These Highlights Do Not Include All The Information Needed To Use Dupixent Safely And Effectively. See Full Prescribing Information For Dupixent.
595f437d-2729-40bb-9c62-c8ece1f82780
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage, Chronic Spontaneous Urticaria ( 1.7 ) 04/2025 Indications and Usage, Bullous Pemphigoid ( 1.8 ) 06/2025 Indications and Usage, Allergic Fungal Rhinosinusitis ( 1.9 ) 02/2026 Dosage and Administration, Chronic Spontaneous Urticaria ( 2.9 ) 04/2025 Dosage and Administration, Bullous Pemphigoid ( 2.10 ) 06/2025 Dosage and Administration, Allergic Fungal Rhinosinusitis ( 2.11 ) 02/2026 Dosage and Administration, Missed Doses ( 2.12 ) 06/2025 Warnings and Precautions, Hypersensitivity ( 5.1 ) 06/2025 Warnings and Precautions, Conjunctivitis and Keratitis ( 5.2 ) 02/2026 Warnings and Precautions ( 5.2 , 5.3 , 5.7 , 5.8 ) 04/2025
Indications and Usage
DUPIXENT is an interleukin-4 receptor alpha antagonist indicated: Atopic Dermatitis for the treatment of adult and pediatric patients aged 6 months and older with moderate-to-severe AD whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids. ( 1.1 ) Asthma as an add-on maintenance treatment of adult and pediatric patients aged 6 years and older with moderate-to-severe asthma characterized by an eosinophilic phenotype or with oral corticosteroid dependent asthma. ( 1.2 ) Limitations of Use: Not for the relief of acute bronchospasm or status asthmaticus. ( 1.2 ) Chronic Rhinosinusitis with Nasal Polyps as an add-on maintenance treatment in adult and pediatric patients aged 12 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP). ( 1.3 ) Eosinophilic Esophagitis for the treatment of adult and pediatric patients aged 1 year and older, weighing at least 15 kg, with eosinophilic esophagitis (EoE). ( 1.4 ) Prurigo Nodularis for the treatment of adult patients with prurigo nodularis (PN). ( 1.5 ) Chronic Obstructive Pulmonary Disease as an add-on maintenance treatment of adult patients with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype. ( 1.6 ) Limitations of Use: Not for the relief of acute bronchospasm. ( 1.6 ) Chronic Spontaneous Urticaria for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment. ( 1.7 ) Limitations of Use: Not indicated for other forms of urticaria. ( 1.7 ) Bullous Pemphigoid for the treatment of adult patients with bullous pemphigoid (BP). ( 1.8 ) Allergic Fungal Rhinosinusitis for the treatment of adult and pediatric patients aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery. ( 1.9 )
Dosage and Administration
DUPIXENT is administered by subcutaneous injection. ( 2.1 ) Atopic Dermatitis Dosage in Adults ( 2.3 ): Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W). Dosage in Pediatric Patients 6 Months to 5 Years of Age ( 2.3 ): Body Weight Initial and Subsequent Dosage 5 to less than 15 kg 200 mg (one 200 mg injection) every 4 weeks (Q4W) 15 to less than 30 kg 300 mg (one 300 mg injection) every 4 weeks (Q4W) Dosage in Pediatric Patients 6 Years of Age and Older ( 2.3 ): Body Weight Initial Loading Dose Subsequent Dosage Q2W – every 2 weeks; Q4W – every 4 weeks 15 to less than 30 kg 600 mg (two 300 mg injections) 300 mg Q4W 30 to less than 60 kg 400 mg (two 200 mg injections) 200 mg Q2W 60 kg or more 600 mg (two 300 mg injections) 300 mg Q2W Asthma Dosage in Adult and Pediatric Patients 12 Years and Older ( 2.4 ): Initial Loading Dose Subsequent Dosage 400 mg (two 200 mg injections) 200 mg every 2 weeks (Q2W) Or 600 mg (two 300 mg injections) 300 mg every 2 weeks (Q2W) Dosage for patients with oral corticosteroid-dependent asthma or with co-morbid moderate-to-severe AD, CRSwNP, or AFRS For pediatric patients 12 to 17 years of age (≥60 kg) and adults with AFRS 600 mg (two 300 mg injections) 300 mg every 2 weeks (Q2W) Dosage in Pediatric Patients 6 to 11 Years of Age ( 2.4 ): Body Weight Initial Dose and Subsequent Dosage 15 to less than 30 kg 300 mg every 4 weeks (Q4W) ≥30 kg 200 mg every 2 weeks (Q2W) For pediatric patients 6 to 11 years old with asthma and co-morbid moderate-to-severe atopic dermatitis, follow the recommended dosage as per Table 2 which includes an initial loading dose. ( 2.3 ) Chronic Rhinosinusitis with Nasal Polyps ( 2.5 ): Recommended dosage for adult and pediatric patients 12 years of age and older is 300 mg given every 2 weeks (Q2W). Eosinophilic Esophagitis ( 2.6 ): Body Weight Recommended Dosage in Adult and Pediatric Patients 1 Year and Older, Weighing At Least 15 kg 15 to less than 30 kg 200 mg every 2 weeks (Q2W) 30 to less than 40 kg 300 mg every 2 weeks (Q2W) 40 kg or more 300 mg every week (QW) Prurigo Nodularis ( 2.7 ): Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W). Chronic Obstructive Pulmonary Disease ( 2.8 ): Recommended dosage for adult patients is 300 mg given every 2 weeks (Q2W). Chronic Spontaneous Urticaria Dosage in Adults ( 2.9 ): Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W). Dosage in Pediatric Patients 12 to 17 Years of Age ( 2.9 ): Body Weight Initial Loading Dose Subsequent Dosage 30 to less than 60 kg 400 mg (two 200 mg injections) 200 mg Q2W 60 kg or more 600 mg (two 300 mg injections) 300 mg Q2W Bullous Pemphigoid ( 2.10 ): Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every other week (Q2W). Use DUPIXENT in combination with a tapering course of oral corticosteroids. Allergic Fungal Rhinosinusitis Dosage in Adults ( 2.11 ): Recommended dosage is 300 mg given every 2 weeks (Q2W). Dosage in Pediatric Patients 6 to 17 Years of Age ( 2.11 ) : Body Weight Recommended Dosage a Q2W – every 2 weeks; Q4W – every 4 weeks 15 to less than 30 kg 300 mg Q4W 30 to less than 60 kg 200 mg Q2W 60 kg or more 300 mg Q2W
Warnings and Precautions
Hypersensitivity: Hypersensitivity reactions including anaphylaxis, acute generalized exanthematous pustulosis (AGEP), serum sickness, angioedema, urticaria, rash, erythema nodosum, and erythema multiforme have occurred. Discontinue DUPIXENT in the event of a hypersensitivity reaction. ( 5.1 ) Conjunctivitis and Keratitis: Advise patients to report new onset or worsening eye symptoms to their healthcare provider. Consider ophthalmological examination, as appropriate. ( 5.2 ) Eosinophilic Conditions: Be alert to vasculitic rash, worsening pulmonary symptoms, cardiac complications, kidney injury and/or neuropathy, especially upon reduction of oral corticosteroids. ( 5.3 ) Reduction of Corticosteroid Dosage: Do not discontinue systemic, topical, or inhaled corticosteroids abruptly upon initiation of DUPIXENT. Decrease steroids gradually, if appropriate. ( 5.5 ) Psoriasis: Advise patients to report new-onset psoriasis symptoms to their healthcare provider. If symptoms persist or worsen, consider dermatologic evaluation and/or discontinuation of DUPIXENT. ( 5.7 ) Arthralgia and Psoriatic Arthritis: Advise patients to report new onset joint symptoms to their healthcare provider. If symptoms persist or worsen, consider rheumatological evaluation and/or discontinuation of DUPIXENT. ( 5.8 ) Parasitic (Helminth) Infections: Treat pre-existing helminth infections before initiating DUPIXENT. If patients become infected while receiving DUPIXENT and do not respond to anti-helminth treatment, discontinue DUPIXENT until the infection resolves. ( 5.9) Vaccinations: Avoid use of live vaccines. ( 5.10 )
Contraindications
DUPIXENT is contraindicated in patients who have known hypersensitivity to dupilumab or any excipients of DUPIXENT [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity [see Warnings and Precautions (5.1) ] Conjunctivitis and Keratitis [see Warnings and Precautions (5.2) ] Psoriasis [see Warnings and Precautions (5.7) ] Arthralgia and Psoriatic Arthritis [see Warnings and Precautions (5.8) ] Parasitic (Helminth) Infections [see Warnings and Precautions (5.9) ]
Medication Information
Warnings and Precautions
Hypersensitivity: Hypersensitivity reactions including anaphylaxis, acute generalized exanthematous pustulosis (AGEP), serum sickness, angioedema, urticaria, rash, erythema nodosum, and erythema multiforme have occurred. Discontinue DUPIXENT in the event of a hypersensitivity reaction. ( 5.1 ) Conjunctivitis and Keratitis: Advise patients to report new onset or worsening eye symptoms to their healthcare provider. Consider ophthalmological examination, as appropriate. ( 5.2 ) Eosinophilic Conditions: Be alert to vasculitic rash, worsening pulmonary symptoms, cardiac complications, kidney injury and/or neuropathy, especially upon reduction of oral corticosteroids. ( 5.3 ) Reduction of Corticosteroid Dosage: Do not discontinue systemic, topical, or inhaled corticosteroids abruptly upon initiation of DUPIXENT. Decrease steroids gradually, if appropriate. ( 5.5 ) Psoriasis: Advise patients to report new-onset psoriasis symptoms to their healthcare provider. If symptoms persist or worsen, consider dermatologic evaluation and/or discontinuation of DUPIXENT. ( 5.7 ) Arthralgia and Psoriatic Arthritis: Advise patients to report new onset joint symptoms to their healthcare provider. If symptoms persist or worsen, consider rheumatological evaluation and/or discontinuation of DUPIXENT. ( 5.8 ) Parasitic (Helminth) Infections: Treat pre-existing helminth infections before initiating DUPIXENT. If patients become infected while receiving DUPIXENT and do not respond to anti-helminth treatment, discontinue DUPIXENT until the infection resolves. ( 5.9) Vaccinations: Avoid use of live vaccines. ( 5.10 )
Indications and Usage
DUPIXENT is an interleukin-4 receptor alpha antagonist indicated: Atopic Dermatitis for the treatment of adult and pediatric patients aged 6 months and older with moderate-to-severe AD whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids. ( 1.1 ) Asthma as an add-on maintenance treatment of adult and pediatric patients aged 6 years and older with moderate-to-severe asthma characterized by an eosinophilic phenotype or with oral corticosteroid dependent asthma. ( 1.2 ) Limitations of Use: Not for the relief of acute bronchospasm or status asthmaticus. ( 1.2 ) Chronic Rhinosinusitis with Nasal Polyps as an add-on maintenance treatment in adult and pediatric patients aged 12 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP). ( 1.3 ) Eosinophilic Esophagitis for the treatment of adult and pediatric patients aged 1 year and older, weighing at least 15 kg, with eosinophilic esophagitis (EoE). ( 1.4 ) Prurigo Nodularis for the treatment of adult patients with prurigo nodularis (PN). ( 1.5 ) Chronic Obstructive Pulmonary Disease as an add-on maintenance treatment of adult patients with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype. ( 1.6 ) Limitations of Use: Not for the relief of acute bronchospasm. ( 1.6 ) Chronic Spontaneous Urticaria for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment. ( 1.7 ) Limitations of Use: Not indicated for other forms of urticaria. ( 1.7 ) Bullous Pemphigoid for the treatment of adult patients with bullous pemphigoid (BP). ( 1.8 ) Allergic Fungal Rhinosinusitis for the treatment of adult and pediatric patients aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery. ( 1.9 )
Dosage and Administration
DUPIXENT is administered by subcutaneous injection. ( 2.1 ) Atopic Dermatitis Dosage in Adults ( 2.3 ): Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W). Dosage in Pediatric Patients 6 Months to 5 Years of Age ( 2.3 ): Body Weight Initial and Subsequent Dosage 5 to less than 15 kg 200 mg (one 200 mg injection) every 4 weeks (Q4W) 15 to less than 30 kg 300 mg (one 300 mg injection) every 4 weeks (Q4W) Dosage in Pediatric Patients 6 Years of Age and Older ( 2.3 ): Body Weight Initial Loading Dose Subsequent Dosage Q2W – every 2 weeks; Q4W – every 4 weeks 15 to less than 30 kg 600 mg (two 300 mg injections) 300 mg Q4W 30 to less than 60 kg 400 mg (two 200 mg injections) 200 mg Q2W 60 kg or more 600 mg (two 300 mg injections) 300 mg Q2W Asthma Dosage in Adult and Pediatric Patients 12 Years and Older ( 2.4 ): Initial Loading Dose Subsequent Dosage 400 mg (two 200 mg injections) 200 mg every 2 weeks (Q2W) Or 600 mg (two 300 mg injections) 300 mg every 2 weeks (Q2W) Dosage for patients with oral corticosteroid-dependent asthma or with co-morbid moderate-to-severe AD, CRSwNP, or AFRS For pediatric patients 12 to 17 years of age (≥60 kg) and adults with AFRS 600 mg (two 300 mg injections) 300 mg every 2 weeks (Q2W) Dosage in Pediatric Patients 6 to 11 Years of Age ( 2.4 ): Body Weight Initial Dose and Subsequent Dosage 15 to less than 30 kg 300 mg every 4 weeks (Q4W) ≥30 kg 200 mg every 2 weeks (Q2W) For pediatric patients 6 to 11 years old with asthma and co-morbid moderate-to-severe atopic dermatitis, follow the recommended dosage as per Table 2 which includes an initial loading dose. ( 2.3 ) Chronic Rhinosinusitis with Nasal Polyps ( 2.5 ): Recommended dosage for adult and pediatric patients 12 years of age and older is 300 mg given every 2 weeks (Q2W). Eosinophilic Esophagitis ( 2.6 ): Body Weight Recommended Dosage in Adult and Pediatric Patients 1 Year and Older, Weighing At Least 15 kg 15 to less than 30 kg 200 mg every 2 weeks (Q2W) 30 to less than 40 kg 300 mg every 2 weeks (Q2W) 40 kg or more 300 mg every week (QW) Prurigo Nodularis ( 2.7 ): Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W). Chronic Obstructive Pulmonary Disease ( 2.8 ): Recommended dosage for adult patients is 300 mg given every 2 weeks (Q2W). Chronic Spontaneous Urticaria Dosage in Adults ( 2.9 ): Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W). Dosage in Pediatric Patients 12 to 17 Years of Age ( 2.9 ): Body Weight Initial Loading Dose Subsequent Dosage 30 to less than 60 kg 400 mg (two 200 mg injections) 200 mg Q2W 60 kg or more 600 mg (two 300 mg injections) 300 mg Q2W Bullous Pemphigoid ( 2.10 ): Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every other week (Q2W). Use DUPIXENT in combination with a tapering course of oral corticosteroids. Allergic Fungal Rhinosinusitis Dosage in Adults ( 2.11 ): Recommended dosage is 300 mg given every 2 weeks (Q2W). Dosage in Pediatric Patients 6 to 17 Years of Age ( 2.11 ) : Body Weight Recommended Dosage a Q2W – every 2 weeks; Q4W – every 4 weeks 15 to less than 30 kg 300 mg Q4W 30 to less than 60 kg 200 mg Q2W 60 kg or more 300 mg Q2W
Contraindications
DUPIXENT is contraindicated in patients who have known hypersensitivity to dupilumab or any excipients of DUPIXENT [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity [see Warnings and Precautions (5.1) ] Conjunctivitis and Keratitis [see Warnings and Precautions (5.2) ] Psoriasis [see Warnings and Precautions (5.7) ] Arthralgia and Psoriatic Arthritis [see Warnings and Precautions (5.8) ] Parasitic (Helminth) Infections [see Warnings and Precautions (5.9) ]
Description
Indications and Usage, Chronic Spontaneous Urticaria ( 1.7 ) 04/2025 Indications and Usage, Bullous Pemphigoid ( 1.8 ) 06/2025 Indications and Usage, Allergic Fungal Rhinosinusitis ( 1.9 ) 02/2026 Dosage and Administration, Chronic Spontaneous Urticaria ( 2.9 ) 04/2025 Dosage and Administration, Bullous Pemphigoid ( 2.10 ) 06/2025 Dosage and Administration, Allergic Fungal Rhinosinusitis ( 2.11 ) 02/2026 Dosage and Administration, Missed Doses ( 2.12 ) 06/2025 Warnings and Precautions, Hypersensitivity ( 5.1 ) 06/2025 Warnings and Precautions, Conjunctivitis and Keratitis ( 5.2 ) 02/2026 Warnings and Precautions ( 5.2 , 5.3 , 5.7 , 5.8 ) 04/2025
Section 42229-5
Limitations of Use
DUPIXENT is not indicated for the relief of acute bronchospasm or status asthmaticus.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: February 2026 | ||
|
PATIENT INFORMATION DUPIXENT® (DU-pix-ent) (dupilumab) injection, for subcutaneous use |
|||
|
What is DUPIXENT?
DUPIXENT is a prescription medicine used: |
|||
|
|||
|
DUPIXENT is not used to relieve sudden breathing problems and will not replace an inhaled rescue medicine or to treat any other forms of hives (urticaria). DUPIXENT works by blocking two proteins that contribute to a type of inflammation that plays a major role in AD, asthma, CRSwNP, EoE, PN, COPD, CSU, BP, and AFRS. It is not known if DUPIXENT is safe and effective in children with: |
|||
|
|
||
| Do not use DUPIXENT if you are allergic to dupilumab or to any of the ingredients in DUPIXENT. See the end of this Patient Information leaflet for a complete list of ingredients in DUPIXENT. | |||
Before using DUPIXENT, tell your healthcare provider about all your medical conditions, including if you:
Especially tell your healthcare provider if you:
|
|||
How should I use DUPIXENT?
|
|||
|
What are the possible side effects of DUPIXENT? DUPIXENT can cause serious side effects, including:
|
|||
|
|
|
|
|
|||
|
|
||
|
|||
|
|
||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of DUPIXENT. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store DUPIXENT?
|
|||
|
General information about the safe and effective use of DUPIXENT.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DUPIXENT for a condition for which it was not prescribed. Do not give DUPIXENT to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DUPIXENT that is written for health professionals. |
|||
|
What are the ingredients in DUPIXENT?
Active ingredient: dupilumab Inactive ingredients: L-arginine hydrochloride, L-histidine, polysorbate 80, sodium acetate, sucrose, and water for injection. Manufactured by: Regeneron Pharmaceuticals, Inc., Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Morristown, NJ 07960) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT® is a registered trademark of Sanofi Biotechnology / © 2026 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. For more information about DUPIXENT, go to www.DUPIXENT.com or call 1-844-DUPIXENT (1-844-387-4936). |
Section 43683-2
| Indications and Usage, Chronic Spontaneous Urticaria (1.7) | 04/2025 |
| Indications and Usage, Bullous Pemphigoid (1.8) | 06/2025 |
| Indications and Usage, Allergic Fungal Rhinosinusitis (1.9) | 02/2026 |
| Dosage and Administration, Chronic Spontaneous Urticaria (2.9) | 04/2025 |
| Dosage and Administration, Bullous Pemphigoid (2.10) | 06/2025 |
| Dosage and Administration, Allergic Fungal Rhinosinusitis (2.11) | 02/2026 |
| Dosage and Administration, Missed Doses (2.12) | 06/2025 |
| Warnings and Precautions, Hypersensitivity (5.1) | 06/2025 |
| Warnings and Precautions, Conjunctivitis and Keratitis (5.2) | 02/2026 |
| Warnings and Precautions (5.2, 5.3, 5.7, 5.8) | 04/2025 |
Section 44425-7
Storage and Handling
DUPIXENT is sterile and preservative-free. Discard any unused portion.
Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light.
If necessary, DUPIXENT may be kept at room temperature up to 25°C (77°F) for a maximum of 14 days. Do not store above 25°C (77°F). After removal from the refrigerator, DUPIXENT must be used within 14 days or discarded.
Do not expose DUPIXENT to heat or direct sunlight.
Do NOT freeze. Do NOT shake.
Section 59845-8
Instructions for Use
DUPIXENT® (DU-pix-ent)
(dupilumab)
injection, for subcutaneous use
Single-Dose Pre-filled Pen (300 mg/2 mL)
This Instructions for Use contains information on how to inject DUPIXENT.
Read this Instructions for Use before using the DUPIXENT Pre-filled Pen. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself for the first time. Keep this Instructions for Use. Call your healthcare provider if you have any questions.
