Yondelis
472bd78e-be17-4b9d-90f4-9482c3aec9ff
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
YONDELIS ® is indicated for the treatment of adult patients with unresectable or metastatic liposarcoma or leiomyosarcoma who received a prior anthracycline-containing regimen [see Clinical Studies (14) ] .
Dosage and Administration
Administer at 1.5 mg/m 2 as a 24-hour intravenous infusion, every 3 weeks through a central venous line ( 2.1 , 2.6 ) Premedication: dexamethasone 20 mg intravenously, 30 min before each infusion ( 2.3 ) Hepatic Impairment: Administer at 0.9 mg/m 2 as a 24-hour intravenous infusion, every 3 weeks through a central venous line in patients with moderate hepatic impairment ( 2.2 )
Contraindications
YONDELIS is contraindicated in patients with known severe hypersensitivity, including anaphylaxis, to trabectedin.
Warnings and Precautions
Neutropenic sepsis: Severe, and fatal, neutropenic sepsis may occur. Monitor neutrophil count during treatment. Withhold YONDELIS for neutrophil count < 1,500/mcL ( 2.4 , 5.1 ) Rhabdomyolysis: Rhabdomyolysis may occur. Monitor creatine phosphokinase (CPK) levels prior to each administration. Withhold YONDELIS for CPK more than 2.5 times the upper limit of normal. ( 2.4 , 5.2 ) Hepatotoxicity: Hepatotoxicity may occur. Monitor and delay and/or reduce dose if needed ( 5.3 ) Cardiomyopathy: Severe and fatal cardiomyopathy can occur. Patients with left ventricular ejection fraction (LVEF) < lower limit of normal, prior cumulative anthracycline dose of ≥300 mg/m 2 , age ≥65 years, or a history of cardiovascular disease may be at increased risk of developing new or worsening cardiac dysfunction. Discontinue YONDELIS in patients who develop decreased LVEF or cardiomyopathy ( 2.4 , 5.4 ) Capillary leak syndrome: Monitor and discontinue YONDELIS for capillary leak syndrome ( 5.5 ) Embryo-fetal toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use effective contraception ( 5.7 , 8.1 , 8.3 )
Adverse Reactions
Permanently discontinue YONDELIS for: Persistent adverse reactions requiring a delay in dosing of more than 3 weeks. Adverse reactions requiring dose reduction following YONDELIS administered at 1.0 mg/m 2 for patients with normal hepatic function or at 0.3 mg/m 2 for patients with pre-existing moderate hepatic impairment. Severe liver dysfunction: bilirubin two times the upper limit of normal, and AST or ALT three times the upper limit of normal, and alkaline phosphatase less than two times the upper limit of normal in the prior treatment cycle for patients with normal liver function at baseline. Exacerbation of liver dysfunction in patients with pre-existing moderate hepatic impairment. Capillary leak syndrome. Rhabdomyolysis. Grade 3 or 4 cardiac adverse events (AEs) indicative of cardiomyopathy or for subjects with an LVEF that decreases below the lower limit of normal. The recommended dose modifications for adverse reactions are listed in Table 1. Once reduced, the dose of YONDELIS should not be increased in subsequent treatment cycles. Table 1: Recommended Dosage Modification Laboratory Result or Adverse Reaction DELAY next dose of YONDELIS for up to 3 weeks REDUCE next dose of YONDELIS by one dose level for adverse reaction(s) during prior cycle Platelets Less than 100,000 platelets/microliter Less than 25,000 platelets/microliter Absolute neutrophil count Less than 1,500 neutrophils/microliter Less than 1,000 neutrophils/microliter with fever/infection Less than 500 neutrophils/microliter lasting more than 5 days Total bilirubin Greater than the upper limit of normal Greater than the upper limit of normal Aspartate aminotransferase (AST) or alanine aminotransferase (ALT) More than 2.5 times the upper limit of normal More than 5 times the upper limit of normal Alkaline phosphatase (ALP) More than 2.5 times the upper limit of normal More than 2.5 times the upper limit of normal Creatine phosphokinase More than 2.5 times the upper limit of normal More than 5 times the upper limit of normal Other non-hematologic adverse reactions Grade 3 or 4 Grade 3 or 4 The recommended starting doses and dose reductions for YONDELIS are listed in Table 2: Table 2: Recommended Starting Doses and Dose Reductions Starting Dose and Dose Reduction For patients with normal hepatic function or mild hepatic impairment Including patients with bilirubin greater than 1 to 1.5 times the upper limit of normal, and any AST or ALT. prior to initiation of YONDELIS treatment For patients with moderate hepatic impairment Including patients with bilirubin levels greater than 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal. prior to initiation of YONDELIS treatment Starting Dose 1.5 mg/m 2 0.9 mg/m 2 Dose Reduction First dose reduction 1.2 mg/m 2 0.6 mg/m 2 Second dose reduction 1.0 mg/m 2 0.3 mg/m 2
Drug Interactions
CYP3A inhibitors: Avoid concomitant strong CYP3A inhibitors ( 7.1 ) CYP3A inducers: Avoid concomitant strong CYP3A inducers ( 7.2 )
How Supplied
YONDELIS ® is supplied in a single-dose glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
Storage and Handling
YONDELIS ® is supplied in a single-dose glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
Description
Warnings and Precautions ( 5.7 ) 12/2025
Medication Information
Warnings and Precautions
Neutropenic sepsis: Severe, and fatal, neutropenic sepsis may occur. Monitor neutrophil count during treatment. Withhold YONDELIS for neutrophil count < 1,500/mcL ( 2.4 , 5.1 ) Rhabdomyolysis: Rhabdomyolysis may occur. Monitor creatine phosphokinase (CPK) levels prior to each administration. Withhold YONDELIS for CPK more than 2.5 times the upper limit of normal. ( 2.4 , 5.2 ) Hepatotoxicity: Hepatotoxicity may occur. Monitor and delay and/or reduce dose if needed ( 5.3 ) Cardiomyopathy: Severe and fatal cardiomyopathy can occur. Patients with left ventricular ejection fraction (LVEF) < lower limit of normal, prior cumulative anthracycline dose of ≥300 mg/m 2 , age ≥65 years, or a history of cardiovascular disease may be at increased risk of developing new or worsening cardiac dysfunction. Discontinue YONDELIS in patients who develop decreased LVEF or cardiomyopathy ( 2.4 , 5.4 ) Capillary leak syndrome: Monitor and discontinue YONDELIS for capillary leak syndrome ( 5.5 ) Embryo-fetal toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use effective contraception ( 5.7 , 8.1 , 8.3 )
Indications and Usage
YONDELIS ® is indicated for the treatment of adult patients with unresectable or metastatic liposarcoma or leiomyosarcoma who received a prior anthracycline-containing regimen [see Clinical Studies (14) ] .
Dosage and Administration
Administer at 1.5 mg/m 2 as a 24-hour intravenous infusion, every 3 weeks through a central venous line ( 2.1 , 2.6 ) Premedication: dexamethasone 20 mg intravenously, 30 min before each infusion ( 2.3 ) Hepatic Impairment: Administer at 0.9 mg/m 2 as a 24-hour intravenous infusion, every 3 weeks through a central venous line in patients with moderate hepatic impairment ( 2.2 )
Contraindications
YONDELIS is contraindicated in patients with known severe hypersensitivity, including anaphylaxis, to trabectedin.
Adverse Reactions
Permanently discontinue YONDELIS for: Persistent adverse reactions requiring a delay in dosing of more than 3 weeks. Adverse reactions requiring dose reduction following YONDELIS administered at 1.0 mg/m 2 for patients with normal hepatic function or at 0.3 mg/m 2 for patients with pre-existing moderate hepatic impairment. Severe liver dysfunction: bilirubin two times the upper limit of normal, and AST or ALT three times the upper limit of normal, and alkaline phosphatase less than two times the upper limit of normal in the prior treatment cycle for patients with normal liver function at baseline. Exacerbation of liver dysfunction in patients with pre-existing moderate hepatic impairment. Capillary leak syndrome. Rhabdomyolysis. Grade 3 or 4 cardiac adverse events (AEs) indicative of cardiomyopathy or for subjects with an LVEF that decreases below the lower limit of normal. The recommended dose modifications for adverse reactions are listed in Table 1. Once reduced, the dose of YONDELIS should not be increased in subsequent treatment cycles. Table 1: Recommended Dosage Modification Laboratory Result or Adverse Reaction DELAY next dose of YONDELIS for up to 3 weeks REDUCE next dose of YONDELIS by one dose level for adverse reaction(s) during prior cycle Platelets Less than 100,000 platelets/microliter Less than 25,000 platelets/microliter Absolute neutrophil count Less than 1,500 neutrophils/microliter Less than 1,000 neutrophils/microliter with fever/infection Less than 500 neutrophils/microliter lasting more than 5 days Total bilirubin Greater than the upper limit of normal Greater than the upper limit of normal Aspartate aminotransferase (AST) or alanine aminotransferase (ALT) More than 2.5 times the upper limit of normal More than 5 times the upper limit of normal Alkaline phosphatase (ALP) More than 2.5 times the upper limit of normal More than 2.5 times the upper limit of normal Creatine phosphokinase More than 2.5 times the upper limit of normal More than 5 times the upper limit of normal Other non-hematologic adverse reactions Grade 3 or 4 Grade 3 or 4 The recommended starting doses and dose reductions for YONDELIS are listed in Table 2: Table 2: Recommended Starting Doses and Dose Reductions Starting Dose and Dose Reduction For patients with normal hepatic function or mild hepatic impairment Including patients with bilirubin greater than 1 to 1.5 times the upper limit of normal, and any AST or ALT. prior to initiation of YONDELIS treatment For patients with moderate hepatic impairment Including patients with bilirubin levels greater than 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal. prior to initiation of YONDELIS treatment Starting Dose 1.5 mg/m 2 0.9 mg/m 2 Dose Reduction First dose reduction 1.2 mg/m 2 0.6 mg/m 2 Second dose reduction 1.0 mg/m 2 0.3 mg/m 2
Drug Interactions
CYP3A inhibitors: Avoid concomitant strong CYP3A inhibitors ( 7.1 ) CYP3A inducers: Avoid concomitant strong CYP3A inducers ( 7.2 )
Storage and Handling
YONDELIS ® is supplied in a single-dose glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
How Supplied
YONDELIS ® is supplied in a single-dose glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
Description
Warnings and Precautions ( 5.7 ) 12/2025
Section 34073-7
Drug Interactions
Section 42229-5
Risk Summary
Based on its mechanism of action, trabectedin can cause fetal harm when administered during pregnancy [see Clinical Pharmacology (12.1)] . There are no available data with the use of YONDELIS during pregnancy. Animal reproductive and developmental studies at relevant doses have not been conducted with trabectedin; however, placental transfer of trabectedin was demonstrated in pregnant rats. Advise pregnant woman of the potential risk to a fetus. The background risk of major birth defects and miscarriage for the indicated population are unknown; however, the background risk in the U.S. general population of major birth defects is 2 to 4% and of miscarriage is 15 to 20% of clinically recognized pregnancies.
