These Highlights Do Not Include All The Information Needed To Use Conexxence Safely And Effectively. See Full Prescribing Information For Conexxence.
39a0a789-778f-4695-b3a9-61643e6396cd
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Patients with advanced chronic kidney disease (eGFR < 30 mL/min/1.73 m 2 ), including dialysis-dependent patients, are at greater risk of severe hypocalcemia following denosumab products administration. Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported [see Warnings and Precautions ( 5.1 )] . The presence of chronic kidney disease-mineral bone disorder (CKD-MBD) markedly increases the risk of hypocalcemia in these patients [see Warnings and Precautions ( 5.1 )] . Prior to initiating Conexxence in patients with advanced chronic kidney disease, evaluate for the presence of CKD-MBD. Treatment with Conexxence in these patients should be supervised by a healthcare provider with expertise in the diagnosis and management of CKD-MBD [see Dosage and Administration ( 2.2 ) and Warnings and Precautions ( 5.1 )].
Indications and Usage
Conexxence is a RANK ligand (RANKL) inhibitor indicated for treatment: of postmenopausal women with osteoporosis at high risk for fracture ( 1.1 ) to increase bone mass in men with osteoporosis at high risk for fracture ( 1.2 ) of glucocorticoid-induced osteoporosis in men and women at high risk for fracture ( 1.3 ) to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy for nonmetastatic prostate cancer ( 1.4 ) to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer ( 1.5 )
Dosage and Administration
Pregnancy must be ruled out prior to administration of Conexxence. ( 2.1 ) Before initiating Conexxence in patients with advanced chronic kidney disease, including dialysis patients, evaluate for the presence of chronic kidney disease mineral and bone disorder with intact parathyroid hormone, serum calcium, 25(OH) vitamin D, and 1,25(OH)2 vitamin D. ( 2.2 , 5.1 , 8.6 ) Conexxence should be administered by a healthcare provider. ( 2.3 ) Administer 60 mg every 6 months as a subcutaneous injection in the upper arm, upper thigh, or abdomen. ( 2.3 ) Instruct patients to take calcium 1000 mg daily and at least 400 IU vitamin D daily. ( 2.3 )
Warnings and Precautions
Hypocalcemia: Pre-existing hypocalcemia must be corrected before initiating Conexxence. May worsen, especially in patients with renal impairment. Adequately supplement all patients with calcium and vitamin D. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk. Evaluate for presence of chronic kidney disease mineral-bone disorder. Monitor serum calcium. ( 5.1 ) Same Active Ingredient: Patients receiving Conexxence should not receive other denosumab products concomitantly ( 5.2 ) Hypersensitivity including anaphylactic reactions may occur. Discontinue permanently if a clinically significant reaction occurs. ( 5.3 ) Osteonecrosis of the jaw: Has been reported with denosumab products. Monitor for symptoms. ( 5.4 ) Atypical femoral fractures: Have been reported. Evaluate patients with thigh or groin pain to rule out a femoral fracture. ( 5.5 ) Multiple vertebral fractures have been reported following treatment discontinuation. Patients should be transitioned to another antiresorptive agent if Conexxence is discontinued. ( 5.6 ) Serious infections including skin infections: May occur, including those leading to hospitalization. Advise patients to seek prompt medical attention if they develop signs or symptoms of infection, including cellulitis. ( 5.7 ) Dermatologic reactions: Dermatitis, rashes, and eczema have been reported. Consider discontinuing Conexxence if severe symptoms develop. ( 5.8 ) Severe bone, joint, muscle pain may occur. Discontinue use if severe symptoms develop. ( 5.9 ) Suppression of bone turnover: Significant suppression has been demonstrated. Monitor for consequences of bone over-suppression. ( 5.10 )
Contraindications
Conexxence is contraindicated in: Patients with hypocalcemia: Pre-existing hypocalcemia must be corrected prior to initiating therapy with Conexxence [see Warnings and Precautions ( 5.1 )] . Pregnant women: Denosumab products may cause fetal harm when administered to a pregnant woman. In women of reproductive potential, pregnancy testing should be performed prior to initiating treatment with Conexxence [see Use in Specific Populations ( 8.1 )] . Patients with hypersensitivity to denosumab products: Conexxence is contraindicated in patients with a history of systemic hypersensitivity to any component of the product. Reactions have included anaphylaxis, facial swelling, and urticaria [see Warnings and Precautions ( 5.3 ), Adverse Reactions ( 6.2 )] .
Adverse Reactions
The following serious adverse reactions are discussed below and also elsewhere in the labeling: Severe Hypocalcemia and Mineral Metabolism Changes [see Warnings and Precautions ( 5.1 )] Hypersensitivity [see Warnings and Precautions ( 5.3 )] Osteonecrosis of the Jaw [see Warnings and Precautions ( 5.4 )] Atypical Subtrochanteric and Diaphyseal Femoral Fractures [see Warnings and Precautions ( 5.5 )] Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation of Treatment [see Warnings and Precautions ( 5.6 )] Serious Infections [see Warnings and Precautions ( 5.7 )] Dermatologic Adverse Reactions [see Warnings and Precautions ( 5.8 )] The most common adverse reactions reported with denosumab products in patients with postmenopausal osteoporosis are back pain, pain in extremity, musculoskeletal pain, hypercholesterolemia, and cystitis. The most common adverse reactions reported with denosumab products in men with osteoporosis are back pain, arthralgia, and nasopharyngitis. The most common adverse reactions reported with denosumab products in patients with glucocorticoid-induced osteoporosis are back pain, hypertension, bronchitis, and headache. The most common (per patient incidence ≥ 10%) adverse reactions reported with denosumab products in patients with bone loss receiving androgen deprivation therapy for prostate cancer or adjuvant aromatase inhibitor therapy for breast cancer are arthralgia and back pain. Pain in extremity and musculoskeletal pain have also been reported in clinical trials. The most common adverse reactions leading to discontinuation of denosumab products in patients with postmenopausal osteoporosis are back pain and constipation.