This DUPIXENT Pre-filled Pen is only for use in adults and children aged 2 years and older.
This DUPIXENT Pre-filled Pen is a single-dose device. It contains 300 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Pre-filled Pen are shown below:
Important Information
|
Storing DUPIXENT
|
| A. Get ready to inject | |
|---|---|
| A1. Gather supplies | |
| Find a clean, flat work surface. Make sure you have the following supplies: |
|
| A2. Check the Pen | |
|
|
| A3. Look at the Label | |
|
|
|
| A4. Check the Medicine | |
|---|---|
|
| A5. Wait 45 minutes | |
|---|---|
|
| B. Choose and prepare your injection site | |
|---|---|
| B1. Wash your hands well with soap and water | |
| B2. Choose an injection site | |
|
|
| B3. Prepare the injection site | |
|---|---|
|
|
| C. Give the injection | |
| C1. Remove Green Cap | |
|
|
| C2. Pinch Skin and Place | ||
|---|---|---|
Pinch the skin before and during the injection.
|
||
| C3. Press down ➜ Watch Window turn fully yellow ➜ Then count to 5 | ||
Press and hold the DUPIXENT Pre-filled Pen firmly against the skin until you cannot see the Yellow Needle Cover.
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and watch the window:
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and count to 5 to make sure you get your full dose. |
| Pinching of the skin is not needed for adults and children aged 12 years and older. | |
|---|---|
| C4. Remove | |
|
| D. Dispose of used DUPIXENT Pre-filled Pen | |
|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: July 2023 |
|
How to Dispose of (throw away) DUPIXENT Pre-filled Pen:
Put the used DUPIXENT Pre-filled Pens and Green Caps in a FDA-cleared sharps disposal container right away after use. Do not dispose of (throw away) DUPIXENT Pre-filled Pens and Green Caps in your household trash. If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Keep your sharps disposal container out of the reach of children. |
|
|
|
|
|
Manufactured by: Regeneron Pharmaceuticals, Inc. Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT ® is a registered trademark of Sanofi Biotechnology © 2023 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. |
1.2 Asthma
DUPIXENT is indicated as an add-on maintenance treatment of adult and pediatric patients aged 6 years and older with moderate-to-severe asthma characterized by an eosinophilic phenotype or with oral corticosteroid dependent asthma.
14.2 Asthma
The asthma development program for patients aged 12 years and older included three randomized, double-blind, placebo-controlled, parallel-group, multicenter trials (DRI12544 (NCT01854047), QUEST (NCT02414854), and VENTURE (NCT02528214)) of 24 to 52 weeks in treatment duration which enrolled a total of 2888 subjects. Subjects enrolled in DRI12544 and QUEST were required to have a history of 1 or more asthma exacerbations that required treatment with systemic corticosteroids or emergency department visit or hospitalization for the treatment of asthma in the year prior to trial entry. Subjects enrolled in VENTURE required dependence on daily oral corticosteroids in addition to regular use of high-dose inhaled corticosteroids plus an additional controller(s). In all 3 trials, subjects were enrolled without requiring a minimum baseline blood eosinophil count. In QUEST and VENTURE, subjects with screening blood eosinophil level of >1500 cells/mcL (<1.3%) were excluded. DUPIXENT was administered as add-on to background asthma treatment. Subjects continued background asthma therapy throughout the duration of the studies, except in VENTURE in which OCS dose was tapered as described below.
10 Overdosage
There is no specific treatment for DUPIXENT overdose. In the event of overdosage, contact Poison Control (1-800-222-1222) for the latest recommendations and monitor the patient for any signs or symptoms of adverse reactions and institute appropriate symptomatic treatment immediately.
5.7 Psoriasis
Cases of new-onset psoriasis have been reported with the use of DUPIXENT for the treatment of atopic dermatitis and asthma, including in patients without a family history of psoriasis. In postmarketing reports, onset of psoriasis varied from weeks to months after the first dose of DUPIXENT and resulted in partial or complete resolution of psoriasis with discontinuation of dupilumab, with or without use of supplemental treatment for psoriasis (topical or systemic). Those who continued on dupilumab received supplemental treatment for psoriasis to improve associated symptoms. Advise patients to report new-onset psoriasis symptoms to their healthcare provider. If symptoms persist or worsen, consider dermatologic evaluation and/or discontinuation of DUPIXENT.
11 Description
Dupilumab, an interleukin-4 receptor alpha antagonist, is a human monoclonal antibody of the IgG4 subclass that binds to the IL-4Rα subunit and inhibits IL-4 and IL-13 signaling. Dupilumab has an approximate molecular weight of 147 kDa.
Dupilumab is produced by recombinant DNA technology in Chinese Hamster Ovary cell suspension culture.
DUPIXENT (dupilumab) Injection is supplied as a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution for subcutaneous injection. DUPIXENT is provided as either a single-dose pre-filled syringe with needle shield or a single-dose pre-filled pen in a siliconized Type-1 clear glass syringe. The needle cap is not made with natural rubber latex.
Each 300 mg pre-filled syringe or pre-filled pen delivers 300 mg dupilumab in 2 mL which also contains L-arginine hydrochloride (10.5 mg), L-histidine (6.2 mg), polysorbate 80 (4 mg), sodium acetate (2 mg), sucrose (100 mg), and water for injection, pH 5.9.
Each 200 mg pre-filled syringe or pre-filled pen delivers 200 mg dupilumab in 1.14 mL which also contains L-arginine hydrochloride (12 mg), L-histidine (3.5 mg), polysorbate 80 (2.3 mg), sodium acetate (1.2 mg), sucrose (57 mg), and water for injection, pH 5.9.
2.12 Missed Doses
If a weekly dose is missed, administer the dose as soon as possible, and start a new weekly schedule from the date of the last administered dose.
If an every 2 week dose is missed, administer the injection within 7 days from the missed dose and then resume the patient's original schedule. If the missed dose is not administered within 7 days, administer the dose, starting a new schedule based on this date.
If an every 4 week dose is missed, administer the injection within 7 days from the missed dose and then resume the patient's original schedule. If the missed dose is not administered within 7 days, administer the dose, starting a new schedule based on this date.
5.10 Vaccinations
Consider completing all age-appropriate vaccinations as recommended by current immunization guidelines prior to initiating treatment with DUPIXENT. Avoid use of live vaccines during treatment with DUPIXENT. It is unknown if administration of live vaccines during DUPIXENT treatment will impact the safety or effectiveness of these vaccines. Limited data are available regarding coadministration of DUPIXENT with non-live vaccines [see Clinical Pharmacology (12.2)].
8.5 Geriatric Use
Of the 1539 subjects with AD exposed to DUPIXENT in a dose-ranging study and placebo-controlled trials, 70 subjects were 65 years or older. Clinical trials of DUPIXENT in AD did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger subjects.
Of the 1977 subjects with asthma exposed to DUPIXENT, a total of 240 subjects were 65 years or older. Efficacy and safety in this age group were similar to the overall study population.
Of the 440 subjects with CRSwNP exposed to DUPIXENT, a total of 79 subjects were 65 years or older. Efficacy and safety in this age group were similar to the overall study population.
Clinical studies of DUPIXENT in EoE did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger adult subjects.
Of the 152 subjects with PN exposed to DUPIXENT, a total of 37 were 65 years or older including 8 subjects 75 years or older. Clinical trials did not include a sufficient number of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
Of the 1874 subjects with COPD randomized in clinical trials of DUPIXENT, a total of 1072 were 65 years or older, while 244 subjects were 75 years or older. No overall differences in safety or effectiveness of DUPIXENT have been observed between subjects 65 years of age and older and younger adult subjects.
Of the 198 subjects with CSU exposed to DUPIXENT, a total of 30 subjects were 65 years or older, including 7 subjects 75 years or older. Efficacy and safety in subjects 65 years or older were similar to the overall study population.
Of the 53 subjects with BP exposed to DUPIXENT, a total of 40 were 65 years or older, including 22 subjects 75 years or older. Ten percent of subjects aged 65 years and older treated with DUPIXENT had an adverse reaction of vision blurred compared to zero in younger adult subjects.
Clinical studies of DUPIXENT in AFRS did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the trials described below with the incidence of anti-drug antibodies in other trials, including those of DUPIXENT or of other dupilumab products.
The anti-drug antibody (ADA) and neutralizing antibody (NAb) incidence rates in subjects treated with DUPIXENT are presented in Table 16.
| Indication and Population | Dose and Duration of Treatment | Anti-drug Antibody (ADA) | Neutralizing Antibody NAb Neutralizing potential is assessed only for ADA-positive samples. The NAb incidence is reported as a percentage of subjects with positive ADA response.
|
|
|---|---|---|---|---|
| Positive ADA Positive ADA: Includes subjects with a treatment-emergent response, defined as a negative or missing result at baseline with at least 1 positive post-baseline result in the ADA assay, and subjects with a treatment-boosted response, defined as a post-baseline ADA-positive result that is ≥ 4-fold higher than baseline titer levels, when baseline results are positive
|
Persistent ADA Persistent ADA: A treatment-emergent ADA positive response with 2 or more consecutive ADA positive samples separated by a greater than 12-week period (>84 days), with no ADA negative result in between.
|
|||
| Atopic Dermatitis | ||||
| Adult | 300 mg Q2W for 52 weeks | 7/105 (7%) | 2/105 (2%) | 1/7 (14%) |
| 12 to 17 years of age | 300 mg or 200 mg Q2W for 16 weeks | 13/81 (16%) | 2/81 (3%) | 4/13 (31%) |
| 6 to 11 years of age | 200 mg Q2W or 300 mg Q4W for 16 weeks | 3/171 (2%) | 0/171 (0%) | 1/3 (33%) |
| 6 months to 5 years of age | 200 mg Q4W, or 300 mg Q4W for 16 weeks | 1/74 (1%) | 0/74 (0%) | 0/1 (0%) |
| Asthma | ||||
| Adult and 12 years of age or older | 300 mg Q2W for 52 weeks | 33/626 (5%) | 13/626 (2%) | 14/33 (42%) |
| 200 mg Q2W for 52 weeks | 58/625 (9%) | 26/625 (4%) | 27/58 (47%) | |
| 6 to 11 years of age | 100 mg Q2W or 200 mg Q2W up to 52 weeks | 17/269 (6%) | 9/269 (3%) | 6/17 (35%) |
| Chronic Rhinosinusitis with Nasal Polyps | ||||
| Adult | 300 mg Q2W for 52 weeks | 8/148 (5%) | 3/148 (2%) | 5/8 (63%) |
| Eosinophilic Esophagitis | ||||
| Adult and 12 years of age or older | 300 mg QW for 52 weeks | 1/108 (1%) | 0/108 (0%) | 0/1 (0%) |
| 1 to 11 years of age | 200 mg Q2W or 300 mg Q2W for 52 weeks Immunogenicity was reported on pooled data for pediatric subjects 1 to 11 years of age with EoE receiving SC dupilumab 100 mg Q2W (in subjects weighing ≥5 kg to <15kg), 200 mg Q2W (in subjects weighing ≥15 kg to <30 kg), 300 mg Q2W (in subjects weighing ≥30 kg to <60 kg), and 300 mg QW (in subjects weighing ≥60 kg).
|
1/37 (3%) | 0/37 (0%) | 0/1 (0%) One subject 1 to 11 years of age with EoE with pre-existing immunoreactivity that was positive for neutralizing antibodies was excluded from the NAb positive subjects.
|
| Prurigo Nodularis | ||||
| Adult | 300 mg Q2W for 24 weeks | 11/143 (8%) | 2/143 (1%) | 4/11 (36%) |
| Chronic Obstructive Pulmonary Disease | ||||
| Adult | 300 mg Q2W for 52 weeks | 78/923 (8%) | 24/922 (3%) | 28/78 (36%) |
| Chronic Spontaneous Urticaria | ||||
| Adult and 12 years of age or older | 200 mg Q2W or 300 mg Q2W through 24 weeks | 9/193 (5%) | 1/193 (1%) | 2/9 (22%) |
| Bullous Pemphigoid | ||||
| Adult | 300 mg Q2W up to 52 weeks | 2/52 (4%) | 0/52 (0%) | 2/2 (100%) |
| Allergic Fungal Rhinosinusitis | ||||
| Adult and 6 years of age or older | 300 mg Q2W or 200 mg Q2W for 52 weeks | 2/22 (9%) | 1/22 (4.5%) | 0/2 (0%) |
Two adult subjects with AD who experienced high titer antibody responses developed serum sickness or serum sickness-like reactions during DUPIXENT therapy [see Warnings and Precautions (5.1)].
The antibody titers detected in subjects who received DUPIXENT were mostly low. In subjects who received DUPIXENT, development of high titer antibodies to DUPIXENT was associated with lower serum dupilumab concentrations [see Clinical Pharmacology (12.3)].
4 Contraindications
DUPIXENT is contraindicated in patients who have known hypersensitivity to dupilumab or any excipients of DUPIXENT [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Conjunctivitis and Keratitis [see Warnings and Precautions (5.2)]
- Psoriasis [see Warnings and Precautions (5.7)]
- Arthralgia and Psoriatic Arthritis [see Warnings and Precautions (5.8)]
- Parasitic (Helminth) Infections [see Warnings and Precautions (5.9)]
5.1 Hypersensitivity
Hypersensitivity reactions, including anaphylaxis, acute generalized exanthematous pustulosis (AGEP), serum sickness or serum sickness-like reactions, angioedema, generalized urticaria, rash, erythema nodosum and erythema multiforme have been reported. A case of AGEP was reported in an adult subject who participated in the bullous pemphigoid development program. If a clinically significant hypersensitivity reaction occurs, institute appropriate therapy and discontinue DUPIXENT [see Adverse Reactions (6.1, 6.2) and Clinical Pharmacology (12.6)].
Instructions for Use
Read this Instructions for Use before using the DUPIXENT Pre-filled Syringe. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself the first time. Keep these instructions for future use. Call your healthcare provider if you have any questions.
This device is a Single-Dose Pre-filled Syringe (called "DUPIXENT Syringe" in these instructions). It contains 300 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Syringe are shown below:
| Important Information | |
|---|---|
|
|
How should I store DUPIXENT?
|
Step 1: Remove
Remove the DUPIXENT Syringe from the carton by holding the middle of the Syringe Body.
Do not pull off the Needle Cap until you are ready to inject.
Do not use the DUPIXENT Syringe if it has been dropped on a hard surface or damaged.
Step 2: Prepare
Ensure you have the following:
- the DUPIXENT Pre-filled Syringe
- 1 alcohol wipe*
- 1 cotton ball or gauze*
- a sharps disposal container* (See Step 13)
*Items not included in the carton
Step 3: Check
When you receive your DUPIXENT Syringes, always check to see that:
- you have the correct medicine and dose.
- the expiration date on the Single-Dose Pre-filled Syringe has not passed.
Do not use the DUPIXENT Syringe if the expiration date has passed.
Step 4: Inspect
Look at the medicine through the Viewing Window on the DUPIXENT Syringe:
Check to see if the liquid is clear and colorless to pale yellow.
Note: You may see an air bubble, this is normal.
Do not use the DUPIXENT Syringe if the liquid is discolored or cloudy, or if it contains visible flakes or particles.
Step 5: Wait 45 minutes
Lay the DUPIXENT Syringe on a flat surface and let it naturally warm to room temperature for at least 45 minutes.
Do not heat the DUPIXENT Syringe.
Do not put the DUPIXENT Syringe into direct sunlight.
Do not keep DUPIXENT Syringes at room temperature for more than 14 days. Throw away (dispose of) any DUPIXENT Syringes that have been left at room temperature for longer than 14 days.
Step 6: Choose your injection site
- You can inject into your thigh or stomach, except for the 2 inches (5 cm) around your belly button (navel).
- If a caregiver injects your dose, they can also use the outer area of the upper arm.
- Choose a different site each time you inject DUPIXENT.
Do not inject into skin that is tender, damaged, bruised or scarred.
Step 7: Clean
Wash your hands.
Clean the injection site with an alcohol wipe.
Let your skin dry before injecting.
Do not touch the injection site again or blow on it before the injection.
Step 8: Remove Needle Cap
Hold the DUPIXENT Syringe in the middle of the Syringe Body with the Needle pointing away from you and pull off the Needle Cap.
Do not put the Needle Cap back on.
Do not touch the Needle.
Inject your medicine right away after removing the Needle Cap.
Step 9: Pinch
Pinch a fold of skin at the injection site (thigh or stomach, except 2 inches around your belly button, or outer area of the upper arm if injected by your caregiver). The figure below shows an example of pinching a fold of skin on your stomach.
Step 10: Insert
Insert the Needle completely into the fold of the skin at about a 45° angle.
Step 11: Push
Relax the pinch.
Push the Plunger Rod down slowly and steadily as far as it will go until the DUPIXENT Syringe is empty.
Note: You will feel some resistance. This is normal.
Step 12: Release and Remove
Lift your thumb to release the Plunger Rod until the Needle is covered by the Needle Shield and then remove the Syringe from the injection site.
Lightly press a cotton ball or gauze on the injection site if you see any blood.
Do not put the Needle Cap back on.
Do not rub your skin after the injection.
Step 13: Dispose
Put your used Needles, DUPIXENT Syringes, and Needle Caps in a FDA-cleared sharps disposal container right away after use.
Do not dispose of (throw away) Needles, DUPIXENT Syringes, and Needle Caps in your household trash.
If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used Needles and Syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Do not put the Needle Cap back on.
Manufactured by: Regeneron Pharmaceuticals, Inc. Tarrytown, NY 10591
U.S. License No. 1760
Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
DUPIXENT
®
is a registered trademark of Sanofi Biotechnology
© 2023 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: July 2023
Instructions for Use
DUPIXENT® (DU-pix-ent)
(dupilumab)
injection, for subcutaneous use
Single-Dose Pre-filled Pen (200 mg/1.14 mL)
This Instructions for Use contains information on how to inject DUPIXENT.
Read this Instructions for Use before using the DUPIXENT Pre-filled Pen. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself for the first time. Keep this Instructions for Use. Call your healthcare provider if you have any questions.
This DUPIXENT Pre-filled Pen is only for use in adults and children aged 2 years and older.
This DUPIXENT Pre-filled Pen is a single-dose device. It contains 200 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Pre-filled Pen are shown below:
Important Information
|
Storing DUPIXENT
|
| A. Get ready to inject | |
|---|---|
| A1. Gather supplies | |
| Find a clean, flat work surface. Make sure you have the following supplies: |
|
| A2. Check the Pen | |
|
|
| A3. Look at the Label | |
|
|
|
| A4. Check the Medicine | |
|---|---|
|
| A5. Wait 30 minutes | |
|---|---|
|
| B. Choose and prepare your injection site | |
|---|---|
| B1. Wash your hands well with soap and water | |
| B2. Choose an injection site | |
|
|
| B3. Prepare the injection site | |
|---|---|
|
|
| C. Give the injection | |
| C1. Remove Yellow Cap | |
|
|
| C2. Pinch Skin and Place | ||
|---|---|---|
Pinch the skin before and during the injection.
|
||
| C3. Press down ➜ Watch Window turn fully yellow ➜ Then count to 5 | ||
Press and hold the DUPIXENT Pre-filled Pen firmly against the skin until you cannot see the Orange Needle Cover.
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and watch the window:
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and count to 5 to make sure you get your full dose. |
| Pinching of the skin is not needed for adults and children aged 12 years and older. | |
|---|---|
| C4. Remove | |
|
| D. Dispose of used DUPIXENT Pre-filled Pen | |
|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: July 2023 |
|
How to Dispose of (throw away) DUPIXENT Pre-filled Pen:
Put the used DUPIXENT Pre-filled Pens and Yellow Caps in a FDA-cleared sharps disposal container right away after use. Do not dispose of (throw away) DUPIXENT Pre-filled Pens and Yellow Caps in your household trash. If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Keep your sharps disposal container out of the reach of children. |
|
|
|
|
|
Manufactured by: Regeneron Pharmaceuticals, Inc. Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT ® is a registered trademark of Sanofi Biotechnology © 2023 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. |
1.1 Atopic Dermatitis
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 6 months and older with moderate-to-severe atopic dermatitis (AD) whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids.
1.5 Prurigo Nodularis
DUPIXENT is indicated for the treatment of adult patients with prurigo nodularis (PN).