Section 42230-3
| PATIENT INFORMATION
YONDELIS ®(yon-DEL-ess) (trabectedin) for injection, for intravenous use |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised 12/2025 | |
|
What is YONDELIS?
YONDELIS is a prescription medicine used to treat adults with liposarcoma or leiomyosarcoma that:
|
||
|
Who should not receive YONDELIS?
Do not receive YONDELIS if you have had a severe allergic reaction to trabectedin or any of the ingredients in YONDELIS. See the end of this leaflet for a complete list of ingredients in YONDELIS. |
||
Before receiving YONDELIS, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using YONDELIS with certain other medicines may affect the way YONDELIS works and may increase your risk of side effects. Know the medicines you take and keep a list of them to show to your healthcare provider or pharmacist when you get a new medicine. |
||
How will I receive YONDELIS?
|
||
|
What are the possible side effects of YONDELIS?
YONDELIS can cause serious side effects, including :
|
||
|
|
|
|
||
|
|
|
The most common severe abnormal blood tests include:
Your healthcare provider may decrease or delay your dose if you develop serious side effects or may permanently stop treatment with YONDELIS if your side effects are severe. YONDELIS may cause fertility problems in males and females, which may affect your ability to have children. Talk to your healthcare provider if this is a concern for you. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of YONDELIS. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about the safe and effective use of YONDELIS
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your healthcare provider or pharmacist for information about YONDELIS that is written for health professionals. |
||
|
What are the ingredients in YONDELIS?
Active ingredient: trabectedin Inactive ingredients:potassium dihydrogen phosphate, sucrose, phosphoric acid and potassium hydroxide. Manufactured for: Janssen Products, LP, Horsham, PA 19044, USA For more information, call 1-800-526-7736 or go to www.YONDELIS.com. For patent information: www.janssenpatents.com |
Section 43683-2
| Warnings and Precautions ( 5.7) | 12/2025 |
Section 44425-7
Storage and Handling
Store YONDELIS vials in a refrigerator at 2 °C to 8 °C (36 °F to 46 °F).
YONDELIS is a hazardous drug. Follow applicable special handling and disposal procedures. 1
10 Overdosage
There is no specific antidote for YONDELIS. Hemodialysis is not expected to enhance the elimination of YONDELIS because trabectedin is highly bound to plasma proteins (97%) and not significantly renally excreted.
15 References
- "OSHA Hazardous Drugs." OSHA.http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
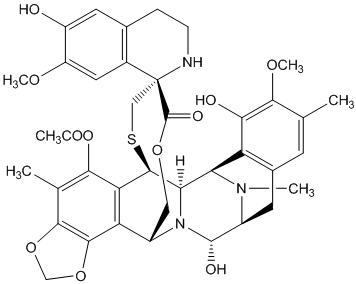
Trabectedin is an alkylating drug with the chemical name (1' R,6 R,6a R,7 R,13 S,14 S,16 R)-5-(acetyloxy)-3',4',6,6a,7,13,14,16-octahydro-6',8,14-trihydroxy-7',9-dimethoxy-4,10,23-trimethyl-spiro[6,16-(epithiopropanoxymethano)-7,13-imino-12 H-1,3-dioxolo[7,8]isoquino[3,2- b][3]benzazocine-20,1'(2' H)-isoquinolin]-19-one. The molecular formula is C 39H 43N 3O 11S. The molecular weight is 761.84 daltons. The chemical structure is shown below:
Trabectedin is hydrophobic and has a low solubility in water.
YONDELIS ®(trabectedin) for injection is supplied as a sterile lyophilized white to off-white powder/cake in a single-dose vial. Each single-dose vial contains 1 mg of trabectedin, 27.2 mg potassium dihydrogen phosphate, 400 mg sucrose, and phosphoric acid and potassium hydroxide (for pH adjustment to 3.6 – 4.2).
2.3 Premedication
Administer dexamethasone 20 mg intravenously 30 minutes prior to each dose of YONDELIS.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Safety (n=61) and efficacy (n=58) were assessed across five open-label studies (NCT00006463, NCT01453283, NCT00005625, NCT00070109, and ET-B-023-00) in pediatric patients (aged 2 to <17 years) with pediatric histotypes of sarcoma (predominantly rhabdomyosarcoma, osteosarcoma, Ewing sarcoma, and non-rhabdomyosarcoma soft tissue sarcoma). No new safety signals were observed in pediatric patients across these studies.
Pharmacokinetic parameters in 17 pediatric patients (aged 3 to 17 years) were within the range of values previously observed in adults given the same dose per body surface area.
8.5 Geriatric Use
Clinical studies of YONDELIS did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
2.6 Administration
- Infuse the reconstituted, diluted solution over 24 hours through a central venous line using an infusion set with a 0.2 micron polyethersulfone (PES) in-line filter.
- Complete infusion within 30 hours of initial reconstitution. Discard any unused portion of the reconstituted product or of the infusion solution.
5.2 Rhabdomyolysis
YONDELIS can cause rhabdomyolysis and musculoskeletal toxicity. In Trial ET743-SAR-3007, rhabdomyolysis leading to death occurred in 3 (0.8%) of the 378 patients receiving YONDELIS. Elevations in creatine phosphokinase (CPK) occurred in 122 (32%) of the 378 patients receiving YONDELIS, including Grade 3 or 4 CPK elevation in 24 patients (6%), compared to 15 (9%) of the 172 patients receiving dacarbazine with any CPK elevation, including 1 patient (0.6%) with Grade 3 CPK elevation. Among the 24 patients receiving YONDELIS with Grade 3 or 4 CPK elevation, renal failure occurred in 11 patients (2.9%); rhabdomyolysis with the complication of renal failure occurred in 4 of these 11 patients (1.1%). The median time to first occurrence of Grade 3 or 4 CPK elevations was 2 months (range: 1 to 11.5 months). The median time to complete resolution was 14 days (range: 5 days to 1 month).
Assess CPK levels prior to each administration of YONDELIS. Withhold, reduce dose, or permanently discontinue based on severity of adverse reaction [see Dosage and Administration (2.4)] .
5.3 Hepatotoxicity
Hepatotoxicity, including hepatic failure, can occur with YONDELIS. Patients with serum bilirubin levels above the upper limit of normal or AST or ALT levels >2.5 × upper limit of normal were not enrolled in Trial ET743-SAR-3007. In Trial ET743-SAR-3007, the incidence of Grade 3–4 elevated liver function tests (LFTs; defined as elevations in ALT, AST, total bilirubin, or alkaline phosphatase) was 35% (134/378) in patients receiving YONDELIS. The median time to development of Grade 3–4 elevation in ALT or AST was 29 days (range: 3 days to 11.5 months). Of the 134 patients with Grade 3–4 elevations in LFTs, 114 (85%) experienced complete resolution with the median time to complete resolution of 13 days (range: 4 days to 4.4 months).
In Trial ET743-SAR-3007, the incidence of drug-induced liver injury (defined as concurrent elevation in ALT or AST of more than three times the upper limit of normal, alkaline phosphatase less than two times the upper limit of normal, and total bilirubin at least two times the upper limit of normal) was 1.3% (5/378) in patients receiving YONDELIS. ALT or AST elevation greater than eight times the upper limit of normal occurred in 18% (67/378) of patients receiving YONDELIS.
Assess LFTs prior to each administration of YONDELIS and as clinically indicated based on underlying severity of pre-existing hepatic impairment. Manage elevated LFTs with treatment interruption, dose reduction, or permanent discontinuation based on severity and duration of LFT abnormality [see Dosage and Administration (2.4)and Use in Specific Populations (8.6)].
5.4 Cardiomyopathy
Cardiomyopathy including cardiac failure, congestive heart failure, ejection fraction decreased, diastolic dysfunction, or right ventricular dysfunction can occur with YONDELIS. In Trial ET743-SAR-3007, a significant decrease in LVEF was defined as an absolute decrease of ≥15% or below the lower limit of normal with an absolute decrease of ≥5%. Patients with a history of New York Heart Association Class II to IV heart failure or abnormal left ventricular ejection fraction (LVEF) at baseline were ineligible. In Trial ET743-SAR-3007, cardiomyopathy occurred in 23 patients (6%) receiving YONDELIS and in four patients (2.3%) receiving dacarbazine. Grade 3 or 4 cardiomyopathy occurred in 15 patients (4%) receiving YONDELIS and 2 patients (1.2%) receiving dacarbazine; cardiomyopathy leading to death occurred in 1 patient (0.3%) receiving YONDELIS and in none of the patients receiving dacarbazine. The median time to development of Grade 3 or 4 cardiomyopathy in patients receiving YONDELIS was 5.3 months (range: 26 days to 15.3 months).
