These Highlights Do Not Include All The Information Needed To Use Igalmi Safely And Effectively. See Full Prescribing Information For Igalmi.
99476e8c-2527-4cb0-9d67-9f9cd91343c6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
IGALMI is indicated for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults.
Indications and Usage
IGALMI is indicated for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults.
Dosage and Administration
IGALMI should be administered under the supervision of a healthcare provider. A healthcare provider should monitor vital signs and alertness after IGALMI administration to prevent falls and syncope. ( 2.1 ) Administer sublingually or buccally. Do not chew or swallow. ( 2.1 ) Recommended dosage ( 2.2 ): Patient Population Agitation Severity Initial Dose See Full Prescribing Information for recommendations on administering up to two additional doses and maximum recommended dosages. Adults Mild or Moderate 120 mcg Severe 180 mcg Mild or Moderate Hepatic Impairment Mild or Moderate 90 mcg Severe 120 mcg Severe Hepatic Impairment Mild or Moderate 60 mcg Severe 90 mcg Geriatric Patients (≥ 65 years old) Mild, Moderate, or Severe 120 mcg IGALMI 120 mcg and 180 mcg dosage strengths may be cut in half to obtain the 60 mcg and 90 mcg doses, respectively. See Full Prescribing Information for preparation and administration instructions. ( 2.3 )
Warnings and Precautions
Hypotension, Orthostatic Hypotension, and Bradycardia: Avoid use of IGALMI in patients with hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope. Ensure that patients are alert and not experiencing orthostatic or symptomatic hypotension prior to resuming ambulation. ( 5.1 ) QT Interval Prolongation: IGALMI prolongs the QT interval; avoid use in patients with risk factors for prolonged QT interval. ( 5.2 ) Somnolence: Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or operating hazardous machinery for at least eight hours after taking IGALMI. ( 5.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in detail in other sections of the labeling: Hypotension, Orthostatic Hypotension, and Bradycardia [see Warnings and Precautions (5.1) ] QT Interval Prolongation [see Warnings and Precautions (5.2) ] Somnolence [see Warnings and Precautions (5.3) ] Risk of Withdrawal Reactions [see Warnings and Precautions (5.4) ] Tolerance and Tachyphylaxis [see Warnings and Precautions (5.5) ]
Drug Interactions
Drugs that Prolong the QT interval: Avoid use. ( 7.1 ) Anesthetics, Sedatives, Hypnotics, Opioids: Concomitant use may cause enhanced CNS depressant effects. Reduction in dosage of IGALMI or the concomitant medication may be required. ( 7.2 )
Medication Information
Warnings and Precautions
Hypotension, Orthostatic Hypotension, and Bradycardia: Avoid use of IGALMI in patients with hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope. Ensure that patients are alert and not experiencing orthostatic or symptomatic hypotension prior to resuming ambulation. ( 5.1 ) QT Interval Prolongation: IGALMI prolongs the QT interval; avoid use in patients with risk factors for prolonged QT interval. ( 5.2 ) Somnolence: Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or operating hazardous machinery for at least eight hours after taking IGALMI. ( 5.3 )
Indications and Usage
IGALMI is indicated for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults.
Dosage and Administration
IGALMI should be administered under the supervision of a healthcare provider. A healthcare provider should monitor vital signs and alertness after IGALMI administration to prevent falls and syncope. ( 2.1 ) Administer sublingually or buccally. Do not chew or swallow. ( 2.1 ) Recommended dosage ( 2.2 ): Patient Population Agitation Severity Initial Dose See Full Prescribing Information for recommendations on administering up to two additional doses and maximum recommended dosages. Adults Mild or Moderate 120 mcg Severe 180 mcg Mild or Moderate Hepatic Impairment Mild or Moderate 90 mcg Severe 120 mcg Severe Hepatic Impairment Mild or Moderate 60 mcg Severe 90 mcg Geriatric Patients (≥ 65 years old) Mild, Moderate, or Severe 120 mcg IGALMI 120 mcg and 180 mcg dosage strengths may be cut in half to obtain the 60 mcg and 90 mcg doses, respectively. See Full Prescribing Information for preparation and administration instructions. ( 2.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in detail in other sections of the labeling: Hypotension, Orthostatic Hypotension, and Bradycardia [see Warnings and Precautions (5.1) ] QT Interval Prolongation [see Warnings and Precautions (5.2) ] Somnolence [see Warnings and Precautions (5.3) ] Risk of Withdrawal Reactions [see Warnings and Precautions (5.4) ] Tolerance and Tachyphylaxis [see Warnings and Precautions (5.5) ]
Drug Interactions
Drugs that Prolong the QT interval: Avoid use. ( 7.1 ) Anesthetics, Sedatives, Hypnotics, Opioids: Concomitant use may cause enhanced CNS depressant effects. Reduction in dosage of IGALMI or the concomitant medication may be required. ( 7.2 )
Description
IGALMI is indicated for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults.
Section 42229-5
Limitations of Use
The safety and effectiveness of IGALMI have not been established beyond 24 hours from the first dose [see Warnings and Precautions (5.4, 5.5)].
Section 44425-7
Storage and Handling
Store at controlled room temperature, 20°C to 25°C (68°F to 77°F). Excursions permitted from 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
Keep IGALMI in the foil pouch until ready to administer.
10 Overdosage
In a tolerability study of intravenous dexmedetomidine in which healthy adult subjects were administered doses at and above the recommended dose of 0.2 to 0.7 mcg/kg/hour, the maximum blood concentration was approximately 13 times the upper boundary of the therapeutic range for the intravenous dexmedetomidine (IGALMI is not approved for intravenous use). The most notable effects observed in two subjects who achieved the highest doses were first degree atrioventricular block and second-degree heart block.
Five adult patients received an overdose of intravenous dexmedetomidine in intensive care unit sedation studies. Two patients who received a 2 mcg/kg loading dose (twice the recommended loading dose) over 10 minutes, experienced bradycardia and/or hypotension.
One patient who received a loading intravenous bolus dose of undiluted dexmedetomidine (19.4 mcg/kg), had cardiac arrest from which he was successfully resuscitated.
Consider contacting a Poison Center (1-800-222-1222) or a medical toxicologist for overdosage management recommendations for IGALMI.
11 Description
IGALMI contains dexmedetomidine, an alpha-2 adrenergic receptor agonist, present as dexmedetomidine hydrochloride, the S-enantiomer of medetomidine chemically described as 4-[(1S)-1- (2, 3-dimethylphenyl) ethyl]-1H-imidazole hydrochloride. The empirical formula is C13H16N2∙HCl with a molecular weight of 236.7 g/mol. The structural formula of dexmedetomidine hydrochloride is:
Dexmedetomidine hydrochloride is a white or almost white powder that is freely soluble in water and has a pKa of 7.1. Its partition coefficient in octanol/water at pH 7.4 is 2.89.
IGALMI is for sublingual or buccal use. Each IGALMI sublingual film contains 120 mcg or 180 mcg of dexmedetomidine equivalent to 141.8 mcg and 212.7 mcg of dexmedetomidine hydrochloride, respectively.
IGALMI contains the following inactive ingredients: FD&C Blue #1 colorant, hydroxypropyl cellulose, peppermint oil, polyethylene oxide, and sucralose.