12.2 Pharmacodynamics
Consistent with inhibition of IL-4 and IL-13 signaling, dupilumab treatment decreased certain biomarkers of inflammation. In asthma subjects, fractional exhaled nitric oxide (FeNO) and circulating concentrations of eotaxin-3, total IgE, allergen specific IgE, TARC, and periostin were decreased relative to placebo. Reductions in these biomarkers were comparable for the 300 mg Q2W and 200 mg Q2W regimens. These markers were near maximal suppression after 2 weeks of treatment, except for IgE which declined more slowly. These effects were sustained throughout treatment. The median percent reduction from baseline in total IgE concentrations with dupilumab treatments was 52% at Week 24 (DRI12544) and 70% at Week 52 (QUEST). For FeNO, the mean percent reduction from baseline at Week 2 was 35% and 24% in DRI12544 and QUEST, respectively, and in the overall safety population, the mean FeNO level decreased to 20 ppb.
A continuous decline in total IgE in serum was observed in CSU trials.
In AFRS subjects, total IgE concentrations showed a progressive decline with dupilumab treatment throughout the 52-week treatment period.
12.3 Pharmacokinetics
The pharmacokinetics of dupilumab is similar in subjects with AD, asthma, CRSwNP, EoE, PN, COPD, CSU, BP, and AFRS.
1.8 Bullous Pemphigoid
DUPIXENT is indicated for the treatment of adult patients with bullous pemphigoid (BP).
14.5 Prurigo Nodularis
The efficacy of DUPIXENT in adults with prurigo nodularis (PN) was evaluated in two 24-week randomized, double-blind, placebo-controlled, multicenter, parallel-group trials (PRIME (NCT04183335) and PRIME 2 (NCT04202679)). These trials enrolled 311 adult subjects with pruritus (WI-NRS ≥ 7 on a scale of 0 to 10) and greater than or equal to 20 nodular lesions. PRIME and PRIME 2 assessed the effect of DUPIXENT on pruritus improvement as well as its effect on PN lesions.
In these two trials, subjects received either subcutaneous DUPIXENT 600 mg (two 300 mg injections) on day 1, followed by 300 mg once every 2 weeks (Q2W) for 24 weeks, or matching placebo.
In these trials, the mean age was 49.5 years, the median weight was 71 kg, 65% of subjects were female, 57% were White, 6% were Black, and 34% were Asian. At baseline, the mean Worst Itch-Numeric Rating Scale (WI-NRS) was 8.5, 66% had 20 to 100 nodules (moderate), and 34% had greater than 100 nodules (severe). Eleven percent (11%) of subjects were taking stable doses of antidepressants at baseline and were instructed to continue taking these medications during the trial. Forty-three percent (43%) had a history of atopy (defined as having a medical history of AD, allergic rhinitis/rhinoconjunctivitis, asthma, or food allergy).
The WI-NRS is comprised of a single item, rated on a scale from 0 ("no itch") to 10 ("worst imaginable itch"). Subjects were asked to rate the intensity of their worst pruritus (itch) over the past 24 hours using this scale. The Investigator's Global Assessment for Prurigo Nodularis-Stage (IGA PN-S) is a scale that measures the approximate number of nodules using a 5-point scale from 0 (clear) to 4 (severe).
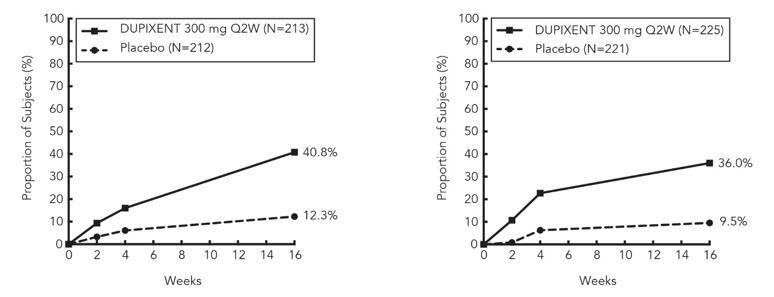
Efficacy was assessed with the proportion of subjects with improvement (reduction) in WI-NRS by ≥4 points, the proportion of subjects with IGA PN-S 0 or 1 (the equivalent of 0-5 nodules), and the proportion of subjects who achieved a response in both WI-NRS and IGA PN-S per the criteria described above.
The efficacy results for PRIME and PRIME2 are presented in Table 32 and Figure 14, Figure 15, and Figure 16.
| PRIME | PRIME2 | |||||
|---|---|---|---|---|---|---|
| Placebo (N=76) |
DUPIXENT 300 mg Q2W (N=75) |
Difference (95% CI) for DUPIXENT vs. Placebo | Placebo (N=82) |
DUPIXENT 300 mg Q2W (N=78) |
Difference (95% CI) for DUPIXENT vs. Placebo | |
| Proportion of subjects with both an improvement (reduction) in WI-NRS by ≥4 points from baseline to Week 24 and an IGA PN-S 0 or 1 at Week 24 Subjects who received rescue treatment earlier or had missing data were considered as non-responders.
|
9.2% | 38.7% | 29.6% (16.4, 42.8) |
8.5% | 32.1% | 25.5% (13.1, 37.9) |
| Proportion of subjects with improvement (reduction) in WI-NRS by ≥4 points from baseline at Week 24 | 18.4% | 60.0% | 42.7% (27.8, 57.7) |
19.5% | 57.7% | 42.6% (29.1, 56.1) |
| Proportion of subjects with IGA PN-S 0 or 1 at Week 24 | 18.4% | 48.0% | 28.3% (13.4, 43.2) |
15.9% | 44.9% | 30.8% (16.4, 45.2) |
| Proportion of subjects with improvement (reduction) in WI-NRS by ≥4 points from baseline at Week 12 | 15.8% Not adjusted for multiplicity in PRIME.
|
44.0% | 29.2% (14.5, 43.8) |
22.0% | 37.2% | 16.8% (2.3, 31.2) |
Figure 14: Proportion of Adult Subjects with PN with Both WI-NRS ≥4-point Improvement and IGA PN-S 0 or 1 Over Time in PRIME and PRIME2
Figure 15: Proportion of Adult Subjects with PN with WI-NRS ≥4-point Improvement Over Time in PRIME and PRIME2
Figure 16: Proportion of Adult Subjects with IGA PN-S 0 or 1 Over Time in PRIME and PRIME 2
The efficacy data did not show differential treatment effect across demographic subgroups.
1 Indications and Usage
DUPIXENT is an interleukin-4 receptor alpha antagonist indicated:
Atopic Dermatitis
for the treatment of adult and pediatric patients aged 6 months and older with moderate-to-severe AD whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids. (1.1)
Asthma
as an add-on maintenance treatment of adult and pediatric patients aged 6 years and older with moderate-to-severe asthma characterized by an eosinophilic phenotype or with oral corticosteroid dependent asthma. (1.2)
Limitations of Use: Not for the relief of acute bronchospasm or status asthmaticus. (1.2)
Chronic Rhinosinusitis with Nasal Polyps
as an add-on maintenance treatment in adult and pediatric patients aged 12 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP). (1.3)
Eosinophilic Esophagitis
for the treatment of adult and pediatric patients aged 1 year and older, weighing at least 15 kg, with eosinophilic esophagitis (EoE). (1.4)
Prurigo Nodularis
for the treatment of adult patients with prurigo nodularis (PN). (1.5)
Chronic Obstructive Pulmonary Disease
as an add-on maintenance treatment of adult patients with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype. (1.6)
Limitations of Use: Not for the relief of acute bronchospasm. (1.6)
Chronic Spontaneous Urticaria
for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment. (1.7)
Limitations of Use: Not indicated for other forms of urticaria. (1.7)
Bullous Pemphigoid
for the treatment of adult patients with bullous pemphigoid (BP). (1.8)
Allergic Fungal Rhinosinusitis
for the treatment of adult and pediatric patients aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery. (1.9)
14.8 Bullous Pemphigoid
The efficacy of DUPIXENT in adults with bullous pemphigoid (BP) was evaluated in a 52-week randomized, double-blind, parallel-group, multicenter, placebo-controlled trial (ADEPT; NCT04206553). This trial enrolled 106 adult subjects with a Bullous Pemphigoid Disease Area Index (BPDAI) activity score ≥24 on a scale of 0-360 and a weekly average Peak Pruritus NRS score ≥4 on a scale of 0-10.
In this trial, subjects were randomized to receive either subcutaneous DUPIXENT 600 mg (two 300 mg injections) on Day 1, followed by 300 mg every other week (Q2W), or matching placebo for 52 weeks. All subjects were also initiated on a standard regimen of oral corticosteroids (prednisone or prednisolone; 0.5 or 0.75 mg/kg/day) on Day 1. After achieving control of disease activity for 2 weeks, oral corticosteroids (OCS) were tapered with the objective to taper them off no later than Week 16 as long as the control of disease activity was maintained. Subjects who experienced a loss of control of disease activity (new lesions form and existing lesions cease to heal) during OCS taper, or who relapsed post-taper, or who used rescue medications were considered treatment failures. During the OCS taper, subjects were permitted to increase their OCS once, with any subsequent increases being considered rescue therapy. Subjects could be rescued during the trial with topical or oral corticosteroids, non-steroidal immunosuppressive medications, or immunomodulating biologics. Subjects were allowed to continue trial treatment if rescued with topical or oral corticosteroids.
Demographics and baseline characteristics were similar between the treatment arms. Of the 106 subjects enrolled, the mean age was 71.3 years; 53% were female; 68% were White, 2% were Black or African American, and 15% were Asian; 3% of subjects identified as Hispanic or Latino. At baseline, 63% of subjects had prior systemic corticosteroid use for BP. The mean Peak Pruritus NRS score was 7.5 at baseline.
The primary endpoint was the proportion of subjects achieving sustained remission at Week 36. Sustained remission was defined as the achievement of complete remission and off OCS no later than Week 16, absence of disease relapse from the time of completion of the corticosteroid taper to Week 36, and absence of rescue therapy during the 36-week double-blind treatment period. Secondary endpoints included total cumulative dose of OCS and proportion of subjects with a reduction in itch defined as at least a 4-point improvement (reduction) in the Peak Pruritus NRS.
The efficacy results for ADEPT are presented in Table 38. The proportion of subjects that received rescue therapy during the 36-week treatment period was 53% in the DUPIXENT group and 79% in the placebo group.
| DUPIXENT 300 mg Q2W + OCS (N=53) |
Placebo + OCS (N=53) |
Difference (95% CI) for DUPIXENT vs. Placebo | |
|---|---|---|---|
| Proportion of subjects achieving sustained remission Sustained remission was defined as the achievement of complete remission and off OCS no later than Week 16, absence of disease relapse from the time of completion of the corticosteroid taper to Week 36, and absence of rescue therapy during the 36-week double-blind treatment period.
|
18.3% | 6.1% | 12.2% (-0.8, 26.1) |
| Proportion of subjects with improvement (reduction) of ≥4 points in Peak Pruritus NRS from baseline | 38.3% | 10.5% | 27.8% (11.6, 43.4) |
The median (min, max) cumulative dose of OCS at Week 36 was 2.8 g (1.2, 22.7) in the DUPIXENT group compared to 4.1 g (1.5, 23.3) in the placebo group.
12.1 Mechanism of Action
Dupilumab is a human monoclonal IgG4 antibody that inhibits interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling by specifically binding to the IL-4Rα subunit shared by the IL-4 and IL-13 receptor complexes. Dupilumab inhibits IL-4 signaling via the Type I receptor and both IL-4 and IL-13 signaling through the Type II receptor.
Inflammation driven by IL-4 and IL-13 is an important component in the pathogenesis of asthma, AD, CRSwNP, EoE, PN, COPD, CSU, BP, and AFRS. Multiple cell types that express IL-4Rα (e.g., mast cells, basophils, eosinophils, macrophages, lymphocytes, epithelial cells, goblet cells) and inflammatory mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines, chemokines) are involved in inflammation. Blocking IL-4Rα with dupilumab inhibits IL-4 and IL-13 cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE. The mechanism of dupilumab action has not been definitively established.
2.13 Preparation for Use
Before injection, remove DUPIXENT from the refrigerator and allow DUPIXENT to reach room temperature (45 minutes for the 300 mg/2 mL pre-filled syringe or pre-filled pen, and 30 minutes for the 200 mg/1.14 mL pre-filled syringe or pre-filled pen) without removing the needle cap. After removal from the refrigerator, DUPIXENT must be used within 14 days or discarded.
Inspect DUPIXENT visually for particulate matter and discoloration prior to administration. DUPIXENT is a clear to slightly opalescent, colorless to pale yellow solution. Do not use if the liquid contains visible particulate matter, is discolored or cloudy (other than clear to slightly opalescent, colorless to pale yellow). DUPIXENT does not contain preservatives; therefore, discard any unused product remaining in the pre-filled syringe or pre-filled pen.
5 Warnings and Precautions
- Hypersensitivity: Hypersensitivity reactions including anaphylaxis, acute generalized exanthematous pustulosis (AGEP), serum sickness, angioedema, urticaria, rash, erythema nodosum, and erythema multiforme have occurred. Discontinue DUPIXENT in the event of a hypersensitivity reaction. (5.1)
- Conjunctivitis and Keratitis: Advise patients to report new onset or worsening eye symptoms to their healthcare provider. Consider ophthalmological examination, as appropriate. (5.2)
- Eosinophilic Conditions: Be alert to vasculitic rash, worsening pulmonary symptoms, cardiac complications, kidney injury and/or neuropathy, especially upon reduction of oral corticosteroids. (5.3)
- Reduction of Corticosteroid Dosage: Do not discontinue systemic, topical, or inhaled corticosteroids abruptly upon initiation of DUPIXENT. Decrease steroids gradually, if appropriate. (5.5)
- Psoriasis: Advise patients to report new-onset psoriasis symptoms to their healthcare provider. If symptoms persist or worsen, consider dermatologic evaluation and/or discontinuation of DUPIXENT. (5.7)
- Arthralgia and Psoriatic Arthritis: Advise patients to report new onset joint symptoms to their healthcare provider. If symptoms persist or worsen, consider rheumatological evaluation and/or discontinuation of DUPIXENT. (5.8)
- Parasitic (Helminth) Infections: Treat pre-existing helminth infections before initiating DUPIXENT. If patients become infected while receiving DUPIXENT and do not respond to anti-helminth treatment, discontinue DUPIXENT until the infection resolves. (5.9)
- Vaccinations: Avoid use of live vaccines. (5.10)
2 Dosage and Administration
DUPIXENT is administered by subcutaneous injection. (2.1)
Atopic Dermatitis
Dosage in Adults (2.3):
-
Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W).
Dosage in Pediatric Patients 6 Months to 5 Years of Age (2.3):
| Body Weight | Initial and Subsequent Dosage |
|---|---|
| 5 to less than 15 kg | 200 mg (one 200 mg injection) every 4 weeks (Q4W) |
| 15 to less than 30 kg | 300 mg (one 300 mg injection) every 4 weeks (Q4W) |
Dosage in Pediatric Patients 6 Years of Age and Older (2.3):
| Body Weight | Initial Loading Dose | Subsequent Dosage Q2W – every 2 weeks; Q4W – every 4 weeks
|
|---|---|---|
| 15 to less than 30 kg | 600 mg (two 300 mg injections) | 300 mg Q4W |
| 30 to less than 60 kg | 400 mg (two 200 mg injections) | 200 mg Q2W |
| 60 kg or more | 600 mg (two 300 mg injections) | 300 mg Q2W |
Asthma
Dosage in Adult and Pediatric Patients 12 Years and Older (2.4):
| Initial Loading Dose | Subsequent Dosage |
|---|---|
| 400 mg (two 200 mg injections) | 200 mg every 2 weeks (Q2W) |
| Or | |
| 600 mg (two 300 mg injections) | 300 mg every 2 weeks (Q2W) |
|
Dosage for patients with oral corticosteroid-dependent asthma or with co-morbid moderate-to-severe AD, CRSwNP, or AFRS For pediatric patients 12 to 17 years of age (≥60 kg) and adults with AFRS
|
|
| 600 mg (two 300 mg injections) | 300 mg every 2 weeks (Q2W) |
Dosage in Pediatric Patients 6 to 11 Years of Age (2.4):
| Body Weight | Initial Dose and Subsequent Dosage |
|---|---|
| 15 to less than 30 kg | 300 mg every 4 weeks (Q4W) |
| ≥30 kg | 200 mg every 2 weeks (Q2W) |
For pediatric patients 6 to 11 years old with asthma and co-morbid moderate-to-severe atopic dermatitis, follow the recommended dosage as per Table 2 which includes an initial loading dose. (2.3)
Chronic Rhinosinusitis with Nasal Polyps (2.5):
-
Recommended dosage for adult and pediatric patients 12 years of age and older is 300 mg given every 2 weeks (Q2W).
Eosinophilic Esophagitis (2.6):
| Body Weight | Recommended Dosage in Adult and Pediatric Patients 1 Year and Older, Weighing At Least 15 kg |
|---|---|
| 15 to less than 30 kg | 200 mg every 2 weeks (Q2W) |
| 30 to less than 40 kg | 300 mg every 2 weeks (Q2W) |
| 40 kg or more | 300 mg every week (QW) |
Prurigo Nodularis (2.7):
-
Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W).
Chronic Obstructive Pulmonary Disease (2.8):
-
Recommended dosage for adult patients is 300 mg given every 2 weeks (Q2W).
Chronic Spontaneous Urticaria
Dosage in Adults (2.9):
-
Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W).
Dosage in Pediatric Patients 12 to 17 Years of Age (2.9):
| Body Weight | Initial Loading Dose | Subsequent Dosage |
|---|---|---|
| 30 to less than 60 kg | 400 mg (two 200 mg injections) | 200 mg Q2W |
| 60 kg or more | 600 mg (two 300 mg injections) | 300 mg Q2W |
Bullous Pemphigoid (2.10):
Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every other week (Q2W). Use DUPIXENT in combination with a tapering course of oral corticosteroids.
Allergic Fungal Rhinosinusitis
Dosage in Adults (2.11):
-
Recommended dosage is 300 mg given every 2 weeks (Q2W).
Dosage in Pediatric Patients 6 to 17 Years of Age (2.11):
| Body Weight | Recommended Dosage |
|---|---|
| a Q2W – every 2 weeks; Q4W – every 4 weeks | |
| 15 to less than 30 kg | 300 mg Q4W |
| 30 to less than 60 kg | 200 mg Q2W |
| 60 kg or more | 300 mg Q2W |
5.3 Eosinophilic Conditions
Patients being treated for asthma may present with clinical features of eosinophilic pneumonia or eosinophilic granulomatosis with polyangiitis (EGPA). These events may be associated with the reduction of oral corticosteroid therapy. Healthcare providers should be alert to vasculitic rash, worsening pulmonary symptoms, cardiac complications, kidney injury, and/or neuropathy presenting in their patients with eosinophilia. Cases of eosinophilic pneumonia were reported in adults who participated in the asthma development program. Cases of EGPA have been reported with DUPIXENT in adults who participated in the asthma development program as well as in adults with co-morbid asthma in the CRSwNP development program. Advise patients to report signs of eosinophilic pneumonia and EGPA to their healthcare provider. Consider withholding DUPIXENT if eosinophilic pneumonia or EGPA are suspected.
1.4 Eosinophilic Esophagitis
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 1 year and older, weighing at least 15 kg, with eosinophilic esophagitis (EoE).
3 Dosage Forms and Strengths
DUPIXENT is a clear to slightly opalescent, colorless to pale yellow solution in a:
Single-dose pre-filled syringe with needle shield as:
- Injection: 300 mg/2 mL (150 mg/mL)
- Injection: 200 mg/1.14 mL (175 mg/mL)
Single-dose pre-filled pen as:
- Injection: 300 mg/2 mL (150 mg/mL)
- Injection: 200 mg/1.14 mL (175 mg/mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of DUPIXENT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Immune system disorders: angioedema
- Musculoskeletal system disorders: psoriatic arthritis
- Skin and subcutaneous tissue disorders: Facial skin reactions, including erythema, rash, scaling, edema, papules, pruritus, burning, and pain; new-onset psoriasis, vasculitis
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.2 Conjunctivitis and Keratitis
Conjunctivitis and keratitis adverse reactions have been reported in clinical trials [see Adverse Reactions (6.1)].
Conjunctivitis and keratitis occurred more frequently in AD subjects who received DUPIXENT compared to those who received placebo. Conjunctivitis was the most frequently reported eye disorder. Most subjects with conjunctivitis or keratitis recovered or were recovering during the treatment period.
Among subjects with asthma, the frequencies of conjunctivitis and keratitis were similar between DUPIXENT and placebo.
In adult subjects with CRSwNP, the frequency of conjunctivitis was 2% in the DUPIXENT group compared to 1% in the placebo group in the 24-week safety pool; these subjects recovered.
Among subjects with EoE, there were no reports of conjunctivitis and keratitis in the DUPIXENT group in placebo-controlled trials.
In subjects with PN, the frequency of conjunctivitis was 4% in the DUPIXENT group compared to 1% in the placebo group; these subjects recovered or were recovering during the treatment period.
