These Highlights Do Not Include All The Information Needed To Use Tretinoin Gel Microsphere Safely And Effectively. See Full Prescribing Information For Tretinoin Gel Microsphere.
d3c528d2-429d-4f37-9ebf-eee8b9fd3050
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Tretinoin Gel Microsphere is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 12 years of age and older.
Indications and Usage
Tretinoin Gel Microsphere is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 12 years of age and older.
Dosage and Administration
For topical use only. Not for oral, ophthalmic, or intravaginal use. • Prior to Tretinoin Gel Microsphere use, thoroughly cleanse area(s) with a mild, non-medicated cleanser then pat the skin dry. • When applying Tretinoin Gel Microsphere, keep away from the eyes, the mouth, paranasal creases of the nose, and mucous membranes. • Apply a thin layer of Tretinoin Gel Microsphere (0.04%, 0.08%, or 0.1%) to skin where acne lesions appear (cover the entire affected area), once daily in the evening. Do not apply more than a thin layer [see Warning and Precautions (5.1) ] . Improvements in acne lesions may be noticed after two weeks of Tretinoin Gel Microsphere therapy, but more than seven weeks of therapy may be needed for sustained benefit. If Tretinoin Gel Microsphere was temporarily discontinued due to local adverse reactions, Tretinoin Gel Microsphere therapy may be resumed upon resolution of local adverse reactions.
Warnings and Precautions
• Local Skin Irritation : Tretinoin Gel Microsphere can cause local skin irritation, including excessive dryness, redness, swelling, peeling, itching, blistering, burning, or stinging. ( 5.1 ) o Avoid use on eczematous skin or during weather extremes, such as severe wind or cold. o To reduce the risk of local skin irritation, wash the treated skin gently, using a mild, non-medicated soap, avoid washing the treated skin too often or scrubbing it hard when washing, and apply a topical moisturizer. o If severe local skin irritation occurs, discontinue use temporarily or permanently. • Initial Worsening of Inflammatory Acne Vulgaris : During the early weeks of Tretinoin Gel Microsphere treatment, an apparent exacerbation of inflammatory lesions may occur. If Tretinoin Gel Microsphere is tolerated, this should not be considered a reason to discontinue therapy. ( 5.2 ) • Photosensitivity : Tretinoin Gel Microsphere can cause photosensitivity. Advise patients to avoid or minimize unnecessary exposure to UV light, including sunlight and sunlamps. Advise patients to use sunscreen (SPF ≥15) and sun-protective clothing if UV light exposure cannot be avoided. Avoid use on sunburn skin. ( 5.3 )
Contraindications
None.
Adverse Reactions
Most common adverse reactions were skin irritation, skin burning, erythema, peeling, dryness, itching, and dermatitis. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
How Supplied Tretinoin Gel Microsphere (tretinoin) is opaque and white to very pale yellow in color and is supplied as follows: Strength Amount of Tretinoin in One Gram of Gel Quantity/Package Type NDC 0.04% 0.4 mg 20 gram tube 68682-514-92 45 gram tube 68682-514-94 50 gram pump 68682-514-95 0.08% 0.8 mg 50 gram pump 68682-515-95 0.1% 1 mg 20 gram tube 68682-513-82 45 gram tube 68682-513-84 50 gram pump 68682-513-85 Storage and Handling Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Store pump upright.
How Supplied
How Supplied Tretinoin Gel Microsphere (tretinoin) is opaque and white to very pale yellow in color and is supplied as follows: Strength Amount of Tretinoin in One Gram of Gel Quantity/Package Type NDC 0.04% 0.4 mg 20 gram tube 68682-514-92 45 gram tube 68682-514-94 50 gram pump 68682-514-95 0.08% 0.8 mg 50 gram pump 68682-515-95 0.1% 1 mg 20 gram tube 68682-513-82 45 gram tube 68682-513-84 50 gram pump 68682-513-85 Storage and Handling Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Store pump upright.
Medication Information
Warnings and Precautions
• Local Skin Irritation : Tretinoin Gel Microsphere can cause local skin irritation, including excessive dryness, redness, swelling, peeling, itching, blistering, burning, or stinging. ( 5.1 ) o Avoid use on eczematous skin or during weather extremes, such as severe wind or cold. o To reduce the risk of local skin irritation, wash the treated skin gently, using a mild, non-medicated soap, avoid washing the treated skin too often or scrubbing it hard when washing, and apply a topical moisturizer. o If severe local skin irritation occurs, discontinue use temporarily or permanently. • Initial Worsening of Inflammatory Acne Vulgaris : During the early weeks of Tretinoin Gel Microsphere treatment, an apparent exacerbation of inflammatory lesions may occur. If Tretinoin Gel Microsphere is tolerated, this should not be considered a reason to discontinue therapy. ( 5.2 ) • Photosensitivity : Tretinoin Gel Microsphere can cause photosensitivity. Advise patients to avoid or minimize unnecessary exposure to UV light, including sunlight and sunlamps. Advise patients to use sunscreen (SPF ≥15) and sun-protective clothing if UV light exposure cannot be avoided. Avoid use on sunburn skin. ( 5.3 )
Indications and Usage
Tretinoin Gel Microsphere is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 12 years of age and older.
Dosage and Administration
For topical use only. Not for oral, ophthalmic, or intravaginal use. • Prior to Tretinoin Gel Microsphere use, thoroughly cleanse area(s) with a mild, non-medicated cleanser then pat the skin dry. • When applying Tretinoin Gel Microsphere, keep away from the eyes, the mouth, paranasal creases of the nose, and mucous membranes. • Apply a thin layer of Tretinoin Gel Microsphere (0.04%, 0.08%, or 0.1%) to skin where acne lesions appear (cover the entire affected area), once daily in the evening. Do not apply more than a thin layer [see Warning and Precautions (5.1) ] . Improvements in acne lesions may be noticed after two weeks of Tretinoin Gel Microsphere therapy, but more than seven weeks of therapy may be needed for sustained benefit. If Tretinoin Gel Microsphere was temporarily discontinued due to local adverse reactions, Tretinoin Gel Microsphere therapy may be resumed upon resolution of local adverse reactions.
Contraindications
None.
Adverse Reactions
Most common adverse reactions were skin irritation, skin burning, erythema, peeling, dryness, itching, and dermatitis. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
How Supplied Tretinoin Gel Microsphere (tretinoin) is opaque and white to very pale yellow in color and is supplied as follows: Strength Amount of Tretinoin in One Gram of Gel Quantity/Package Type NDC 0.04% 0.4 mg 20 gram tube 68682-514-92 45 gram tube 68682-514-94 50 gram pump 68682-514-95 0.08% 0.8 mg 50 gram pump 68682-515-95 0.1% 1 mg 20 gram tube 68682-513-82 45 gram tube 68682-513-84 50 gram pump 68682-513-85 Storage and Handling Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Store pump upright.
How Supplied
How Supplied Tretinoin Gel Microsphere (tretinoin) is opaque and white to very pale yellow in color and is supplied as follows: Strength Amount of Tretinoin in One Gram of Gel Quantity/Package Type NDC 0.04% 0.4 mg 20 gram tube 68682-514-92 45 gram tube 68682-514-94 50 gram pump 68682-514-95 0.08% 0.8 mg 50 gram pump 68682-515-95 0.1% 1 mg 20 gram tube 68682-513-82 45 gram tube 68682-513-84 50 gram pump 68682-513-85 Storage and Handling Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Store pump upright.
Description
Tretinoin Gel Microsphere is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 12 years of age and older.
Section 42230-3
|
Patient Information Tretinoin Gel Microsphere, 0.1%, 0.08% and 0.04% for topical use |
|
Important information: Tretinoin Gel Microsphere is for use on skin (topical use) only. Keep Tretinoin Gel Microsphere away from your eyes, mouth, vagina, or the corners of your nose. |
|
What is Tretinoin Gel Microsphere? Tretinoin Gel Microsphere is a prescription medicine used on the skin (topical) to treat acne in adults and children 12 years of age and older. Acne is a condition in which the skin has blackheads, whiteheads, and other pimples. It is not known if Tretinoin Gel Microsphere is safe and effective in children under 12 years of age. |
|
Before using Tretinoin Gel Microsphere, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you use any skin products that contain benzoyl peroxide, sulfur, resorcinol, or salicylic acid. Using these topical skin products may increase the irritation of your skin when used with Tretinoin Gel Microsphere. |
|
How should I use Tretinoin Gel Microsphere?