5.3 Somnolence
IGALMI can cause somnolence. In placebo-controlled clinical studies in adults with agitation associated with schizophrenia or bipolar I or II disorder, somnolence (including fatigue and sluggishness) was reported in 23% and 22% of patients treated with IGALMI 180 mcg and 120 mcg, respectively, compared to 6% of placebo-treated patients. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or operating hazardous machinery, for at least eight hours after taking IGALMI [see Adverse Reactions (6.1)].
8.4 Pediatric Use
The safety and effectiveness of IGALMI have not been established in pediatric patients.
8.5 Geriatric Use
Fifteen geriatric patients (≥ 65 years of age) were enrolled (no patients were 75 years of age and older) in the clinical studies for acute treatment of agitation associated with schizophrenia or bipolar I or II disorder. Of the total number of IGALMI-treated patients in these clinical studies, 11/507 (2.2%) were 65 years of age and older [see Clinical Studies (14)].
Dosage reduction of IGALMI is recommended in geriatric patients [see Dosage and Administration (2.2)]. A higher incidence of bradycardia and hypotension was observed in geriatric patients compared to younger adult patients after intravenous administration of another dexmedetomidine product [see Warnings and Precautions (5.1)]. The pharmacokinetic profile of intravenous dexmedetomidine was not altered in geriatric subjects [see Clinical Pharmacology (12.3)].
Clinical studies of IGALMI did not include sufficient numbers of patients 65 years of age and older to determine whether there were differences in the effectiveness of IGALMI in the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder compared to younger adult patients.
14 Clinical Studies
The effectiveness of IGALMI for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults was established in two randomized, double-blind, placebo-controlled, fixed-dose studies (Studies 1 and 2):
- Study 1 (NCT04268303) included 380 patients who met DSM-5 criteria for schizophrenia, schizoaffective or schizophreniform disorder. The population was 18 to 71 years of age (mean age was 46 years old); 37% female and 63% male; 78% Black, 20% White, 1% multiracial, and 1% Asian.
- Study 2 (NCT04276883) included 378 patients who met DSM-5 criteria for bipolar I or II disorder. The population was 18 to 70 years of age (mean age was 47 years old); 55% female and 45% male; 56% Black, 41% White, 1% Asian, 1% multiracial, and 1% other.
The Positive and Negative Syndrome Scale-Excited Component (PEC) is an investigator-rated instrument consisting of 5 items: poor impulse control, tension, hostility, uncooperativeness, and excitement. Each item is scored on a scale from 1 to 7 (1=absent, 2=minimal, 3=mild, 4=moderate, 5=moderate-severe, 6=severe, 7=extremely severe). The total PEC score ranges from 5 to 35, with higher scores reflecting greater overall symptom severity. For enrollment in the studies, patients had to be judged to be clinically agitated with a total PEC score of ≥ 14, with at least one individual item score ≥ 4. In both studies, patients were admitted to a clinical research unit or a hospital and remained under medical supervision for at least 24 hours following treatment.
Patients were randomized to receive a single sublingual dose of 180 mcg of IGALMI, 120 mcg of IGALMI, or placebo. The primary efficacy endpoint in both studies was the change from baseline in the PEC score, assessed two hours following the initial dose. The key secondary endpoint was the time to effect onset, assessed by measuring the change from baseline in PEC score at 10, 20, 30, 45, 60, and 90 minutes after the initial dose administration.
In both studies, mean baseline PEC scores were similar in all treatment groups (Table 5). The mean change from baseline in the PEC total score at two hours after the first dose in patients treated with 180 mcg and 120 mcg of IGALMI was statistically greater than patients who received placebo (Table 5).
Examination of population subsets (race and sex) on the primary endpoint did not show evidence for differential responsiveness between White and Black or female and male patients. The clinical studies did not include enough patients of other races or patients ≥65 years of age to determine whether there were differences in effectiveness for those groups.
| Study | Treatment Group | Number of Patients | Mean Baseline PEC Score (SD) |
LS Mean Change from Baseline to 2 hour Post First Dose (SE) |
LS Mean Difference (95% CI) |
|---|---|---|---|---|---|
| SD=standard deviation; SE=standard error; LS Mean=least-squares mean; CI=unadjusted confidence interval; PEC=Positive and Negative Syndrome Scale-Excited Component | |||||
| Study 1 | IGALMI 180 mcg IGALMI doses that were statistically significantly superior to placebo after adjusting for multiplicity.
|
125 | 17.6 (2.7) | -10.3 (0.4) | -5.5 (-6.5, -4.4) |
| IGALMI 120 mcg | 129 | 17.5 (2.5) | -8.5 (0.4) | -3.7 (-4.8, -2.7) | |
| Placebo | 126 | 17.6 (2.3) | -4.8 (0.4) | - | |
| Study 2 | IGALMI 180 mcg | 126 | 18.0 (3.0) | -10.4 (0.4) | -5.4 (-6.5, -4.3) |
| IGALMI 120 mcg | 126 | 18.0 (2.7) | -9.1 (0.4) | -4.1 (-5.1, -3.0) | |
| Placebo | 126 | 17.9 (2.9) | -5.0 (0.4) | - |
Figures 1 and 2 show the changes from baseline in PEC score at each time point assessed up to two hours following the initial dose. In Study 1, the decrease in agitation with IGALMI, compared to placebo, was statistically significant beginning at 20 minutes following dosing with the 180 mcg dose and 30 minutes after the 120 mcg dose. In Study 2, the decrease in agitation with IGALMI, compared to placebo, was statistically significant beginning at 20 minutes after treatment with both 120 mcg and 180 mcg doses.
Figure 1: Mean Change from Baseline in PEC Score Through Two Hours after a Single Dose in Agitated Patients with Schizophrenia (Study 1)
Figure 2: Mean Change from Baseline in PEC Score Through Two Hours after a Single Dose in Agitated Patients with Bipolar I or II Disorder (Study 2)
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in detail in other sections of the labeling:
- Hypotension, Orthostatic Hypotension, and Bradycardia [see Warnings and Precautions (5.1)]
- QT Interval Prolongation [see Warnings and Precautions (5.2)]
- Somnolence [see Warnings and Precautions (5.3)]
- Risk of Withdrawal Reactions [see Warnings and Precautions (5.4)]
- Tolerance and Tachyphylaxis [see Warnings and Precautions (5.5)]
7 Drug Interactions
12.2 Pharmacodynamics
Dexmedetomidine acts as an agonist at alpha-2 adrenergic receptors with binding affinities (Ki values) of 4 to 6 nM at the alpha-2 adrenergic receptor subtypes.
12.3 Pharmacokinetics
Dexmedetomidine exposure (Cmax and AUC) increased in a dose proportional manner in the dose range of 20 mcg (0.17 times the lowest recommended initial dose of 120 mcg) to 180 mcg after single sublingual administration of IGALMI.
The mean time for film to dissolve in the mouth was about 6 to 8 minutes and 18 minutes following sublingual and buccal administration, respectively. Dexmedetomidine was quantifiable in plasma generally after 5 to 20 minutes post dosing.
2.2 Recommended Dosage
Table 1 includes dosage recommendations for IGALMI based on agitation severity for adults, patients with hepatic impairment, and geriatric patients. Lower dosages are recommended for patients with hepatic impairment and geriatric patients [see Warnings and Precautions (5.1) and Use in Specific Populations (8.5, 8.6)].
If agitation persists after the initial dose, up to two additional doses may be administered at least two hours apart. The dosage recommendations for additional doses vary depending upon the patient population and agitation severity (see Table 1). Assess vital signs including orthostatic measurements prior to the administration of any subsequent doses.