Among subjects with COPD, the frequency of conjunctivitis and keratitis was 1.4% and 0.1% in the DUPIXENT group and 1% and 0% in the placebo group, respectively.
In subjects with CSU, the frequency of conjunctivitis was similar between DUPIXENT and placebo.
Among subjects with BP, the frequency of conjunctivitis and keratitis was 7.5% and 3.8% in the DUPIXENT group and 0% and 0% in the placebo group, respectively.
Conjunctivitis and keratitis adverse events have also been reported with DUPIXENT in postmarketing settings, predominantly in AD patients. Some patients reported visual disturbances (e.g., blurred vision) associated with conjunctivitis or keratitis.
Advise patients or their caregivers to report new onset or worsening eye symptoms to their healthcare provider. Consider ophthalmological examination for patients who develop conjunctivitis that does not resolve following standard treatment or signs and symptoms suggestive of keratitis, as appropriate.
1.7 Chronic Spontaneous Urticaria
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
1.9 Allergic Fungal Rhinosinusitis
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery.
14.7 Chronic Spontaneous Urticaria
The efficacy of DUPIXENT for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria who remain symptomatic despite H1 antihistamine treatment was evaluated in a master protocol clinical trial (CUPID [NCT04180488]) that included three 24-week, randomized, double-blind, parallel-group, multicenter, placebo-controlled trials (CUPID Study A, Study B, and Study C), followed by 12-week blinded safety follow-up periods. CUPID Study A and Study C included subjects who remained symptomatic despite H1 antihistamine treatment and were anti-IgE treatment naïve, while CUPID Study B included patients who remained symptomatic despite H1 antihistamine and anti-IgE treatments. The efficacy of DUPIXENT was based only on CUPID Study A and Study C; Study B did not meet the primary endpoint.
2.2 Vaccination Prior to Treatment
Consider completing all age-appropriate vaccinations as recommended by current immunization guidelines prior to initiating treatment with DUPIXENT [see Warnings and Precautions (5.10)].
5.6 Patients With Co Morbid Asthma
Advise patients with co-morbid asthma not to adjust or stop their asthma treatments without consultation with their physicians.
14.9 Allergic Fungal Rhinosinusitis
The efficacy of DUPIXENT for the treatment of adult and pediatric subjects aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery was evaluated in a randomized, double-blind, parallel-group, placebo-controlled trial (AIMS [NCT04684524]) of 52 weeks duration. This trial included a total of 62 adult and pediatric subjects aged 6 years and older.
The AIMS trial enrolled patients with AFRS who had evidence of sinus opacification on the Lund Mackay (LMK) sinus CT scan with a LMK score of ≥9 for subjects with unilateral polyps and >12 for subjects with bilateral polyps. There were 79% of subjects who had opacification of all sinuses. Additionally, subjects were required to have a nasal polyps score (NPS) of ≥2 for unilateral polyps or ≥3 for bilateral polyps. Rescue with systemic corticosteroids or surgery was allowed during the study at the investigator's discretion. In the AIMS trial, subjects were randomized 1:1 to receive either DUPIXENT or placebo subcutaneously for 52 weeks. In the DUPIXENT group, adults and pediatric subjects who weighed ≥60 kg received 300 mg every 2 weeks; pediatric subjects who weighed ≥30 kg to <60 kg received 200 mg every 2 weeks. The demographics and baseline characteristics of AIMS are provided in Table 39 below.
| Parameter | AIMS (N=62) |
|---|---|
| SD = standard deviation; LMK = Lund Mackay score; NC = nasal congestion/obstruction; NPS = nasal polyps score; AM = morning | |
| Mean age (years) (SD) | 39.8 (16.0) |
| >17 years, n | 56 |
| 12 to 17 years, n | 5 |
| 6 to <12 years, n | 1 |
| % Male | 72.6 |
| White, N (%) | 27 (43.5) |
| Black, N (%) | 8 (12.9) |
| Asian, N (%) | 25 (40.3) |
| Ethnicity Hispanic or Latino | 14 (22.6) |
| Subjects with systemic corticosteroid use in the previous 2 years, N (%) | 13 (27.7) |
| Mean LMK sinus CT total score Higher scores indicate greater disease severity (SD), range 0 24 |
17.9 (3.6) |
| Mean NC score (SD), range 0-3 | 1.9 (0.9) |
| Mean NPS (SD), range 0-8 | 5.2 (1.9) |
| Mean loss of smell score (AM), (SD) range 0-3 | 2.1 (1.1) |
| Mean blood eosinophils (cells/mcL) (SD) | 0.5 (0.3) |
| Mean total IgE IU/mL (SD) | 1356.4 (1535.0) |
| Prior sino-nasal surgery, N (%) | 61 (98.4) |
| 1 sino-nasal surgery, n (%) | 30 (48.4) |
| ≥2 sino-nasal surgery, n (%) | 31 (50.0) |
| Comorbid asthma, N (%) | 29 (46.8) |
| History of allergic comorbidity, N (%) | 50 (80.6) |
The primary efficacy endpoint was the change from baseline in sinus opacification assessed by the LMK sinus CT scan score at Week 52. The LMK sinus CT scan score evaluated the opacification of each sinus using a 0 to 2 scale (0=normal; 1=partial opacification; 2=total opacification) deriving a maximum score of 12 per side and a total maximum score of 24 (higher scores indicate more opacification).
Secondary endpoints included change from baseline at Week 24 and Week 52 in monthly average nasal congestion score (NC) and endoscopic nasal polyps score (NPS); and change from baseline at Week 24 in sinus opacification assessed by the LMK sinus CT scan score and loss of smell score. Description of the NCS, NPS, and loss of smell score endpoints is the same as for the indication of CRSwNP [see Clinical Studies (14.3)]. The proportion of participants receiving systemic corticosteroids for any reason and/or undergoing/planning to undergo surgery for AFRS (sino-nasal surgery) was evaluated as a secondary endpoint.
Statistically significant efficacy was observed in AIMS for the primary and secondary endpoints as presented in Table 40.
| AIMS Trial | |||||
|---|---|---|---|---|---|
| Placebo (N=29) |
DUPIXENT (N=33) |
LS mean difference vs. Placebo (95% CI) |
|||
| A reduction in score indicates improvement. | |||||
| LMK = Lund Mackay score; NC = nasal congestion/obstruction; NPS = nasal polyps score | |||||
| Scores | Baseline mean |
LS mean change |
Baseline mean |
LS mean change |
|
| Primary Endpoint at Week 52 | |||||
| LMK sinus CT scan score | 18.45 | -1.81 | 17.50 | -9.17 | -7.36 (-9.38, -5.35) |
| Secondary Endpoints at Week 52 | |||||
| NC | 2.05 | -0.17 | 1.86 | -1.57 | -1.40 (-1.77, -1.02) |
| NPS | 5.38 | -0.55 | 5.12 | -3.32 | -2.77 (-3.82, -1.72) |
| Secondary Endpoints at Week 24 | |||||
| LMK sinus CT scan score | 18.45 | -1.93 | 17.50 | -7.38 | -5.45 (-7.48, -3.43) |
| NC | 2.05 | -0.43 | 1.86 | -1.30 | -0.87 (-1.18, -0.56) |
| NPS | 5.38 | -0.80 | 5.12 | -3.16 | -2.36 (-3.31, -1.41) |
| Loss of smell | 2.15 | -0.39 | 1.97 | -1.28 | -0.89 (-1.29, -0.49) |
Figure 19 and Figure 20 demonstrate the time course of improvement in the mean change from baseline in NPS and NCS.
Figure 19: LS Mean Change from Baseline in Monthly Average Nasal Congestion (NC) Score Up to Week 52 in Subjects 6 Years of Age and Older with AFRS (AIMS - ITT Population)
Figure 20: LS Mean Change from Baseline in Nasal Polyp Score (NPS) Up to Week 52 in Subjects 6 Years of Age and Older with AFRS (AIMS - ITT Population)
5.9 Parasitic (helminth) Infections
Patients with known helminth infections were excluded from participation in clinical studies. It is unknown if DUPIXENT will influence the immune response against helminth infections.
Treat patients with pre-existing helminth infections before initiating therapy with DUPIXENT. If patients become infected while receiving treatment with DUPIXENT and do not respond to anti-helminth treatment, discontinue treatment with DUPIXENT until the infection resolves. Adverse reactions of helminth infections (5 cases of enterobiasis and 1 case of ascariasis) were reported in pediatric subjects 6 to 11 years old who participated in the pediatric asthma development program [see Adverse Reactions (6.1)].
5.8 Arthralgia and Psoriatic Arthritis
Arthralgia has been reported with the use of DUPIXENT with some patients reporting gait disturbances or decreased mobility associated with joint symptoms; some cases resulted in hospitalization [see Adverse Reactions (6.1)]. In postmarketing reports, onset of arthralgia was variable, ranging from days to months after the first dose of DUPIXENT.
Cases of new-onset psoriatic arthritis requiring systemic treatment have been reported with the use of DUPIXENT.
Some patients' symptoms resolved while continuing treatment with DUPIXENT, and other patients recovered or were recovering following discontinuation of DUPIXENT.
Advise patients to report new onset or worsening joint symptoms to their healthcare provider. If symptoms persist or worsen, consider rheumatological evaluation and/or discontinuation of DUPIXENT.
1.6 Chronic Obstructive Pulmonary Disease
DUPIXENT is indicated as an add-on maintenance treatment of adult patients with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype.
2.1 Important Administration Instructions
DUPIXENT is administered by subcutaneous injection.
DUPIXENT is intended for use under the guidance of a healthcare provider. Provide proper training to patients and/or caregivers on the preparation and administration of DUPIXENT prior to use according to the "Instructions for Use".
14.6 Chronic Obstructive Pulmonary Disease
The efficacy of DUPIXENT as add-on maintenance treatment of adult patients with inadequately controlled COPD and an eosinophilic phenotype was evaluated in two randomized, double-blind, multicenter, parallel-group, placebo-controlled trials (BOREAS [NCT03930732] and NOTUS [NCT04456673]) of 52 weeks duration. The two trials enrolled a total of 1874 adult subjects with COPD.
Both trials enrolled subjects with a diagnosis of COPD with moderate to severe airflow limitation (post-bronchodilator FEV1/FVC ratio <0.7 and post-bronchodilator FEV1 of 30% to 70% predicted) and a minimum blood eosinophil count of 300 cells/mcL at screening. Trial enrollment required an exacerbation history of at least 2 moderate or 1 severe exacerbation(s) in the previous year despite receiving maintenance triple therapy consisting of a long-acting muscarinic antagonist (LAMA), long-acting beta agonist (LABA), and inhaled corticosteroid (ICS), and symptoms of chronic productive cough for at least 3 months in the past year. Greater than or equal to 95% of subjects in each trial had chronic bronchitis. Subjects also had a Medical Research Council (MRC) dyspnea score ≥2 (range 0-4). Exacerbations of COPD were defined as clinically significant worsening of COPD symptoms including increases in dyspnea, wheezing, cough, sputum volume, and/or increase in sputum purulence. Exacerbation severity was further defined as moderate if treatment with systemic corticosteroids and/or antibiotics was required, or severe if they resulted in hospitalization or observation for over 24 hours in an emergency department or urgent care facility.
In both trials, subjects were randomized to receive DUPIXENT 300 mg subcutaneously every 2 weeks (Q2W) or placebo in addition to their background maintenance therapy for 52 weeks.
The demographics and baseline characteristics of BOREAS and NOTUS trial populations are provided in Table 33 below.
| Parameter | BOREAS (N = 939) |
NOTUS (N = 935) |
|---|---|---|
| ICS = inhaled corticosteroid; LAMA = long-acting muscarinic antagonist; LABA = long-acting beta agonist; FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity |
||
| Mean age (years) (± SD) | 65.1 (8.1) | 65.0 (8.3) |
| Male (%) | 66.0 | 67.6 |
| White, N (%) | 790 (84.1) | 838 (89.6) |
| Asian, N (%) | 134 (14.3) | 10 (1.1) |
| Black, N (%) | 5 (0.5) | 12 (1.3) |
| American Indian or Alaska Native, N (%) | 7 (0.7) | 48 (5.1) |
| Other/Multiple, N (%) | 3 (0.3) | 27 (2.9) |
| Ethnicity Hispanic/Latino, N (%) | 261 (27.8) | 300 (32.1) |
| Mean smoking history (pack-years) (± SD) | 40.5 (23.4) | 40.3 (27.2) |
| Current smokers (%) | 30.0 | 29.5 |
| Chronic Bronchitis (%) | 95.0 | 99.9 |
| Emphysema (%) | 32.6 | 30.4 |
| Mean number of moderate Exacerbations treated with either systemic corticosteroids and/or antibiotics. or severeExacerbations requiring hospitalization or observation for over 24 hours in an emergency department or urgent care facility. exacerbations in previous year (± SD) |
2.3 (1.0) | 2.1 (0.9) |
| Background COPD medications at randomization: ICS/LAMA/LABA (%) |
97.6 | 98.8 |
| Mean post-bronchodilator FEV1/FVC ratio (± SD) | 0.49 (0.12) | 0.50 (0.12) |
| Mean post-bronchodilator FEV1 (L) (± SD) | 1.40 (0.47) | 1.45 (0.49) |
| Mean percent predicted post-bronchodilator FEV1 (%) (± SD) | 50.6 (13.1) | 50.1 (12.6) |
| Mean SGRQ total score (± SD) | 48.4 (17.4) | 51.5 (17.0) |
| Mean screening blood eosinophil count Reported screening eosinophil value is the highest values from up to three retests (cells/mcL) (± SD) |
521 (307) | 538 (333) |
| Mean baseline blood eosinophil count Reported baseline eosinophil value was obtained within 4 weeks of screening value (cells/mcL) (± SD) |
401 (298) | 407 (336) |
1.3 Chronic Rhinosinusitis With Nasal Polyps
DUPIXENT is indicated as an add-on maintenance treatment in adult and pediatric patients aged 12 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP).
2.7 Recommended Dosage for Prurigo Nodularis
The recommended dosage of DUPIXENT for adult patients is an initial dose of 600 mg (two 300 mg injections) followed by 300 mg given every 2 weeks (Q2W).
14.3 Chronic Rhinosinusitis With Nasal Polyps
The efficacy of DUPIXENT as an add-on maintenance treatment in adults with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP) was evaluated in two randomized, double-blind, parallel-group, multicenter, placebo-controlled studies (SINUS-24 (NCT02912468) and SINUS-52 (NCT02898454)) in 724 adult subjects 18 years of age and older on background intranasal corticosteroids (INCS). These studies included subjects with CRSwNP despite prior sino-nasal surgery or treatment with, or who were ineligible to receive or were intolerant to, systemic corticosteroids in the past 2 years. Subjects with chronic rhinosinusitis without nasal polyps were not included in these trials. Rescue with systemic corticosteroids or surgery was allowed during the studies at the investigator's discretion. In SINUS-24, a total of 276 subjects were randomized to receive either 300 mg DUPIXENT (N=143) or placebo (N=133) every 2 weeks for 24 weeks. In SINUS-52, 448 subjects were randomized to receive either 300 mg DUPIXENT (N=150) every 2 weeks for 52 weeks, 300 mg DUPIXENT (N=145) every 2 weeks until Week 24 followed by 300 mg DUPIXENT every 4 weeks until Week 52, or placebo (N=153). All subjects had evidence of sinus opacification on the Lund Mackay (LMK) sinus CT scan and 73% to 90% of subjects had opacification of all sinuses. Subjects were stratified based on their histories of prior surgery and co-morbid asthma/nonsteroidal anti-inflammatory drug exacerbated respiratory disease (NSAID-ERD). A total of 63% of subjects reported previous sinus surgery, with a mean number of 2.0 prior surgeries, 74% used systemic corticosteroids in the previous 2 years with a mean number of 1.6 systemic corticosteroid courses in the previous 2 years, 59% had co-morbid asthma, and 28% had NSAID-ERD.
The co-primary efficacy endpoints were change from baseline to Week 24 in bilateral endoscopic nasal polyps score (NPS; 0-8 scale) as graded by central blinded readers, and change from baseline to Week 24 in nasal congestion/obstruction score averaged over 28 days (NC; 0-3 scale), as determined by subjects using a daily diary. For NPS, polyps on each side of the nose were graded on a categorical scale (0=no polyps; 1=small polyps in the middle meatus not reaching below the inferior border of the middle turbinate; 2=polyps reaching below the lower border of the middle turbinate; 3=large polyps reaching the lower border of the inferior turbinate or polyps medial to the middle turbinate; 4=large polyps causing complete obstruction of the inferior nasal cavity). The total score was the sum of the right and left scores. Nasal congestion was rated daily by the subjects on a 0 to 3 categorical severity scale (0=no symptoms; 1=mild symptoms; 2=moderate symptoms; 3=severe symptoms).
In both studies, key secondary endpoints at Week 24 included change from baseline in: LMK sinus CT scan score, daily loss of smell, and 22-item sino-nasal outcome test (SNOT-22). The LMK sinus CT scan score evaluated the opacification of each sinus using a 0 to 2 scale (0=normal; 1=partial opacification; 2=total opacification) deriving a maximum score of 12 per side and a total maximum score of 24 (higher scores indicate more opacification). Loss of smell was scored reflectively by the subject every morning on a 0-3 scale (0=no symptoms, 1=mild symptoms, 2=moderate symptoms, 3=severe symptoms). SNOT-22 includes 22 items assessing symptoms and symptom impact associated with CRSwNP with each item scored from 0 (no problem) to 5 (problem as bad as it can be) with a global score ranging from 0 to 110. SNOT-22 had a 2 week recall period. In the pooled efficacy results, the reduction in the proportion of subjects rescued with systemic corticosteroids and/or sino-nasal surgery (up to Week 52) were evaluated.
The demographics and baseline characteristics of these 2 trials are provided in Table 28 below.
| Parameter | SINUS-24 (N=276) |
SINUS-52 (N=448) |
|---|---|---|
| SD = standard deviation; AM = morning; NPS = nasal polyps score; SNOT-22 = 22-item sino-nasal outcome test; NSAID-ERD = asthma/nonsteroidal anti-inflammatory drug exacerbated respiratory disease | ||
| Mean age (years) (SD) | 50 (13) | 52 (12) |
| % Male | 57 | 62 |
| Mean CRSwNP duration (years) (SD) | 11 (9) | 11 (10) |
| Subjects with ≥1 prior surgery (%) | 72 | 58 |
| Subjects with systemic corticosteroid use in the previous 2 years (%) | 65 | 80 |
| Mean Bilateral endoscopic NPS Higher scores indicate greater disease severity (SD), range 0-8 |
5.8 (1.3) | 6.1 (1.2) |
| Mean Nasal congestion (NC) score (SD), range 0-3 | 2.4 (0.6) | 2.4 (0.6) |
| Mean LMK sinus CT total score (SD), range 0-24 | 19 (4.4) | 18 (3.8) |
| Mean loss of smell score (AM), (SD) range 0-3 | 2.7 (0.5) | 2.8 (0.5) |
| Mean SNOT-22 total score (SD), range 0-110 | 49.4 (20.2) | 51.9 (20.9) |
| Mean blood eosinophils (cells/mcL) (SD) | 440 (330) | 430 (350) |
| Mean total IgE IU/mL (SD) | 212 (276) | 240 (342) |
| Atopic Medical History % Overall |
75 | 82 |
| Asthma (%) | 58 | 60 |
| NSAID-ERD (%) | 30 | 27 |
Clinical Response (SINUS-24 and SINUS-52)
The results for primary endpoints in CRSwNP studies are presented in Table 29.
| SINUS-24 | SINUS-52 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Placebo (n=133) |
DUPIXENT 300 mg Q2W (n=143) |
LS mean difference vs. Placebo (95% CI) |
Placebo (n=153) |
DUPIXENT 300 mg Q2W (n=295) |
LS mean difference vs. Placebo (95% CI) |
|||||
| Primary Endpoints at Week 24 | ||||||||||
| Scores | Baseline mean | LS mean change | Baseline mean | LS mean change | Baseline mean | LS mean change | Baseline mean | LS mean change | ||
| A reduction in score indicates improvement. NPS = nasal polyps score; NC = nasal congestion/obstruction |
||||||||||
| NPS | 5.86 | 0.17 | 5.64 | -1.89 | -2.06 (-2.43, -1.69) |
5.96 | 0.10 | 6.18 | -1.71 | -1.80 (-2.10, -1.51) |
| NC | 2.45 | -0.45 | 2.26 | -1.34 | -0.89 (-1.07, -0.71) |
2.38 | -0.38 | 2.46 | -1.25 | -0.87 (-1.03, -0.71) |
Statistically significant efficacy was observed in SINUS-52 with regard to improvement in bilateral endoscopic NPS score at week 24 and week 52 (see Figure 11).