What should I avoid while using Tretinoin Gel Microsphere?
|
|
What are the possible side effects of Tretinoin Gel Microsphere? Tretinoin Gel Microsphere can cause serious side effects, including:
The most common side effects of Tretinoin Gel Microsphere include skin redness, pain, and itching. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Tretinoin Gel Microsphere. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store Tretinoin Gel Microsphere?
Keep Tretinoin Gel Microsphere and all medicines out of the reach of children. |
|
General information about the safe and effective use of Tretinoin Gel Microsphere. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Tretinoin Gel Microsphere for a condition for which it was not prescribed. Do not give Tretinoin Gel Microsphere to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Tretinoin Gel Microsphere that is written for health professionals. |
|
What are the ingredients of Tretinoin Gel Microsphere? Active ingredient: tretinoin Inactive ingredients: benzyl alcohol, butylated hydroxytoluene, carbomer 974P, cyclomethicone and dimethicone copolyol, disodium EDTA, glycerin, methyl methacrylate/glycol dimethacrylate crosspolymer, PPG-20 methyl glucose ether distearate, propylene glycol, propylene glycol dicaprylate/dicaprate, purified water, sorbic acid, and trolamine |
|
Distributed by: Oceanside Pharmaceuticals, a division of Bausch Health US, LLC, Bridgewater, NJ 08807 USA Manufactured by: Bausch Health Companies Inc., Laval, Quebec H7L 4A8, Canada Microsponge is a registered trademark of AMCOL International Corporation. All other products/brand names and/or logos are trademarks of the respective owners. © 2025 Bausch Health Companies Inc. or its affiliates For more information, call 1-800-321-4576. |
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 09/2025
9515306
10 Overdosage
Oral ingestion of large amounts of Tretinoin Gel Microsphere may lead to the same adverse reactions as those associated with excessive oral intake of Vitamin A.
Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for overdose recommendations.
8.1 Pregnancy
Risk Summary
Available data from published prospective observational studies and retrospective cohort studies over decades of use of topical tretinoin in pregnant women have not established a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes (see Data).
In animal reproduction studies with pregnant rats, alterations in the vertebrae and ribs of offspring were observed with daily topical dosing of 0.1% Tretinoin Gel (microsponge) during organogenesis at 5 to 10 times the maximum recommended human dose (MRHD). In animal reproduction studies with pregnant rabbits, fetal malformations, such as domed head and hydrocephaly, were observed in the offspring with daily topical dosing of 0.1% Tretinoin Gel (microsponge) during organogenesis at 10 to 19 times the MRHD [see Data].
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4%, and 15% to 20%, respectively.
Data
Human Data: Published data from prospective observational studies and retrospective cohort studies on the use of topical tretinoin products during pregnancy have not identified an association with topical tretinoin and major birth defects or miscarriage. The available studies have methodologic limitations, including potential misclassification of exposure, small sample size and in some cases, lack of physical exam by an expert in birth defects.
Animal Data: For purposes of comparison of the animal exposure to systemic human exposure, the MRHD applied topically is defined as 1 gram of Tretinoin Gel Microsphere, 0.1%, applied daily to a 60 kg person (0.017 mg tretinoin/kg body weight).
Pregnant rats were treated with 0.1% Tretinoin Gel (microsponge) at daily dermal doses of 0.2, 0.5, and 1 mg/kg/day tretinoin on gestation days 6-15. Alterations were seen in the vertebrae and ribs of the affected offsprings at 0.5 mg/kg/day tretinoin, 5 to 10 times the MRHD based on body surface area (BSA) comparison.
Pregnant rabbits were treated with 0.1% Tretinoin Gel (microsponge) at daily dermal doses of 0.2, 0.5, and 1 mg/kg/day tretinoin on gestation days 7-19. Doses were administered topically for 24 hours a day while wearing Elizabethan collars to prevent ingestion of the drug. Increased incidences of certain alterations, including domed head and hydrocephaly, typical of retinoid-induced fetal malformations in this species were observed at doses of 0.5 and 1 mg/kg/day. Similar malformations were not observed in the offspring at 0.2 mg/kg/day, 4 times the MRHD based on BSA comparison. In a second rabbit study, pregnant rabbits were treated with 0.1% Tretinoin Gel (microsponge) at daily dermal doses of 0.5 or 1 mg/kg/day tretinoin on gestation days 7-19. Doses were administered topically for six hours per day while pregnant rabbits were restrained in stocks to prevent ingestion. The offspring of pregnant rabbits exposed to 0.5 or 1 mg/kg/day tretinoin did not show any malformations at doses up to 19 times (1.0 mg/kg/day) the MRHD based on BSA comparison, but fetal resorptions were increased at 0.5 mg/kg (10 times the MRHD based on BSA comparison).
Malformations (shortened or kinked tail) were observed in the offspring of pregnant rats treated with topical tretinoin at doses greater than 1 mg/kg/day during the period of organogenesis (10 times the MRHD based on BSA comparison). Anomalies (humerus: short 13%, bent 6%, os parietal incompletely ossified 14%) have also been reported in offspring when 10 mg/kg/day was topically applied to pregnant rats during the period of organogenesis. Supernumerary ribs have been a consistent finding in newborn rats when dams were treated topically or orally with retinoids.
Oral administration of tretinoin during organogenesis has been shown to cause malformations in the offspring of rats, mice, rabbits, hamsters, and nonhuman primates. Fetal malformations and death were observed when tretinoin was orally administered to pregnant rats during organogenesis at doses greater than 1 mg/kg/day (10 times the MRHD based on BSA comparison). Fetal malformations were reported at doses of 10 mg/kg/day or greater when administered to pregnant cynomolgus monkeys, but none were observed at 5 mg/kg/day (95 times the MRHD based on BSA comparison), although increased skeletal variations were observed at all doses. Dose-dependent increases in embryolethality and abortion also were reported. Similar results have also been reported in pigtail macaques.
In peri- and postnatal development studies in rats with oral tretinoin, decreased survival of neonates and growth retardation were observed at doses in excess of 2 mg/kg/day (19 times the MRHD based on BSA comparison).
Oral tretinoin has been shown to be fetotoxic in rats when administered at doses 24 times the MRHD based on BSA comparison.
Topical tretinoin has been shown to be fetotoxic in rabbits when administered at doses 10 times the MRHD based on BSA comparison.
8.2 Lactation
Risk Summary
There are no data on the presence of tretinoin or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. It is not known whether topical administration of tretinoin could result in sufficient systemic absorption to produce detectable quantities in human milk (see Clinical Considerations).
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Tretinoin Gel Microsphere and any potential adverse effects on the breastfed infant from the Tretinoin Gel Microsphere or from the underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the infant via human milk, during breastfeeding, use Tretinoin Gel Microsphere on the smallest area of skin with acne and for the shortest duration possible. Advise breastfeeding women not to apply Tretinoin Gel Microsphere directly to the nipple and areola to avoid direct infant exposure.
11 Description
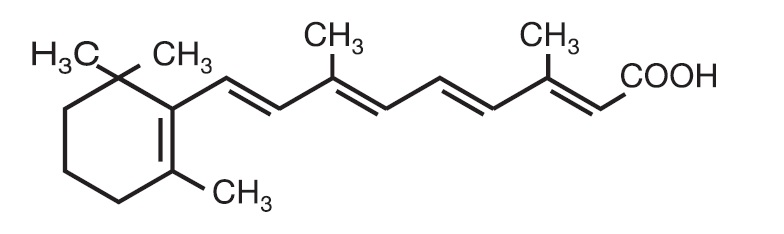
Chemically, tretinoin is all-trans-retinoic acid, also known as (all-E)-3,7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid. Tretinoin is a retinoid and a metabolite of naturally occurring Vitamin A. Tretinoin has a molecular weight of 300.44, a molecular formula of C20H28O2 and the following chemical structure:
Tretinoin Gel Microsphere (tretinoin) gel is for topical use. Each gram of Tretinoin Gel Microsphere gel:
-
•0.04%, contains 0.4 mg of tretinoin
-
•0.08%, contains 0.8 mg of tretinoin
-
•0.1%, contains 1 mg of tretinoin
The formulation contains methyl methacrylate/glycol dimethacrylate crosspolymer (MICROSPONGE® System), propylene glycol dicaprylate/dicaprate and butylated hydroxytoluene. Other components consist of benzyl alcohol, butylated hydroxytoluene, carbomer 974P, cyclomethicone and dimethicone copolyol, disodium EDTA, glycerin, PPG-20 methyl glucose ether distearate, propylene glycol, purified water, sorbic acid, and trolamine.