Due to risk of hypotension, additional half-doses are not recommended in patients with systolic blood pressure (SBP) less than 90 mmHg, diastolic blood pressure (DBP) less than 60 mmHg, heart rate (HR) less than 60 beats per minute, or postural decrease in SBP ≥ 20 mmHg or in DBP ≥ 10 mmHg.
| Patient Population | Agitation Severity | Initial Dose IGALMI 120 mcg and 180 mcg dosage strengths may be cut in half to obtain the 60 mcg and 90 mcg doses, respectively [see Dosage and Administration (2.3)].
|
Optional 2nd/3rd Doses | Maximum Recommended Total Daily Dosage |
|---|---|---|---|---|
| Adults | Mild or Moderate | 120 mcg | 60 mcg | 240 mcg |
| Severe | 180 mcg | 90 mcg | 360 mcg | |
| Patients with Mild or Moderate Hepatic Impairment Hepatic impairment: Mild (Child-Pugh Class A); Moderate (Child-Pugh Class B); Severe (Child-Pugh Class C)
|
Mild or Moderate | 90 mcg | 60 mcg | 210 mcg |
| Severe | 120 mcg | 60 mcg | 240 mcg | |
| Patients with Severe Hepatic Impairment | Mild or Moderate | 60 mcg | 60 mcg | 180 mcg |
| Severe | 90 mcg | 60 mcg | 210 mcg | |
| Geriatric Patients (≥ 65 years old) |
Mild, Moderate, or Severe | 120 mcg | 60 mcg | 240 mcg |
8.6 Hepatic Impairment
Dexmedetomidine clearance was decreased in patients with hepatic impairment (Child-Pugh Class A, B, or C). Thus, a dosage reduction of IGALMI is recommended in patients with hepatic impairment compared to patients with normal hepatic function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
1 Indications and Usage
IGALMI is indicated for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults.
12.1 Mechanism of Action
Dexmedetomidine is an alpha-2 adrenergic receptor agonist. The mechanism of action of IGALMI in the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder is thought to be due to activation of presynaptic alpha-2 adrenergic receptors.
9.1 Controlled Substance
IGALMI contains dexmedetomidine, which is not a controlled substance.
5 Warnings and Precautions
- Hypotension, Orthostatic Hypotension, and Bradycardia: Avoid use of IGALMI in patients with hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope. Ensure that patients are alert and not experiencing orthostatic or symptomatic hypotension prior to resuming ambulation. (5.1)
- QT Interval Prolongation: IGALMI prolongs the QT interval; avoid use in patients with risk factors for prolonged QT interval. (5.2)
- Somnolence: Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or operating hazardous machinery for at least eight hours after taking IGALMI. (5.3)
2 Dosage and Administration
- IGALMI should be administered under the supervision of a healthcare provider. A healthcare provider should monitor vital signs and alertness after IGALMI administration to prevent falls and syncope. (2.1)
- Administer sublingually or buccally. Do not chew or swallow. (2.1)
- Recommended dosage (2.2):
Patient Population Agitation Severity Initial Dose See Full Prescribing Information for recommendations on administering up to two additional doses and maximum recommended dosages.Adults Mild or Moderate 120 mcg Severe 180 mcg Mild or Moderate Hepatic Impairment Mild or Moderate 90 mcg Severe 120 mcg Severe Hepatic Impairment Mild or Moderate 60 mcg Severe 90 mcg Geriatric Patients
(≥ 65 years old)Mild, Moderate, or Severe 120 mcg - IGALMI 120 mcg and 180 mcg dosage strengths may be cut in half to obtain the 60 mcg and 90 mcg doses, respectively. See Full Prescribing Information for preparation and administration instructions. (2.3)
3 Dosage Forms and Strengths
IGALMI is a blue rectangular sublingual film containing on its surface two darker blue spots in dose strengths of 120 mcg and 180 mcg.
5.2 Qt Interval Prolongation
IGALMI prolongs the QT interval. Avoid use of IGALMI in patients at risk of torsades de pointes or sudden death including those with known QT prolongation, a history of other arrhythmias, symptomatic bradycardia, hypokalemia or hypomagnesemia, and in patients receiving other drugs known to prolong the QT interval [see Drug Interactions (7.1) and Clinical Pharmacology (12.2)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of another dexmedetomidine product given intravenously (IGALMI is not approved for intravenous use). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Blood and Lymphatic System Disorders: Anemia
- Cardiac Disorders: Arrhythmia, atrial fibrillation, atrioventricular block, bradycardia, cardiac arrest, cardiac disorder, extrasystoles, myocardial infarction, supraventricular tachycardia, tachycardia, ventricular arrhythmia, ventricular tachycardia
- Eye Disorders: Photopsia, visual impairment
- Gastrointestinal Disorders: Abdominal pain, diarrhea, nausea, vomiting
- General Disorders and Administration Site Conditions: Chills, hyperpyrexia, pain, pyrexia, thirst
- Hepatobiliary Disorders: Hepatic function abnormal, hyperbilirubinemia
- Investigations: Alanine aminotransferase increased, aspartate aminotransferase increased, blood alkaline phosphatase increased, blood urea increased, electrocardiogram T wave inversion, gammaglutamyltransferase increased, electrocardiogram QT prolonged
- Metabolism and Nutrition Disorders: Acidosis, hyperkalemia, hypoglycemia, hypovolemia, hypernatremia
- Nervous System Disorders: Convulsion, dizziness, headache, neuralgia, neuritis, speech disorder
- Psychiatric Disorders: Agitation, confusional state, delirium, hallucination, illusion
- Renal and Urinary Disorders: Oliguria, polyuria
- Respiratory, Thoracic and Mediastinal Disorders: Apnea, bronchospasm, dyspnea, hypercapnia, hypoventilation, hypoxia, pulmonary congestion, respiratory acidosis
- Skin and Subcutaneous Tissue Disorders: Hyperhidrosis, pruritus, rash, urticaria
- Surgical and Medical Procedures: Light anesthesia
- Vascular Disorders: Blood pressure fluctuation, hemorrhage, hypertension, hypotension
5.5 Tolerance and Tachyphylaxis
Use of another dexmedetomidine product administered intravenously beyond 24 hours has been associated with tolerance and tachyphylaxis and a dose-related increase in adverse reactions. The effectiveness of IGALMI beyond 24 hours after the first dose has not been established. There may be a risk of tolerance and tachyphylaxis if IGALMI is administered at higher doses or for a longer duration than were evaluated, or in a manner other than indicated [see Dosage and Administration (2.2) and Drug Abuse and Dependence (9.3)].
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
The safety of IGALMI was evaluated in 507 adult patients with agitation associated with schizophrenia (N=255) or bipolar I or II disorder (N=252) in two randomized, placebo-controlled studies (Studies 1 and 2) [see Clinical Studies (14)]. In both studies, patients were admitted to a clinical research unit or a hospital and remained under medical supervision for at least 24 hours following treatment. Patients were 18 to 71 years of age (mean age was 46 years old); 45% were female and 55% were male; 66% were Black, 31% were White, 2% were multiracial, and 1% were other.