Figure 11: LS Mean Change from Baseline in Bilateral Nasal Polyps Score (NPS) up to Week 52 in Subjects 18 Years of Age and Older with CRSwNP (SINUS-52 - ITT Population)
Similar results were seen in SINUS-24 at Week 24. In the post-treatment period when subjects were off DUPIXENT, the treatment effect diminished over time (see Figure 12).
Figure 12: LS Mean Change from Baseline in Bilateral Nasal Polyps Score (NPS) up to Week 48 in Subjects 18 Years of Age and Older with CRSwNP (SINUS-24 - ITT Population)
At Week 52, the LS mean difference for nasal congestion in the DUPIXENT group versus placebo was -0.98 (95% CI -1.17, -0.79). In both studies, significant improvements in nasal congestion were observed as early as the first assessment at Week 4. The LS mean difference for nasal congestion at Week 4 in the DUPIXENT group versus placebo was -0.41 (95% CI: -0.52, -0.30) in SINUS-24 and -0.37 (95% CI: -0.46, -0.27) in SINUS-52.
A significant decrease in the LMK sinus CT scan score was observed. The LS mean difference for LMK sinus CT scan score at Week 24 in the DUPIXENT group versus placebo was -7.44 (95% CI: -8.35, -6.53) in SINUS-24 and -5.13 (95% CI: -5.80, -4.46) in SINUS-52. At Week 52, in SINUS-52 the LS mean difference for LMK sinus CT scan score in the DUPIXENT group versus placebo was -6.94 (95% CI: -7.87, -6.01).
Dupilumab significantly improved the loss of smell compared to placebo. The LS mean difference for loss of smell at Week 24 in the DUPIXENT group versus placebo was -1.12 (95% CI: -1.31, -0.93) in SINUS-24 and -0.98 (95% CI: -1.15, -0.81) in SINUS-52. At Week 52, the LS mean difference for loss of smell in the DUPIXENT group versus placebo was -1.10 (95% CI -1.31, -0.89). In both studies, significant improvements in daily loss of smell severity were observed as early as the first assessment at Week 4.
Dupilumab significantly decreased sino-nasal symptoms as measured by SNOT-22 compared to placebo. The LS mean difference for SNOT-22 at Week 24 in the DUPIXENT group versus placebo was -21.12 (95% CI: -25.17, -17.06) in SINUS-24 and -17.36 (95% CI: -20.87, -13.85) in SINUS-52. At Week 52, the LS mean difference in the DUPIXENT group versus placebo was -20.96 (95% CI -25.03, -16.89).
In the pre-specified multiplicity-adjusted pooled analysis of two studies, treatment with DUPIXENT resulted in significant reduction of systemic corticosteroid use and need for sino-nasal surgery versus placebo (HR of 0.24; 95% CI: 0.17, 0.35) (see Figure 13). The proportion of subjects who required systemic corticosteroids was reduced by 74% (HR of 0.26; 95% CI: 0.18, 0.38). The total number of systemic corticosteroid courses per year was reduced by 75% (RR of 0.25; 95% CI: 0.17, 0.37). The proportion of subjects who required surgery was reduced by 83% (HR of 0.17; 95% CI: 0.07, 0.46).
Figure 13: Kaplan Meier Curve for Time to First Systemic Corticosteroid Use and/or Sino-Nasal Surgery During Treatment Period in Subjects 18 Years of Age and Older with CRSwNP (SINUS-24 and SINUS-52 Pooled - ITT Population)
The effects of DUPIXENT on the primary endpoints of NPS and nasal congestion and the key secondary endpoint of LMK sinus CT scan score were consistent in subjects with prior surgery and without prior surgery.
In subjects with co-morbid asthma, improvements in pre-bronchodilator FEV1 were similar to subjects in the asthma program.
2.10 Recommended Dosage for Bullous Pemphigoid
The recommended dosage of DUPIXENT for adult patients is an initial dose of 600 mg (two 300 mg injections) followed by 300 mg given every other week (Q2W).
Principal Display Panel 300 Mg/2 Ml Pen Carton
NDC 0024-5915-02
Rx Only
DUPIXENT®
(dupilumab)
Injection
Single-dose Pre-filled Pen
300 mg/2 mL (150 mg/mL)
For Subcutaneous Use Only.
2
Single-dose
Pre-filled Pens
Keep out of reach of children. Do not use
after expiration. Do not use if seal is broken
or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Do NOT freeze. Do NOT heat.
Dosage and Administration: See package insert for dosage information
and directions for use.
300 mg/2 mL
Per Pre-filled Pen
sanofi | REGENERON
2.6 Recommended Dosage for Eosinophilic Esophagitis
The recommended dosage of DUPIXENT for adult and pediatric patients 1 year of age and older, weighing at least 15 kg, is specified in Table 5.
| Body Weight | Recommended Dosage |
|---|---|
| 15 to less than 30 kg | 200 mg every 2 weeks (Q2W) |
| 30 to less than 40 kg | 300 mg every 2 weeks (Q2W) |
| 40 kg or more | 300 mg every week (QW) |
Principal Display Panel 200 Mg/1.14 Ml Pen Carton
NDC 0024-5919-02
Rx Only
DUPIXENT®
(dupilumab)
Injection
Single-dose Pre-filled Pen
200 mg/1.14 mL (175 mg/mL)
For Subcutaneous Use Only.
2
Single-dose
Pre-filled Pens
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in
the original carton. Do not freeze. Do not heat.
Dosage and Administration: See package insert
for dosage information and directions for use.
200 mg/1.14 mL
Per Pre-filled Pen
sanofi | REGENERON
Principal Display Panel 300 Mg/2 Ml Syringe Carton
NDC 0024-5914-01
Rx Only
DUPIXENT®
(dupilumab)
Injection
300 mg/2 mL (150 mg/mL)
For Subcutaneous Use Only.
300 mg/2 mL
Per Pre-filled Syringe
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
sanofi | REGENERON
Principal Display Panel 100 Mg/0.67 Ml Syringe Carton
NDC 0024-5911-02
Rx Only
DUPIXENT®
(dupilumab)
Injection
100 mg/0.67 mL (150 mg/mL)
For Subcutaneous Use Only.
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
100 mg/0.67 mL
sanofi | REGENERON
Principal Display Panel 200 Mg/1.14 Ml Syringe Carton
NDC 0024-5918-01
Rx Only
DUPIXENT®
(dupilumab)
Injection
200 mg/1.14 mL (175 mg/mL)
For Subcutaneous Use Only.
200 mg/1.14 mL
Per Pre-filled Syringe
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
sanofi | REGENERON
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been conducted to evaluate the carcinogenic or mutagenic potential of dupilumab.
No effects on fertility parameters such as reproductive organs, menstrual cycle length, or sperm analysis were observed in sexually mature mice that were subcutaneously administered a homologous antibody against IL-4Rα at doses up to 200 mg/kg/week.
2.8 Recommended Dosage for Chronic Obstructive Pulmonary Disease
The recommended dosage of DUPIXENT for adult patients is 300 mg given every 2 weeks (Q2W).
5.5 Risk Associated With Abrupt Reduction of Corticosteroid Dosage
Do not discontinue systemic, topical, or inhaled corticosteroids abruptly upon initiation of therapy with DUPIXENT. Reductions in corticosteroid dose, if appropriate, should be gradual and performed under the direct supervision of a healthcare provider. Reduction in corticosteroid dose may be associated with systemic withdrawal symptoms and/or unmask conditions previously suppressed by systemic corticosteroid therapy.
2.5 Recommended Dosage for Chronic Rhinosinusitis With Nasal Polyps
The recommended dosage of DUPIXENT for adult and pediatric patients 12 years of age and older is 300 mg given every 2 weeks (Q2W).
5.4 Acute Symptoms of Asthma Or Chronic Obstructive Pulmonary Disease Or Acute Deteriorating Disease
DUPIXENT should not be used to treat acute symptoms or acute exacerbations of asthma or COPD. Do not use DUPIXENT to treat acute bronchospasm or status asthmaticus. Patients should seek medical advice if their asthma or COPD remains uncontrolled or worsens after initiation of treatment with DUPIXENT.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
DUPIXENT is not indicated for the relief of acute bronchospasm or status asthmaticus.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: February 2026 | ||
|
PATIENT INFORMATION DUPIXENT® (DU-pix-ent) (dupilumab) injection, for subcutaneous use |
|||
|
What is DUPIXENT?
DUPIXENT is a prescription medicine used: |
|||
|
|||
|
DUPIXENT is not used to relieve sudden breathing problems and will not replace an inhaled rescue medicine or to treat any other forms of hives (urticaria). DUPIXENT works by blocking two proteins that contribute to a type of inflammation that plays a major role in AD, asthma, CRSwNP, EoE, PN, COPD, CSU, BP, and AFRS. It is not known if DUPIXENT is safe and effective in children with: |
|||
|
|
||
| Do not use DUPIXENT if you are allergic to dupilumab or to any of the ingredients in DUPIXENT. See the end of this Patient Information leaflet for a complete list of ingredients in DUPIXENT. | |||
Before using DUPIXENT, tell your healthcare provider about all your medical conditions, including if you:
Especially tell your healthcare provider if you:
|
|||
How should I use DUPIXENT?
|
|||
|
What are the possible side effects of DUPIXENT? DUPIXENT can cause serious side effects, including:
|
|||
|
|
|
|
|
|||
|
|
||
|
|||
|
|
||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of DUPIXENT. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store DUPIXENT?
|
|||
|
General information about the safe and effective use of DUPIXENT.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DUPIXENT for a condition for which it was not prescribed. Do not give DUPIXENT to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DUPIXENT that is written for health professionals. |
|||
|
What are the ingredients in DUPIXENT?
Active ingredient: dupilumab Inactive ingredients: L-arginine hydrochloride, L-histidine, polysorbate 80, sodium acetate, sucrose, and water for injection. Manufactured by: Regeneron Pharmaceuticals, Inc., Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Morristown, NJ 07960) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT® is a registered trademark of Sanofi Biotechnology / © 2026 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. For more information about DUPIXENT, go to www.DUPIXENT.com or call 1-844-DUPIXENT (1-844-387-4936). |
Section 43683-2 (43683-2)
| Indications and Usage, Chronic Spontaneous Urticaria (1.7) | 04/2025 |
| Indications and Usage, Bullous Pemphigoid (1.8) | 06/2025 |
| Indications and Usage, Allergic Fungal Rhinosinusitis (1.9) | 02/2026 |
| Dosage and Administration, Chronic Spontaneous Urticaria (2.9) | 04/2025 |
| Dosage and Administration, Bullous Pemphigoid (2.10) | 06/2025 |
| Dosage and Administration, Allergic Fungal Rhinosinusitis (2.11) | 02/2026 |
| Dosage and Administration, Missed Doses (2.12) | 06/2025 |
| Warnings and Precautions, Hypersensitivity (5.1) | 06/2025 |
| Warnings and Precautions, Conjunctivitis and Keratitis (5.2) | 02/2026 |
| Warnings and Precautions (5.2, 5.3, 5.7, 5.8) | 04/2025 |
Section 44425-7 (44425-7)
Storage and Handling
DUPIXENT is sterile and preservative-free. Discard any unused portion.
Store refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light.
If necessary, DUPIXENT may be kept at room temperature up to 25°C (77°F) for a maximum of 14 days. Do not store above 25°C (77°F). After removal from the refrigerator, DUPIXENT must be used within 14 days or discarded.
Do not expose DUPIXENT to heat or direct sunlight.
Do NOT freeze. Do NOT shake.
Section 59845-8 (59845-8)
Instructions for Use
DUPIXENT® (DU-pix-ent)
(dupilumab)
injection, for subcutaneous use
Single-Dose Pre-filled Pen (300 mg/2 mL)
This Instructions for Use contains information on how to inject DUPIXENT.
Read this Instructions for Use before using the DUPIXENT Pre-filled Pen. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself for the first time. Keep this Instructions for Use. Call your healthcare provider if you have any questions.
This DUPIXENT Pre-filled Pen is only for use in adults and children aged 2 years and older.
This DUPIXENT Pre-filled Pen is a single-dose device. It contains 300 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Pre-filled Pen are shown below:
Important Information
|
Storing DUPIXENT
|
| A. Get ready to inject | |
|---|---|
| A1. Gather supplies | |
| Find a clean, flat work surface. Make sure you have the following supplies: |
|
| A2. Check the Pen | |
|
|
| A3. Look at the Label | |
|
|
|
| A4. Check the Medicine | |
|---|---|
|
| A5. Wait 45 minutes | |
|---|---|
|
| B. Choose and prepare your injection site | |
|---|---|
| B1. Wash your hands well with soap and water | |
| B2. Choose an injection site | |
|
|
| B3. Prepare the injection site | |
|---|---|
|
|
| C. Give the injection | |
| C1. Remove Green Cap | |
|
|
| C2. Pinch Skin and Place | ||
|---|---|---|
Pinch the skin before and during the injection.
|
||
| C3. Press down ➜ Watch Window turn fully yellow ➜ Then count to 5 | ||
Press and hold the DUPIXENT Pre-filled Pen firmly against the skin until you cannot see the Yellow Needle Cover.
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and watch the window:
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and count to 5 to make sure you get your full dose. |
| Pinching of the skin is not needed for adults and children aged 12 years and older. | |
|---|---|
| C4. Remove | |
|
| D. Dispose of used DUPIXENT Pre-filled Pen | |
|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: July 2023 |
|
How to Dispose of (throw away) DUPIXENT Pre-filled Pen:
Put the used DUPIXENT Pre-filled Pens and Green Caps in a FDA-cleared sharps disposal container right away after use. Do not dispose of (throw away) DUPIXENT Pre-filled Pens and Green Caps in your household trash. If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Keep your sharps disposal container out of the reach of children. |
|
|
|
|
|
Manufactured by: Regeneron Pharmaceuticals, Inc. Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT ® is a registered trademark of Sanofi Biotechnology © 2023 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. |
1.2 Asthma
DUPIXENT is indicated as an add-on maintenance treatment of adult and pediatric patients aged 6 years and older with moderate-to-severe asthma characterized by an eosinophilic phenotype or with oral corticosteroid dependent asthma.
14.2 Asthma
The asthma development program for patients aged 12 years and older included three randomized, double-blind, placebo-controlled, parallel-group, multicenter trials (DRI12544 (NCT01854047), QUEST (NCT02414854), and VENTURE (NCT02528214)) of 24 to 52 weeks in treatment duration which enrolled a total of 2888 subjects. Subjects enrolled in DRI12544 and QUEST were required to have a history of 1 or more asthma exacerbations that required treatment with systemic corticosteroids or emergency department visit or hospitalization for the treatment of asthma in the year prior to trial entry. Subjects enrolled in VENTURE required dependence on daily oral corticosteroids in addition to regular use of high-dose inhaled corticosteroids plus an additional controller(s). In all 3 trials, subjects were enrolled without requiring a minimum baseline blood eosinophil count. In QUEST and VENTURE, subjects with screening blood eosinophil level of >1500 cells/mcL (<1.3%) were excluded. DUPIXENT was administered as add-on to background asthma treatment. Subjects continued background asthma therapy throughout the duration of the studies, except in VENTURE in which OCS dose was tapered as described below.
10 Overdosage (10 OVERDOSAGE)
There is no specific treatment for DUPIXENT overdose. In the event of overdosage, contact Poison Control (1-800-222-1222) for the latest recommendations and monitor the patient for any signs or symptoms of adverse reactions and institute appropriate symptomatic treatment immediately.
5.7 Psoriasis
Cases of new-onset psoriasis have been reported with the use of DUPIXENT for the treatment of atopic dermatitis and asthma, including in patients without a family history of psoriasis. In postmarketing reports, onset of psoriasis varied from weeks to months after the first dose of DUPIXENT and resulted in partial or complete resolution of psoriasis with discontinuation of dupilumab, with or without use of supplemental treatment for psoriasis (topical or systemic). Those who continued on dupilumab received supplemental treatment for psoriasis to improve associated symptoms. Advise patients to report new-onset psoriasis symptoms to their healthcare provider. If symptoms persist or worsen, consider dermatologic evaluation and/or discontinuation of DUPIXENT.
11 Description (11 DESCRIPTION)
Dupilumab, an interleukin-4 receptor alpha antagonist, is a human monoclonal antibody of the IgG4 subclass that binds to the IL-4Rα subunit and inhibits IL-4 and IL-13 signaling. Dupilumab has an approximate molecular weight of 147 kDa.
Dupilumab is produced by recombinant DNA technology in Chinese Hamster Ovary cell suspension culture.
DUPIXENT (dupilumab) Injection is supplied as a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution for subcutaneous injection. DUPIXENT is provided as either a single-dose pre-filled syringe with needle shield or a single-dose pre-filled pen in a siliconized Type-1 clear glass syringe. The needle cap is not made with natural rubber latex.
Each 300 mg pre-filled syringe or pre-filled pen delivers 300 mg dupilumab in 2 mL which also contains L-arginine hydrochloride (10.5 mg), L-histidine (6.2 mg), polysorbate 80 (4 mg), sodium acetate (2 mg), sucrose (100 mg), and water for injection, pH 5.9.
Each 200 mg pre-filled syringe or pre-filled pen delivers 200 mg dupilumab in 1.14 mL which also contains L-arginine hydrochloride (12 mg), L-histidine (3.5 mg), polysorbate 80 (2.3 mg), sodium acetate (1.2 mg), sucrose (57 mg), and water for injection, pH 5.9.
2.12 Missed Doses
If a weekly dose is missed, administer the dose as soon as possible, and start a new weekly schedule from the date of the last administered dose.
If an every 2 week dose is missed, administer the injection within 7 days from the missed dose and then resume the patient's original schedule. If the missed dose is not administered within 7 days, administer the dose, starting a new schedule based on this date.
If an every 4 week dose is missed, administer the injection within 7 days from the missed dose and then resume the patient's original schedule. If the missed dose is not administered within 7 days, administer the dose, starting a new schedule based on this date.
5.10 Vaccinations
Consider completing all age-appropriate vaccinations as recommended by current immunization guidelines prior to initiating treatment with DUPIXENT. Avoid use of live vaccines during treatment with DUPIXENT. It is unknown if administration of live vaccines during DUPIXENT treatment will impact the safety or effectiveness of these vaccines. Limited data are available regarding coadministration of DUPIXENT with non-live vaccines [see Clinical Pharmacology (12.2)].
8.5 Geriatric Use
Of the 1539 subjects with AD exposed to DUPIXENT in a dose-ranging study and placebo-controlled trials, 70 subjects were 65 years or older. Clinical trials of DUPIXENT in AD did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger subjects.
Of the 1977 subjects with asthma exposed to DUPIXENT, a total of 240 subjects were 65 years or older. Efficacy and safety in this age group were similar to the overall study population.
Of the 440 subjects with CRSwNP exposed to DUPIXENT, a total of 79 subjects were 65 years or older. Efficacy and safety in this age group were similar to the overall study population.
Clinical studies of DUPIXENT in EoE did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger adult subjects.
Of the 152 subjects with PN exposed to DUPIXENT, a total of 37 were 65 years or older including 8 subjects 75 years or older. Clinical trials did not include a sufficient number of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
Of the 1874 subjects with COPD randomized in clinical trials of DUPIXENT, a total of 1072 were 65 years or older, while 244 subjects were 75 years or older. No overall differences in safety or effectiveness of DUPIXENT have been observed between subjects 65 years of age and older and younger adult subjects.
Of the 198 subjects with CSU exposed to DUPIXENT, a total of 30 subjects were 65 years or older, including 7 subjects 75 years or older. Efficacy and safety in subjects 65 years or older were similar to the overall study population.
Of the 53 subjects with BP exposed to DUPIXENT, a total of 40 were 65 years or older, including 22 subjects 75 years or older. Ten percent of subjects aged 65 years and older treated with DUPIXENT had an adverse reaction of vision blurred compared to zero in younger adult subjects.
Clinical studies of DUPIXENT in AFRS did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the trials described below with the incidence of anti-drug antibodies in other trials, including those of DUPIXENT or of other dupilumab products.