Patients with LVEF < lower limit of normal, prior cumulative anthracycline dose of ≥300 mg/m 2, age ≥65 years, or a history of cardiovascular disease may be at increased risk of cardiac dysfunction. Assess LVEF by echocardiogram (ECHO) or multigated acquisition (MUGA) scan before initiation of YONDELIS and at 2- to 3-month intervals thereafter until YONDELIS is discontinued. Discontinue treatment with YONDELIS based on severity of adverse reaction [see Dosage and Administration (2.4)] .
14 Clinical Studies
The clinical efficacy and safety of YONDELIS in patients with metastatic or recurrent leiomyosarcoma or liposarcoma were demonstrated in Trial ET743-SAR-3007 (NCT01343277), a randomized (2:1), open-label, active-controlled trial comparing treatment with YONDELIS 1.5 mg/m 2as a 24-hour continuous intravenous infusion once every 3 weeks to dacarbazine 1000 mg/m 2intravenous infusion (20 to 120 minutes) once every 3 weeks. Treatment continued in both arms until disease progression or unacceptable toxicity; all patients in the YONDELIS arm were required to receive dexamethasone 20 mg intravenous injection prior to each YONDELIS infusion. Patients were required to have unresectable, locally advanced or metastatic leiomyosarcoma or liposarcoma (dedifferentiated, myxoid round cell, or pleomorphic) and previous treatment with an anthracycline- and ifosfamide-containing regimen or an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen. Randomization was stratified by subtype of soft tissue sarcoma (leiomyosarcoma vs. liposarcoma), ECOG performance status (0 vs. 1), and number of prior chemotherapy regimens (1 vs. ≥2). The efficacy outcome measures were investigator-assessed progression-free survival (PFS) according to the Response Evaluation Criteria in Solid Tumors (RECIST v1.1), overall survival (OS), objective response rate (ORR), and duration of response (DOR). Patients in the dacarbazine arm were not offered YONDELIS at the time of disease progression.
A total of 518 patients were randomized, 345 to the YONDELIS arm and 173 patients to the dacarbazine arm. The median patient age was 56 years (range: 17 to 81); 30% were male; 76% White, 12% Black, and 4% Asian; 73% had leiomyosarcomas and 27% liposarcomas; 49% had an ECOG PS of 0; and 89% received ≥2 prior chemotherapy regimens. The most common (≥20%) pre-study chemotherapeutic agents administered were doxorubicin (90%), gemcitabine (81%), docetaxel (74%), and ifosfamide (59%). Approximately 10% of patients had received pazopanib.
Trial ET743-SAR-3007 demonstrated a statistically significant improvement in PFS. An exploratory analysis of independent radiology committee-determined PFS, in a subgroup consisting of approximately 60% of the total population, provided similar results to the investigator-determined PFS. Efficacy results from Trial ET743-SAR-3007 are presented in the table below.
| Efficacy Endpoint | YONDELIS
N=345 |
Dacarbazine
N=173 |
|---|---|---|
| CR=Complete Response; PR=Partial Response; CI=Confidence Interval, HR=hazard ratio, NE=not estimable. | ||
| Progression-free survival | ||
| PFS Events, n (%) | 217 (63%) | 112 (65%) |
| Disease progression | 204 | 109 |
| Death | 13 | 3 |
| Median (95% CI) (months) | 4.2 (3.0, 4.8) | 1.5 (1.5, 2.6) |
| HR (95% CI)
Cox proportional hazards model with treatment group as the only covariate.
|
0.55 (0.44, 0.70) | |
| p-value
Unstratified log rank test.
|
<0.001 | |
|
Overall survival
Based on 384 patients randomized to YONDELIS arm and 193 patients randomized to dacarbazine.
|
||
| Events, n (%) | 258 (67%) | 123 (64%) |
| Median (95% CI) (months) | 13.7 (12.2, 16.0) | 13.1 (9.1, 16.2) |
| HR (95% CI) | 0.93 (0.75, 1.15) | |
| p-value | 0.49 | |
| Objective Response Rate (ORR: CR+PR) | ||
| Number of patients (%) | 23 (7%) | 10 (6%) |
| 95% CI
Fisher's exact CI.
|
(4.3, 9.8) | (2.8, 10.4) |
| Duration of Response (CR+ PR) | ||
| Median (95% CI) (months) | 6.9 (4.5, 7.6) | 4.2 (2.9, NE) |
Figure 1: Kaplan-Meier Curves of Progression-Free Survival in Trial ET743-SAR-3007
4 Contraindications
YONDELIS is contraindicated in patients with known severe hypersensitivity, including anaphylaxis, to trabectedin.
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Anaphylaxis [see Contraindications (4)]
- Neutropenic Sepsis [see Warnings and Precautions (5.1)]
- Rhabdomyolysis [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
- Cardiomyopathy [see Warnings and Precautions (5.4)]
- Capillary Leak Syndrome [see Warnings and Precautions (5.5)]
- Extravasation Resulting in Tissue Necrosis [see Warnings and Precautions (5.6)]
7 Drug Interactions
8.7 Renal Impairment
No dose adjustment is recommended in patients with mild [creatinine clearance (CLcr) 60–89 mL/min] or moderate (CLcr of 30–59 mL/min) renal impairment.
The pharmacokinetics of trabectedin has not been evaluated in patients with severe renal impairment (CLcr <30 mL/min) or end stage renal disease [see Clinical Pharmacology (12.3)] .
12.3 Pharmacokinetics
The pharmacokinetics of trabectedin is characterized by a rapid decline phase at the end of the infusion and slower exponential phases. Population pharmacokinetic analyses suggest that the pharmacokinetics of trabectedin is dose-proportional (over the dose range of 0.024 to 1.8 mg/m 2) and exposure is time-independent. No accumulation of trabectedin in plasma is observed upon repeated administrations every 3 weeks.
2.1 Recommended Dosage
The recommended dose is 1.5 mg/m 2administered as an intravenous infusion over 24 hours through a central venous line every 21 days (3 weeks), until disease progression or unacceptable toxicity.
5.1 Neutropenic Sepsis
Neutropenic sepsis, including fatal cases, can occur with YONDELIS. In Trial ET743-SAR-3007, the incidence of Grade 3 or 4 neutropenia, based on laboratory values, in patients receiving YONDELIS was 43% (161/378). The median time to the first occurrence of Grade 3 or 4 neutropenia was 16 days (range: 8 days to 9.7 months); the median time to complete resolution of neutropenia was 13 days (range: 3 days to 2.3 months). Febrile neutropenia (fever ≥38.5 °C with Grade 3 or 4 neutropenia) occurred in 18 patients (5%) treated with YONDELIS. Ten patients (2.6%) experienced neutropenic sepsis, 5 of whom had febrile neutropenia, which was fatal in 4 patients (1.1%).
Assess neutrophil count prior to administration of each dose of YONDELIS and periodically throughout the treatment cycle. Withhold or reduce dose of YONDELIS based on severity of adverse reaction [see Dosage and Administration (2.4)] .
8.6 Hepatic Impairment
The mean trabectedin exposure was (97%) higher in patients with moderate (bilirubin levels greater than 1.5 to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal) hepatic impairment compared to patients with normal (total bilirubin ≤ the upper limit of normal, and AST and ALT ≤ the upper limit of normal) liver function. Reduce YONDELIS dose in patients with moderate hepatic impairment [see Dosage and Administration (2.2)and Clinical Pharmacology (12.3)] .
Do not administer YONDELIS to patients with severe hepatic impairment (bilirubin levels above 3 times the upper limit of normal, and any AST and ALT) [see Warnings and Precautions (5.3)] .
1 Indications and Usage
YONDELIS ®is indicated for the treatment of adult patients with unresectable or metastatic liposarcoma or leiomyosarcoma who received a prior anthracycline-containing regimen [see Clinical Studies (14)] .
12.1 Mechanism of Action
Trabectedin is an alkylating drug that binds guanine residues in the minor groove of DNA, forming adducts and resulting in a bending of the DNA helix towards the major groove. Adduct formation triggers a cascade of events that can affect the subsequent activity of DNA binding proteins, including some transcription factors, and DNA repair pathways, resulting in perturbation of the cell cycle and eventual cell death.
5.7 Embryo Fetal Toxicity
Based on its mechanism of action, YONDELIS can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during therapy and for at least 8 months after the last dose of YONDELIS. Advise males with female partners of reproductive potential to use effective contraception during therapy and for at least 5 months after the last dose of YONDELIS [see Use in Specific Populations (8.1, 8.3)] .