In these studies, patients received an initial dose of IGALMI 180 mcg (N=252), IGALMI 120 mcg (N=255), or placebo (N=252). Patients who were hemodynamically stable (i.e., those with systolic blood pressure (SBP) > 90 mmHg, diastolic blood pressure (DBP) > 60 mmHg, and heart rate (HR) > 60 beats per minute) and without orthostatic hypotension (i.e., reduction in SBP < 20 mmHg or DBP < 10 mmHg upon standing) were eligible for an additional dose after 2 hours. An additional half dose (90 mcg, 60 mcg, or placebo) was given to 7.1% (18/252), 22.7% (58/255) and 44.0% (111/252) of patients in the IGALMI 180 mcg, IGALMI 120 mcg or placebo arms, respectively. After at least an additional 2 hours, an additional second half dose (total IGALMI dose of 360 mcg, total IGALMI dose of 240 mcg, or placebo, respectively) was given to 3.2% (8/252), 9.4% (24/255), and 21.0% (53/252) of patients in the IGALMI 180 mcg, IGALMI 120 mcg or placebo arms, respectively.
In these studies, one patient discontinued treatment due to an adverse reaction of oropharyngeal pain.
The most common adverse reactions (incidence ≥ 5% and at least twice the rate of placebo) were: somnolence, oral paresthesia or oral hypoesthesia, dizziness, dry mouth, hypotension, and orthostatic hypotension.
Table 2 presents the adverse reactions that occurred in IGALMI-treated patients at a rate of at least 2% and at a higher rate than in placebo-treated patients in Studies 1 and 2.
| Adverse Reaction | IGALMI 180 mcg N=252 % |
IGALMI 120 mcg N=255 % |
Placebo N=252 % |
|---|---|---|---|
| Somnolence Somnolence includes the terms fatigue and sluggishness
|
23 | 22 | 6 |
| Paresthesia oral or hypoesthesia oral | 7 | 6 | 1 |
| Dizziness | 6 | 4 | 1 |
| Hypotension | 5 | 5 | 0 |
| Orthostatic hypotension | 5 | 3 | <1 |
| Dry mouth | 4 | 7 | 1 |
| Nausea | 3 | 2 | 2 |
| Bradycardia | 2 | 2 | 0 |
| Abdominal discomfort Abdominal discomfort includes dyspepsia, gastroesophageal reflux disease
|
2 | 0 | 1 |
5.4 Risk of Withdrawal Reactions
Symptoms of withdrawal have been observed after treatment with another dexmedetomidine product administered as a continuous IV infusion in the ICU for another indication. In this study, 12 (5%) adult patients who received intravenous dexmedetomidine up to 7 days (regardless of dose) experienced at least 1 event related to withdrawal within the first 24 hours after discontinuing dexmedetomidine and 7 (3%) adult patients who received intravenous dexmedetomidine experienced at least 1 event related with withdrawal 24 to 48 hours after discontinuing dexmedetomidine. The most common withdrawal reactions were nausea, vomiting, and agitation. In these subjects, tachycardia and hypertension requiring intervention occurred at a frequency of <5% in the 48 hours following intravenous dexmedetomidine discontinuation. Withdrawal symptoms have been reported in pediatric patients who received another dexmedetomidine product intravenously for another indication. IGALMI is not approved for pediatric use.
Withdrawal symptoms reported in published literature include cardiovascular symptoms, such as hypertension, tachycardia, arrhythmias, and in severe cases, hypertensive crisis; neurological symptoms, such as agitation, anxiety, tremors, seizures, and altered mental status; and autonomic symptoms such as diaphoresis, nausea, vomiting, hyperthermia, and mydriasis. In a randomized, double-blind, placebo-controlled, multiple ascending dose study assessing IGALMI at doses up to 120 mcg per day administered over 7 days in healthy volunteers, elevated heart rate and systolic blood pressure and vomiting occurred at a higher frequency upon abrupt discontinuation of 120-mcg doses of IGALMI as compared to placebo. There may be a risk of physical dependence and a withdrawal syndrome if IGALMI is used in a manner other than indicated [see Dosage and Administration (2.2) and Drug Abuse and Dependence (9.3)].
7.1 Drugs That Prolong the Qt Interval
Concomitant use of drugs that prolong the QT interval may add to the QT-prolonging effects of IGALMI and increase the risk of cardiac arrhythmia. Avoid the use of IGALMI in combination with other drugs known to prolong the QT interval [see Warnings and Precautions (5.2)].
13.2 Animal Toxicology And/or Pharmacology
Twice daily sublingual administration of 120 to 320 mcg/day of dexmedetomidine to dogs for 28 days caused decreased heart rate and moderate sedation up to 3.5 hours post dose. A single male dog (out of 32 treated dogs) dosed 320 mcg/day (equivalent to the MRHD of 360 mcg/day) exhibited inflammation, necrosis, myofiber degeneration, and hemorrhage at the sublingual treatment site. No adverse effects were noted at 240 mcg/day (less than the MRHD of 360 mcg/day).
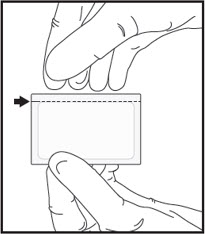
2.3 Preparation and Administration Instructions
Keep IGALMI in the foil pouch until ready to administer. IGALMI should be immediately administered once the pouch is opened and the dose prepared.
Prepare and administer IGALMI under the supervision of a healthcare provider as follows:
| Healthcare Professional: Prepare IGALMI Dose for Patient | |||
| 1 | Open the sealed foil pouch by tearing straight across at the notch. |
|
|
|
|||
| 2a | Remove the film from the pouch with clean dry hands. |
|
|
| 2b | Cut the film in half between the dots with clean, dry scissors. |
|
|
| 2c | Discard unused half in waste container. | ||
| 2d | Place the half film for administration to the patient back into the pouch. |
|
|
| 3 | Immediately give the pouch to the patient. |
|
|
| 4 | Instruct patient to remove the film from the pouch with clean dry hands. |
|
|
| 5 |
For sublingual administration: Instruct patient to place film under the tongue. The film will stick in place. Note: Patient may not eat or drink for 15 minutes after sublingual administration. |
|
|
|
For buccal administration: Instruct patient to place film behind lower lip. The film will stick in place. Note: Patient may not eat or drink for one hour after buccal administration. |
|
||
| 6 | Instruct patient to:
|
7.2 Anesthetics, Sedatives, Hypnotics, and Opioids
Concomitant use of IGALMI with anesthetics, sedatives, hypnotics, or opioids is likely to lead to enhanced CNS depressant effects. Specific studies with another dexmedetomidine product given intravenously have confirmed these effects with sevoflurane, isoflurane, propofol, alfentanil, and midazolam. Due to possible enhanced CNS effects when given concomitantly with IGALMI, consider a reduction in dosage of IGALMI or the concomitant anesthetic, sedative, hypnotic, or opioid.
Principal Display Panel 120 Mcg Film Pouch Carton
Rx Only
Igalmi™
(dexmedetomidine) sublingual film
For sublingual or buccal use
Keep IGALMI in pouch until administration
Do not chew or swallow the film
120 mcg
10 pouches
Each containing
1 film
NDC 81092-1120-1
Principal Display Panel 180 Mcg Film Pouch Carton
Rx Only
Igalmi™
(dexmedetomidine) sublingual film
For sublingual or buccal use
Keep IGALMI in pouch until administration
Do not chew or swallow the film
180 mcg
10 pouches
Each containing
1 film
NDC 81092-1180-1
5.1 Hypotension, Orthostatic Hypotension, and Bradycardia
IGALMI causes dose-dependent hypotension, orthostatic hypotension, and bradycardia. In clinical studies, 18%, 16%, and 9% of patients treated with 180 mcg of IGALMI, 120 mcg of IGALMI, and placebo, respectively, experienced orthostatic hypotension (defined as SBP decrease ≥ 20 mmHg or DBP decrease ≥ 10 mmHg after 1, 3, or 5 minutes of standing) at 2 hours post-dose. In those studies, 7%, 6%, and 1% of patients treated with 180 mcg of IGALMI, 120 mcg of IGALMI, and placebo, respectively, experienced HR ≤ 50 beats per minute within 2 hours of dosing [see Adverse Reactions (6.1)]. In clinical studies with IGALMI, patients were excluded if they had treatment with alpha-1 noradrenergic blockers, benzodiazepines, other hypnotics or antipsychotic drugs four hours prior to study drug administration; had a history of syncope or syncopal attacks; SBP < 110 mmHg; DBP < 70 mmHg; HR < 55 beats per minute; or had evidence of hypovolemia or orthostatic hypotension.