The anti-drug antibody (ADA) and neutralizing antibody (NAb) incidence rates in subjects treated with DUPIXENT are presented in Table 16.
| Indication and Population | Dose and Duration of Treatment | Anti-drug Antibody (ADA) | Neutralizing Antibody NAb Neutralizing potential is assessed only for ADA-positive samples. The NAb incidence is reported as a percentage of subjects with positive ADA response.
|
|
|---|---|---|---|---|
| Positive ADA Positive ADA: Includes subjects with a treatment-emergent response, defined as a negative or missing result at baseline with at least 1 positive post-baseline result in the ADA assay, and subjects with a treatment-boosted response, defined as a post-baseline ADA-positive result that is ≥ 4-fold higher than baseline titer levels, when baseline results are positive
|
Persistent ADA Persistent ADA: A treatment-emergent ADA positive response with 2 or more consecutive ADA positive samples separated by a greater than 12-week period (>84 days), with no ADA negative result in between.
|
|||
| Atopic Dermatitis | ||||
| Adult | 300 mg Q2W for 52 weeks | 7/105 (7%) | 2/105 (2%) | 1/7 (14%) |
| 12 to 17 years of age | 300 mg or 200 mg Q2W for 16 weeks | 13/81 (16%) | 2/81 (3%) | 4/13 (31%) |
| 6 to 11 years of age | 200 mg Q2W or 300 mg Q4W for 16 weeks | 3/171 (2%) | 0/171 (0%) | 1/3 (33%) |
| 6 months to 5 years of age | 200 mg Q4W, or 300 mg Q4W for 16 weeks | 1/74 (1%) | 0/74 (0%) | 0/1 (0%) |
| Asthma | ||||
| Adult and 12 years of age or older | 300 mg Q2W for 52 weeks | 33/626 (5%) | 13/626 (2%) | 14/33 (42%) |
| 200 mg Q2W for 52 weeks | 58/625 (9%) | 26/625 (4%) | 27/58 (47%) | |
| 6 to 11 years of age | 100 mg Q2W or 200 mg Q2W up to 52 weeks | 17/269 (6%) | 9/269 (3%) | 6/17 (35%) |
| Chronic Rhinosinusitis with Nasal Polyps | ||||
| Adult | 300 mg Q2W for 52 weeks | 8/148 (5%) | 3/148 (2%) | 5/8 (63%) |
| Eosinophilic Esophagitis | ||||
| Adult and 12 years of age or older | 300 mg QW for 52 weeks | 1/108 (1%) | 0/108 (0%) | 0/1 (0%) |
| 1 to 11 years of age | 200 mg Q2W or 300 mg Q2W for 52 weeks Immunogenicity was reported on pooled data for pediatric subjects 1 to 11 years of age with EoE receiving SC dupilumab 100 mg Q2W (in subjects weighing ≥5 kg to <15kg), 200 mg Q2W (in subjects weighing ≥15 kg to <30 kg), 300 mg Q2W (in subjects weighing ≥30 kg to <60 kg), and 300 mg QW (in subjects weighing ≥60 kg).
|
1/37 (3%) | 0/37 (0%) | 0/1 (0%) One subject 1 to 11 years of age with EoE with pre-existing immunoreactivity that was positive for neutralizing antibodies was excluded from the NAb positive subjects.
|
| Prurigo Nodularis | ||||
| Adult | 300 mg Q2W for 24 weeks | 11/143 (8%) | 2/143 (1%) | 4/11 (36%) |
| Chronic Obstructive Pulmonary Disease | ||||
| Adult | 300 mg Q2W for 52 weeks | 78/923 (8%) | 24/922 (3%) | 28/78 (36%) |
| Chronic Spontaneous Urticaria | ||||
| Adult and 12 years of age or older | 200 mg Q2W or 300 mg Q2W through 24 weeks | 9/193 (5%) | 1/193 (1%) | 2/9 (22%) |
| Bullous Pemphigoid | ||||
| Adult | 300 mg Q2W up to 52 weeks | 2/52 (4%) | 0/52 (0%) | 2/2 (100%) |
| Allergic Fungal Rhinosinusitis | ||||
| Adult and 6 years of age or older | 300 mg Q2W or 200 mg Q2W for 52 weeks | 2/22 (9%) | 1/22 (4.5%) | 0/2 (0%) |
Two adult subjects with AD who experienced high titer antibody responses developed serum sickness or serum sickness-like reactions during DUPIXENT therapy [see Warnings and Precautions (5.1)].
The antibody titers detected in subjects who received DUPIXENT were mostly low. In subjects who received DUPIXENT, development of high titer antibodies to DUPIXENT was associated with lower serum dupilumab concentrations [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
DUPIXENT is contraindicated in patients who have known hypersensitivity to dupilumab or any excipients of DUPIXENT [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Conjunctivitis and Keratitis [see Warnings and Precautions (5.2)]
- Psoriasis [see Warnings and Precautions (5.7)]
- Arthralgia and Psoriatic Arthritis [see Warnings and Precautions (5.8)]
- Parasitic (Helminth) Infections [see Warnings and Precautions (5.9)]
5.1 Hypersensitivity
Hypersensitivity reactions, including anaphylaxis, acute generalized exanthematous pustulosis (AGEP), serum sickness or serum sickness-like reactions, angioedema, generalized urticaria, rash, erythema nodosum and erythema multiforme have been reported. A case of AGEP was reported in an adult subject who participated in the bullous pemphigoid development program. If a clinically significant hypersensitivity reaction occurs, institute appropriate therapy and discontinue DUPIXENT [see Adverse Reactions (6.1, 6.2) and Clinical Pharmacology (12.6)].
Instructions for Use (INSTRUCTIONS FOR USE)
Read this Instructions for Use before using the DUPIXENT Pre-filled Syringe. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself the first time. Keep these instructions for future use. Call your healthcare provider if you have any questions.
This device is a Single-Dose Pre-filled Syringe (called "DUPIXENT Syringe" in these instructions). It contains 300 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Syringe are shown below:
| Important Information | |
|---|---|
|
|
How should I store DUPIXENT?
|
Step 1: Remove
Remove the DUPIXENT Syringe from the carton by holding the middle of the Syringe Body.
Do not pull off the Needle Cap until you are ready to inject.
Do not use the DUPIXENT Syringe if it has been dropped on a hard surface or damaged.
Step 2: Prepare
Ensure you have the following:
- the DUPIXENT Pre-filled Syringe
- 1 alcohol wipe*
- 1 cotton ball or gauze*
- a sharps disposal container* (See Step 13)
*Items not included in the carton
Step 3: Check
When you receive your DUPIXENT Syringes, always check to see that:
- you have the correct medicine and dose.
- the expiration date on the Single-Dose Pre-filled Syringe has not passed.
Do not use the DUPIXENT Syringe if the expiration date has passed.
Step 4: Inspect
Look at the medicine through the Viewing Window on the DUPIXENT Syringe:
Check to see if the liquid is clear and colorless to pale yellow.
Note: You may see an air bubble, this is normal.
Do not use the DUPIXENT Syringe if the liquid is discolored or cloudy, or if it contains visible flakes or particles.
Step 5: Wait 45 minutes
Lay the DUPIXENT Syringe on a flat surface and let it naturally warm to room temperature for at least 45 minutes.
Do not heat the DUPIXENT Syringe.
Do not put the DUPIXENT Syringe into direct sunlight.
Do not keep DUPIXENT Syringes at room temperature for more than 14 days. Throw away (dispose of) any DUPIXENT Syringes that have been left at room temperature for longer than 14 days.
Step 6: Choose your injection site
- You can inject into your thigh or stomach, except for the 2 inches (5 cm) around your belly button (navel).
- If a caregiver injects your dose, they can also use the outer area of the upper arm.
- Choose a different site each time you inject DUPIXENT.
Do not inject into skin that is tender, damaged, bruised or scarred.
Step 7: Clean
Wash your hands.
Clean the injection site with an alcohol wipe.
Let your skin dry before injecting.
Do not touch the injection site again or blow on it before the injection.
Step 8: Remove Needle Cap
Hold the DUPIXENT Syringe in the middle of the Syringe Body with the Needle pointing away from you and pull off the Needle Cap.
Do not put the Needle Cap back on.
Do not touch the Needle.
Inject your medicine right away after removing the Needle Cap.
Step 9: Pinch
Pinch a fold of skin at the injection site (thigh or stomach, except 2 inches around your belly button, or outer area of the upper arm if injected by your caregiver). The figure below shows an example of pinching a fold of skin on your stomach.
Step 10: Insert
Insert the Needle completely into the fold of the skin at about a 45° angle.
Step 11: Push
Relax the pinch.
Push the Plunger Rod down slowly and steadily as far as it will go until the DUPIXENT Syringe is empty.
Note: You will feel some resistance. This is normal.
Step 12: Release and Remove
Lift your thumb to release the Plunger Rod until the Needle is covered by the Needle Shield and then remove the Syringe from the injection site.
Lightly press a cotton ball or gauze on the injection site if you see any blood.
Do not put the Needle Cap back on.
Do not rub your skin after the injection.
Step 13: Dispose
Put your used Needles, DUPIXENT Syringes, and Needle Caps in a FDA-cleared sharps disposal container right away after use.
Do not dispose of (throw away) Needles, DUPIXENT Syringes, and Needle Caps in your household trash.
If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used Needles and Syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Do not put the Needle Cap back on.
Manufactured by: Regeneron Pharmaceuticals, Inc. Tarrytown, NY 10591
U.S. License No. 1760
Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
DUPIXENT
®
is a registered trademark of Sanofi Biotechnology
© 2023 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: July 2023
Instructions for Use
DUPIXENT® (DU-pix-ent)
(dupilumab)
injection, for subcutaneous use
Single-Dose Pre-filled Pen (200 mg/1.14 mL)
This Instructions for Use contains information on how to inject DUPIXENT.
Read this Instructions for Use before using the DUPIXENT Pre-filled Pen. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself for the first time. Keep this Instructions for Use. Call your healthcare provider if you have any questions.
This DUPIXENT Pre-filled Pen is only for use in adults and children aged 2 years and older.
This DUPIXENT Pre-filled Pen is a single-dose device. It contains 200 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Pre-filled Pen are shown below:
Important Information
|
Storing DUPIXENT
|
| A. Get ready to inject | |
|---|---|
| A1. Gather supplies | |
| Find a clean, flat work surface. Make sure you have the following supplies: |
|
| A2. Check the Pen | |
|
|
| A3. Look at the Label | |
|
|
|
| A4. Check the Medicine | |
|---|---|
|
| A5. Wait 30 minutes | |
|---|---|
|
| B. Choose and prepare your injection site | |
|---|---|
| B1. Wash your hands well with soap and water | |
| B2. Choose an injection site | |
|
|
| B3. Prepare the injection site | |
|---|---|
|
|
| C. Give the injection | |
| C1. Remove Yellow Cap | |
|
|
| C2. Pinch Skin and Place | ||
|---|---|---|
Pinch the skin before and during the injection.
|
||
| C3. Press down ➜ Watch Window turn fully yellow ➜ Then count to 5 | ||
Press and hold the DUPIXENT Pre-filled Pen firmly against the skin until you cannot see the Orange Needle Cover.
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and watch the window:
|
Keep pressing the DUPIXENT Pre-filled Pen against the skin and count to 5 to make sure you get your full dose. |
| Pinching of the skin is not needed for adults and children aged 12 years and older. | |
|---|---|
| C4. Remove | |
|
| D. Dispose of used DUPIXENT Pre-filled Pen | |
|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: July 2023 |
|
How to Dispose of (throw away) DUPIXENT Pre-filled Pen:
Put the used DUPIXENT Pre-filled Pens and Yellow Caps in a FDA-cleared sharps disposal container right away after use. Do not dispose of (throw away) DUPIXENT Pre-filled Pens and Yellow Caps in your household trash. If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Keep your sharps disposal container out of the reach of children. |
|
|
|
|
|
Manufactured by: Regeneron Pharmaceuticals, Inc. Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT ® is a registered trademark of Sanofi Biotechnology © 2023 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. |
1.1 Atopic Dermatitis
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 6 months and older with moderate-to-severe atopic dermatitis (AD) whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids.
1.5 Prurigo Nodularis
DUPIXENT is indicated for the treatment of adult patients with prurigo nodularis (PN).
12.2 Pharmacodynamics
Consistent with inhibition of IL-4 and IL-13 signaling, dupilumab treatment decreased certain biomarkers of inflammation. In asthma subjects, fractional exhaled nitric oxide (FeNO) and circulating concentrations of eotaxin-3, total IgE, allergen specific IgE, TARC, and periostin were decreased relative to placebo. Reductions in these biomarkers were comparable for the 300 mg Q2W and 200 mg Q2W regimens. These markers were near maximal suppression after 2 weeks of treatment, except for IgE which declined more slowly. These effects were sustained throughout treatment. The median percent reduction from baseline in total IgE concentrations with dupilumab treatments was 52% at Week 24 (DRI12544) and 70% at Week 52 (QUEST). For FeNO, the mean percent reduction from baseline at Week 2 was 35% and 24% in DRI12544 and QUEST, respectively, and in the overall safety population, the mean FeNO level decreased to 20 ppb.
A continuous decline in total IgE in serum was observed in CSU trials.
In AFRS subjects, total IgE concentrations showed a progressive decline with dupilumab treatment throughout the 52-week treatment period.
12.3 Pharmacokinetics
The pharmacokinetics of dupilumab is similar in subjects with AD, asthma, CRSwNP, EoE, PN, COPD, CSU, BP, and AFRS.
1.8 Bullous Pemphigoid
DUPIXENT is indicated for the treatment of adult patients with bullous pemphigoid (BP).
14.5 Prurigo Nodularis
The efficacy of DUPIXENT in adults with prurigo nodularis (PN) was evaluated in two 24-week randomized, double-blind, placebo-controlled, multicenter, parallel-group trials (PRIME (NCT04183335) and PRIME 2 (NCT04202679)). These trials enrolled 311 adult subjects with pruritus (WI-NRS ≥ 7 on a scale of 0 to 10) and greater than or equal to 20 nodular lesions. PRIME and PRIME 2 assessed the effect of DUPIXENT on pruritus improvement as well as its effect on PN lesions.
In these two trials, subjects received either subcutaneous DUPIXENT 600 mg (two 300 mg injections) on day 1, followed by 300 mg once every 2 weeks (Q2W) for 24 weeks, or matching placebo.
In these trials, the mean age was 49.5 years, the median weight was 71 kg, 65% of subjects were female, 57% were White, 6% were Black, and 34% were Asian. At baseline, the mean Worst Itch-Numeric Rating Scale (WI-NRS) was 8.5, 66% had 20 to 100 nodules (moderate), and 34% had greater than 100 nodules (severe). Eleven percent (11%) of subjects were taking stable doses of antidepressants at baseline and were instructed to continue taking these medications during the trial. Forty-three percent (43%) had a history of atopy (defined as having a medical history of AD, allergic rhinitis/rhinoconjunctivitis, asthma, or food allergy).
The WI-NRS is comprised of a single item, rated on a scale from 0 ("no itch") to 10 ("worst imaginable itch"). Subjects were asked to rate the intensity of their worst pruritus (itch) over the past 24 hours using this scale. The Investigator's Global Assessment for Prurigo Nodularis-Stage (IGA PN-S) is a scale that measures the approximate number of nodules using a 5-point scale from 0 (clear) to 4 (severe).
Efficacy was assessed with the proportion of subjects with improvement (reduction) in WI-NRS by ≥4 points, the proportion of subjects with IGA PN-S 0 or 1 (the equivalent of 0-5 nodules), and the proportion of subjects who achieved a response in both WI-NRS and IGA PN-S per the criteria described above.
The efficacy results for PRIME and PRIME2 are presented in Table 32 and Figure 14, Figure 15, and Figure 16.
| PRIME | PRIME2 | |||||
|---|---|---|---|---|---|---|
| Placebo (N=76) |
DUPIXENT 300 mg Q2W (N=75) |
Difference (95% CI) for DUPIXENT vs. Placebo | Placebo (N=82) |
DUPIXENT 300 mg Q2W (N=78) |
Difference (95% CI) for DUPIXENT vs. Placebo | |
| Proportion of subjects with both an improvement (reduction) in WI-NRS by ≥4 points from baseline to Week 24 and an IGA PN-S 0 or 1 at Week 24 Subjects who received rescue treatment earlier or had missing data were considered as non-responders.
|
9.2% | 38.7% | 29.6% (16.4, 42.8) |
8.5% | 32.1% | 25.5% (13.1, 37.9) |
| Proportion of subjects with improvement (reduction) in WI-NRS by ≥4 points from baseline at Week 24 | 18.4% | 60.0% | 42.7% (27.8, 57.7) |
19.5% | 57.7% | 42.6% (29.1, 56.1) |
| Proportion of subjects with IGA PN-S 0 or 1 at Week 24 | 18.4% | 48.0% | 28.3% (13.4, 43.2) |
15.9% | 44.9% | 30.8% (16.4, 45.2) |
| Proportion of subjects with improvement (reduction) in WI-NRS by ≥4 points from baseline at Week 12 | 15.8% Not adjusted for multiplicity in PRIME.
|
44.0% | 29.2% (14.5, 43.8) |
22.0% | 37.2% | 16.8% (2.3, 31.2) |
Figure 14: Proportion of Adult Subjects with PN with Both WI-NRS ≥4-point Improvement and IGA PN-S 0 or 1 Over Time in PRIME and PRIME2
Figure 15: Proportion of Adult Subjects with PN with WI-NRS ≥4-point Improvement Over Time in PRIME and PRIME2
Figure 16: Proportion of Adult Subjects with IGA PN-S 0 or 1 Over Time in PRIME and PRIME 2
The efficacy data did not show differential treatment effect across demographic subgroups.
1 Indications and Usage (1 INDICATIONS AND USAGE)
DUPIXENT is an interleukin-4 receptor alpha antagonist indicated:
Atopic Dermatitis
for the treatment of adult and pediatric patients aged 6 months and older with moderate-to-severe AD whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids. (1.1)
Asthma
as an add-on maintenance treatment of adult and pediatric patients aged 6 years and older with moderate-to-severe asthma characterized by an eosinophilic phenotype or with oral corticosteroid dependent asthma. (1.2)
Limitations of Use: Not for the relief of acute bronchospasm or status asthmaticus. (1.2)
Chronic Rhinosinusitis with Nasal Polyps
as an add-on maintenance treatment in adult and pediatric patients aged 12 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP). (1.3)
Eosinophilic Esophagitis
for the treatment of adult and pediatric patients aged 1 year and older, weighing at least 15 kg, with eosinophilic esophagitis (EoE). (1.4)
Prurigo Nodularis
for the treatment of adult patients with prurigo nodularis (PN). (1.5)
Chronic Obstructive Pulmonary Disease
as an add-on maintenance treatment of adult patients with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype. (1.6)
Limitations of Use: Not for the relief of acute bronchospasm. (1.6)
Chronic Spontaneous Urticaria
for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment. (1.7)
Limitations of Use: Not indicated for other forms of urticaria. (1.7)
Bullous Pemphigoid
for the treatment of adult patients with bullous pemphigoid (BP). (1.8)
Allergic Fungal Rhinosinusitis
for the treatment of adult and pediatric patients aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery. (1.9)
14.8 Bullous Pemphigoid
The efficacy of DUPIXENT in adults with bullous pemphigoid (BP) was evaluated in a 52-week randomized, double-blind, parallel-group, multicenter, placebo-controlled trial (ADEPT; NCT04206553). This trial enrolled 106 adult subjects with a Bullous Pemphigoid Disease Area Index (BPDAI) activity score ≥24 on a scale of 0-360 and a weekly average Peak Pruritus NRS score ≥4 on a scale of 0-10.
In this trial, subjects were randomized to receive either subcutaneous DUPIXENT 600 mg (two 300 mg injections) on Day 1, followed by 300 mg every other week (Q2W), or matching placebo for 52 weeks. All subjects were also initiated on a standard regimen of oral corticosteroids (prednisone or prednisolone; 0.5 or 0.75 mg/kg/day) on Day 1. After achieving control of disease activity for 2 weeks, oral corticosteroids (OCS) were tapered with the objective to taper them off no later than Week 16 as long as the control of disease activity was maintained. Subjects who experienced a loss of control of disease activity (new lesions form and existing lesions cease to heal) during OCS taper, or who relapsed post-taper, or who used rescue medications were considered treatment failures. During the OCS taper, subjects were permitted to increase their OCS once, with any subsequent increases being considered rescue therapy. Subjects could be rescued during the trial with topical or oral corticosteroids, non-steroidal immunosuppressive medications, or immunomodulating biologics. Subjects were allowed to continue trial treatment if rescued with topical or oral corticosteroids.
Demographics and baseline characteristics were similar between the treatment arms. Of the 106 subjects enrolled, the mean age was 71.3 years; 53% were female; 68% were White, 2% were Black or African American, and 15% were Asian; 3% of subjects identified as Hispanic or Latino. At baseline, 63% of subjects had prior systemic corticosteroid use for BP. The mean Peak Pruritus NRS score was 7.5 at baseline.