8.4 Pediatric Use
The safety and effectiveness of Tretinoin Gel Microsphere for topical application for the treatment of acne vulgaris have been established in pediatric patients aged 12 years and older [see Clinical Studies (14)]. The safety and effectiveness of Tretinoin Gel Microsphere have not been established in pediatric patients younger than 12 years of age.
8.5 Geriatric Use
Clinical trials of Tretinoin Gel Microsphere did not include sufficient numbers of subjects aged 65 and over to determine whether they responded differently from younger adult subjects. Acne vulgaris is largely a disease of pediatric and young adult patients. Clinical studies of Tretinoin Gel Microsphere did not include patients 65 years of age and older.
4 Contraindications
None.
6 Adverse Reactions
Most common adverse reactions were skin irritation, skin burning, erythema, peeling, dryness, itching, and dermatitis. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
5.3 Photosensitivity
Tretinoin Gel Microsphere can cause photosensitivity. Advise patients to avoid or minimize unnecessary exposure to ultraviolet (UV) light, including sunlight and sunlamps, while using Tretinoin Gel Microsphere. Advise patients with sunburn to not use Tretinoin Gel Microsphere until the sunburn fully recovers.
Advise patients, especially those who may be required to have extended periods of UV light exposure (e.g., due to occupation or sports), those with inherent sensitivity to the sun, or those using drugs that cause photosensitivity, to use sun protection daily in the form of sunscreen (sun protection factor [SPF] ≥ 15) and sun-protective clothing, when UV exposure cannot be avoided, even on days when it is not sunny or inside activities are expected.
12.3 Pharmacokinetics
Absorption
Tretinoin is a metabolite of Vitamin A. Percutaneous absorption, as determined by the cumulative excretion of radiolabeled drug into urine and feces, was assessed in 44 healthy males and females after single and repeated daily tretinoin applications up to 28 days of 500 mg of a 0.1% tretinoin gel product. In this assessment, estimates of in vivo bioavailability, mean (SD) were 0.82 (0.11)% and 1.41 (0.54)%, for single and multiple daily topical applications, respectively. The plasma concentrations of tretinoin and its metabolites, 13-cis-retinoic acid, all-trans-4-oxo-retinoic acid, and 13-cis-4-oxo-retinoic acid, generally ranged from 1 to 3 ng/mL and were essentially unaltered after either single or multiple daily applications of Tretinoin Gel Microsphere, 0.1%, relative to baseline levels.
Clinical pharmacokinetic studies have not been performed with Tretinoin Gel Microsphere, 0.08%, and 0.04%.
1 Indications and Usage
Tretinoin Gel Microsphere is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 12 years of age and older.
12.1 Mechanism of Action
Although tretinoin activates three members of the retinoic acid (RAR) nuclear receptors (RARα, RARβ, and RARγ) which may act to modify gene expression, subsequent protein synthesis, and epithelial cell growth and differentiation, it has not been established whether the clinical effects of tretinoin are mediated through activation of retinoic acid receptors and/or other mechanisms.
The exact mechanism of action of topical tretinoin for treatment of acne vulgaris is unknown. Current evidence suggests that topical tretinoin decreases cohesiveness of follicular epithelial cells with decreased microcomedone formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.
5.1 Local Skin Irritation
Tretinoin Gel Microsphere can cause local skin irritation, including excessive dryness, redness, swelling, peeling, itching, blistering, burning, or stinging [see Adverse Reactions (6.1)]. Use of Tretinoin Gel Microsphere in greater than the recommended dosage (more frequent than once daily application or excessive application) will not result in more rapid or improved acne results and may result in marked redness, peeling, or discomfort.
Tretinoin has been reported to cause severe local skin irritation on eczematous skin.
Weather extremes, such as severe wind or cold, may increase the risk of skin irritation in patients using Tretinoin Gel Microsphere.
To reduce the risk of local skin irritation, instruct Tretinoin Gel Microsphere-treated patients to:
-
•Avoid use of Tretinoin Gel Microsphere in areas affected by eczema.
-
•Minimize or avoid use of Tretinoin Gel Microsphere with weather extremes.
-
•Wash the treated skin gently, using a mild, non-medicated soap, pat it dry, and avoid washing the treated skin too often or scrubbing it hard when washing. Tretinoin Gel Microsphere is not recommended with concomitant use of medicated or abrasive soaps and cleansers, products that have a strong drying effect, products with high concentrations of alcohol, astringents, spices, or lime peels.
-
•Apply a topical moisturizer.
Advise patients that concomitant use of topical over the counter (OTC) acne products containing benzoyl peroxide, sulfur, resorcinol, or salicylic acid with Tretinoin Gel Microsphere may increase the risk for local skin irritation including dryness, erythema, and peeling. Consider withholding the use of topical OTC acne products if signs of skin irritation develop. Advise patients to allow the skin irritation effects of the topical OTC acne products to subside before initiation of Tretinoin Gel Microsphere treatment.
If severe local skin irritation occurs, discontinue Tretinoin Gel Microsphere use temporarily or permanently.
Efficacy of Tretinoin Gel Microsphere at reduced frequencies of application has not been established.
5 Warnings and Precautions
-
•Local Skin Irritation: Tretinoin Gel Microsphere can cause local skin irritation, including excessive dryness, redness, swelling, peeling, itching, blistering, burning, or stinging. (5.1)
-
oAvoid use on eczematous skin or during weather extremes, such as severe wind or cold.
-
oTo reduce the risk of local skin irritation, wash the treated skin gently, using a mild, non-medicated soap, avoid washing the treated skin too often or scrubbing it hard when washing, and apply a topical moisturizer.
-
oIf severe local skin irritation occurs, discontinue use temporarily or permanently.
-
•Initial Worsening of Inflammatory Acne Vulgaris: During the early weeks of Tretinoin Gel Microsphere treatment, an apparent exacerbation of inflammatory lesions may occur. If Tretinoin Gel Microsphere is tolerated, this should not be considered a reason to discontinue therapy. (5.2)
-
•Photosensitivity: Tretinoin Gel Microsphere can cause photosensitivity. Advise patients to avoid or minimize unnecessary exposure to UV light, including sunlight and sunlamps. Advise patients to use sunscreen (SPF ≥15) and sun-protective clothing if UV light exposure cannot be avoided. Avoid use on sunburn skin. (5.3)
2 Dosage and Administration
For topical use only. Not for oral, ophthalmic, or intravaginal use.
-
•Prior to Tretinoin Gel Microsphere use, thoroughly cleanse area(s) with a mild, non-medicated cleanser then pat the skin dry.
-
•When applying Tretinoin Gel Microsphere, keep away from the eyes, the mouth, paranasal creases of the nose, and mucous membranes.
-
•Apply a thin layer of Tretinoin Gel Microsphere (0.04%, 0.08%, or 0.1%) to skin where acne lesions appear (cover the entire affected area), once daily in the evening. Do not apply more than a thin layer [see Warning and Precautions (5.1)].
Improvements in acne lesions may be noticed after two weeks of Tretinoin Gel Microsphere therapy, but more than seven weeks of therapy may be needed for sustained benefit.
If Tretinoin Gel Microsphere was temporarily discontinued due to local adverse reactions, Tretinoin Gel Microsphere therapy may be resumed upon resolution of local adverse reactions.
Package/label Display Panel
NDC 68682-513-85
Rx only
TRETINOIN GEL
(MICROSPHERE)
0.1%
PUMP
For Topical Use Only
Net Wt. 50 g
OCEANSIDE
PHARMACEUTICALS
9515706
20001068G
3 Dosage Forms and Strengths
Gel (white to very pale yellow and opaque):
-
•0.04% (0.4 mg of tretinoin per gram)
-
•0.08% (0.8 mg of tretinoin per gram)
-
•0.1% (1 mg of tretinoin per gram).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Tretinoin Gel Microsphere and other topical tretinoin products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
-
•Hyper- or hypopigmentation has been reported with repeated application of tretinoin.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Tretinoin Gel Microsphere, 0.1%
The safety of Tretinoin Gel Microsphere, 0.1% for the treatment of acne vulgaris was evaluated in two multicenter, double-blind, randomized, vehicle-controlled clinical trials (Studies 1 and 2). A total of 347 subjects with acne vulgaris were treated in Studies 1 and 2 in which 172 subjects received Tretinoin Gel Microsphere, 0.1% and 175 subjects received vehicle, applied topically once daily in the evening, for 12 weeks. Mean age was 19 years (range 11-40) and 55% were female [see Clinical Studies (14.1)]. Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)].