Reports of hypotension and bradycardia, including some resulting in fatalities, have been associated with the use of another dexmedetomidine product given intravenously (IGALMI is for sublingual or buccal use and is not approved for intravenous use). Clinically significant episodes of bradycardia and sinus arrest have been reported after administration of this other dexmedetomidine product to young, healthy adult volunteers with high vagal tone and when this product was given by rapid intravenous or bolus administration.
Because IGALMI decreases sympathetic nervous system activity, hypotension and/or bradycardia may be more pronounced in patients with hypovolemia, diabetes mellitus, or chronic hypertension, and in geriatric patients [see Dosage and Administration (2.2) and Use in Specific Populations (8.5)].
Avoid use of IGALMI in patients with hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope. After IGALMI administration, patients should be adequately hydrated and should sit or lie down until vital signs are within normal range. If a patient is unable to remain seated or lying down, precautions should be taken to reduce the risk of falls. Ensure that a patient is alert and not experiencing orthostatic hypotension or symptomatic hypotension prior to allowing them to resume ambulation [see Dosage and Administration (2.1)].
2.1 Important Recommendations Prior to Initiating Igalmi and During Therapy
IGALMI should be administered under the supervision of a healthcare provider. A healthcare provider should monitor vital signs and alertness after IGALMI administration to prevent falls and syncope [see Warnings and Precautions (5.5)].
IGALMI is for sublingual or buccal administration. Do not chew or swallow IGALMI. Do not eat or drink for at least 15 minutes after sublingual administration, or at least one hour after buccal administration.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
The safety and effectiveness of IGALMI have not been established beyond 24 hours from the first dose [see Warnings and Precautions (5.4, 5.5)].
Section 44425-7 (44425-7)
Storage and Handling
Store at controlled room temperature, 20°C to 25°C (68°F to 77°F). Excursions permitted from 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
Keep IGALMI in the foil pouch until ready to administer.
10 Overdosage (10 OVERDOSAGE)
In a tolerability study of intravenous dexmedetomidine in which healthy adult subjects were administered doses at and above the recommended dose of 0.2 to 0.7 mcg/kg/hour, the maximum blood concentration was approximately 13 times the upper boundary of the therapeutic range for the intravenous dexmedetomidine (IGALMI is not approved for intravenous use). The most notable effects observed in two subjects who achieved the highest doses were first degree atrioventricular block and second-degree heart block.
Five adult patients received an overdose of intravenous dexmedetomidine in intensive care unit sedation studies. Two patients who received a 2 mcg/kg loading dose (twice the recommended loading dose) over 10 minutes, experienced bradycardia and/or hypotension.
One patient who received a loading intravenous bolus dose of undiluted dexmedetomidine (19.4 mcg/kg), had cardiac arrest from which he was successfully resuscitated.
Consider contacting a Poison Center (1-800-222-1222) or a medical toxicologist for overdosage management recommendations for IGALMI.
11 Description (11 DESCRIPTION)
IGALMI contains dexmedetomidine, an alpha-2 adrenergic receptor agonist, present as dexmedetomidine hydrochloride, the S-enantiomer of medetomidine chemically described as 4-[(1S)-1- (2, 3-dimethylphenyl) ethyl]-1H-imidazole hydrochloride. The empirical formula is C13H16N2∙HCl with a molecular weight of 236.7 g/mol. The structural formula of dexmedetomidine hydrochloride is:
Dexmedetomidine hydrochloride is a white or almost white powder that is freely soluble in water and has a pKa of 7.1. Its partition coefficient in octanol/water at pH 7.4 is 2.89.
IGALMI is for sublingual or buccal use. Each IGALMI sublingual film contains 120 mcg or 180 mcg of dexmedetomidine equivalent to 141.8 mcg and 212.7 mcg of dexmedetomidine hydrochloride, respectively.
IGALMI contains the following inactive ingredients: FD&C Blue #1 colorant, hydroxypropyl cellulose, peppermint oil, polyethylene oxide, and sucralose.
5.3 Somnolence
IGALMI can cause somnolence. In placebo-controlled clinical studies in adults with agitation associated with schizophrenia or bipolar I or II disorder, somnolence (including fatigue and sluggishness) was reported in 23% and 22% of patients treated with IGALMI 180 mcg and 120 mcg, respectively, compared to 6% of placebo-treated patients. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or operating hazardous machinery, for at least eight hours after taking IGALMI [see Adverse Reactions (6.1)].
8.4 Pediatric Use
The safety and effectiveness of IGALMI have not been established in pediatric patients.
8.5 Geriatric Use
Fifteen geriatric patients (≥ 65 years of age) were enrolled (no patients were 75 years of age and older) in the clinical studies for acute treatment of agitation associated with schizophrenia or bipolar I or II disorder. Of the total number of IGALMI-treated patients in these clinical studies, 11/507 (2.2%) were 65 years of age and older [see Clinical Studies (14)].
Dosage reduction of IGALMI is recommended in geriatric patients [see Dosage and Administration (2.2)]. A higher incidence of bradycardia and hypotension was observed in geriatric patients compared to younger adult patients after intravenous administration of another dexmedetomidine product [see Warnings and Precautions (5.1)]. The pharmacokinetic profile of intravenous dexmedetomidine was not altered in geriatric subjects [see Clinical Pharmacology (12.3)].
Clinical studies of IGALMI did not include sufficient numbers of patients 65 years of age and older to determine whether there were differences in the effectiveness of IGALMI in the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder compared to younger adult patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of IGALMI for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults was established in two randomized, double-blind, placebo-controlled, fixed-dose studies (Studies 1 and 2):
- Study 1 (NCT04268303) included 380 patients who met DSM-5 criteria for schizophrenia, schizoaffective or schizophreniform disorder. The population was 18 to 71 years of age (mean age was 46 years old); 37% female and 63% male; 78% Black, 20% White, 1% multiracial, and 1% Asian.
- Study 2 (NCT04276883) included 378 patients who met DSM-5 criteria for bipolar I or II disorder. The population was 18 to 70 years of age (mean age was 47 years old); 55% female and 45% male; 56% Black, 41% White, 1% Asian, 1% multiracial, and 1% other.
The Positive and Negative Syndrome Scale-Excited Component (PEC) is an investigator-rated instrument consisting of 5 items: poor impulse control, tension, hostility, uncooperativeness, and excitement. Each item is scored on a scale from 1 to 7 (1=absent, 2=minimal, 3=mild, 4=moderate, 5=moderate-severe, 6=severe, 7=extremely severe). The total PEC score ranges from 5 to 35, with higher scores reflecting greater overall symptom severity. For enrollment in the studies, patients had to be judged to be clinically agitated with a total PEC score of ≥ 14, with at least one individual item score ≥ 4. In both studies, patients were admitted to a clinical research unit or a hospital and remained under medical supervision for at least 24 hours following treatment.
