.
Overall survival was evaluated in a subset of the pooled population from Trial 1 and Trial 2, referred to as the pooled efficacy population. This efficacy population included only patients with Menkes disease who carried a severe pathogenic variant of the ATP7A gene (duplication/deletion, nonsense, or a canonical splice junction variant) and were born after 1999. There were 83 pediatric patients (66 ZYCUBO; 17 external control) in this pooled efficacy population: 21 patients (21 ZYCUBO) from Trial 1 and 62 patients (45 ZYCUBO; 17 external control) from Trial 2.
Patients in the pooled efficacy population were assigned to 1 of 4 cohorts as described in Table 2.
|
All values are in median (range) |
||||
|
1 Within 4 weeks of birth or within 4 weeks of birth corrected for prematurity (i.e., < 40 weeks' gestation) |
||||
|
2 After 4 weeks of birth or after 4 weeks of birth corrected for prematurity (i.e., < 40 weeks' gestation) |
||||
| Treated Cohorts | Untreated Cohorts | |||
|
ZYCUBO- Early Treatment
(ZYCUBO-ET) n=31 |
ZYCUBO- Late Treatment
(ZYCUBO-LT) n=35 |
External Control-Early Treatment
(EC-ET) n=17 |
External Control-Late Treatment
(EC-LT) n=16 |
|
| Eligibility | Started ZYCUBO treatment within 4 weeks of birth [1] | Started ZYCUBO treatment after 4 weeks of birth [2] |
|
|
| Age at diagnosis (months) | 0.1 (-4.5 – 1.9) | 4.8 (0.4 – 29.4) | 4.7 (2.1 – 22.2) | 5.6 (2.1 – 22.2) |
| Age at treatment initiation (months) | 0.4 (0.1 – 1.9) | 7.1 (1.3 – 31.4) | NA | NA |
| Treatment duration (months) | 34.1 (1.1 – 36) | 20 (1.3 – 36) | NA | NA |
In the 4 cohorts, 81 patients were male (98%) except for 2 female (2%) patients in ZYCUBO-LT. The pooled efficacy population included patients with the following race and ethnicity: 52 White (63%), 11 Hispanic (13%), 8 Black or African American (10%), 6 Unknown (7%), 4 Other (5%), and 2 Asian or Pacific Islander (2%). The majority of patients in all 4 cohorts were born prematurely: ZYCUBO-ET = 77%, ZYCUBO-LT = 66%, EC-ET = 82%, and EC-LT = 81%.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Copper Accumulation and Risk of Toxicity: Drug-Induced Kidney Injury, Liver Dysfunction, Hematological Abnormalities [see Warnings and Precautions (5.1)]
12.2 Pharmacodynamics
Patients with Menkes disease have low serum concentrations of copper and ceruloplasmin. Treatment with ZYCUBO increases serum copper and ceruloplasmin concentrations in patients with Menkes disease.
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of ZYCUBO have not been fully characterized.
12.3 Pharmacokinetics
The geometric mean (CV%) maximum serum concentration (Cmax) of copper histidinate was 67 (36%) ng/mL, the geometric mean (CV%) area under the concentration-time curve from time 0 to 24 hours (AUC0-24hr) was 186 (21%) ng•hr/mL, and the geometric mean (CV%) area under the concentration-time curve from time 0 to infinity (AUC0-inf) was 296 (15%) ng•hr/mL following a single subcutaneous dose of 3 mg copper histidinate (approximately twice the approved recommended dose for Menkes disease patients 1 year old or older) in healthy adult subjects.
At the recommended dosage, the mean (SD) serum copper concentration increased from a baseline concentration of 30 (25) mcg/dL to 114 (38) mcg/dL at 12 months, and gradually decreased over the 36-month treatment period, with a mean (SD) serum copper concentration of 63 (31) mcg/dL at 36 months. The mean (SD) serum ceruloplasmin concentration was 12 (12) mg/dL at baseline, 33 (11) mg/dL at 12 months, and 20 (8) mg/dL at 36 months [see Clinical Studies (14)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZYCUBO is indicated for the treatment of Menkes disease in pediatric patients.