In Studies 1 and 2, subjects treated with Tretinoin Gel Microsphere, 0.1% had increased cutaneous irritation scores for erythema, peeling, dryness, burning/stinging, or itching that peaked during the initial two weeks of therapy and decreased thereafter, compared to those treated with vehicle [see Warnings and Precautions (5.1)]. During the 12-week treatment period, no more than 3% of Tretinoin Gel Microsphere, 0.1%-treated subjects had cutaneous irritation scores indicative of severe cutaneous irritation and 6% (14/224) of Tretinoin Gel Microsphere 0.1%-treated subjects discontinued treatment due to cutaneous irritation. Of these 14 subjects, four had severe cutaneous irritation after 3 to 5 days of treatment, with blistering in one subject.
Tretinoin Gel Microsphere, 0.04%
The safety of Tretinoin Gel Microsphere, 0.04% for the treatment of acne vulgaris was evaluated in two multicenter, double-blind, randomized, vehicle-controlled clinical trials (Studies 3 and 4). A total of 451 subjects with acne vulgaris were treated in Studies 3 and 4 in which 225 subjects received Tretinoin Gel Microsphere, 0.04% and 226 subjects received vehicle, applied once daily in the evening, for 12 weeks. Mean age was 19 years (range 11-49) and 57% were female [see Clinical Studies (14.2)]. Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)].
In Studies 3 and 4, subjects treated with Tretinoin Gel Microsphere, 0.04% had increased cutaneous irritation scores for erythema, peeling, dryness, burning/stinging, or itching that peaked during the initial two weeks of therapy and decreased thereafter, compared to those treated with vehicle [see Warnings and Precautions (5.1)]. Approximately half of the 225 subjects in the Tretinoin Gel Microsphere, 0.04%-treated group had cutaneous irritation at Week 2. Of the subjects who experienced cutaneous irritation, most had signs or symptoms that were mild in severity (severity was ranked on a 4-point ordinal scale: 0=none, 1=mild, 2=moderate, and 3=severe). Less than 10% of Tretinoin Gel Microsphere, 0.04%-treated subjects experienced moderate cutaneous irritation, and none had severe cutaneous irritation at Week 2.
In Studies 3 and 4, during the 12-week treatment period, the majority of Tretinoin Gel Microsphere, 0.04%-treated subjects experienced cutaneous irritation (mild, moderate, or severe), of which, 1% (2/225) of subjects had cutaneous irritation scores indicative of a severe irritation and 1.3% (3/225) of subjects discontinued treatment due to cutaneous irritation, which included dryness in one subject and peeling and urticaria in another.
Tretinoin Gel Microsphere, 0.04% and 0.1%
In a double-blind trial, 156 subjects with acne vulgaris were treated for 12-weeks with Tretinoin Gel Microsphere 0.04% (n=78) or 0.1% (n=78) topically once daily. In this trial, the most frequently reported adverse events affected the skin and subcutaneous tissue (15% in the 0.04% group, and 21% in the 0.1% group). The most prevalent events in the 0.04% group were skin irritation (6%); and in the 0.1% group, skin burning (8%), erythema (5%), skin irritation (4%), and dermatitis (4%). In this trial, 63% of the adverse events were of mild intensity, and 34% were of moderate intensity.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Administration
Instruct the patient to [see Dosage and Administration (2)]:
-
•Thoroughly cleanse treatment area(s) with a mild, non-medicated cleanser then pat the skin dry prior to application of Tretinoin Gel Microsphere.
-
•Avoid contact with eyes, mouth, paranal creases of nose, and mucous membranes when applying Tretinoin Gel Microsphere.
-
•Apply a thin layer of Tretinoin Gel Microsphere to cover the affected area(s).
Local Skin Irritation:
Advise the patient that use of Tretinoin Gel Microsphere greater than the recommended dosage will not result in more rapid of improved acne results and may result in marked redness, peeling or discomfort [see Warnings and Precautions (5.1)].
To reduce the risk of local skin irritation, advise the patient to:
-
•Minimize or avoid use of Tretinoin Gel Microsphere during weather extremes, such as severe wind or cold.
-
•Wash the treated skin gently, using a mild, non-medicated soap, and pat it dry, and avoid washing the treated skin too often or scrubbing it hard when washing.
-
•Not use medicated or abrasive soaps and cleansers, products that have a strong drying effect, products with high concentrations of alcohol, astringents, spices, and lime peels while using Tretinoin Gel Microsphere.
-
•Apply a topical moisturizer when using Tretinoin Gel Microsphere.
Advise the patient that concomitant use of topical OTC acne products containing benzoyl peroxide, sulfur, resorcinol, or salicylic acid with Tretinoin Gel Microsphere may increase the risk of local skin irritation. Advise the patient to withhold the use of topical OTC acne products if signs of skin irritation develop. Advise the patient to allow the skin irritation effects of the topical OTC acne products to subside before initiation of Tretinoin Gel Microsphere [see Warning and Precautions (5.1)].
Photosensitivity
Advise the patient to avoid or minimize unnecessary exposure to ultraviolet (UV) light, including sunlight and sunlamps, while using Tretinoin Gel Microsphere.
Advise the patient to not use Tretinoin Gel Microsphere on sunburn skin.
Advise the patient to use sun protection in the form of sunscreen (SPF ≥15) and sun-protective clothing if UV exposure cannot be avoided [see Warnings and Precautions (5.3)].
Lactation
Advise the female patient to use Tretinoin Gel Microsphere on the smallest area of skin with acne vulgaris and for the shortest duration possible while breastfeeding. To avoid direct infant exposure, instruct the patient who is breastfeeding not to apply Tretinoin Gel Microsphere directly to the nipple and areola [see Use in Specific Populations (8.2)].
Distributed by:
Oceanside Pharmaceuticals, a division of
Bausch Health US, LLC
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch Health Companies Inc.
Laval, Quebec H7L 4A8, Canada
MICROSPONGE is a registered trademark of AMCOL International Corporation.
All other products/brand names and/or logos are trademarks of the respective owners.
© 2025 Bausch Health Companies Inc. or its affiliates
9515306
16 How Supplied/storage and Handling
How Supplied
Tretinoin Gel Microsphere (tretinoin) is opaque and white to very pale yellow in color and is supplied as follows:
|
Strength |
Amount of Tretinoin |
Quantity/Package Type |
NDC |
|
0.04% |
0.4 mg |
20 gram tube |
68682-514-92 |
|
45 gram tube |
68682-514-94 |
||
|
50 gram pump |
68682-514-95 |
||
|
0.08% |
0.8 mg |
50 gram pump |
68682-515-95 |
|
0.1% |
1 mg |
20 gram tube |
68682-513-82 |
|
45 gram tube |
68682-513-84 |
||
|
50 gram pump |
68682-513-85 |
Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Store pump upright.
5.2 Initial Worsening of Inflammatory Acne Vulgaris
During the early weeks of Tretinoin Gel Microsphere treatment, an apparent exacerbation of inflammatory acne vulgaris lesions may occur. If Tretinoin Gel Microsphere is tolerated, initial worsening of inflammatory acne vulgaris lesions should not be considered a reason to discontinue therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Dermal carcinogenicity testing has not been performed with Tretinoin Gel Microsphere, 0.04%, 0.08%, or 0.1%.
In a 91-week dermal study in which CD-1 mice were administered 0.017% and 0.035% formulations of tretinoin, cutaneous squamous cell carcinomas and papillomas in the treatment area were observed in some female mice. These concentrations are near the tretinoin concentration of the 0.04% and 0.1% clinical formulations. A dose-dependent incidence of liver tumors in male mice was observed at those same doses. The maximum systemic doses associated with the administered 0.017% and 0.035% formulations are 0.5 and 1 mg/kg/day tretinoin, respectively. These doses are two and four times the MRHD based on BSA comparison.
The biological significance of these findings is not clear because they occurred at doses that exceeded the dermal maximally tolerated dose of tretinoin and because they were within the background natural occurrence rate for these tumors in this strain of mice. There was no evidence of carcinogenic potential when 0.025 mg/kg/day of tretinoin was administered topically to mice (0.1 times the MRHD based on BSA comparison). Studies in hairless albino mice suggest that concurrent exposure to tretinoin may enhance the tumorigenic potential of carcinogenic doses of UVB and UVA light from a solar simulator. This effect has been confirmed in a later study in pigmented mice, and dark pigmentation did not overcome the enhancement of photocarcinogenesis by 0.05% tretinoin. Although the significance of these studies to humans is not clear, patients should minimize exposure to sunlight or artificial ultraviolet irradiation sources [see Warnings and Precautions (5.3)].