5 Warnings and Precautions
- Neutropenic sepsis: Severe, and fatal, neutropenic sepsis may occur. Monitor neutrophil count during treatment. Withhold YONDELIS for neutrophil count < 1,500/mcL ( 2.4, 5.1)
- Rhabdomyolysis: Rhabdomyolysis may occur. Monitor creatine phosphokinase (CPK) levels prior to each administration. Withhold YONDELIS for CPK more than 2.5 times the upper limit of normal. ( 2.4, 5.2)
- Hepatotoxicity: Hepatotoxicity may occur. Monitor and delay and/or reduce dose if needed ( 5.3)
- Cardiomyopathy: Severe and fatal cardiomyopathy can occur. Patients with left ventricular ejection fraction (LVEF) < lower limit of normal, prior cumulative anthracycline dose of ≥300 mg/m 2, age ≥65 years, or a history of cardiovascular disease may be at increased risk of developing new or worsening cardiac dysfunction. Discontinue YONDELIS in patients who develop decreased LVEF or cardiomyopathy ( 2.4, 5.4)
- Capillary leak syndrome: Monitor and discontinue YONDELIS for capillary leak syndrome ( 5.5)
- Embryo-fetal toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use effective contraception ( 5.7, 8.1, 8.3)
2 Dosage and Administration
- Administer at 1.5 mg/m 2as a 24-hour intravenous infusion, every 3 weeks through a central venous line ( 2.1, 2.6)
- Premedication: dexamethasone 20 mg intravenously, 30 min before each infusion ( 2.3)
- Hepatic Impairment: Administer at 0.9 mg/m 2as a 24-hour intravenous infusion, every 3 weeks through a central venous line in patients with moderate hepatic impairment ( 2.2)
5.5 Capillary Leak Syndrome
Capillary leak syndrome (CLS) characterized by hypotension, edema, and hypoalbuminemia has been reported with YONDELIS, including serious CLS resulting in death. Monitor for signs and symptoms of CLS. Discontinue YONDELIS and promptly initiate standard management for patients with CLS, which may include a need for intensive care [see Adverse Reactions (6.2)] .
3 Dosage Forms and Strengths
For injection: 1 mg, lyophilized powder in single-dose vial for reconstitution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of YONDELIS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Vascular disorders: capillary leak syndrome
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to YONDELIS in 755 patients with soft tissue sarcoma including 197 (26%) patients exposed to YONDELIS for greater than or equal to 6 months and 57 (8%) patients exposed to YONDELIS for greater than or equal to 1 year. The safety of YONDELIS was evaluated in six open-label, single-arm trials, in which 377 patients received YONDELIS and one open-label, randomized, active-controlled clinical trial in which 378 patients received YONDELIS (Trial ET743-SAR-3007). All patients received YONDELIS at the recommended dosing regimen of 1.5 mg/m 2administered as an intravenous infusion over 24 hours once every 3 weeks (q3wk, 24-h). The median age was 54 years (range: 18 to 81 years), 63% were female, and all patients had metastatic soft tissue sarcoma.
Tables 3 and 4 present selected adverse reactions and laboratory abnormalities, respectively, observed in Trial ET743-SAR-3007, an open-label, randomized (2:1), active-controlled trial in which 550 patients with previously treated leiomyosarcoma or liposarcoma (dedifferentiated, myxoid round cell, or pleomorphic) received YONDELIS 1.5 mg/m 2intravenous infusion over 24 hours once every 3 weeks (n=378) or dacarbazine 1000 mg/m 2intravenous infusion over 20 to 120 minutes once every 3 weeks (n=172) [see Clinical Studies (14)] . All patients treated with YONDELIS were required to receive dexamethasone 20 mg intravenous injection 30 minutes prior to start of the YONDELIS infusion.
In Trial ET743-SAR-3007, patients had been previously treated with an anthracycline- and ifosfamide-containing regimen or with an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen. The trial excluded patients with known central nervous system metastasis, elevated serum bilirubin or significant chronic liver disease, such as cirrhosis or active hepatitis, and history of myocardial infarction within 6 months, history of New York Heart Association Class II to IV heart failure, or abnormal left ventricular ejection fraction at baseline. The median age of patients in Trial ET743-SAR-3007 was 57 years (range: 17 to 81 years), with 69% female, 77% White, 12% Black or African American, 4% Asian, and <1% American Indian or Alaska Native. The median duration of exposure to trabectedin was 13 weeks (range: 1 to 127 weeks) with 30% of patients exposed to YONDELIS for greater than 6 months and 7% of patients exposed to YONDELIS for greater than 1 year.
In Trial ET743-SAR-3007, adverse reactions resulting in permanent discontinuation of YONDELIS occurred in 26% (98/378) of patients; the most common were increased liver tests (defined as ALT, AST, alkaline phosphatase, bilirubin) (5.6%), thrombocytopenia (3.4%), fatigue (1.6%), increased creatine phosphokinase (1.1%), and decreased ejection fraction (1.1%). Adverse reactions that led to dose reductions occurred in 42% (158/378) of patients treated with YONDELIS; the most common were increased liver tests (24%), neutropenia (including febrile neutropenia) (8%), thrombocytopenia (4.2%), fatigue (3.7%), increased creatine phosphokinase (2.4%), nausea (1.1%), and vomiting (1.1%). Adverse reactions led to dose interruptions in 52% (198/378) of patients treated with YONDELIS; the most common were neutropenia (31%), thrombocytopenia (15%), increased liver tests (6%), fatigue (2.9%), anemia (2.6%), increased creatinine (1.1%), and nausea (1.1%).
The most common adverse reactions (≥20%) were nausea, fatigue, vomiting, constipation, decreased appetite, diarrhea, peripheral edema, dyspnea, and headache. The most common laboratory abnormalities (≥20%) were increases in AST or ALT, increased alkaline phosphatase, hypoalbuminemia, increased creatinine, increased creatine phosphokinase, anemia, neutropenia, and thrombocytopenia.
| YONDELIS
(N=378) |
Dacarbazine
(N=172) |
|||
|---|---|---|---|---|
| System Organ Class
Adverse Reaction |
All Grades
Toxicity grade is based on NCI common toxicity criteria, version 4.0.
(%) |
Grades 3–4
(%) |
All Grades
(%) |
Grades 3–4
(%) |
| Gastrointestinal disorders | ||||
| Nausea | 75 | 7 | 50 | 1.7 |
| Vomiting | 46 | 6 | 22 | 1.2 |
| Constipation | 37 | 0.8 | 31 | 0.6 |
| Diarrhea | 35 | 1.6 | 23 | 0 |
| General disorders and administration site conditions | ||||
| Fatigue
Fatigue is a composite of the following adverse event terms: fatigue, asthenia, and malaise.
|
69 | 8 | 52 | 1.7 |
| Peripheral edema | 28 | 0.8 | 13 | 0.6 |
| Metabolism and nutrition disorders | ||||
| Decreased appetite | 37 | 1.9 | 21 | 0.6 |
| Respiratory, thoracic and mediastinal disorders | ||||
| Dyspnea | 25 | 4.2 | 20 | 1.2 |
| Nervous system disorders | ||||
| Headache | 25 | 0.3 | 19 | 0 |
| Musculoskeletal and connective tissue disorders | ||||
| Arthralgia | 15 | 0 | 8 | 1.2 |
| Myalgia | 12 | 0 | 6 | 0 |
| Psychiatric disorders | ||||
| Insomnia | 15 | 0.3 | 9 | 0 |
Other clinically important adverse reactions observed in <10% of patients (N=755) with soft tissue sarcoma receiving YONDELIS were:
Nervous system disorders: peripheral neuropathy, paresthesia, hypoesthesia.
Respiratory, thoracic, and mediastinal disorders: pulmonary embolism.
General disorders and administration site conditions: mucosal inflammation
| Laboratory Abnormalities | YONDELIS | Dacarbazine | ||
|---|---|---|---|---|
| All Grades
(%) |
Grades 3–4
(%) |
All Grades
(%) |
Grades 3–4
(%) |
|
| YONDELIS group (range: 373 to 377 patients) and dacarbazine group (range: 166 to 168 patients). | ||||
| Chemistry | ||||
| Increased ALT | 90 | 31 | 33 | 0.6 |
| Increased AST | 84 | 17 | 32 | 1.2 |
| Increased alkaline phosphatase | 70 | 1.6 | 60 | 0.6 |
| Hypoalbuminemia | 63 | 3.7 | 51 | 3.0 |
| Increased creatinine | 46 | 4.2 | 29 | 1.2 |
| Increased creatine phosphokinase | 33 | 6.4 | 9 | 0.6 |
| Hyperbilirubinemia | 13 | 1.9 | 5 | 0.6 |
| Hematology | ||||
| Anemia | 96 | 19 | 79 | 12 |
| Neutropenia | 66 | 43 | 47 | 26 |
| Thrombocytopenia | 59 | 21 | 57 | 20 |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.5 Preparation for Administration
- YONDELIS is a hazardous drug. Follow applicable special handling and disposal procedures. 1
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Using aseptic technique, inject 20 mL of Sterile Water for Injection, USP into the vial. Shake the vial until complete dissolution. The reconstituted solution is clear, colorless to pale brownish-yellow, and contains 0.05 mg/mL of trabectedin.
- Inspect for particulate matter and discoloration prior to further dilution. Discard vial if particles or discoloration are observed.
- Immediately following reconstitution, withdraw the calculated volume of trabectedin and further dilute in 500 mL of 0.9% Sodium Chloride, USP or 5% Dextrose Injection, USP.
- Do not mix YONDELIS with other drugs.
- Discard any remaining solution within 30 hours of reconstituting the lyophilized powder.
- YONDELIS diluted solution is compatible with Type I colorless glass vials, polyvinylchloride (PVC) and polyethylene (PE) bags and tubing, PE and polypropylene (PP) mixture bags, polyethersulfone (PES) in-line filters, titanium, platinum or plastic ports, silicone and polyurethane catheters, and pumps having contact surfaces made of PVC, PE, or PE/PP.
16 How Supplied/storage and Handling
YONDELIS ®is supplied in a single-dose glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
7.2 Effect of Cytochrome Cyp3a Inducers
Coadministration of YONDELIS with rifampin, a strong CYP3A inducer, decreased systemic exposure of trabectedin by 31%. Avoid using strong CYP3A inducers (e.g., rifampin, phenobarbital, St. John's wort) in patients taking YONDELIS [see Clinical Pharmacology (12.3)] .