Patients were randomized to receive a single sublingual dose of 180 mcg of IGALMI, 120 mcg of IGALMI, or placebo. The primary efficacy endpoint in both studies was the change from baseline in the PEC score, assessed two hours following the initial dose. The key secondary endpoint was the time to effect onset, assessed by measuring the change from baseline in PEC score at 10, 20, 30, 45, 60, and 90 minutes after the initial dose administration.
In both studies, mean baseline PEC scores were similar in all treatment groups (Table 5). The mean change from baseline in the PEC total score at two hours after the first dose in patients treated with 180 mcg and 120 mcg of IGALMI was statistically greater than patients who received placebo (Table 5).
Examination of population subsets (race and sex) on the primary endpoint did not show evidence for differential responsiveness between White and Black or female and male patients. The clinical studies did not include enough patients of other races or patients ≥65 years of age to determine whether there were differences in effectiveness for those groups.
| Study | Treatment Group | Number of Patients | Mean Baseline PEC Score (SD) |
LS Mean Change from Baseline to 2 hour Post First Dose (SE) |
LS Mean Difference (95% CI) |
|---|---|---|---|---|---|
| SD=standard deviation; SE=standard error; LS Mean=least-squares mean; CI=unadjusted confidence interval; PEC=Positive and Negative Syndrome Scale-Excited Component | |||||
| Study 1 | IGALMI 180 mcg IGALMI doses that were statistically significantly superior to placebo after adjusting for multiplicity.
|
125 | 17.6 (2.7) | -10.3 (0.4) | -5.5 (-6.5, -4.4) |
| IGALMI 120 mcg | 129 | 17.5 (2.5) | -8.5 (0.4) | -3.7 (-4.8, -2.7) | |
| Placebo | 126 | 17.6 (2.3) | -4.8 (0.4) | - | |
| Study 2 | IGALMI 180 mcg | 126 | 18.0 (3.0) | -10.4 (0.4) | -5.4 (-6.5, -4.3) |
| IGALMI 120 mcg | 126 | 18.0 (2.7) | -9.1 (0.4) | -4.1 (-5.1, -3.0) | |
| Placebo | 126 | 17.9 (2.9) | -5.0 (0.4) | - |
Figures 1 and 2 show the changes from baseline in PEC score at each time point assessed up to two hours following the initial dose. In Study 1, the decrease in agitation with IGALMI, compared to placebo, was statistically significant beginning at 20 minutes following dosing with the 180 mcg dose and 30 minutes after the 120 mcg dose. In Study 2, the decrease in agitation with IGALMI, compared to placebo, was statistically significant beginning at 20 minutes after treatment with both 120 mcg and 180 mcg doses.
Figure 1: Mean Change from Baseline in PEC Score Through Two Hours after a Single Dose in Agitated Patients with Schizophrenia (Study 1)
Figure 2: Mean Change from Baseline in PEC Score Through Two Hours after a Single Dose in Agitated Patients with Bipolar I or II Disorder (Study 2)
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in detail in other sections of the labeling:
- Hypotension, Orthostatic Hypotension, and Bradycardia [see Warnings and Precautions (5.1)]
- QT Interval Prolongation [see Warnings and Precautions (5.2)]
- Somnolence [see Warnings and Precautions (5.3)]
- Risk of Withdrawal Reactions [see Warnings and Precautions (5.4)]
- Tolerance and Tachyphylaxis [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
Dexmedetomidine acts as an agonist at alpha-2 adrenergic receptors with binding affinities (Ki values) of 4 to 6 nM at the alpha-2 adrenergic receptor subtypes.
12.3 Pharmacokinetics
Dexmedetomidine exposure (Cmax and AUC) increased in a dose proportional manner in the dose range of 20 mcg (0.17 times the lowest recommended initial dose of 120 mcg) to 180 mcg after single sublingual administration of IGALMI.
The mean time for film to dissolve in the mouth was about 6 to 8 minutes and 18 minutes following sublingual and buccal administration, respectively. Dexmedetomidine was quantifiable in plasma generally after 5 to 20 minutes post dosing.
2.2 Recommended Dosage
Table 1 includes dosage recommendations for IGALMI based on agitation severity for adults, patients with hepatic impairment, and geriatric patients. Lower dosages are recommended for patients with hepatic impairment and geriatric patients [see Warnings and Precautions (5.1) and Use in Specific Populations (8.5, 8.6)].
If agitation persists after the initial dose, up to two additional doses may be administered at least two hours apart. The dosage recommendations for additional doses vary depending upon the patient population and agitation severity (see Table 1). Assess vital signs including orthostatic measurements prior to the administration of any subsequent doses.
Due to risk of hypotension, additional half-doses are not recommended in patients with systolic blood pressure (SBP) less than 90 mmHg, diastolic blood pressure (DBP) less than 60 mmHg, heart rate (HR) less than 60 beats per minute, or postural decrease in SBP ≥ 20 mmHg or in DBP ≥ 10 mmHg.
| Patient Population | Agitation Severity | Initial Dose IGALMI 120 mcg and 180 mcg dosage strengths may be cut in half to obtain the 60 mcg and 90 mcg doses, respectively [see Dosage and Administration (2.3)].
|
Optional 2nd/3rd Doses | Maximum Recommended Total Daily Dosage |
|---|---|---|---|---|
| Adults | Mild or Moderate | 120 mcg | 60 mcg | 240 mcg |
| Severe | 180 mcg | 90 mcg | 360 mcg | |
| Patients with Mild or Moderate Hepatic Impairment Hepatic impairment: Mild (Child-Pugh Class A); Moderate (Child-Pugh Class B); Severe (Child-Pugh Class C)
|
Mild or Moderate | 90 mcg | 60 mcg | 210 mcg |
| Severe | 120 mcg | 60 mcg | 240 mcg | |
| Patients with Severe Hepatic Impairment | Mild or Moderate | 60 mcg | 60 mcg | 180 mcg |
| Severe | 90 mcg | 60 mcg | 210 mcg | |
| Geriatric Patients (≥ 65 years old) |
Mild, Moderate, or Severe | 120 mcg | 60 mcg | 240 mcg |
8.6 Hepatic Impairment
Dexmedetomidine clearance was decreased in patients with hepatic impairment (Child-Pugh Class A, B, or C). Thus, a dosage reduction of IGALMI is recommended in patients with hepatic impairment compared to patients with normal hepatic function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
IGALMI is indicated for the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder in adults.
12.1 Mechanism of Action
Dexmedetomidine is an alpha-2 adrenergic receptor agonist. The mechanism of action of IGALMI in the acute treatment of agitation associated with schizophrenia or bipolar I or II disorder is thought to be due to activation of presynaptic alpha-2 adrenergic receptors.