The genotoxic potential of tretinoin was evaluated in the Ames assay and an in vivo mouse micronucleus assay. Both tests were negative.
The components of the microspheres have shown potential for genetic toxicity and fetal malformation. EGDMA, a component of the excipient acrylates copolymer, was positive for induction of structural chromosomal aberrations in an in vitro chromosomal aberration assay in mammalian cells in the absence of metabolic activation, and negative for genetic toxicity in the Ames assay, and an in vivo mouse micronucleus assay.
In fertility studies in rats with oral tretinoin, the no-observable effect level was 2 mg/kg/day (19 times the MRHD based on BSA comparison).
14.1 Clinical Studies With Tretinoin Gel Microsphere, 0.1%
The efficacy of Tretinoin Gel Microsphere, 0.1 was evaluated in two double-blind, randomized, vehicle-controlled trials in adult and pediatric subjects 11 years of age and older with acne vulgaris (Studies 1 and 2). Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)]. Tretinoin Gel Microsphere, 0.1%, applied topically once daily in the evening, was superior to vehicle gel, applied topically once daily, in reducing the acne lesion counts. The mean reduction in lesion counts from baseline after treatment for 12 weeks are shown in Table 1:
|
Study 1 |
Study 2 |
|||
|
Tretinoin Gel Microsphere, 0.1% |
Vehicle Gel (n=71) |
Tretinoin Gel Microsphere, 0.1% (n=72) |
Vehicle Gel |
|
|
Non-inflammatory lesion counts |
49% |
22% |
32% |
3% |
|
Inflammatory lesion counts |
37% |
18% |
29% |
24% |
|
Total lesion counts |
45% |
23% |
32% |
16% |
Efficacy was also assessed by the investigator’s global evaluation, which included categories of “excellent”, “good”, “fair”, “no change”, and “poor”. Tretinoin Gel Microsphere, 0.1%, was significantly superior (p<0.001) to the vehicle in the investigator’s global evaluation of the clinical response:
-
•In Study #1, 35% of Tretinoin Gel Microsphere, 0.1% -treated subjects achieved an “excellent” result, as compared to 11% of vehicle-treated subjects.
-
•In Study #2, 28% of Tretinoin Gel Microsphere, 0.1%-treated subjects achieved an “excellent” result, as compared to 9% of vehicle-treated subjects.
14.2 Clinical Studies With Tretinoin Gel Microsphere, 0.04%
-
In two vehicle-controlled clinical trials in adult and pediatric subjects 11 years of age and older with acne vulgaris (Studies 3 and 4). Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)]. Tretinoin Gel Microsphere, 0.04%, applied topically once daily in the evening, was more effective (p<0.05) than vehicle gel, applied topically once daily, in reducing the acne lesion counts. The mean reduction in lesion counts from baseline after treatment for 12 weeks are shown in Table 2:
| Study 3 | Study 4 | |||
|---|---|---|---|---|
|
Tretinoin Gel Microsphere, 0.04% (n=108) |
Vehicle Gel (n=111) |
Tretinoin Gel Microsphere, 0.04% (n=110) |
Vehicle Gel
(n=103) |
|
|
Non-inflammatory |
37% |
-2% -2% represents a mean percent increase of 2%
|
29% |
14% |
|
Inflammatory lesion |
44% |
13% |
41% |
30% |
|
Total lesion counts |
40% |
8% |
35% |
20% |
Efficacy was also assessed by the investigator’s global evaluation, which included categories of “excellent”, “good”, “fair”, “no change”, and “poor”. Tretinoin Gel Microsphere, 0.04% was superior (p<0.05) to the vehicle in the investigator's global evaluation of the clinical response:
-
•In Study #3, 14% of Tretinoin Gel Microsphere, 0.04%-treated subjects achieved an “excellent” result, as compared to 5% of vehicle-treated subjects.
-
•In Study #4, 19% of Tretinoin Gel Microsphere, 0.04%-treated subjects achieved an “excellent” result, as compared to 9% of vehicle-treated subjects.
Structured Label Content
Section 42230-3 (42230-3)
|
Patient Information Tretinoin Gel Microsphere, 0.1%, 0.08% and 0.04% for topical use |
|
Important information: Tretinoin Gel Microsphere is for use on skin (topical use) only. Keep Tretinoin Gel Microsphere away from your eyes, mouth, vagina, or the corners of your nose. |
|
What is Tretinoin Gel Microsphere? Tretinoin Gel Microsphere is a prescription medicine used on the skin (topical) to treat acne in adults and children 12 years of age and older. Acne is a condition in which the skin has blackheads, whiteheads, and other pimples. It is not known if Tretinoin Gel Microsphere is safe and effective in children under 12 years of age. |
|
Before using Tretinoin Gel Microsphere, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you use any skin products that contain benzoyl peroxide, sulfur, resorcinol, or salicylic acid. Using these topical skin products may increase the irritation of your skin when used with Tretinoin Gel Microsphere. |
|
How should I use Tretinoin Gel Microsphere?
What should I avoid while using Tretinoin Gel Microsphere?
|
|
What are the possible side effects of Tretinoin Gel Microsphere? Tretinoin Gel Microsphere can cause serious side effects, including:
The most common side effects of Tretinoin Gel Microsphere include skin redness, pain, and itching. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Tretinoin Gel Microsphere. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store Tretinoin Gel Microsphere?
Keep Tretinoin Gel Microsphere and all medicines out of the reach of children. |
|
General information about the safe and effective use of Tretinoin Gel Microsphere. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Tretinoin Gel Microsphere for a condition for which it was not prescribed. Do not give Tretinoin Gel Microsphere to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Tretinoin Gel Microsphere that is written for health professionals. |
|
What are the ingredients of Tretinoin Gel Microsphere? Active ingredient: tretinoin Inactive ingredients: benzyl alcohol, butylated hydroxytoluene, carbomer 974P, cyclomethicone and dimethicone copolyol, disodium EDTA, glycerin, methyl methacrylate/glycol dimethacrylate crosspolymer, PPG-20 methyl glucose ether distearate, propylene glycol, propylene glycol dicaprylate/dicaprate, purified water, sorbic acid, and trolamine |
|
Distributed by: Oceanside Pharmaceuticals, a division of Bausch Health US, LLC, Bridgewater, NJ 08807 USA Manufactured by: Bausch Health Companies Inc., Laval, Quebec H7L 4A8, Canada Microsponge is a registered trademark of AMCOL International Corporation. All other products/brand names and/or logos are trademarks of the respective owners. © 2025 Bausch Health Companies Inc. or its affiliates For more information, call 1-800-321-4576. |
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 09/2025
9515306
10 Overdosage (10 OVERDOSAGE)
Oral ingestion of large amounts of Tretinoin Gel Microsphere may lead to the same adverse reactions as those associated with excessive oral intake of Vitamin A.
Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for overdose recommendations.
8.1 Pregnancy
Risk Summary
Available data from published prospective observational studies and retrospective cohort studies over decades of use of topical tretinoin in pregnant women have not established a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes (see Data).
In animal reproduction studies with pregnant rats, alterations in the vertebrae and ribs of offspring were observed with daily topical dosing of 0.1% Tretinoin Gel (microsponge) during organogenesis at 5 to 10 times the maximum recommended human dose (MRHD). In animal reproduction studies with pregnant rabbits, fetal malformations, such as domed head and hydrocephaly, were observed in the offspring with daily topical dosing of 0.1% Tretinoin Gel (microsponge) during organogenesis at 10 to 19 times the MRHD [see Data].
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4%, and 15% to 20%, respectively.
Data
Human Data: Published data from prospective observational studies and retrospective cohort studies on the use of topical tretinoin products during pregnancy have not identified an association with topical tretinoin and major birth defects or miscarriage. The available studies have methodologic limitations, including potential misclassification of exposure, small sample size and in some cases, lack of physical exam by an expert in birth defects.
Animal Data: For purposes of comparison of the animal exposure to systemic human exposure, the MRHD applied topically is defined as 1 gram of Tretinoin Gel Microsphere, 0.1%, applied daily to a 60 kg person (0.017 mg tretinoin/kg body weight).
Pregnant rats were treated with 0.1% Tretinoin Gel (microsponge) at daily dermal doses of 0.2, 0.5, and 1 mg/kg/day tretinoin on gestation days 6-15. Alterations were seen in the vertebrae and ribs of the affected offsprings at 0.5 mg/kg/day tretinoin, 5 to 10 times the MRHD based on body surface area (BSA) comparison.