Principal Display Panel 1 Vial Carton
NDC 59676-610-01
Yondelis
®
(trabectedin)
for Injection
1 mg per vial
For Intravenous Infusion Only
Reconstitute before further dilution
Each vial contains 1 mg of trabectedin as a
sterile lyophilized powder.
Rx only
Single-dose vial
Discard any unused portion
Cytotoxic
7.1 Effect of Cytochrome Cyp3a Inhibitors
Coadministration of YONDELIS with ketoconazole, a strong CYP3A inhibitor, increased systemic exposure of trabectedin by 66%. Avoid using strong CYP3A inhibitors (e.g., oral ketoconazole, itraconazole, posaconazole, voriconazole, clarithromycin, telithromycin, indinavir, lopinavir, ritonavir, boceprevir, nelfinavir, saquinavir, telaprevir, nefazodone, conivaptan) in patients taking YONDELIS. If a strong CYP3A inhibitor for short-term use (i.e., less than 14 days) must be used, administer the strong CYP3A inhibitor 1 week after the YONDELIS infusion, and discontinue it the day prior to the next YONDELIS infusion [see Clinical Pharmacology (12.3)] .
2.4 Dosage Modifications for Adverse Reactions
Permanently discontinue YONDELIS for:
- Persistent adverse reactions requiring a delay in dosing of more than 3 weeks.
- Adverse reactions requiring dose reduction following YONDELIS administered at 1.0 mg/m 2for patients with normal hepatic function or at 0.3 mg/m 2for patients with pre-existing moderate hepatic impairment.
- Severe liver dysfunction: bilirubin two times the upper limit of normal, and AST or ALT three times the upper limit of normal, and alkaline phosphatase less than two times the upper limit of normal in the prior treatment cycle for patients with normal liver function at baseline.
- Exacerbation of liver dysfunction in patients with pre-existing moderate hepatic impairment.
- Capillary leak syndrome.
- Rhabdomyolysis.
- Grade 3 or 4 cardiac adverse events (AEs) indicative of cardiomyopathy or for subjects with an LVEF that decreases below the lower limit of normal.
The recommended dose modifications for adverse reactions are listed in Table 1. Once reduced, the dose of YONDELIS should not be increased in subsequent treatment cycles.
| Laboratory Result or Adverse Reaction | DELAY next dose of YONDELIS for up to 3 weeks | REDUCE next dose of YONDELIS by one dose level for adverse reaction(s) during prior cycle |
|---|---|---|
| Platelets | Less than 100,000 platelets/microliter | Less than 25,000 platelets/microliter |
| Absolute neutrophil count | Less than 1,500 neutrophils/microliter |
|
| Total bilirubin | Greater than the upper limit of normal | Greater than the upper limit of normal |
| Aspartate aminotransferase (AST) or alanine aminotransferase (ALT) | More than 2.5 times the upper limit of normal | More than 5 times the upper limit of normal |
| Alkaline phosphatase (ALP) | More than 2.5 times the upper limit of normal | More than 2.5 times the upper limit of normal |
| Creatine phosphokinase | More than 2.5 times the upper limit of normal | More than 5 times the upper limit of normal |
| Other non-hematologic adverse reactions | Grade 3 or 4 | Grade 3 or 4 |
The recommended starting doses and dose reductions for YONDELIS are listed in Table 2:
| Starting Dose and Dose Reduction | For patients with normal hepatic function or mild hepatic impairment
Including patients with bilirubin greater than 1 to 1.5 times the upper limit of normal, and any AST or ALT. prior to initiation of YONDELIS treatment
|
For patients with moderate hepatic impairment
Including patients with bilirubin levels greater than 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal. prior to initiation of YONDELIS treatment
|
|---|---|---|
| Starting Dose | 1.5 mg/m 2 | 0.9 mg/m 2 |
| Dose Reduction | ||
| First dose reduction | 1.2 mg/m 2 | 0.6 mg/m 2 |
| Second dose reduction | 1.0 mg/m 2 | 0.3 mg/m 2 |
5.6 Extravasation Resulting in Tissue Necrosis
Extravasation of YONDELIS, resulting in tissue necrosis requiring debridement, can occur. Evidence of tissue necrosis can occur more than 1 week after the extravasation. There is no specific antidote for extravasation of YONDELIS. Administer YONDELIS through a central venous line [see Dosage and Administration (2.6)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Trabectedin is genotoxic in both in vitroand in vivostudies. Long-term carcinogenicity studies have not been performed.
Fertility studies with trabectedin were not performed. In male rats there were limited histopathological signs of hemorrhage and degeneration in the testes following repeated administration of trabectedin at doses approximately 0.2 times the 1.5 mg/m 2human dose based on body surface area.
2.2 Recommended Dosage in Patients With Hepatic Impairment
The recommended dosage of YONDELIS in patients with moderate hepatic impairment (bilirubin levels greater than 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal) is 0.9 mg/m 2every 21 days (3 weeks).
Do not administer YONDELIS to patients with severe hepatic impairment (bilirubin levels above 3 times the upper limit of normal, and any AST and ALT) [ see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
Structured Label Content
Section 34073-7 (34073-7)
Drug Interactions
Section 42229-5 (42229-5)
Risk Summary
Based on its mechanism of action, trabectedin can cause fetal harm when administered during pregnancy [see Clinical Pharmacology (12.1)] . There are no available data with the use of YONDELIS during pregnancy. Animal reproductive and developmental studies at relevant doses have not been conducted with trabectedin; however, placental transfer of trabectedin was demonstrated in pregnant rats. Advise pregnant woman of the potential risk to a fetus. The background risk of major birth defects and miscarriage for the indicated population are unknown; however, the background risk in the U.S. general population of major birth defects is 2 to 4% and of miscarriage is 15 to 20% of clinically recognized pregnancies.
Section 42230-3 (42230-3)
| PATIENT INFORMATION
YONDELIS ®(yon-DEL-ess) (trabectedin) for injection, for intravenous use |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised 12/2025 | |
|
What is YONDELIS?
YONDELIS is a prescription medicine used to treat adults with liposarcoma or leiomyosarcoma that:
|
||
|
Who should not receive YONDELIS?
Do not receive YONDELIS if you have had a severe allergic reaction to trabectedin or any of the ingredients in YONDELIS. See the end of this leaflet for a complete list of ingredients in YONDELIS. |
||
Before receiving YONDELIS, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using YONDELIS with certain other medicines may affect the way YONDELIS works and may increase your risk of side effects. Know the medicines you take and keep a list of them to show to your healthcare provider or pharmacist when you get a new medicine. |
||
How will I receive YONDELIS?
|
||
|
What are the possible side effects of YONDELIS?
YONDELIS can cause serious side effects, including :
|
||
|
|
|
|
||
|
|
|
The most common severe abnormal blood tests include:
Your healthcare provider may decrease or delay your dose if you develop serious side effects or may permanently stop treatment with YONDELIS if your side effects are severe. YONDELIS may cause fertility problems in males and females, which may affect your ability to have children. Talk to your healthcare provider if this is a concern for you. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of YONDELIS. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about the safe and effective use of YONDELIS
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your healthcare provider or pharmacist for information about YONDELIS that is written for health professionals. |
||
|
What are the ingredients in YONDELIS?
Active ingredient: trabectedin Inactive ingredients:potassium dihydrogen phosphate, sucrose, phosphoric acid and potassium hydroxide. Manufactured for: Janssen Products, LP, Horsham, PA 19044, USA For more information, call 1-800-526-7736 or go to www.YONDELIS.com. For patent information: www.janssenpatents.com |
Section 43683-2 (43683-2)
| Warnings and Precautions ( 5.7) | 12/2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store YONDELIS vials in a refrigerator at 2 °C to 8 °C (36 °F to 46 °F).
YONDELIS is a hazardous drug. Follow applicable special handling and disposal procedures. 1
10 Overdosage (10 OVERDOSAGE)
There is no specific antidote for YONDELIS. Hemodialysis is not expected to enhance the elimination of YONDELIS because trabectedin is highly bound to plasma proteins (97%) and not significantly renally excreted.
15 References (15 REFERENCES)
- "OSHA Hazardous Drugs." OSHA.http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Trabectedin is an alkylating drug with the chemical name (1' R,6 R,6a R,7 R,13 S,14 S,16 R)-5-(acetyloxy)-3',4',6,6a,7,13,14,16-octahydro-6',8,14-trihydroxy-7',9-dimethoxy-4,10,23-trimethyl-spiro[6,16-(epithiopropanoxymethano)-7,13-imino-12 H-1,3-dioxolo[7,8]isoquino[3,2- b][3]benzazocine-20,1'(2' H)-isoquinolin]-19-one. The molecular formula is C 39H 43N 3O 11S. The molecular weight is 761.84 daltons. The chemical structure is shown below:
Trabectedin is hydrophobic and has a low solubility in water.
YONDELIS ®(trabectedin) for injection is supplied as a sterile lyophilized white to off-white powder/cake in a single-dose vial. Each single-dose vial contains 1 mg of trabectedin, 27.2 mg potassium dihydrogen phosphate, 400 mg sucrose, and phosphoric acid and potassium hydroxide (for pH adjustment to 3.6 – 4.2).