The primary endpoint was the proportion of subjects achieving sustained remission at Week 36. Sustained remission was defined as the achievement of complete remission and off OCS no later than Week 16, absence of disease relapse from the time of completion of the corticosteroid taper to Week 36, and absence of rescue therapy during the 36-week double-blind treatment period. Secondary endpoints included total cumulative dose of OCS and proportion of subjects with a reduction in itch defined as at least a 4-point improvement (reduction) in the Peak Pruritus NRS.
The efficacy results for ADEPT are presented in Table 38. The proportion of subjects that received rescue therapy during the 36-week treatment period was 53% in the DUPIXENT group and 79% in the placebo group.
| DUPIXENT 300 mg Q2W + OCS (N=53) |
Placebo + OCS (N=53) |
Difference (95% CI) for DUPIXENT vs. Placebo | |
|---|---|---|---|
| Proportion of subjects achieving sustained remission Sustained remission was defined as the achievement of complete remission and off OCS no later than Week 16, absence of disease relapse from the time of completion of the corticosteroid taper to Week 36, and absence of rescue therapy during the 36-week double-blind treatment period.
|
18.3% | 6.1% | 12.2% (-0.8, 26.1) |
| Proportion of subjects with improvement (reduction) of ≥4 points in Peak Pruritus NRS from baseline | 38.3% | 10.5% | 27.8% (11.6, 43.4) |
The median (min, max) cumulative dose of OCS at Week 36 was 2.8 g (1.2, 22.7) in the DUPIXENT group compared to 4.1 g (1.5, 23.3) in the placebo group.
12.1 Mechanism of Action
Dupilumab is a human monoclonal IgG4 antibody that inhibits interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling by specifically binding to the IL-4Rα subunit shared by the IL-4 and IL-13 receptor complexes. Dupilumab inhibits IL-4 signaling via the Type I receptor and both IL-4 and IL-13 signaling through the Type II receptor.
Inflammation driven by IL-4 and IL-13 is an important component in the pathogenesis of asthma, AD, CRSwNP, EoE, PN, COPD, CSU, BP, and AFRS. Multiple cell types that express IL-4Rα (e.g., mast cells, basophils, eosinophils, macrophages, lymphocytes, epithelial cells, goblet cells) and inflammatory mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines, chemokines) are involved in inflammation. Blocking IL-4Rα with dupilumab inhibits IL-4 and IL-13 cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE. The mechanism of dupilumab action has not been definitively established.
2.13 Preparation for Use
Before injection, remove DUPIXENT from the refrigerator and allow DUPIXENT to reach room temperature (45 minutes for the 300 mg/2 mL pre-filled syringe or pre-filled pen, and 30 minutes for the 200 mg/1.14 mL pre-filled syringe or pre-filled pen) without removing the needle cap. After removal from the refrigerator, DUPIXENT must be used within 14 days or discarded.
Inspect DUPIXENT visually for particulate matter and discoloration prior to administration. DUPIXENT is a clear to slightly opalescent, colorless to pale yellow solution. Do not use if the liquid contains visible particulate matter, is discolored or cloudy (other than clear to slightly opalescent, colorless to pale yellow). DUPIXENT does not contain preservatives; therefore, discard any unused product remaining in the pre-filled syringe or pre-filled pen.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity: Hypersensitivity reactions including anaphylaxis, acute generalized exanthematous pustulosis (AGEP), serum sickness, angioedema, urticaria, rash, erythema nodosum, and erythema multiforme have occurred. Discontinue DUPIXENT in the event of a hypersensitivity reaction. (5.1)
- Conjunctivitis and Keratitis: Advise patients to report new onset or worsening eye symptoms to their healthcare provider. Consider ophthalmological examination, as appropriate. (5.2)
- Eosinophilic Conditions: Be alert to vasculitic rash, worsening pulmonary symptoms, cardiac complications, kidney injury and/or neuropathy, especially upon reduction of oral corticosteroids. (5.3)
- Reduction of Corticosteroid Dosage: Do not discontinue systemic, topical, or inhaled corticosteroids abruptly upon initiation of DUPIXENT. Decrease steroids gradually, if appropriate. (5.5)
- Psoriasis: Advise patients to report new-onset psoriasis symptoms to their healthcare provider. If symptoms persist or worsen, consider dermatologic evaluation and/or discontinuation of DUPIXENT. (5.7)
- Arthralgia and Psoriatic Arthritis: Advise patients to report new onset joint symptoms to their healthcare provider. If symptoms persist or worsen, consider rheumatological evaluation and/or discontinuation of DUPIXENT. (5.8)
- Parasitic (Helminth) Infections: Treat pre-existing helminth infections before initiating DUPIXENT. If patients become infected while receiving DUPIXENT and do not respond to anti-helminth treatment, discontinue DUPIXENT until the infection resolves. (5.9)
- Vaccinations: Avoid use of live vaccines. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
DUPIXENT is administered by subcutaneous injection. (2.1)
Atopic Dermatitis
Dosage in Adults (2.3):
-
Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W).
Dosage in Pediatric Patients 6 Months to 5 Years of Age (2.3):
| Body Weight | Initial and Subsequent Dosage |
|---|---|
| 5 to less than 15 kg | 200 mg (one 200 mg injection) every 4 weeks (Q4W) |
| 15 to less than 30 kg | 300 mg (one 300 mg injection) every 4 weeks (Q4W) |
Dosage in Pediatric Patients 6 Years of Age and Older (2.3):
| Body Weight | Initial Loading Dose | Subsequent Dosage Q2W – every 2 weeks; Q4W – every 4 weeks
|
|---|---|---|
| 15 to less than 30 kg | 600 mg (two 300 mg injections) | 300 mg Q4W |
| 30 to less than 60 kg | 400 mg (two 200 mg injections) | 200 mg Q2W |
| 60 kg or more | 600 mg (two 300 mg injections) | 300 mg Q2W |
Asthma
Dosage in Adult and Pediatric Patients 12 Years and Older (2.4):
| Initial Loading Dose | Subsequent Dosage |
|---|---|
| 400 mg (two 200 mg injections) | 200 mg every 2 weeks (Q2W) |
| Or | |
| 600 mg (two 300 mg injections) | 300 mg every 2 weeks (Q2W) |
|
Dosage for patients with oral corticosteroid-dependent asthma or with co-morbid moderate-to-severe AD, CRSwNP, or AFRS For pediatric patients 12 to 17 years of age (≥60 kg) and adults with AFRS
|
|
| 600 mg (two 300 mg injections) | 300 mg every 2 weeks (Q2W) |
Dosage in Pediatric Patients 6 to 11 Years of Age (2.4):
| Body Weight | Initial Dose and Subsequent Dosage |
|---|---|
| 15 to less than 30 kg | 300 mg every 4 weeks (Q4W) |
| ≥30 kg | 200 mg every 2 weeks (Q2W) |
For pediatric patients 6 to 11 years old with asthma and co-morbid moderate-to-severe atopic dermatitis, follow the recommended dosage as per Table 2 which includes an initial loading dose. (2.3)
Chronic Rhinosinusitis with Nasal Polyps (2.5):
-
Recommended dosage for adult and pediatric patients 12 years of age and older is 300 mg given every 2 weeks (Q2W).
Eosinophilic Esophagitis (2.6):
| Body Weight | Recommended Dosage in Adult and Pediatric Patients 1 Year and Older, Weighing At Least 15 kg |
|---|---|
| 15 to less than 30 kg | 200 mg every 2 weeks (Q2W) |
| 30 to less than 40 kg | 300 mg every 2 weeks (Q2W) |
| 40 kg or more | 300 mg every week (QW) |
Prurigo Nodularis (2.7):
-
Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W).
Chronic Obstructive Pulmonary Disease (2.8):
-
Recommended dosage for adult patients is 300 mg given every 2 weeks (Q2W).
Chronic Spontaneous Urticaria
Dosage in Adults (2.9):
-
Recommended dosage is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every 2 weeks (Q2W).
Dosage in Pediatric Patients 12 to 17 Years of Age (2.9):
| Body Weight | Initial Loading Dose | Subsequent Dosage |
|---|---|---|
| 30 to less than 60 kg | 400 mg (two 200 mg injections) | 200 mg Q2W |
| 60 kg or more | 600 mg (two 300 mg injections) | 300 mg Q2W |
Bullous Pemphigoid (2.10):
Recommended dosage for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every other week (Q2W). Use DUPIXENT in combination with a tapering course of oral corticosteroids.
Allergic Fungal Rhinosinusitis
Dosage in Adults (2.11):
-
Recommended dosage is 300 mg given every 2 weeks (Q2W).
Dosage in Pediatric Patients 6 to 17 Years of Age (2.11):
| Body Weight | Recommended Dosage |
|---|---|
| a Q2W – every 2 weeks; Q4W – every 4 weeks | |
| 15 to less than 30 kg | 300 mg Q4W |
| 30 to less than 60 kg | 200 mg Q2W |
| 60 kg or more | 300 mg Q2W |
5.3 Eosinophilic Conditions
Patients being treated for asthma may present with clinical features of eosinophilic pneumonia or eosinophilic granulomatosis with polyangiitis (EGPA). These events may be associated with the reduction of oral corticosteroid therapy. Healthcare providers should be alert to vasculitic rash, worsening pulmonary symptoms, cardiac complications, kidney injury, and/or neuropathy presenting in their patients with eosinophilia. Cases of eosinophilic pneumonia were reported in adults who participated in the asthma development program. Cases of EGPA have been reported with DUPIXENT in adults who participated in the asthma development program as well as in adults with co-morbid asthma in the CRSwNP development program. Advise patients to report signs of eosinophilic pneumonia and EGPA to their healthcare provider. Consider withholding DUPIXENT if eosinophilic pneumonia or EGPA are suspected.
1.4 Eosinophilic Esophagitis
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 1 year and older, weighing at least 15 kg, with eosinophilic esophagitis (EoE).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
DUPIXENT is a clear to slightly opalescent, colorless to pale yellow solution in a:
Single-dose pre-filled syringe with needle shield as:
- Injection: 300 mg/2 mL (150 mg/mL)
- Injection: 200 mg/1.14 mL (175 mg/mL)
Single-dose pre-filled pen as:
- Injection: 300 mg/2 mL (150 mg/mL)
- Injection: 200 mg/1.14 mL (175 mg/mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of DUPIXENT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Immune system disorders: angioedema
- Musculoskeletal system disorders: psoriatic arthritis
- Skin and subcutaneous tissue disorders: Facial skin reactions, including erythema, rash, scaling, edema, papules, pruritus, burning, and pain; new-onset psoriasis, vasculitis
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.2 Conjunctivitis and Keratitis
Conjunctivitis and keratitis adverse reactions have been reported in clinical trials [see Adverse Reactions (6.1)].
Conjunctivitis and keratitis occurred more frequently in AD subjects who received DUPIXENT compared to those who received placebo. Conjunctivitis was the most frequently reported eye disorder. Most subjects with conjunctivitis or keratitis recovered or were recovering during the treatment period.
Among subjects with asthma, the frequencies of conjunctivitis and keratitis were similar between DUPIXENT and placebo.
In adult subjects with CRSwNP, the frequency of conjunctivitis was 2% in the DUPIXENT group compared to 1% in the placebo group in the 24-week safety pool; these subjects recovered.
Among subjects with EoE, there were no reports of conjunctivitis and keratitis in the DUPIXENT group in placebo-controlled trials.
In subjects with PN, the frequency of conjunctivitis was 4% in the DUPIXENT group compared to 1% in the placebo group; these subjects recovered or were recovering during the treatment period.
Among subjects with COPD, the frequency of conjunctivitis and keratitis was 1.4% and 0.1% in the DUPIXENT group and 1% and 0% in the placebo group, respectively.
In subjects with CSU, the frequency of conjunctivitis was similar between DUPIXENT and placebo.
Among subjects with BP, the frequency of conjunctivitis and keratitis was 7.5% and 3.8% in the DUPIXENT group and 0% and 0% in the placebo group, respectively.
Conjunctivitis and keratitis adverse events have also been reported with DUPIXENT in postmarketing settings, predominantly in AD patients. Some patients reported visual disturbances (e.g., blurred vision) associated with conjunctivitis or keratitis.
Advise patients or their caregivers to report new onset or worsening eye symptoms to their healthcare provider. Consider ophthalmological examination for patients who develop conjunctivitis that does not resolve following standard treatment or signs and symptoms suggestive of keratitis, as appropriate.
1.7 Chronic Spontaneous Urticaria
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
1.9 Allergic Fungal Rhinosinusitis
DUPIXENT is indicated for the treatment of adult and pediatric patients aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery.
14.7 Chronic Spontaneous Urticaria
The efficacy of DUPIXENT for the treatment of adult and pediatric patients aged 12 years and older with chronic spontaneous urticaria who remain symptomatic despite H1 antihistamine treatment was evaluated in a master protocol clinical trial (CUPID [NCT04180488]) that included three 24-week, randomized, double-blind, parallel-group, multicenter, placebo-controlled trials (CUPID Study A, Study B, and Study C), followed by 12-week blinded safety follow-up periods. CUPID Study A and Study C included subjects who remained symptomatic despite H1 antihistamine treatment and were anti-IgE treatment naïve, while CUPID Study B included patients who remained symptomatic despite H1 antihistamine and anti-IgE treatments. The efficacy of DUPIXENT was based only on CUPID Study A and Study C; Study B did not meet the primary endpoint.
2.2 Vaccination Prior to Treatment
Consider completing all age-appropriate vaccinations as recommended by current immunization guidelines prior to initiating treatment with DUPIXENT [see Warnings and Precautions (5.10)].
5.6 Patients With Co Morbid Asthma (5.6 Patients with Co-morbid Asthma)
Advise patients with co-morbid asthma not to adjust or stop their asthma treatments without consultation with their physicians.
14.9 Allergic Fungal Rhinosinusitis
The efficacy of DUPIXENT for the treatment of adult and pediatric subjects aged 6 years and older with allergic fungal rhinosinusitis (AFRS) who have a history of sino-nasal surgery was evaluated in a randomized, double-blind, parallel-group, placebo-controlled trial (AIMS [NCT04684524]) of 52 weeks duration. This trial included a total of 62 adult and pediatric subjects aged 6 years and older.
The AIMS trial enrolled patients with AFRS who had evidence of sinus opacification on the Lund Mackay (LMK) sinus CT scan with a LMK score of ≥9 for subjects with unilateral polyps and >12 for subjects with bilateral polyps. There were 79% of subjects who had opacification of all sinuses. Additionally, subjects were required to have a nasal polyps score (NPS) of ≥2 for unilateral polyps or ≥3 for bilateral polyps. Rescue with systemic corticosteroids or surgery was allowed during the study at the investigator's discretion. In the AIMS trial, subjects were randomized 1:1 to receive either DUPIXENT or placebo subcutaneously for 52 weeks. In the DUPIXENT group, adults and pediatric subjects who weighed ≥60 kg received 300 mg every 2 weeks; pediatric subjects who weighed ≥30 kg to <60 kg received 200 mg every 2 weeks. The demographics and baseline characteristics of AIMS are provided in Table 39 below.
| Parameter | AIMS (N=62) |
|---|---|
| SD = standard deviation; LMK = Lund Mackay score; NC = nasal congestion/obstruction; NPS = nasal polyps score; AM = morning | |
| Mean age (years) (SD) | 39.8 (16.0) |
| >17 years, n | 56 |
| 12 to 17 years, n | 5 |
| 6 to <12 years, n | 1 |
| % Male | 72.6 |
| White, N (%) | 27 (43.5) |
| Black, N (%) | 8 (12.9) |
| Asian, N (%) | 25 (40.3) |
| Ethnicity Hispanic or Latino | 14 (22.6) |
| Subjects with systemic corticosteroid use in the previous 2 years, N (%) | 13 (27.7) |
| Mean LMK sinus CT total score Higher scores indicate greater disease severity (SD), range 0 24 |
17.9 (3.6) |
| Mean NC score (SD), range 0-3 | 1.9 (0.9) |
| Mean NPS (SD), range 0-8 | 5.2 (1.9) |
| Mean loss of smell score (AM), (SD) range 0-3 | 2.1 (1.1) |
| Mean blood eosinophils (cells/mcL) (SD) | 0.5 (0.3) |
| Mean total IgE IU/mL (SD) | 1356.4 (1535.0) |
| Prior sino-nasal surgery, N (%) | 61 (98.4) |
| 1 sino-nasal surgery, n (%) | 30 (48.4) |
| ≥2 sino-nasal surgery, n (%) | 31 (50.0) |
| Comorbid asthma, N (%) | 29 (46.8) |
| History of allergic comorbidity, N (%) | 50 (80.6) |
The primary efficacy endpoint was the change from baseline in sinus opacification assessed by the LMK sinus CT scan score at Week 52. The LMK sinus CT scan score evaluated the opacification of each sinus using a 0 to 2 scale (0=normal; 1=partial opacification; 2=total opacification) deriving a maximum score of 12 per side and a total maximum score of 24 (higher scores indicate more opacification).
Secondary endpoints included change from baseline at Week 24 and Week 52 in monthly average nasal congestion score (NC) and endoscopic nasal polyps score (NPS); and change from baseline at Week 24 in sinus opacification assessed by the LMK sinus CT scan score and loss of smell score. Description of the NCS, NPS, and loss of smell score endpoints is the same as for the indication of CRSwNP [see Clinical Studies (14.3)]. The proportion of participants receiving systemic corticosteroids for any reason and/or undergoing/planning to undergo surgery for AFRS (sino-nasal surgery) was evaluated as a secondary endpoint.
Statistically significant efficacy was observed in AIMS for the primary and secondary endpoints as presented in Table 40.
| AIMS Trial | |||||
|---|---|---|---|---|---|
| Placebo (N=29) |
DUPIXENT (N=33) |
LS mean difference vs. Placebo (95% CI) |
|||
| A reduction in score indicates improvement. | |||||
| LMK = Lund Mackay score; NC = nasal congestion/obstruction; NPS = nasal polyps score | |||||
| Scores | Baseline mean |
LS mean change |
Baseline mean |
LS mean change |
|
| Primary Endpoint at Week 52 | |||||
| LMK sinus CT scan score | 18.45 | -1.81 | 17.50 | -9.17 | -7.36 (-9.38, -5.35) |
| Secondary Endpoints at Week 52 | |||||
| NC | 2.05 | -0.17 | 1.86 | -1.57 | -1.40 (-1.77, -1.02) |
| NPS | 5.38 | -0.55 | 5.12 | -3.32 | -2.77 (-3.82, -1.72) |
| Secondary Endpoints at Week 24 | |||||
| LMK sinus CT scan score | 18.45 | -1.93 | 17.50 | -7.38 | -5.45 (-7.48, -3.43) |
| NC | 2.05 | -0.43 | 1.86 | -1.30 | -0.87 (-1.18, -0.56) |
| NPS | 5.38 | -0.80 | 5.12 | -3.16 | -2.36 (-3.31, -1.41) |
| Loss of smell | 2.15 | -0.39 | 1.97 | -1.28 | -0.89 (-1.29, -0.49) |
Figure 19 and Figure 20 demonstrate the time course of improvement in the mean change from baseline in NPS and NCS.
Figure 19: LS Mean Change from Baseline in Monthly Average Nasal Congestion (NC) Score Up to Week 52 in Subjects 6 Years of Age and Older with AFRS (AIMS - ITT Population)
Figure 20: LS Mean Change from Baseline in Nasal Polyp Score (NPS) Up to Week 52 in Subjects 6 Years of Age and Older with AFRS (AIMS - ITT Population)
5.9 Parasitic (helminth) Infections (5.9 Parasitic (Helminth) Infections)
Patients with known helminth infections were excluded from participation in clinical studies. It is unknown if DUPIXENT will influence the immune response against helminth infections.
Treat patients with pre-existing helminth infections before initiating therapy with DUPIXENT. If patients become infected while receiving treatment with DUPIXENT and do not respond to anti-helminth treatment, discontinue treatment with DUPIXENT until the infection resolves. Adverse reactions of helminth infections (5 cases of enterobiasis and 1 case of ascariasis) were reported in pediatric subjects 6 to 11 years old who participated in the pediatric asthma development program [see Adverse Reactions (6.1)].
5.8 Arthralgia and Psoriatic Arthritis
Arthralgia has been reported with the use of DUPIXENT with some patients reporting gait disturbances or decreased mobility associated with joint symptoms; some cases resulted in hospitalization [see Adverse Reactions (6.1)]. In postmarketing reports, onset of arthralgia was variable, ranging from days to months after the first dose of DUPIXENT.
Cases of new-onset psoriatic arthritis requiring systemic treatment have been reported with the use of DUPIXENT.