Pregnant rabbits were treated with 0.1% Tretinoin Gel (microsponge) at daily dermal doses of 0.2, 0.5, and 1 mg/kg/day tretinoin on gestation days 7-19. Doses were administered topically for 24 hours a day while wearing Elizabethan collars to prevent ingestion of the drug. Increased incidences of certain alterations, including domed head and hydrocephaly, typical of retinoid-induced fetal malformations in this species were observed at doses of 0.5 and 1 mg/kg/day. Similar malformations were not observed in the offspring at 0.2 mg/kg/day, 4 times the MRHD based on BSA comparison. In a second rabbit study, pregnant rabbits were treated with 0.1% Tretinoin Gel (microsponge) at daily dermal doses of 0.5 or 1 mg/kg/day tretinoin on gestation days 7-19. Doses were administered topically for six hours per day while pregnant rabbits were restrained in stocks to prevent ingestion. The offspring of pregnant rabbits exposed to 0.5 or 1 mg/kg/day tretinoin did not show any malformations at doses up to 19 times (1.0 mg/kg/day) the MRHD based on BSA comparison, but fetal resorptions were increased at 0.5 mg/kg (10 times the MRHD based on BSA comparison).
Malformations (shortened or kinked tail) were observed in the offspring of pregnant rats treated with topical tretinoin at doses greater than 1 mg/kg/day during the period of organogenesis (10 times the MRHD based on BSA comparison). Anomalies (humerus: short 13%, bent 6%, os parietal incompletely ossified 14%) have also been reported in offspring when 10 mg/kg/day was topically applied to pregnant rats during the period of organogenesis. Supernumerary ribs have been a consistent finding in newborn rats when dams were treated topically or orally with retinoids.
Oral administration of tretinoin during organogenesis has been shown to cause malformations in the offspring of rats, mice, rabbits, hamsters, and nonhuman primates. Fetal malformations and death were observed when tretinoin was orally administered to pregnant rats during organogenesis at doses greater than 1 mg/kg/day (10 times the MRHD based on BSA comparison). Fetal malformations were reported at doses of 10 mg/kg/day or greater when administered to pregnant cynomolgus monkeys, but none were observed at 5 mg/kg/day (95 times the MRHD based on BSA comparison), although increased skeletal variations were observed at all doses. Dose-dependent increases in embryolethality and abortion also were reported. Similar results have also been reported in pigtail macaques.
In peri- and postnatal development studies in rats with oral tretinoin, decreased survival of neonates and growth retardation were observed at doses in excess of 2 mg/kg/day (19 times the MRHD based on BSA comparison).
Oral tretinoin has been shown to be fetotoxic in rats when administered at doses 24 times the MRHD based on BSA comparison.
Topical tretinoin has been shown to be fetotoxic in rabbits when administered at doses 10 times the MRHD based on BSA comparison.
8.2 Lactation
Risk Summary
There are no data on the presence of tretinoin or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. It is not known whether topical administration of tretinoin could result in sufficient systemic absorption to produce detectable quantities in human milk (see Clinical Considerations).
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Tretinoin Gel Microsphere and any potential adverse effects on the breastfed infant from the Tretinoin Gel Microsphere or from the underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the infant via human milk, during breastfeeding, use Tretinoin Gel Microsphere on the smallest area of skin with acne and for the shortest duration possible. Advise breastfeeding women not to apply Tretinoin Gel Microsphere directly to the nipple and areola to avoid direct infant exposure.
11 Description (11 DESCRIPTION)
Chemically, tretinoin is all-trans-retinoic acid, also known as (all-E)-3,7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid. Tretinoin is a retinoid and a metabolite of naturally occurring Vitamin A. Tretinoin has a molecular weight of 300.44, a molecular formula of C20H28O2 and the following chemical structure:
Tretinoin Gel Microsphere (tretinoin) gel is for topical use. Each gram of Tretinoin Gel Microsphere gel:
-
•0.04%, contains 0.4 mg of tretinoin
-
•0.08%, contains 0.8 mg of tretinoin
-
•0.1%, contains 1 mg of tretinoin
The formulation contains methyl methacrylate/glycol dimethacrylate crosspolymer (MICROSPONGE® System), propylene glycol dicaprylate/dicaprate and butylated hydroxytoluene. Other components consist of benzyl alcohol, butylated hydroxytoluene, carbomer 974P, cyclomethicone and dimethicone copolyol, disodium EDTA, glycerin, PPG-20 methyl glucose ether distearate, propylene glycol, purified water, sorbic acid, and trolamine.
8.4 Pediatric Use
The safety and effectiveness of Tretinoin Gel Microsphere for topical application for the treatment of acne vulgaris have been established in pediatric patients aged 12 years and older [see Clinical Studies (14)]. The safety and effectiveness of Tretinoin Gel Microsphere have not been established in pediatric patients younger than 12 years of age.
8.5 Geriatric Use
Clinical trials of Tretinoin Gel Microsphere did not include sufficient numbers of subjects aged 65 and over to determine whether they responded differently from younger adult subjects. Acne vulgaris is largely a disease of pediatric and young adult patients. Clinical studies of Tretinoin Gel Microsphere did not include patients 65 years of age and older.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions were skin irritation, skin burning, erythema, peeling, dryness, itching, and dermatitis. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
5.3 Photosensitivity
Tretinoin Gel Microsphere can cause photosensitivity. Advise patients to avoid or minimize unnecessary exposure to ultraviolet (UV) light, including sunlight and sunlamps, while using Tretinoin Gel Microsphere. Advise patients with sunburn to not use Tretinoin Gel Microsphere until the sunburn fully recovers.
Advise patients, especially those who may be required to have extended periods of UV light exposure (e.g., due to occupation or sports), those with inherent sensitivity to the sun, or those using drugs that cause photosensitivity, to use sun protection daily in the form of sunscreen (sun protection factor [SPF] ≥ 15) and sun-protective clothing, when UV exposure cannot be avoided, even on days when it is not sunny or inside activities are expected.
12.3 Pharmacokinetics
Absorption
Tretinoin is a metabolite of Vitamin A. Percutaneous absorption, as determined by the cumulative excretion of radiolabeled drug into urine and feces, was assessed in 44 healthy males and females after single and repeated daily tretinoin applications up to 28 days of 500 mg of a 0.1% tretinoin gel product. In this assessment, estimates of in vivo bioavailability, mean (SD) were 0.82 (0.11)% and 1.41 (0.54)%, for single and multiple daily topical applications, respectively. The plasma concentrations of tretinoin and its metabolites, 13-cis-retinoic acid, all-trans-4-oxo-retinoic acid, and 13-cis-4-oxo-retinoic acid, generally ranged from 1 to 3 ng/mL and were essentially unaltered after either single or multiple daily applications of Tretinoin Gel Microsphere, 0.1%, relative to baseline levels.
Clinical pharmacokinetic studies have not been performed with Tretinoin Gel Microsphere, 0.08%, and 0.04%.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tretinoin Gel Microsphere is indicated for the topical treatment of acne vulgaris in adults and pediatric patients 12 years of age and older.
12.1 Mechanism of Action
Although tretinoin activates three members of the retinoic acid (RAR) nuclear receptors (RARα, RARβ, and RARγ) which may act to modify gene expression, subsequent protein synthesis, and epithelial cell growth and differentiation, it has not been established whether the clinical effects of tretinoin are mediated through activation of retinoic acid receptors and/or other mechanisms.
The exact mechanism of action of topical tretinoin for treatment of acne vulgaris is unknown. Current evidence suggests that topical tretinoin decreases cohesiveness of follicular epithelial cells with decreased microcomedone formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.
5.1 Local Skin Irritation
Tretinoin Gel Microsphere can cause local skin irritation, including excessive dryness, redness, swelling, peeling, itching, blistering, burning, or stinging [see Adverse Reactions (6.1)]. Use of Tretinoin Gel Microsphere in greater than the recommended dosage (more frequent than once daily application or excessive application) will not result in more rapid or improved acne results and may result in marked redness, peeling, or discomfort.
Tretinoin has been reported to cause severe local skin irritation on eczematous skin.
Weather extremes, such as severe wind or cold, may increase the risk of skin irritation in patients using Tretinoin Gel Microsphere.
To reduce the risk of local skin irritation, instruct Tretinoin Gel Microsphere-treated patients to:
-
•Avoid use of Tretinoin Gel Microsphere in areas affected by eczema.
-
•Minimize or avoid use of Tretinoin Gel Microsphere with weather extremes.