2.3 Premedication
Administer dexamethasone 20 mg intravenously 30 minutes prior to each dose of YONDELIS.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Safety (n=61) and efficacy (n=58) were assessed across five open-label studies (NCT00006463, NCT01453283, NCT00005625, NCT00070109, and ET-B-023-00) in pediatric patients (aged 2 to <17 years) with pediatric histotypes of sarcoma (predominantly rhabdomyosarcoma, osteosarcoma, Ewing sarcoma, and non-rhabdomyosarcoma soft tissue sarcoma). No new safety signals were observed in pediatric patients across these studies.
Pharmacokinetic parameters in 17 pediatric patients (aged 3 to 17 years) were within the range of values previously observed in adults given the same dose per body surface area.
8.5 Geriatric Use
Clinical studies of YONDELIS did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
2.6 Administration
- Infuse the reconstituted, diluted solution over 24 hours through a central venous line using an infusion set with a 0.2 micron polyethersulfone (PES) in-line filter.
- Complete infusion within 30 hours of initial reconstitution. Discard any unused portion of the reconstituted product or of the infusion solution.
5.2 Rhabdomyolysis
YONDELIS can cause rhabdomyolysis and musculoskeletal toxicity. In Trial ET743-SAR-3007, rhabdomyolysis leading to death occurred in 3 (0.8%) of the 378 patients receiving YONDELIS. Elevations in creatine phosphokinase (CPK) occurred in 122 (32%) of the 378 patients receiving YONDELIS, including Grade 3 or 4 CPK elevation in 24 patients (6%), compared to 15 (9%) of the 172 patients receiving dacarbazine with any CPK elevation, including 1 patient (0.6%) with Grade 3 CPK elevation. Among the 24 patients receiving YONDELIS with Grade 3 or 4 CPK elevation, renal failure occurred in 11 patients (2.9%); rhabdomyolysis with the complication of renal failure occurred in 4 of these 11 patients (1.1%). The median time to first occurrence of Grade 3 or 4 CPK elevations was 2 months (range: 1 to 11.5 months). The median time to complete resolution was 14 days (range: 5 days to 1 month).
Assess CPK levels prior to each administration of YONDELIS. Withhold, reduce dose, or permanently discontinue based on severity of adverse reaction [see Dosage and Administration (2.4)] .
5.3 Hepatotoxicity
Hepatotoxicity, including hepatic failure, can occur with YONDELIS. Patients with serum bilirubin levels above the upper limit of normal or AST or ALT levels >2.5 × upper limit of normal were not enrolled in Trial ET743-SAR-3007. In Trial ET743-SAR-3007, the incidence of Grade 3–4 elevated liver function tests (LFTs; defined as elevations in ALT, AST, total bilirubin, or alkaline phosphatase) was 35% (134/378) in patients receiving YONDELIS. The median time to development of Grade 3–4 elevation in ALT or AST was 29 days (range: 3 days to 11.5 months). Of the 134 patients with Grade 3–4 elevations in LFTs, 114 (85%) experienced complete resolution with the median time to complete resolution of 13 days (range: 4 days to 4.4 months).
In Trial ET743-SAR-3007, the incidence of drug-induced liver injury (defined as concurrent elevation in ALT or AST of more than three times the upper limit of normal, alkaline phosphatase less than two times the upper limit of normal, and total bilirubin at least two times the upper limit of normal) was 1.3% (5/378) in patients receiving YONDELIS. ALT or AST elevation greater than eight times the upper limit of normal occurred in 18% (67/378) of patients receiving YONDELIS.
Assess LFTs prior to each administration of YONDELIS and as clinically indicated based on underlying severity of pre-existing hepatic impairment. Manage elevated LFTs with treatment interruption, dose reduction, or permanent discontinuation based on severity and duration of LFT abnormality [see Dosage and Administration (2.4)and Use in Specific Populations (8.6)].
5.4 Cardiomyopathy
Cardiomyopathy including cardiac failure, congestive heart failure, ejection fraction decreased, diastolic dysfunction, or right ventricular dysfunction can occur with YONDELIS. In Trial ET743-SAR-3007, a significant decrease in LVEF was defined as an absolute decrease of ≥15% or below the lower limit of normal with an absolute decrease of ≥5%. Patients with a history of New York Heart Association Class II to IV heart failure or abnormal left ventricular ejection fraction (LVEF) at baseline were ineligible. In Trial ET743-SAR-3007, cardiomyopathy occurred in 23 patients (6%) receiving YONDELIS and in four patients (2.3%) receiving dacarbazine. Grade 3 or 4 cardiomyopathy occurred in 15 patients (4%) receiving YONDELIS and 2 patients (1.2%) receiving dacarbazine; cardiomyopathy leading to death occurred in 1 patient (0.3%) receiving YONDELIS and in none of the patients receiving dacarbazine. The median time to development of Grade 3 or 4 cardiomyopathy in patients receiving YONDELIS was 5.3 months (range: 26 days to 15.3 months).
Patients with LVEF < lower limit of normal, prior cumulative anthracycline dose of ≥300 mg/m 2, age ≥65 years, or a history of cardiovascular disease may be at increased risk of cardiac dysfunction. Assess LVEF by echocardiogram (ECHO) or multigated acquisition (MUGA) scan before initiation of YONDELIS and at 2- to 3-month intervals thereafter until YONDELIS is discontinued. Discontinue treatment with YONDELIS based on severity of adverse reaction [see Dosage and Administration (2.4)] .
14 Clinical Studies (14 CLINICAL STUDIES)
The clinical efficacy and safety of YONDELIS in patients with metastatic or recurrent leiomyosarcoma or liposarcoma were demonstrated in Trial ET743-SAR-3007 (NCT01343277), a randomized (2:1), open-label, active-controlled trial comparing treatment with YONDELIS 1.5 mg/m 2as a 24-hour continuous intravenous infusion once every 3 weeks to dacarbazine 1000 mg/m 2intravenous infusion (20 to 120 minutes) once every 3 weeks. Treatment continued in both arms until disease progression or unacceptable toxicity; all patients in the YONDELIS arm were required to receive dexamethasone 20 mg intravenous injection prior to each YONDELIS infusion. Patients were required to have unresectable, locally advanced or metastatic leiomyosarcoma or liposarcoma (dedifferentiated, myxoid round cell, or pleomorphic) and previous treatment with an anthracycline- and ifosfamide-containing regimen or an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen. Randomization was stratified by subtype of soft tissue sarcoma (leiomyosarcoma vs. liposarcoma), ECOG performance status (0 vs. 1), and number of prior chemotherapy regimens (1 vs. ≥2). The efficacy outcome measures were investigator-assessed progression-free survival (PFS) according to the Response Evaluation Criteria in Solid Tumors (RECIST v1.1), overall survival (OS), objective response rate (ORR), and duration of response (DOR). Patients in the dacarbazine arm were not offered YONDELIS at the time of disease progression.
A total of 518 patients were randomized, 345 to the YONDELIS arm and 173 patients to the dacarbazine arm. The median patient age was 56 years (range: 17 to 81); 30% were male; 76% White, 12% Black, and 4% Asian; 73% had leiomyosarcomas and 27% liposarcomas; 49% had an ECOG PS of 0; and 89% received ≥2 prior chemotherapy regimens. The most common (≥20%) pre-study chemotherapeutic agents administered were doxorubicin (90%), gemcitabine (81%), docetaxel (74%), and ifosfamide (59%). Approximately 10% of patients had received pazopanib.
Trial ET743-SAR-3007 demonstrated a statistically significant improvement in PFS. An exploratory analysis of independent radiology committee-determined PFS, in a subgroup consisting of approximately 60% of the total population, provided similar results to the investigator-determined PFS. Efficacy results from Trial ET743-SAR-3007 are presented in the table below.
| Efficacy Endpoint | YONDELIS
N=345 |
Dacarbazine
N=173 |
|---|---|---|
| CR=Complete Response; PR=Partial Response; CI=Confidence Interval, HR=hazard ratio, NE=not estimable. | ||
| Progression-free survival | ||
| PFS Events, n (%) | 217 (63%) | 112 (65%) |
| Disease progression | 204 | 109 |
| Death | 13 | 3 |
| Median (95% CI) (months) | 4.2 (3.0, 4.8) | 1.5 (1.5, 2.6) |
| HR (95% CI)
Cox proportional hazards model with treatment group as the only covariate.
|
0.55 (0.44, 0.70) | |
| p-value
Unstratified log rank test.
|
<0.001 | |
|
Overall survival
Based on 384 patients randomized to YONDELIS arm and 193 patients randomized to dacarbazine.
|
||
| Events, n (%) | 258 (67%) | 123 (64%) |
| Median (95% CI) (months) | 13.7 (12.2, 16.0) | 13.1 (9.1, 16.2) |
| HR (95% CI) | 0.93 (0.75, 1.15) | |
| p-value | 0.49 | |
| Objective Response Rate (ORR: CR+PR) | ||
| Number of patients (%) | 23 (7%) | 10 (6%) |
| 95% CI
Fisher's exact CI.
|
(4.3, 9.8) | (2.8, 10.4) |
| Duration of Response (CR+ PR) | ||
| Median (95% CI) (months) | 6.9 (4.5, 7.6) | 4.2 (2.9, NE) |
Figure 1: Kaplan-Meier Curves of Progression-Free Survival in Trial ET743-SAR-3007
4 Contraindications (4 CONTRAINDICATIONS)
YONDELIS is contraindicated in patients with known severe hypersensitivity, including anaphylaxis, to trabectedin.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Anaphylaxis [see Contraindications (4)]
- Neutropenic Sepsis [see Warnings and Precautions (5.1)]
- Rhabdomyolysis [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
- Cardiomyopathy [see Warnings and Precautions (5.4)]
- Capillary Leak Syndrome [see Warnings and Precautions (5.5)]
- Extravasation Resulting in Tissue Necrosis [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
No dose adjustment is recommended in patients with mild [creatinine clearance (CLcr) 60–89 mL/min] or moderate (CLcr of 30–59 mL/min) renal impairment.