9.1 Controlled Substance
IGALMI contains dexmedetomidine, which is not a controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypotension, Orthostatic Hypotension, and Bradycardia: Avoid use of IGALMI in patients with hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope. Ensure that patients are alert and not experiencing orthostatic or symptomatic hypotension prior to resuming ambulation. (5.1)
- QT Interval Prolongation: IGALMI prolongs the QT interval; avoid use in patients with risk factors for prolonged QT interval. (5.2)
- Somnolence: Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or operating hazardous machinery for at least eight hours after taking IGALMI. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- IGALMI should be administered under the supervision of a healthcare provider. A healthcare provider should monitor vital signs and alertness after IGALMI administration to prevent falls and syncope. (2.1)
- Administer sublingually or buccally. Do not chew or swallow. (2.1)
- Recommended dosage (2.2):
Patient Population Agitation Severity Initial Dose See Full Prescribing Information for recommendations on administering up to two additional doses and maximum recommended dosages.Adults Mild or Moderate 120 mcg Severe 180 mcg Mild or Moderate Hepatic Impairment Mild or Moderate 90 mcg Severe 120 mcg Severe Hepatic Impairment Mild or Moderate 60 mcg Severe 90 mcg Geriatric Patients
(≥ 65 years old)Mild, Moderate, or Severe 120 mcg - IGALMI 120 mcg and 180 mcg dosage strengths may be cut in half to obtain the 60 mcg and 90 mcg doses, respectively. See Full Prescribing Information for preparation and administration instructions. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
IGALMI is a blue rectangular sublingual film containing on its surface two darker blue spots in dose strengths of 120 mcg and 180 mcg.
5.2 Qt Interval Prolongation (5.2 QT Interval Prolongation)
IGALMI prolongs the QT interval. Avoid use of IGALMI in patients at risk of torsades de pointes or sudden death including those with known QT prolongation, a history of other arrhythmias, symptomatic bradycardia, hypokalemia or hypomagnesemia, and in patients receiving other drugs known to prolong the QT interval [see Drug Interactions (7.1) and Clinical Pharmacology (12.2)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of another dexmedetomidine product given intravenously (IGALMI is not approved for intravenous use). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Blood and Lymphatic System Disorders: Anemia
- Cardiac Disorders: Arrhythmia, atrial fibrillation, atrioventricular block, bradycardia, cardiac arrest, cardiac disorder, extrasystoles, myocardial infarction, supraventricular tachycardia, tachycardia, ventricular arrhythmia, ventricular tachycardia
- Eye Disorders: Photopsia, visual impairment
- Gastrointestinal Disorders: Abdominal pain, diarrhea, nausea, vomiting
- General Disorders and Administration Site Conditions: Chills, hyperpyrexia, pain, pyrexia, thirst
- Hepatobiliary Disorders: Hepatic function abnormal, hyperbilirubinemia
- Investigations: Alanine aminotransferase increased, aspartate aminotransferase increased, blood alkaline phosphatase increased, blood urea increased, electrocardiogram T wave inversion, gammaglutamyltransferase increased, electrocardiogram QT prolonged
- Metabolism and Nutrition Disorders: Acidosis, hyperkalemia, hypoglycemia, hypovolemia, hypernatremia
- Nervous System Disorders: Convulsion, dizziness, headache, neuralgia, neuritis, speech disorder
- Psychiatric Disorders: Agitation, confusional state, delirium, hallucination, illusion
- Renal and Urinary Disorders: Oliguria, polyuria
- Respiratory, Thoracic and Mediastinal Disorders: Apnea, bronchospasm, dyspnea, hypercapnia, hypoventilation, hypoxia, pulmonary congestion, respiratory acidosis
- Skin and Subcutaneous Tissue Disorders: Hyperhidrosis, pruritus, rash, urticaria
- Surgical and Medical Procedures: Light anesthesia
- Vascular Disorders: Blood pressure fluctuation, hemorrhage, hypertension, hypotension
5.5 Tolerance and Tachyphylaxis
Use of another dexmedetomidine product administered intravenously beyond 24 hours has been associated with tolerance and tachyphylaxis and a dose-related increase in adverse reactions. The effectiveness of IGALMI beyond 24 hours after the first dose has not been established. There may be a risk of tolerance and tachyphylaxis if IGALMI is administered at higher doses or for a longer duration than were evaluated, or in a manner other than indicated [see Dosage and Administration (2.2) and Drug Abuse and Dependence (9.3)].
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
The safety of IGALMI was evaluated in 507 adult patients with agitation associated with schizophrenia (N=255) or bipolar I or II disorder (N=252) in two randomized, placebo-controlled studies (Studies 1 and 2) [see Clinical Studies (14)]. In both studies, patients were admitted to a clinical research unit or a hospital and remained under medical supervision for at least 24 hours following treatment. Patients were 18 to 71 years of age (mean age was 46 years old); 45% were female and 55% were male; 66% were Black, 31% were White, 2% were multiracial, and 1% were other.
In these studies, patients received an initial dose of IGALMI 180 mcg (N=252), IGALMI 120 mcg (N=255), or placebo (N=252). Patients who were hemodynamically stable (i.e., those with systolic blood pressure (SBP) > 90 mmHg, diastolic blood pressure (DBP) > 60 mmHg, and heart rate (HR) > 60 beats per minute) and without orthostatic hypotension (i.e., reduction in SBP < 20 mmHg or DBP < 10 mmHg upon standing) were eligible for an additional dose after 2 hours. An additional half dose (90 mcg, 60 mcg, or placebo) was given to 7.1% (18/252), 22.7% (58/255) and 44.0% (111/252) of patients in the IGALMI 180 mcg, IGALMI 120 mcg or placebo arms, respectively. After at least an additional 2 hours, an additional second half dose (total IGALMI dose of 360 mcg, total IGALMI dose of 240 mcg, or placebo, respectively) was given to 3.2% (8/252), 9.4% (24/255), and 21.0% (53/252) of patients in the IGALMI 180 mcg, IGALMI 120 mcg or placebo arms, respectively.
In these studies, one patient discontinued treatment due to an adverse reaction of oropharyngeal pain.
The most common adverse reactions (incidence ≥ 5% and at least twice the rate of placebo) were: somnolence, oral paresthesia or oral hypoesthesia, dizziness, dry mouth, hypotension, and orthostatic hypotension.
Table 2 presents the adverse reactions that occurred in IGALMI-treated patients at a rate of at least 2% and at a higher rate than in placebo-treated patients in Studies 1 and 2.
| Adverse Reaction | IGALMI 180 mcg N=252 % |
IGALMI 120 mcg N=255 % |
Placebo N=252 % |
|---|---|---|---|
| Somnolence Somnolence includes the terms fatigue and sluggishness
|
23 | 22 | 6 |
| Paresthesia oral or hypoesthesia oral | 7 | 6 | 1 |
| Dizziness | 6 | 4 | 1 |
| Hypotension | 5 | 5 | 0 |
| Orthostatic hypotension | 5 | 3 | <1 |
| Dry mouth | 4 | 7 | 1 |
| Nausea | 3 | 2 | 2 |
| Bradycardia | 2 | 2 | 0 |
| Abdominal discomfort Abdominal discomfort includes dyspepsia, gastroesophageal reflux disease
|
2 | 0 | 1 |
5.4 Risk of Withdrawal Reactions
Symptoms of withdrawal have been observed after treatment with another dexmedetomidine product administered as a continuous IV infusion in the ICU for another indication. In this study, 12 (5%) adult patients who received intravenous dexmedetomidine up to 7 days (regardless of dose) experienced at least 1 event related to withdrawal within the first 24 hours after discontinuing dexmedetomidine and 7 (3%) adult patients who received intravenous dexmedetomidine experienced at least 1 event related with withdrawal 24 to 48 hours after discontinuing dexmedetomidine. The most common withdrawal reactions were nausea, vomiting, and agitation. In these subjects, tachycardia and hypertension requiring intervention occurred at a frequency of <5% in the 48 hours following intravenous dexmedetomidine discontinuation. Withdrawal symptoms have been reported in pediatric patients who received another dexmedetomidine product intravenously for another indication. IGALMI is not approved for pediatric use.