-
•Wash the treated skin gently, using a mild, non-medicated soap, pat it dry, and avoid washing the treated skin too often or scrubbing it hard when washing. Tretinoin Gel Microsphere is not recommended with concomitant use of medicated or abrasive soaps and cleansers, products that have a strong drying effect, products with high concentrations of alcohol, astringents, spices, or lime peels.
-
•Apply a topical moisturizer.
Advise patients that concomitant use of topical over the counter (OTC) acne products containing benzoyl peroxide, sulfur, resorcinol, or salicylic acid with Tretinoin Gel Microsphere may increase the risk for local skin irritation including dryness, erythema, and peeling. Consider withholding the use of topical OTC acne products if signs of skin irritation develop. Advise patients to allow the skin irritation effects of the topical OTC acne products to subside before initiation of Tretinoin Gel Microsphere treatment.
If severe local skin irritation occurs, discontinue Tretinoin Gel Microsphere use temporarily or permanently.
Efficacy of Tretinoin Gel Microsphere at reduced frequencies of application has not been established.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Local Skin Irritation: Tretinoin Gel Microsphere can cause local skin irritation, including excessive dryness, redness, swelling, peeling, itching, blistering, burning, or stinging. (5.1)
-
oAvoid use on eczematous skin or during weather extremes, such as severe wind or cold.
-
oTo reduce the risk of local skin irritation, wash the treated skin gently, using a mild, non-medicated soap, avoid washing the treated skin too often or scrubbing it hard when washing, and apply a topical moisturizer.
-
oIf severe local skin irritation occurs, discontinue use temporarily or permanently.
-
•Initial Worsening of Inflammatory Acne Vulgaris: During the early weeks of Tretinoin Gel Microsphere treatment, an apparent exacerbation of inflammatory lesions may occur. If Tretinoin Gel Microsphere is tolerated, this should not be considered a reason to discontinue therapy. (5.2)
-
•Photosensitivity: Tretinoin Gel Microsphere can cause photosensitivity. Advise patients to avoid or minimize unnecessary exposure to UV light, including sunlight and sunlamps. Advise patients to use sunscreen (SPF ≥15) and sun-protective clothing if UV light exposure cannot be avoided. Avoid use on sunburn skin. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For topical use only. Not for oral, ophthalmic, or intravaginal use.
-
•Prior to Tretinoin Gel Microsphere use, thoroughly cleanse area(s) with a mild, non-medicated cleanser then pat the skin dry.
-
•When applying Tretinoin Gel Microsphere, keep away from the eyes, the mouth, paranasal creases of the nose, and mucous membranes.
-
•Apply a thin layer of Tretinoin Gel Microsphere (0.04%, 0.08%, or 0.1%) to skin where acne lesions appear (cover the entire affected area), once daily in the evening. Do not apply more than a thin layer [see Warning and Precautions (5.1)].
Improvements in acne lesions may be noticed after two weeks of Tretinoin Gel Microsphere therapy, but more than seven weeks of therapy may be needed for sustained benefit.
If Tretinoin Gel Microsphere was temporarily discontinued due to local adverse reactions, Tretinoin Gel Microsphere therapy may be resumed upon resolution of local adverse reactions.
Package/label Display Panel (Package/Label Display Panel)
NDC 68682-513-85
Rx only
TRETINOIN GEL
(MICROSPHERE)
0.1%
PUMP
For Topical Use Only
Net Wt. 50 g
OCEANSIDE
PHARMACEUTICALS
9515706
20001068G
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Gel (white to very pale yellow and opaque):
-
•0.04% (0.4 mg of tretinoin per gram)
-
•0.08% (0.8 mg of tretinoin per gram)
-
•0.1% (1 mg of tretinoin per gram).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Tretinoin Gel Microsphere and other topical tretinoin products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
-
•Hyper- or hypopigmentation has been reported with repeated application of tretinoin.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Tretinoin Gel Microsphere, 0.1%
The safety of Tretinoin Gel Microsphere, 0.1% for the treatment of acne vulgaris was evaluated in two multicenter, double-blind, randomized, vehicle-controlled clinical trials (Studies 1 and 2). A total of 347 subjects with acne vulgaris were treated in Studies 1 and 2 in which 172 subjects received Tretinoin Gel Microsphere, 0.1% and 175 subjects received vehicle, applied topically once daily in the evening, for 12 weeks. Mean age was 19 years (range 11-40) and 55% were female [see Clinical Studies (14.1)]. Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)].
In Studies 1 and 2, subjects treated with Tretinoin Gel Microsphere, 0.1% had increased cutaneous irritation scores for erythema, peeling, dryness, burning/stinging, or itching that peaked during the initial two weeks of therapy and decreased thereafter, compared to those treated with vehicle [see Warnings and Precautions (5.1)]. During the 12-week treatment period, no more than 3% of Tretinoin Gel Microsphere, 0.1%-treated subjects had cutaneous irritation scores indicative of severe cutaneous irritation and 6% (14/224) of Tretinoin Gel Microsphere 0.1%-treated subjects discontinued treatment due to cutaneous irritation. Of these 14 subjects, four had severe cutaneous irritation after 3 to 5 days of treatment, with blistering in one subject.
Tretinoin Gel Microsphere, 0.04%
The safety of Tretinoin Gel Microsphere, 0.04% for the treatment of acne vulgaris was evaluated in two multicenter, double-blind, randomized, vehicle-controlled clinical trials (Studies 3 and 4). A total of 451 subjects with acne vulgaris were treated in Studies 3 and 4 in which 225 subjects received Tretinoin Gel Microsphere, 0.04% and 226 subjects received vehicle, applied once daily in the evening, for 12 weeks. Mean age was 19 years (range 11-49) and 57% were female [see Clinical Studies (14.2)]. Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)].
In Studies 3 and 4, subjects treated with Tretinoin Gel Microsphere, 0.04% had increased cutaneous irritation scores for erythema, peeling, dryness, burning/stinging, or itching that peaked during the initial two weeks of therapy and decreased thereafter, compared to those treated with vehicle [see Warnings and Precautions (5.1)]. Approximately half of the 225 subjects in the Tretinoin Gel Microsphere, 0.04%-treated group had cutaneous irritation at Week 2. Of the subjects who experienced cutaneous irritation, most had signs or symptoms that were mild in severity (severity was ranked on a 4-point ordinal scale: 0=none, 1=mild, 2=moderate, and 3=severe). Less than 10% of Tretinoin Gel Microsphere, 0.04%-treated subjects experienced moderate cutaneous irritation, and none had severe cutaneous irritation at Week 2.
In Studies 3 and 4, during the 12-week treatment period, the majority of Tretinoin Gel Microsphere, 0.04%-treated subjects experienced cutaneous irritation (mild, moderate, or severe), of which, 1% (2/225) of subjects had cutaneous irritation scores indicative of a severe irritation and 1.3% (3/225) of subjects discontinued treatment due to cutaneous irritation, which included dryness in one subject and peeling and urticaria in another.
Tretinoin Gel Microsphere, 0.04% and 0.1%
In a double-blind trial, 156 subjects with acne vulgaris were treated for 12-weeks with Tretinoin Gel Microsphere 0.04% (n=78) or 0.1% (n=78) topically once daily. In this trial, the most frequently reported adverse events affected the skin and subcutaneous tissue (15% in the 0.04% group, and 21% in the 0.1% group). The most prevalent events in the 0.04% group were skin irritation (6%); and in the 0.1% group, skin burning (8%), erythema (5%), skin irritation (4%), and dermatitis (4%). In this trial, 63% of the adverse events were of mild intensity, and 34% were of moderate intensity.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Administration
Instruct the patient to [see Dosage and Administration (2)]:
-
•Thoroughly cleanse treatment area(s) with a mild, non-medicated cleanser then pat the skin dry prior to application of Tretinoin Gel Microsphere.
-
•Avoid contact with eyes, mouth, paranal creases of nose, and mucous membranes when applying Tretinoin Gel Microsphere.
-
•Apply a thin layer of Tretinoin Gel Microsphere to cover the affected area(s).
Local Skin Irritation:
Advise the patient that use of Tretinoin Gel Microsphere greater than the recommended dosage will not result in more rapid of improved acne results and may result in marked redness, peeling or discomfort [see Warnings and Precautions (5.1)].
To reduce the risk of local skin irritation, advise the patient to:
-
•Minimize or avoid use of Tretinoin Gel Microsphere during weather extremes, such as severe wind or cold.
-
•Wash the treated skin gently, using a mild, non-medicated soap, and pat it dry, and avoid washing the treated skin too often or scrubbing it hard when washing.
-
•Not use medicated or abrasive soaps and cleansers, products that have a strong drying effect, products with high concentrations of alcohol, astringents, spices, and lime peels while using Tretinoin Gel Microsphere.