The pharmacokinetics of trabectedin has not been evaluated in patients with severe renal impairment (CLcr <30 mL/min) or end stage renal disease [see Clinical Pharmacology (12.3)] .
12.3 Pharmacokinetics
The pharmacokinetics of trabectedin is characterized by a rapid decline phase at the end of the infusion and slower exponential phases. Population pharmacokinetic analyses suggest that the pharmacokinetics of trabectedin is dose-proportional (over the dose range of 0.024 to 1.8 mg/m 2) and exposure is time-independent. No accumulation of trabectedin in plasma is observed upon repeated administrations every 3 weeks.
2.1 Recommended Dosage
The recommended dose is 1.5 mg/m 2administered as an intravenous infusion over 24 hours through a central venous line every 21 days (3 weeks), until disease progression or unacceptable toxicity.
5.1 Neutropenic Sepsis
Neutropenic sepsis, including fatal cases, can occur with YONDELIS. In Trial ET743-SAR-3007, the incidence of Grade 3 or 4 neutropenia, based on laboratory values, in patients receiving YONDELIS was 43% (161/378). The median time to the first occurrence of Grade 3 or 4 neutropenia was 16 days (range: 8 days to 9.7 months); the median time to complete resolution of neutropenia was 13 days (range: 3 days to 2.3 months). Febrile neutropenia (fever ≥38.5 °C with Grade 3 or 4 neutropenia) occurred in 18 patients (5%) treated with YONDELIS. Ten patients (2.6%) experienced neutropenic sepsis, 5 of whom had febrile neutropenia, which was fatal in 4 patients (1.1%).
Assess neutrophil count prior to administration of each dose of YONDELIS and periodically throughout the treatment cycle. Withhold or reduce dose of YONDELIS based on severity of adverse reaction [see Dosage and Administration (2.4)] .
8.6 Hepatic Impairment
The mean trabectedin exposure was (97%) higher in patients with moderate (bilirubin levels greater than 1.5 to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal) hepatic impairment compared to patients with normal (total bilirubin ≤ the upper limit of normal, and AST and ALT ≤ the upper limit of normal) liver function. Reduce YONDELIS dose in patients with moderate hepatic impairment [see Dosage and Administration (2.2)and Clinical Pharmacology (12.3)] .
Do not administer YONDELIS to patients with severe hepatic impairment (bilirubin levels above 3 times the upper limit of normal, and any AST and ALT) [see Warnings and Precautions (5.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
YONDELIS ®is indicated for the treatment of adult patients with unresectable or metastatic liposarcoma or leiomyosarcoma who received a prior anthracycline-containing regimen [see Clinical Studies (14)] .
12.1 Mechanism of Action
Trabectedin is an alkylating drug that binds guanine residues in the minor groove of DNA, forming adducts and resulting in a bending of the DNA helix towards the major groove. Adduct formation triggers a cascade of events that can affect the subsequent activity of DNA binding proteins, including some transcription factors, and DNA repair pathways, resulting in perturbation of the cell cycle and eventual cell death.
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on its mechanism of action, YONDELIS can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during therapy and for at least 8 months after the last dose of YONDELIS. Advise males with female partners of reproductive potential to use effective contraception during therapy and for at least 5 months after the last dose of YONDELIS [see Use in Specific Populations (8.1, 8.3)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Neutropenic sepsis: Severe, and fatal, neutropenic sepsis may occur. Monitor neutrophil count during treatment. Withhold YONDELIS for neutrophil count < 1,500/mcL ( 2.4, 5.1)
- Rhabdomyolysis: Rhabdomyolysis may occur. Monitor creatine phosphokinase (CPK) levels prior to each administration. Withhold YONDELIS for CPK more than 2.5 times the upper limit of normal. ( 2.4, 5.2)
- Hepatotoxicity: Hepatotoxicity may occur. Monitor and delay and/or reduce dose if needed ( 5.3)
- Cardiomyopathy: Severe and fatal cardiomyopathy can occur. Patients with left ventricular ejection fraction (LVEF) < lower limit of normal, prior cumulative anthracycline dose of ≥300 mg/m 2, age ≥65 years, or a history of cardiovascular disease may be at increased risk of developing new or worsening cardiac dysfunction. Discontinue YONDELIS in patients who develop decreased LVEF or cardiomyopathy ( 2.4, 5.4)
- Capillary leak syndrome: Monitor and discontinue YONDELIS for capillary leak syndrome ( 5.5)
- Embryo-fetal toxicity: Can cause fetal harm. Advise of potential risk to a fetus and use effective contraception ( 5.7, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer at 1.5 mg/m 2as a 24-hour intravenous infusion, every 3 weeks through a central venous line ( 2.1, 2.6)
- Premedication: dexamethasone 20 mg intravenously, 30 min before each infusion ( 2.3)
- Hepatic Impairment: Administer at 0.9 mg/m 2as a 24-hour intravenous infusion, every 3 weeks through a central venous line in patients with moderate hepatic impairment ( 2.2)
5.5 Capillary Leak Syndrome
Capillary leak syndrome (CLS) characterized by hypotension, edema, and hypoalbuminemia has been reported with YONDELIS, including serious CLS resulting in death. Monitor for signs and symptoms of CLS. Discontinue YONDELIS and promptly initiate standard management for patients with CLS, which may include a need for intensive care [see Adverse Reactions (6.2)] .
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 1 mg, lyophilized powder in single-dose vial for reconstitution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of YONDELIS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Vascular disorders: capillary leak syndrome
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to YONDELIS in 755 patients with soft tissue sarcoma including 197 (26%) patients exposed to YONDELIS for greater than or equal to 6 months and 57 (8%) patients exposed to YONDELIS for greater than or equal to 1 year. The safety of YONDELIS was evaluated in six open-label, single-arm trials, in which 377 patients received YONDELIS and one open-label, randomized, active-controlled clinical trial in which 378 patients received YONDELIS (Trial ET743-SAR-3007). All patients received YONDELIS at the recommended dosing regimen of 1.5 mg/m 2administered as an intravenous infusion over 24 hours once every 3 weeks (q3wk, 24-h). The median age was 54 years (range: 18 to 81 years), 63% were female, and all patients had metastatic soft tissue sarcoma.
Tables 3 and 4 present selected adverse reactions and laboratory abnormalities, respectively, observed in Trial ET743-SAR-3007, an open-label, randomized (2:1), active-controlled trial in which 550 patients with previously treated leiomyosarcoma or liposarcoma (dedifferentiated, myxoid round cell, or pleomorphic) received YONDELIS 1.5 mg/m 2intravenous infusion over 24 hours once every 3 weeks (n=378) or dacarbazine 1000 mg/m 2intravenous infusion over 20 to 120 minutes once every 3 weeks (n=172) [see Clinical Studies (14)] . All patients treated with YONDELIS were required to receive dexamethasone 20 mg intravenous injection 30 minutes prior to start of the YONDELIS infusion.
In Trial ET743-SAR-3007, patients had been previously treated with an anthracycline- and ifosfamide-containing regimen or with an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen. The trial excluded patients with known central nervous system metastasis, elevated serum bilirubin or significant chronic liver disease, such as cirrhosis or active hepatitis, and history of myocardial infarction within 6 months, history of New York Heart Association Class II to IV heart failure, or abnormal left ventricular ejection fraction at baseline. The median age of patients in Trial ET743-SAR-3007 was 57 years (range: 17 to 81 years), with 69% female, 77% White, 12% Black or African American, 4% Asian, and <1% American Indian or Alaska Native. The median duration of exposure to trabectedin was 13 weeks (range: 1 to 127 weeks) with 30% of patients exposed to YONDELIS for greater than 6 months and 7% of patients exposed to YONDELIS for greater than 1 year.
In Trial ET743-SAR-3007, adverse reactions resulting in permanent discontinuation of YONDELIS occurred in 26% (98/378) of patients; the most common were increased liver tests (defined as ALT, AST, alkaline phosphatase, bilirubin) (5.6%), thrombocytopenia (3.4%), fatigue (1.6%), increased creatine phosphokinase (1.1%), and decreased ejection fraction (1.1%). Adverse reactions that led to dose reductions occurred in 42% (158/378) of patients treated with YONDELIS; the most common were increased liver tests (24%), neutropenia (including febrile neutropenia) (8%), thrombocytopenia (4.2%), fatigue (3.7%), increased creatine phosphokinase (2.4%), nausea (1.1%), and vomiting (1.1%). Adverse reactions led to dose interruptions in 52% (198/378) of patients treated with YONDELIS; the most common were neutropenia (31%), thrombocytopenia (15%), increased liver tests (6%), fatigue (2.9%), anemia (2.6%), increased creatinine (1.1%), and nausea (1.1%).
The most common adverse reactions (≥20%) were nausea, fatigue, vomiting, constipation, decreased appetite, diarrhea, peripheral edema, dyspnea, and headache. The most common laboratory abnormalities (≥20%) were increases in AST or ALT, increased alkaline phosphatase, hypoalbuminemia, increased creatinine, increased creatine phosphokinase, anemia, neutropenia, and thrombocytopenia.
| YONDELIS
(N=378) |
Dacarbazine
(N=172) |
|||
|---|---|---|---|---|
| System Organ Class
Adverse Reaction |
All Grades
Toxicity grade is based on NCI common toxicity criteria, version 4.0.