12.1 Mechanism of Action
Menkes disease is an X-linked recessive disorder caused by pathogenic variants in the copper transport ATPase encoded by ATP7A. Patients with Menkes disease have impaired absorption of copper from their diet, impaired transport of copper across the blood-brain barrier, and dysregulation of many copper-dependent enzymes. ZYCUBO is a bioavailable copper replacement therapy that is administered as a subcutaneous injection to bypass the impaired gastrointestinal absorption observed in patients with Menkes disease.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Copper Accumulation and Risk of Toxicity: Exogenous administration of copper with ZYCUBO may lead to further copper accumulation and has the potential to result in drug-induced kidney injury, liver dysfunction, and hematological abnormalities. Monitor patients during ZYCUBO treatment. Adjust dosage if necessary. (2.2, 5.1, 6.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Before initiating ZYCUBO, obtain baseline serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count. (2.1)
- The recommended dosage of ZYCUBO in pediatric patients:
- Monitor serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC). (2.3)
- Reconstitute ZYCUBO and administer subcutaneously. (2.4, 2.6)
- See Full Prescribing Information for additional preparation, storage, and administration instructions. (2.4, 2.5, 2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For Injection: 2.9 mg of copper histidinate (equivalent to 0.5 mg elemental copper) as a blue lyophilized powder or cake in a single-dose vial for reconstitution.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety analysis from 2 open-label, single-arm clinical trials included a total of 129 ZYCUBO-treated patients with an age range from 0 to 48 months. Patients less than 1 year of age received ZYCUBO 1.45 mg twice daily, and patients 1 year of age and older received ZYCUBO 1.45 mg once daily. The median exposure duration was 24 months (range: 1 to 39 months) [see Clinical Studies (14)].
2.6 Administration Instructions
A caregiver may administer ZYCUBO to patients after proper training in subcutaneous injection technique if a healthcare provider determines that it is appropriate (see Instructions for Use ). Administer ZYCUBO using a sterile disposable 1 mL syringe and 1/2 inch injection needle (between 23 to 27 gauge).
Slowly withdraw 0.5 mL of reconstituted ZYCUBO solution from the vial and inject subcutaneously.
Administer ZYCUBO by subcutaneous injection at separate sites in the abdominal area (2 inches from the navel), buttocks, and the outer lateral aspect of the upper arm or thigh. Rotate injection sites with each injection to reduce the risk of lipodystrophy. Do not give injections into areas where the skin is scarred, tender, bruised, red, or hard.
Discard unused portion after each single use. Do not administer more than one dose from the vial.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Instructions for Use).
2.5 Storage of Reconstituted Solution
If the reconstituted ZYCUBO vial is not used immediately, store the vial refrigerated at 2°C to 8°C (36° to 46°F) for up to 24 hours or at controlled room temperature at 20°C to 25°C (68°F to 77°F) for up to 4 hours.
Discard the reconstituted ZYCUBO vial if not used within 24 hours of refrigeration or within 4 hours at room temperature.
2.2 Recommended Dosage and Administration
The recommended dosage of ZYCUBO in pediatric patients:
- Less than 1 year of age is 1.45 mg administered subcutaneously twice daily (8-12 hours between injections).
- 1 year of age to less than 17 years of age is 1.45 mg administered subcutaneously once daily.
5.1 Copper Accumulation and Risk of Toxicity
Impaired copper transport in patients with Menkes disease can lead to copper accumulation and organ impairment in the kidneys, liver, and hematopoietic system. Treatment with ZYCUBO may lead to further copper accumulation and related toxicity, especially in the first two years of life given renal and hepatic immaturity.
Obtain baseline serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC). After initiating ZYCUBO, monitor laboratory values every 6 weeks for the first 6 months, then every 3 months for 18 months, and then every 6 months thereafter during ZYCUBO treatment. If laboratory abnormalities are detected, consider reducing the frequency of ZYCUBO administration or temporarily withholding or permanently discontinuing ZYCUBO. Return to increased frequency of laboratory monitoring when resuming a dosage as clinically indicated.
2.1 Recommended Testing Before Initiating Zycubo (2.1 Recommended Testing Before Initiating ZYCUBO)
Before initiating ZYCUBO, obtain baseline serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC) [see Warnings and Precautions (5.1)].
2.3 Dosage and Administration Modifications and Monitoring
Monitor serum copper and ceruloplasmin levels, serum electrolytes, kidney and liver function, and complete blood count (CBC) every 6 weeks for the first 6 months, then every 3 months for 18 months, and then every 6 months thereafter during ZYCUBO treatment. If laboratory abnormalities are detected, consider reducing the frequency of ZYCUBO administration or temporarily withholding or permanently discontinuing ZYCUBO. Return to increased frequency of laboratory evaluation when resuming a dosage as clinically indicated [see Warnings and Precautions (5.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:36.959402 · Updated: 2026-03-14T22:44:06.575433