Some patients' symptoms resolved while continuing treatment with DUPIXENT, and other patients recovered or were recovering following discontinuation of DUPIXENT.
Advise patients to report new onset or worsening joint symptoms to their healthcare provider. If symptoms persist or worsen, consider rheumatological evaluation and/or discontinuation of DUPIXENT.
1.6 Chronic Obstructive Pulmonary Disease
DUPIXENT is indicated as an add-on maintenance treatment of adult patients with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype.
2.1 Important Administration Instructions
DUPIXENT is administered by subcutaneous injection.
DUPIXENT is intended for use under the guidance of a healthcare provider. Provide proper training to patients and/or caregivers on the preparation and administration of DUPIXENT prior to use according to the "Instructions for Use".
14.6 Chronic Obstructive Pulmonary Disease
The efficacy of DUPIXENT as add-on maintenance treatment of adult patients with inadequately controlled COPD and an eosinophilic phenotype was evaluated in two randomized, double-blind, multicenter, parallel-group, placebo-controlled trials (BOREAS [NCT03930732] and NOTUS [NCT04456673]) of 52 weeks duration. The two trials enrolled a total of 1874 adult subjects with COPD.
Both trials enrolled subjects with a diagnosis of COPD with moderate to severe airflow limitation (post-bronchodilator FEV1/FVC ratio <0.7 and post-bronchodilator FEV1 of 30% to 70% predicted) and a minimum blood eosinophil count of 300 cells/mcL at screening. Trial enrollment required an exacerbation history of at least 2 moderate or 1 severe exacerbation(s) in the previous year despite receiving maintenance triple therapy consisting of a long-acting muscarinic antagonist (LAMA), long-acting beta agonist (LABA), and inhaled corticosteroid (ICS), and symptoms of chronic productive cough for at least 3 months in the past year. Greater than or equal to 95% of subjects in each trial had chronic bronchitis. Subjects also had a Medical Research Council (MRC) dyspnea score ≥2 (range 0-4). Exacerbations of COPD were defined as clinically significant worsening of COPD symptoms including increases in dyspnea, wheezing, cough, sputum volume, and/or increase in sputum purulence. Exacerbation severity was further defined as moderate if treatment with systemic corticosteroids and/or antibiotics was required, or severe if they resulted in hospitalization or observation for over 24 hours in an emergency department or urgent care facility.
In both trials, subjects were randomized to receive DUPIXENT 300 mg subcutaneously every 2 weeks (Q2W) or placebo in addition to their background maintenance therapy for 52 weeks.
The demographics and baseline characteristics of BOREAS and NOTUS trial populations are provided in Table 33 below.
| Parameter | BOREAS (N = 939) |
NOTUS (N = 935) |
|---|---|---|
| ICS = inhaled corticosteroid; LAMA = long-acting muscarinic antagonist; LABA = long-acting beta agonist; FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity |
||
| Mean age (years) (± SD) | 65.1 (8.1) | 65.0 (8.3) |
| Male (%) | 66.0 | 67.6 |
| White, N (%) | 790 (84.1) | 838 (89.6) |
| Asian, N (%) | 134 (14.3) | 10 (1.1) |
| Black, N (%) | 5 (0.5) | 12 (1.3) |
| American Indian or Alaska Native, N (%) | 7 (0.7) | 48 (5.1) |
| Other/Multiple, N (%) | 3 (0.3) | 27 (2.9) |
| Ethnicity Hispanic/Latino, N (%) | 261 (27.8) | 300 (32.1) |
| Mean smoking history (pack-years) (± SD) | 40.5 (23.4) | 40.3 (27.2) |
| Current smokers (%) | 30.0 | 29.5 |
| Chronic Bronchitis (%) | 95.0 | 99.9 |
| Emphysema (%) | 32.6 | 30.4 |
| Mean number of moderate Exacerbations treated with either systemic corticosteroids and/or antibiotics. or severeExacerbations requiring hospitalization or observation for over 24 hours in an emergency department or urgent care facility. exacerbations in previous year (± SD) |
2.3 (1.0) | 2.1 (0.9) |
| Background COPD medications at randomization: ICS/LAMA/LABA (%) |
97.6 | 98.8 |
| Mean post-bronchodilator FEV1/FVC ratio (± SD) | 0.49 (0.12) | 0.50 (0.12) |
| Mean post-bronchodilator FEV1 (L) (± SD) | 1.40 (0.47) | 1.45 (0.49) |
| Mean percent predicted post-bronchodilator FEV1 (%) (± SD) | 50.6 (13.1) | 50.1 (12.6) |
| Mean SGRQ total score (± SD) | 48.4 (17.4) | 51.5 (17.0) |
| Mean screening blood eosinophil count Reported screening eosinophil value is the highest values from up to three retests (cells/mcL) (± SD) |
521 (307) | 538 (333) |
| Mean baseline blood eosinophil count Reported baseline eosinophil value was obtained within 4 weeks of screening value (cells/mcL) (± SD) |
401 (298) | 407 (336) |
1.3 Chronic Rhinosinusitis With Nasal Polyps (1.3 Chronic Rhinosinusitis with Nasal Polyps)
DUPIXENT is indicated as an add-on maintenance treatment in adult and pediatric patients aged 12 years and older with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP).
2.7 Recommended Dosage for Prurigo Nodularis
The recommended dosage of DUPIXENT for adult patients is an initial dose of 600 mg (two 300 mg injections) followed by 300 mg given every 2 weeks (Q2W).
14.3 Chronic Rhinosinusitis With Nasal Polyps (14.3 Chronic Rhinosinusitis with Nasal Polyps)
The efficacy of DUPIXENT as an add-on maintenance treatment in adults with inadequately controlled chronic rhinosinusitis with nasal polyps (CRSwNP) was evaluated in two randomized, double-blind, parallel-group, multicenter, placebo-controlled studies (SINUS-24 (NCT02912468) and SINUS-52 (NCT02898454)) in 724 adult subjects 18 years of age and older on background intranasal corticosteroids (INCS). These studies included subjects with CRSwNP despite prior sino-nasal surgery or treatment with, or who were ineligible to receive or were intolerant to, systemic corticosteroids in the past 2 years. Subjects with chronic rhinosinusitis without nasal polyps were not included in these trials. Rescue with systemic corticosteroids or surgery was allowed during the studies at the investigator's discretion. In SINUS-24, a total of 276 subjects were randomized to receive either 300 mg DUPIXENT (N=143) or placebo (N=133) every 2 weeks for 24 weeks. In SINUS-52, 448 subjects were randomized to receive either 300 mg DUPIXENT (N=150) every 2 weeks for 52 weeks, 300 mg DUPIXENT (N=145) every 2 weeks until Week 24 followed by 300 mg DUPIXENT every 4 weeks until Week 52, or placebo (N=153). All subjects had evidence of sinus opacification on the Lund Mackay (LMK) sinus CT scan and 73% to 90% of subjects had opacification of all sinuses. Subjects were stratified based on their histories of prior surgery and co-morbid asthma/nonsteroidal anti-inflammatory drug exacerbated respiratory disease (NSAID-ERD). A total of 63% of subjects reported previous sinus surgery, with a mean number of 2.0 prior surgeries, 74% used systemic corticosteroids in the previous 2 years with a mean number of 1.6 systemic corticosteroid courses in the previous 2 years, 59% had co-morbid asthma, and 28% had NSAID-ERD.
The co-primary efficacy endpoints were change from baseline to Week 24 in bilateral endoscopic nasal polyps score (NPS; 0-8 scale) as graded by central blinded readers, and change from baseline to Week 24 in nasal congestion/obstruction score averaged over 28 days (NC; 0-3 scale), as determined by subjects using a daily diary. For NPS, polyps on each side of the nose were graded on a categorical scale (0=no polyps; 1=small polyps in the middle meatus not reaching below the inferior border of the middle turbinate; 2=polyps reaching below the lower border of the middle turbinate; 3=large polyps reaching the lower border of the inferior turbinate or polyps medial to the middle turbinate; 4=large polyps causing complete obstruction of the inferior nasal cavity). The total score was the sum of the right and left scores. Nasal congestion was rated daily by the subjects on a 0 to 3 categorical severity scale (0=no symptoms; 1=mild symptoms; 2=moderate symptoms; 3=severe symptoms).
In both studies, key secondary endpoints at Week 24 included change from baseline in: LMK sinus CT scan score, daily loss of smell, and 22-item sino-nasal outcome test (SNOT-22). The LMK sinus CT scan score evaluated the opacification of each sinus using a 0 to 2 scale (0=normal; 1=partial opacification; 2=total opacification) deriving a maximum score of 12 per side and a total maximum score of 24 (higher scores indicate more opacification). Loss of smell was scored reflectively by the subject every morning on a 0-3 scale (0=no symptoms, 1=mild symptoms, 2=moderate symptoms, 3=severe symptoms). SNOT-22 includes 22 items assessing symptoms and symptom impact associated with CRSwNP with each item scored from 0 (no problem) to 5 (problem as bad as it can be) with a global score ranging from 0 to 110. SNOT-22 had a 2 week recall period. In the pooled efficacy results, the reduction in the proportion of subjects rescued with systemic corticosteroids and/or sino-nasal surgery (up to Week 52) were evaluated.
The demographics and baseline characteristics of these 2 trials are provided in Table 28 below.
| Parameter | SINUS-24 (N=276) |
SINUS-52 (N=448) |
|---|---|---|
| SD = standard deviation; AM = morning; NPS = nasal polyps score; SNOT-22 = 22-item sino-nasal outcome test; NSAID-ERD = asthma/nonsteroidal anti-inflammatory drug exacerbated respiratory disease | ||
| Mean age (years) (SD) | 50 (13) | 52 (12) |
| % Male | 57 | 62 |
| Mean CRSwNP duration (years) (SD) | 11 (9) | 11 (10) |
| Subjects with ≥1 prior surgery (%) | 72 | 58 |
| Subjects with systemic corticosteroid use in the previous 2 years (%) | 65 | 80 |
| Mean Bilateral endoscopic NPS Higher scores indicate greater disease severity (SD), range 0-8 |
5.8 (1.3) | 6.1 (1.2) |
| Mean Nasal congestion (NC) score (SD), range 0-3 | 2.4 (0.6) | 2.4 (0.6) |
| Mean LMK sinus CT total score (SD), range 0-24 | 19 (4.4) | 18 (3.8) |
| Mean loss of smell score (AM), (SD) range 0-3 | 2.7 (0.5) | 2.8 (0.5) |
| Mean SNOT-22 total score (SD), range 0-110 | 49.4 (20.2) | 51.9 (20.9) |
| Mean blood eosinophils (cells/mcL) (SD) | 440 (330) | 430 (350) |
| Mean total IgE IU/mL (SD) | 212 (276) | 240 (342) |
| Atopic Medical History % Overall |
75 | 82 |
| Asthma (%) | 58 | 60 |
| NSAID-ERD (%) | 30 | 27 |
Clinical Response (SINUS-24 and SINUS-52)
The results for primary endpoints in CRSwNP studies are presented in Table 29.
| SINUS-24 | SINUS-52 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Placebo (n=133) |
DUPIXENT 300 mg Q2W (n=143) |
LS mean difference vs. Placebo (95% CI) |
Placebo (n=153) |
DUPIXENT 300 mg Q2W (n=295) |
LS mean difference vs. Placebo (95% CI) |
|||||
| Primary Endpoints at Week 24 | ||||||||||
| Scores | Baseline mean | LS mean change | Baseline mean | LS mean change | Baseline mean | LS mean change | Baseline mean | LS mean change | ||
| A reduction in score indicates improvement. NPS = nasal polyps score; NC = nasal congestion/obstruction |
||||||||||
| NPS | 5.86 | 0.17 | 5.64 | -1.89 | -2.06 (-2.43, -1.69) |
5.96 | 0.10 | 6.18 | -1.71 | -1.80 (-2.10, -1.51) |
| NC | 2.45 | -0.45 | 2.26 | -1.34 | -0.89 (-1.07, -0.71) |
2.38 | -0.38 | 2.46 | -1.25 | -0.87 (-1.03, -0.71) |
Statistically significant efficacy was observed in SINUS-52 with regard to improvement in bilateral endoscopic NPS score at week 24 and week 52 (see Figure 11).
Figure 11: LS Mean Change from Baseline in Bilateral Nasal Polyps Score (NPS) up to Week 52 in Subjects 18 Years of Age and Older with CRSwNP (SINUS-52 - ITT Population)
Similar results were seen in SINUS-24 at Week 24. In the post-treatment period when subjects were off DUPIXENT, the treatment effect diminished over time (see Figure 12).
Figure 12: LS Mean Change from Baseline in Bilateral Nasal Polyps Score (NPS) up to Week 48 in Subjects 18 Years of Age and Older with CRSwNP (SINUS-24 - ITT Population)
At Week 52, the LS mean difference for nasal congestion in the DUPIXENT group versus placebo was -0.98 (95% CI -1.17, -0.79). In both studies, significant improvements in nasal congestion were observed as early as the first assessment at Week 4. The LS mean difference for nasal congestion at Week 4 in the DUPIXENT group versus placebo was -0.41 (95% CI: -0.52, -0.30) in SINUS-24 and -0.37 (95% CI: -0.46, -0.27) in SINUS-52.
A significant decrease in the LMK sinus CT scan score was observed. The LS mean difference for LMK sinus CT scan score at Week 24 in the DUPIXENT group versus placebo was -7.44 (95% CI: -8.35, -6.53) in SINUS-24 and -5.13 (95% CI: -5.80, -4.46) in SINUS-52. At Week 52, in SINUS-52 the LS mean difference for LMK sinus CT scan score in the DUPIXENT group versus placebo was -6.94 (95% CI: -7.87, -6.01).
Dupilumab significantly improved the loss of smell compared to placebo. The LS mean difference for loss of smell at Week 24 in the DUPIXENT group versus placebo was -1.12 (95% CI: -1.31, -0.93) in SINUS-24 and -0.98 (95% CI: -1.15, -0.81) in SINUS-52. At Week 52, the LS mean difference for loss of smell in the DUPIXENT group versus placebo was -1.10 (95% CI -1.31, -0.89). In both studies, significant improvements in daily loss of smell severity were observed as early as the first assessment at Week 4.
Dupilumab significantly decreased sino-nasal symptoms as measured by SNOT-22 compared to placebo. The LS mean difference for SNOT-22 at Week 24 in the DUPIXENT group versus placebo was -21.12 (95% CI: -25.17, -17.06) in SINUS-24 and -17.36 (95% CI: -20.87, -13.85) in SINUS-52. At Week 52, the LS mean difference in the DUPIXENT group versus placebo was -20.96 (95% CI -25.03, -16.89).
In the pre-specified multiplicity-adjusted pooled analysis of two studies, treatment with DUPIXENT resulted in significant reduction of systemic corticosteroid use and need for sino-nasal surgery versus placebo (HR of 0.24; 95% CI: 0.17, 0.35) (see Figure 13). The proportion of subjects who required systemic corticosteroids was reduced by 74% (HR of 0.26; 95% CI: 0.18, 0.38). The total number of systemic corticosteroid courses per year was reduced by 75% (RR of 0.25; 95% CI: 0.17, 0.37). The proportion of subjects who required surgery was reduced by 83% (HR of 0.17; 95% CI: 0.07, 0.46).
Figure 13: Kaplan Meier Curve for Time to First Systemic Corticosteroid Use and/or Sino-Nasal Surgery During Treatment Period in Subjects 18 Years of Age and Older with CRSwNP (SINUS-24 and SINUS-52 Pooled - ITT Population)
The effects of DUPIXENT on the primary endpoints of NPS and nasal congestion and the key secondary endpoint of LMK sinus CT scan score were consistent in subjects with prior surgery and without prior surgery.
In subjects with co-morbid asthma, improvements in pre-bronchodilator FEV1 were similar to subjects in the asthma program.
2.10 Recommended Dosage for Bullous Pemphigoid
The recommended dosage of DUPIXENT for adult patients is an initial dose of 600 mg (two 300 mg injections) followed by 300 mg given every other week (Q2W).
Principal Display Panel 300 Mg/2 Ml Pen Carton (PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Pen Carton)
NDC 0024-5915-02
Rx Only
DUPIXENT®
(dupilumab)
Injection
Single-dose Pre-filled Pen
300 mg/2 mL (150 mg/mL)
For Subcutaneous Use Only.
2
Single-dose
Pre-filled Pens
Keep out of reach of children. Do not use
after expiration. Do not use if seal is broken
or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Do NOT freeze. Do NOT heat.
Dosage and Administration: See package insert for dosage information
and directions for use.
300 mg/2 mL
Per Pre-filled Pen
sanofi | REGENERON
2.6 Recommended Dosage for Eosinophilic Esophagitis
The recommended dosage of DUPIXENT for adult and pediatric patients 1 year of age and older, weighing at least 15 kg, is specified in Table 5.
| Body Weight | Recommended Dosage |
|---|---|
| 15 to less than 30 kg | 200 mg every 2 weeks (Q2W) |
| 30 to less than 40 kg | 300 mg every 2 weeks (Q2W) |
| 40 kg or more | 300 mg every week (QW) |
Principal Display Panel 200 Mg/1.14 Ml Pen Carton (PRINCIPAL DISPLAY PANEL - 200 mg/1.14 mL Pen Carton)
NDC 0024-5919-02
Rx Only
DUPIXENT®
(dupilumab)
Injection
Single-dose Pre-filled Pen
200 mg/1.14 mL (175 mg/mL)
For Subcutaneous Use Only.
2
Single-dose
Pre-filled Pens
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in
the original carton. Do not freeze. Do not heat.
Dosage and Administration: See package insert
for dosage information and directions for use.
200 mg/1.14 mL
Per Pre-filled Pen
sanofi | REGENERON
Principal Display Panel 300 Mg/2 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Syringe Carton)
NDC 0024-5914-01
Rx Only
DUPIXENT®
(dupilumab)
Injection
300 mg/2 mL (150 mg/mL)
For Subcutaneous Use Only.
300 mg/2 mL
Per Pre-filled Syringe
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
sanofi | REGENERON
Principal Display Panel 100 Mg/0.67 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 100 mg/0.67 mL Syringe Carton)
NDC 0024-5911-02
Rx Only
DUPIXENT®
(dupilumab)
Injection
100 mg/0.67 mL (150 mg/mL)
For Subcutaneous Use Only.
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
100 mg/0.67 mL
sanofi | REGENERON
Principal Display Panel 200 Mg/1.14 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 200 mg/1.14 mL Syringe Carton)
NDC 0024-5918-01
Rx Only
DUPIXENT®
(dupilumab)
Injection
200 mg/1.14 mL (175 mg/mL)
For Subcutaneous Use Only.
200 mg/1.14 mL
Per Pre-filled Syringe
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
sanofi | REGENERON
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been conducted to evaluate the carcinogenic or mutagenic potential of dupilumab.
No effects on fertility parameters such as reproductive organs, menstrual cycle length, or sperm analysis were observed in sexually mature mice that were subcutaneously administered a homologous antibody against IL-4Rα at doses up to 200 mg/kg/week.
2.8 Recommended Dosage for Chronic Obstructive Pulmonary Disease
The recommended dosage of DUPIXENT for adult patients is 300 mg given every 2 weeks (Q2W).
5.5 Risk Associated With Abrupt Reduction of Corticosteroid Dosage (5.5 Risk Associated with Abrupt Reduction of Corticosteroid Dosage)
Do not discontinue systemic, topical, or inhaled corticosteroids abruptly upon initiation of therapy with DUPIXENT. Reductions in corticosteroid dose, if appropriate, should be gradual and performed under the direct supervision of a healthcare provider. Reduction in corticosteroid dose may be associated with systemic withdrawal symptoms and/or unmask conditions previously suppressed by systemic corticosteroid therapy.
2.5 Recommended Dosage for Chronic Rhinosinusitis With Nasal Polyps (2.5 Recommended Dosage for Chronic Rhinosinusitis with Nasal Polyps)
The recommended dosage of DUPIXENT for adult and pediatric patients 12 years of age and older is 300 mg given every 2 weeks (Q2W).
5.4 Acute Symptoms of Asthma Or Chronic Obstructive Pulmonary Disease Or Acute Deteriorating Disease (5.4 Acute Symptoms of Asthma or Chronic Obstructive Pulmonary Disease or Acute Deteriorating Disease)
DUPIXENT should not be used to treat acute symptoms or acute exacerbations of asthma or COPD. Do not use DUPIXENT to treat acute bronchospasm or status asthmaticus. Patients should seek medical advice if their asthma or COPD remains uncontrolled or worsens after initiation of treatment with DUPIXENT.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:49.678513 · Updated: 2026-03-14T22:51:27.803574