-
•Apply a topical moisturizer when using Tretinoin Gel Microsphere.
Advise the patient that concomitant use of topical OTC acne products containing benzoyl peroxide, sulfur, resorcinol, or salicylic acid with Tretinoin Gel Microsphere may increase the risk of local skin irritation. Advise the patient to withhold the use of topical OTC acne products if signs of skin irritation develop. Advise the patient to allow the skin irritation effects of the topical OTC acne products to subside before initiation of Tretinoin Gel Microsphere [see Warning and Precautions (5.1)].
Photosensitivity
Advise the patient to avoid or minimize unnecessary exposure to ultraviolet (UV) light, including sunlight and sunlamps, while using Tretinoin Gel Microsphere.
Advise the patient to not use Tretinoin Gel Microsphere on sunburn skin.
Advise the patient to use sun protection in the form of sunscreen (SPF ≥15) and sun-protective clothing if UV exposure cannot be avoided [see Warnings and Precautions (5.3)].
Lactation
Advise the female patient to use Tretinoin Gel Microsphere on the smallest area of skin with acne vulgaris and for the shortest duration possible while breastfeeding. To avoid direct infant exposure, instruct the patient who is breastfeeding not to apply Tretinoin Gel Microsphere directly to the nipple and areola [see Use in Specific Populations (8.2)].
Distributed by:
Oceanside Pharmaceuticals, a division of
Bausch Health US, LLC
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch Health Companies Inc.
Laval, Quebec H7L 4A8, Canada
MICROSPONGE is a registered trademark of AMCOL International Corporation.
All other products/brand names and/or logos are trademarks of the respective owners.
© 2025 Bausch Health Companies Inc. or its affiliates
9515306
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
Tretinoin Gel Microsphere (tretinoin) is opaque and white to very pale yellow in color and is supplied as follows:
|
Strength |
Amount of Tretinoin |
Quantity/Package Type |
NDC |
|
0.04% |
0.4 mg |
20 gram tube |
68682-514-92 |
|
45 gram tube |
68682-514-94 |
||
|
50 gram pump |
68682-514-95 |
||
|
0.08% |
0.8 mg |
50 gram pump |
68682-515-95 |
|
0.1% |
1 mg |
20 gram tube |
68682-513-82 |
|
45 gram tube |
68682-513-84 |
||
|
50 gram pump |
68682-513-85 |
Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Store pump upright.
5.2 Initial Worsening of Inflammatory Acne Vulgaris
During the early weeks of Tretinoin Gel Microsphere treatment, an apparent exacerbation of inflammatory acne vulgaris lesions may occur. If Tretinoin Gel Microsphere is tolerated, initial worsening of inflammatory acne vulgaris lesions should not be considered a reason to discontinue therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Dermal carcinogenicity testing has not been performed with Tretinoin Gel Microsphere, 0.04%, 0.08%, or 0.1%.
In a 91-week dermal study in which CD-1 mice were administered 0.017% and 0.035% formulations of tretinoin, cutaneous squamous cell carcinomas and papillomas in the treatment area were observed in some female mice. These concentrations are near the tretinoin concentration of the 0.04% and 0.1% clinical formulations. A dose-dependent incidence of liver tumors in male mice was observed at those same doses. The maximum systemic doses associated with the administered 0.017% and 0.035% formulations are 0.5 and 1 mg/kg/day tretinoin, respectively. These doses are two and four times the MRHD based on BSA comparison.
The biological significance of these findings is not clear because they occurred at doses that exceeded the dermal maximally tolerated dose of tretinoin and because they were within the background natural occurrence rate for these tumors in this strain of mice. There was no evidence of carcinogenic potential when 0.025 mg/kg/day of tretinoin was administered topically to mice (0.1 times the MRHD based on BSA comparison). Studies in hairless albino mice suggest that concurrent exposure to tretinoin may enhance the tumorigenic potential of carcinogenic doses of UVB and UVA light from a solar simulator. This effect has been confirmed in a later study in pigmented mice, and dark pigmentation did not overcome the enhancement of photocarcinogenesis by 0.05% tretinoin. Although the significance of these studies to humans is not clear, patients should minimize exposure to sunlight or artificial ultraviolet irradiation sources [see Warnings and Precautions (5.3)].
The genotoxic potential of tretinoin was evaluated in the Ames assay and an in vivo mouse micronucleus assay. Both tests were negative.
The components of the microspheres have shown potential for genetic toxicity and fetal malformation. EGDMA, a component of the excipient acrylates copolymer, was positive for induction of structural chromosomal aberrations in an in vitro chromosomal aberration assay in mammalian cells in the absence of metabolic activation, and negative for genetic toxicity in the Ames assay, and an in vivo mouse micronucleus assay.
In fertility studies in rats with oral tretinoin, the no-observable effect level was 2 mg/kg/day (19 times the MRHD based on BSA comparison).
14.1 Clinical Studies With Tretinoin Gel Microsphere, 0.1% (14.1 Clinical Studies with Tretinoin Gel Microsphere, 0.1%)
The efficacy of Tretinoin Gel Microsphere, 0.1 was evaluated in two double-blind, randomized, vehicle-controlled trials in adult and pediatric subjects 11 years of age and older with acne vulgaris (Studies 1 and 2). Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)]. Tretinoin Gel Microsphere, 0.1%, applied topically once daily in the evening, was superior to vehicle gel, applied topically once daily, in reducing the acne lesion counts. The mean reduction in lesion counts from baseline after treatment for 12 weeks are shown in Table 1:
|
Study 1 |
Study 2 |
|||
|
Tretinoin Gel Microsphere, 0.1% |
Vehicle Gel (n=71) |
Tretinoin Gel Microsphere, 0.1% (n=72) |
Vehicle Gel |
|
|
Non-inflammatory lesion counts |
49% |
22% |
32% |
3% |
|
Inflammatory lesion counts |
37% |
18% |
29% |
24% |
|
Total lesion counts |
45% |
23% |
32% |
16% |
Efficacy was also assessed by the investigator’s global evaluation, which included categories of “excellent”, “good”, “fair”, “no change”, and “poor”. Tretinoin Gel Microsphere, 0.1%, was significantly superior (p<0.001) to the vehicle in the investigator’s global evaluation of the clinical response:
-
•In Study #1, 35% of Tretinoin Gel Microsphere, 0.1% -treated subjects achieved an “excellent” result, as compared to 11% of vehicle-treated subjects.
-
•In Study #2, 28% of Tretinoin Gel Microsphere, 0.1%-treated subjects achieved an “excellent” result, as compared to 9% of vehicle-treated subjects.
14.2 Clinical Studies With Tretinoin Gel Microsphere, 0.04% (14.2 Clinical Studies with Tretinoin Gel Microsphere, 0.04%)
-
In two vehicle-controlled clinical trials in adult and pediatric subjects 11 years of age and older with acne vulgaris (Studies 3 and 4). Tretinoin Gel Microsphere is not approved for use in pediatric patients younger than 12 years of age [see Indications and Usage (1)]. Tretinoin Gel Microsphere, 0.04%, applied topically once daily in the evening, was more effective (p<0.05) than vehicle gel, applied topically once daily, in reducing the acne lesion counts. The mean reduction in lesion counts from baseline after treatment for 12 weeks are shown in Table 2:
| Study 3 | Study 4 | |||
|---|---|---|---|---|
|
Tretinoin Gel Microsphere, 0.04% (n=108) |
Vehicle Gel (n=111) |
Tretinoin Gel Microsphere, 0.04% (n=110) |
Vehicle Gel
(n=103) |
|
|
Non-inflammatory |
37% |
-2% -2% represents a mean percent increase of 2%
|
29% |
14% |
|
Inflammatory lesion |
44% |
13% |
41% |
30% |
|
Total lesion counts |
40% |
8% |
35% |
20% |
Efficacy was also assessed by the investigator’s global evaluation, which included categories of “excellent”, “good”, “fair”, “no change”, and “poor”. Tretinoin Gel Microsphere, 0.04% was superior (p<0.05) to the vehicle in the investigator's global evaluation of the clinical response:
-
•In Study #3, 14% of Tretinoin Gel Microsphere, 0.04%-treated subjects achieved an “excellent” result, as compared to 5% of vehicle-treated subjects.
-
•In Study #4, 19% of Tretinoin Gel Microsphere, 0.04%-treated subjects achieved an “excellent” result, as compared to 9% of vehicle-treated subjects.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:55.345868 · Updated: 2026-03-14T22:50:56.475417