Withdrawal symptoms reported in published literature include cardiovascular symptoms, such as hypertension, tachycardia, arrhythmias, and in severe cases, hypertensive crisis; neurological symptoms, such as agitation, anxiety, tremors, seizures, and altered mental status; and autonomic symptoms such as diaphoresis, nausea, vomiting, hyperthermia, and mydriasis. In a randomized, double-blind, placebo-controlled, multiple ascending dose study assessing IGALMI at doses up to 120 mcg per day administered over 7 days in healthy volunteers, elevated heart rate and systolic blood pressure and vomiting occurred at a higher frequency upon abrupt discontinuation of 120-mcg doses of IGALMI as compared to placebo. There may be a risk of physical dependence and a withdrawal syndrome if IGALMI is used in a manner other than indicated [see Dosage and Administration (2.2) and Drug Abuse and Dependence (9.3)].
7.1 Drugs That Prolong the Qt Interval (7.1 Drugs that Prolong the QT Interval)
Concomitant use of drugs that prolong the QT interval may add to the QT-prolonging effects of IGALMI and increase the risk of cardiac arrhythmia. Avoid the use of IGALMI in combination with other drugs known to prolong the QT interval [see Warnings and Precautions (5.2)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Twice daily sublingual administration of 120 to 320 mcg/day of dexmedetomidine to dogs for 28 days caused decreased heart rate and moderate sedation up to 3.5 hours post dose. A single male dog (out of 32 treated dogs) dosed 320 mcg/day (equivalent to the MRHD of 360 mcg/day) exhibited inflammation, necrosis, myofiber degeneration, and hemorrhage at the sublingual treatment site. No adverse effects were noted at 240 mcg/day (less than the MRHD of 360 mcg/day).
2.3 Preparation and Administration Instructions
Keep IGALMI in the foil pouch until ready to administer. IGALMI should be immediately administered once the pouch is opened and the dose prepared.
Prepare and administer IGALMI under the supervision of a healthcare provider as follows:
| Healthcare Professional: Prepare IGALMI Dose for Patient | |||
| 1 | Open the sealed foil pouch by tearing straight across at the notch. |
|
|
|
|||
| 2a | Remove the film from the pouch with clean dry hands. |
|
|
| 2b | Cut the film in half between the dots with clean, dry scissors. |
|
|
| 2c | Discard unused half in waste container. | ||
| 2d | Place the half film for administration to the patient back into the pouch. |
|
|
| 3 | Immediately give the pouch to the patient. |
|
|
| 4 | Instruct patient to remove the film from the pouch with clean dry hands. |
|
|
| 5 |
For sublingual administration: Instruct patient to place film under the tongue. The film will stick in place. Note: Patient may not eat or drink for 15 minutes after sublingual administration. |
|
|
|
For buccal administration: Instruct patient to place film behind lower lip. The film will stick in place. Note: Patient may not eat or drink for one hour after buccal administration. |
|
||
| 6 | Instruct patient to:
|
7.2 Anesthetics, Sedatives, Hypnotics, and Opioids
Concomitant use of IGALMI with anesthetics, sedatives, hypnotics, or opioids is likely to lead to enhanced CNS depressant effects. Specific studies with another dexmedetomidine product given intravenously have confirmed these effects with sevoflurane, isoflurane, propofol, alfentanil, and midazolam. Due to possible enhanced CNS effects when given concomitantly with IGALMI, consider a reduction in dosage of IGALMI or the concomitant anesthetic, sedative, hypnotic, or opioid.
Principal Display Panel 120 Mcg Film Pouch Carton (PRINCIPAL DISPLAY PANEL - 120 mcg Film Pouch Carton)
Rx Only
Igalmi™
(dexmedetomidine) sublingual film
For sublingual or buccal use
Keep IGALMI in pouch until administration
Do not chew or swallow the film
120 mcg
10 pouches
Each containing
1 film
NDC 81092-1120-1
Principal Display Panel 180 Mcg Film Pouch Carton (PRINCIPAL DISPLAY PANEL - 180 mcg Film Pouch Carton)
Rx Only
Igalmi™
(dexmedetomidine) sublingual film
For sublingual or buccal use
Keep IGALMI in pouch until administration
Do not chew or swallow the film
180 mcg
10 pouches
Each containing
1 film
NDC 81092-1180-1
5.1 Hypotension, Orthostatic Hypotension, and Bradycardia
IGALMI causes dose-dependent hypotension, orthostatic hypotension, and bradycardia. In clinical studies, 18%, 16%, and 9% of patients treated with 180 mcg of IGALMI, 120 mcg of IGALMI, and placebo, respectively, experienced orthostatic hypotension (defined as SBP decrease ≥ 20 mmHg or DBP decrease ≥ 10 mmHg after 1, 3, or 5 minutes of standing) at 2 hours post-dose. In those studies, 7%, 6%, and 1% of patients treated with 180 mcg of IGALMI, 120 mcg of IGALMI, and placebo, respectively, experienced HR ≤ 50 beats per minute within 2 hours of dosing [see Adverse Reactions (6.1)]. In clinical studies with IGALMI, patients were excluded if they had treatment with alpha-1 noradrenergic blockers, benzodiazepines, other hypnotics or antipsychotic drugs four hours prior to study drug administration; had a history of syncope or syncopal attacks; SBP < 110 mmHg; DBP < 70 mmHg; HR < 55 beats per minute; or had evidence of hypovolemia or orthostatic hypotension.
Reports of hypotension and bradycardia, including some resulting in fatalities, have been associated with the use of another dexmedetomidine product given intravenously (IGALMI is for sublingual or buccal use and is not approved for intravenous use). Clinically significant episodes of bradycardia and sinus arrest have been reported after administration of this other dexmedetomidine product to young, healthy adult volunteers with high vagal tone and when this product was given by rapid intravenous or bolus administration.
Because IGALMI decreases sympathetic nervous system activity, hypotension and/or bradycardia may be more pronounced in patients with hypovolemia, diabetes mellitus, or chronic hypertension, and in geriatric patients [see Dosage and Administration (2.2) and Use in Specific Populations (8.5)].
Avoid use of IGALMI in patients with hypotension, orthostatic hypotension, advanced heart block, severe ventricular dysfunction, or history of syncope. After IGALMI administration, patients should be adequately hydrated and should sit or lie down until vital signs are within normal range. If a patient is unable to remain seated or lying down, precautions should be taken to reduce the risk of falls. Ensure that a patient is alert and not experiencing orthostatic hypotension or symptomatic hypotension prior to allowing them to resume ambulation [see Dosage and Administration (2.1)].
2.1 Important Recommendations Prior to Initiating Igalmi and During Therapy (2.1 Important Recommendations Prior to Initiating IGALMI and During Therapy)
IGALMI should be administered under the supervision of a healthcare provider. A healthcare provider should monitor vital signs and alertness after IGALMI administration to prevent falls and syncope [see Warnings and Precautions (5.5)].
IGALMI is for sublingual or buccal administration. Do not chew or swallow IGALMI. Do not eat or drink for at least 15 minutes after sublingual administration, or at least one hour after buccal administration.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:32.692208 · Updated: 2026-03-14T22:37:51.668409