(%) |
Grades 3–4
(%) |
All Grades
(%) |
Grades 3–4
(%) |
| Gastrointestinal disorders | ||||
| Nausea | 75 | 7 | 50 | 1.7 |
| Vomiting | 46 | 6 | 22 | 1.2 |
| Constipation | 37 | 0.8 | 31 | 0.6 |
| Diarrhea | 35 | 1.6 | 23 | 0 |
| General disorders and administration site conditions | ||||
| Fatigue
Fatigue is a composite of the following adverse event terms: fatigue, asthenia, and malaise.
|
69 | 8 | 52 | 1.7 |
| Peripheral edema | 28 | 0.8 | 13 | 0.6 |
| Metabolism and nutrition disorders | ||||
| Decreased appetite | 37 | 1.9 | 21 | 0.6 |
| Respiratory, thoracic and mediastinal disorders | ||||
| Dyspnea | 25 | 4.2 | 20 | 1.2 |
| Nervous system disorders | ||||
| Headache | 25 | 0.3 | 19 | 0 |
| Musculoskeletal and connective tissue disorders | ||||
| Arthralgia | 15 | 0 | 8 | 1.2 |
| Myalgia | 12 | 0 | 6 | 0 |
| Psychiatric disorders | ||||
| Insomnia | 15 | 0.3 | 9 | 0 |
Other clinically important adverse reactions observed in <10% of patients (N=755) with soft tissue sarcoma receiving YONDELIS were:
Nervous system disorders: peripheral neuropathy, paresthesia, hypoesthesia.
Respiratory, thoracic, and mediastinal disorders: pulmonary embolism.
General disorders and administration site conditions: mucosal inflammation
| Laboratory Abnormalities | YONDELIS | Dacarbazine | ||
|---|---|---|---|---|
| All Grades
(%) |
Grades 3–4
(%) |
All Grades
(%) |
Grades 3–4
(%) |
|
| YONDELIS group (range: 373 to 377 patients) and dacarbazine group (range: 166 to 168 patients). | ||||
| Chemistry | ||||
| Increased ALT | 90 | 31 | 33 | 0.6 |
| Increased AST | 84 | 17 | 32 | 1.2 |
| Increased alkaline phosphatase | 70 | 1.6 | 60 | 0.6 |
| Hypoalbuminemia | 63 | 3.7 | 51 | 3.0 |
| Increased creatinine | 46 | 4.2 | 29 | 1.2 |
| Increased creatine phosphokinase | 33 | 6.4 | 9 | 0.6 |
| Hyperbilirubinemia | 13 | 1.9 | 5 | 0.6 |
| Hematology | ||||
| Anemia | 96 | 19 | 79 | 12 |
| Neutropenia | 66 | 43 | 47 | 26 |
| Thrombocytopenia | 59 | 21 | 57 | 20 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.5 Preparation for Administration
- YONDELIS is a hazardous drug. Follow applicable special handling and disposal procedures. 1
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Using aseptic technique, inject 20 mL of Sterile Water for Injection, USP into the vial. Shake the vial until complete dissolution. The reconstituted solution is clear, colorless to pale brownish-yellow, and contains 0.05 mg/mL of trabectedin.
- Inspect for particulate matter and discoloration prior to further dilution. Discard vial if particles or discoloration are observed.
- Immediately following reconstitution, withdraw the calculated volume of trabectedin and further dilute in 500 mL of 0.9% Sodium Chloride, USP or 5% Dextrose Injection, USP.
- Do not mix YONDELIS with other drugs.
- Discard any remaining solution within 30 hours of reconstituting the lyophilized powder.
- YONDELIS diluted solution is compatible with Type I colorless glass vials, polyvinylchloride (PVC) and polyethylene (PE) bags and tubing, PE and polypropylene (PP) mixture bags, polyethersulfone (PES) in-line filters, titanium, platinum or plastic ports, silicone and polyurethane catheters, and pumps having contact surfaces made of PVC, PE, or PE/PP.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
YONDELIS ®is supplied in a single-dose glass vial containing 1 mg trabectedin. Each carton contains one vial (NDC: 59676-610-01).
7.2 Effect of Cytochrome Cyp3a Inducers (7.2 Effect of Cytochrome CYP3A Inducers)
Coadministration of YONDELIS with rifampin, a strong CYP3A inducer, decreased systemic exposure of trabectedin by 31%. Avoid using strong CYP3A inducers (e.g., rifampin, phenobarbital, St. John's wort) in patients taking YONDELIS [see Clinical Pharmacology (12.3)] .
Principal Display Panel 1 Vial Carton (PRINCIPAL DISPLAY PANEL - 1 Vial Carton)
NDC 59676-610-01
Yondelis
®
(trabectedin)
for Injection
1 mg per vial
For Intravenous Infusion Only
Reconstitute before further dilution
Each vial contains 1 mg of trabectedin as a
sterile lyophilized powder.
Rx only
Single-dose vial
Discard any unused portion
Cytotoxic
7.1 Effect of Cytochrome Cyp3a Inhibitors (7.1 Effect of Cytochrome CYP3A Inhibitors)
Coadministration of YONDELIS with ketoconazole, a strong CYP3A inhibitor, increased systemic exposure of trabectedin by 66%. Avoid using strong CYP3A inhibitors (e.g., oral ketoconazole, itraconazole, posaconazole, voriconazole, clarithromycin, telithromycin, indinavir, lopinavir, ritonavir, boceprevir, nelfinavir, saquinavir, telaprevir, nefazodone, conivaptan) in patients taking YONDELIS. If a strong CYP3A inhibitor for short-term use (i.e., less than 14 days) must be used, administer the strong CYP3A inhibitor 1 week after the YONDELIS infusion, and discontinue it the day prior to the next YONDELIS infusion [see Clinical Pharmacology (12.3)] .
2.4 Dosage Modifications for Adverse Reactions
Permanently discontinue YONDELIS for:
- Persistent adverse reactions requiring a delay in dosing of more than 3 weeks.
- Adverse reactions requiring dose reduction following YONDELIS administered at 1.0 mg/m 2for patients with normal hepatic function or at 0.3 mg/m 2for patients with pre-existing moderate hepatic impairment.
- Severe liver dysfunction: bilirubin two times the upper limit of normal, and AST or ALT three times the upper limit of normal, and alkaline phosphatase less than two times the upper limit of normal in the prior treatment cycle for patients with normal liver function at baseline.
- Exacerbation of liver dysfunction in patients with pre-existing moderate hepatic impairment.
- Capillary leak syndrome.
- Rhabdomyolysis.
- Grade 3 or 4 cardiac adverse events (AEs) indicative of cardiomyopathy or for subjects with an LVEF that decreases below the lower limit of normal.
The recommended dose modifications for adverse reactions are listed in Table 1. Once reduced, the dose of YONDELIS should not be increased in subsequent treatment cycles.
| Laboratory Result or Adverse Reaction | DELAY next dose of YONDELIS for up to 3 weeks | REDUCE next dose of YONDELIS by one dose level for adverse reaction(s) during prior cycle |
|---|---|---|
| Platelets | Less than 100,000 platelets/microliter | Less than 25,000 platelets/microliter |
| Absolute neutrophil count | Less than 1,500 neutrophils/microliter |
|
| Total bilirubin | Greater than the upper limit of normal | Greater than the upper limit of normal |
| Aspartate aminotransferase (AST) or alanine aminotransferase (ALT) | More than 2.5 times the upper limit of normal | More than 5 times the upper limit of normal |
| Alkaline phosphatase (ALP) | More than 2.5 times the upper limit of normal | More than 2.5 times the upper limit of normal |
| Creatine phosphokinase | More than 2.5 times the upper limit of normal | More than 5 times the upper limit of normal |
| Other non-hematologic adverse reactions | Grade 3 or 4 | Grade 3 or 4 |
The recommended starting doses and dose reductions for YONDELIS are listed in Table 2:
| Starting Dose and Dose Reduction | For patients with normal hepatic function or mild hepatic impairment
Including patients with bilirubin greater than 1 to 1.5 times the upper limit of normal, and any AST or ALT. prior to initiation of YONDELIS treatment
|
For patients with moderate hepatic impairment
Including patients with bilirubin levels greater than 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal. prior to initiation of YONDELIS treatment
|
|---|---|---|
| Starting Dose | 1.5 mg/m 2 | 0.9 mg/m 2 |
| Dose Reduction | ||
| First dose reduction | 1.2 mg/m 2 | 0.6 mg/m 2 |
| Second dose reduction | 1.0 mg/m 2 | 0.3 mg/m 2 |
5.6 Extravasation Resulting in Tissue Necrosis
Extravasation of YONDELIS, resulting in tissue necrosis requiring debridement, can occur. Evidence of tissue necrosis can occur more than 1 week after the extravasation. There is no specific antidote for extravasation of YONDELIS. Administer YONDELIS through a central venous line [see Dosage and Administration (2.6)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Trabectedin is genotoxic in both in vitroand in vivostudies. Long-term carcinogenicity studies have not been performed.
Fertility studies with trabectedin were not performed. In male rats there were limited histopathological signs of hemorrhage and degeneration in the testes following repeated administration of trabectedin at doses approximately 0.2 times the 1.5 mg/m 2human dose based on body surface area.
2.2 Recommended Dosage in Patients With Hepatic Impairment (2.2 Recommended Dosage in Patients with Hepatic Impairment)
The recommended dosage of YONDELIS in patients with moderate hepatic impairment (bilirubin levels greater than 1.5 times to 3 times the upper limit of normal, and AST and ALT less than 8 times the upper limit of normal) is 0.9 mg/m 2every 21 days (3 weeks).
Do not administer YONDELIS to patients with severe hepatic impairment (bilirubin levels above 3 times the upper limit of normal, and any AST and ALT) [ see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:16.387820 · Updated: 2026-03-14T22:44:06.575433