Medication Information
Warnings and Precautions
Hypocalcemia: Pre-existing hypocalcemia must be corrected before initiating Conexxence. May worsen, especially in patients with renal impairment. Adequately supplement all patients with calcium and vitamin D. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk. Evaluate for presence of chronic kidney disease mineral-bone disorder. Monitor serum calcium. ( 5.1 ) Same Active Ingredient: Patients receiving Conexxence should not receive other denosumab products concomitantly ( 5.2 ) Hypersensitivity including anaphylactic reactions may occur. Discontinue permanently if a clinically significant reaction occurs. ( 5.3 ) Osteonecrosis of the jaw: Has been reported with denosumab products. Monitor for symptoms. ( 5.4 ) Atypical femoral fractures: Have been reported. Evaluate patients with thigh or groin pain to rule out a femoral fracture. ( 5.5 ) Multiple vertebral fractures have been reported following treatment discontinuation. Patients should be transitioned to another antiresorptive agent if Conexxence is discontinued. ( 5.6 ) Serious infections including skin infections: May occur, including those leading to hospitalization. Advise patients to seek prompt medical attention if they develop signs or symptoms of infection, including cellulitis. ( 5.7 ) Dermatologic reactions: Dermatitis, rashes, and eczema have been reported. Consider discontinuing Conexxence if severe symptoms develop. ( 5.8 ) Severe bone, joint, muscle pain may occur. Discontinue use if severe symptoms develop. ( 5.9 ) Suppression of bone turnover: Significant suppression has been demonstrated. Monitor for consequences of bone over-suppression. ( 5.10 )
Indications and Usage
Conexxence is a RANK ligand (RANKL) inhibitor indicated for treatment: of postmenopausal women with osteoporosis at high risk for fracture ( 1.1 ) to increase bone mass in men with osteoporosis at high risk for fracture ( 1.2 ) of glucocorticoid-induced osteoporosis in men and women at high risk for fracture ( 1.3 ) to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy for nonmetastatic prostate cancer ( 1.4 ) to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer ( 1.5 )
Dosage and Administration
Pregnancy must be ruled out prior to administration of Conexxence. ( 2.1 ) Before initiating Conexxence in patients with advanced chronic kidney disease, including dialysis patients, evaluate for the presence of chronic kidney disease mineral and bone disorder with intact parathyroid hormone, serum calcium, 25(OH) vitamin D, and 1,25(OH)2 vitamin D. ( 2.2 , 5.1 , 8.6 ) Conexxence should be administered by a healthcare provider. ( 2.3 ) Administer 60 mg every 6 months as a subcutaneous injection in the upper arm, upper thigh, or abdomen. ( 2.3 ) Instruct patients to take calcium 1000 mg daily and at least 400 IU vitamin D daily. ( 2.3 )
Contraindications
Conexxence is contraindicated in: Patients with hypocalcemia: Pre-existing hypocalcemia must be corrected prior to initiating therapy with Conexxence [see Warnings and Precautions ( 5.1 )] . Pregnant women: Denosumab products may cause fetal harm when administered to a pregnant woman. In women of reproductive potential, pregnancy testing should be performed prior to initiating treatment with Conexxence [see Use in Specific Populations ( 8.1 )] . Patients with hypersensitivity to denosumab products: Conexxence is contraindicated in patients with a history of systemic hypersensitivity to any component of the product. Reactions have included anaphylaxis, facial swelling, and urticaria [see Warnings and Precautions ( 5.3 ), Adverse Reactions ( 6.2 )] .
Adverse Reactions
The following serious adverse reactions are discussed below and also elsewhere in the labeling: Severe Hypocalcemia and Mineral Metabolism Changes [see Warnings and Precautions ( 5.1 )] Hypersensitivity [see Warnings and Precautions ( 5.3 )] Osteonecrosis of the Jaw [see Warnings and Precautions ( 5.4 )] Atypical Subtrochanteric and Diaphyseal Femoral Fractures [see Warnings and Precautions ( 5.5 )] Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation of Treatment [see Warnings and Precautions ( 5.6 )] Serious Infections [see Warnings and Precautions ( 5.7 )] Dermatologic Adverse Reactions [see Warnings and Precautions ( 5.8 )] The most common adverse reactions reported with denosumab products in patients with postmenopausal osteoporosis are back pain, pain in extremity, musculoskeletal pain, hypercholesterolemia, and cystitis. The most common adverse reactions reported with denosumab products in men with osteoporosis are back pain, arthralgia, and nasopharyngitis. The most common adverse reactions reported with denosumab products in patients with glucocorticoid-induced osteoporosis are back pain, hypertension, bronchitis, and headache. The most common (per patient incidence ≥ 10%) adverse reactions reported with denosumab products in patients with bone loss receiving androgen deprivation therapy for prostate cancer or adjuvant aromatase inhibitor therapy for breast cancer are arthralgia and back pain. Pain in extremity and musculoskeletal pain have also been reported in clinical trials. The most common adverse reactions leading to discontinuation of denosumab products in patients with postmenopausal osteoporosis are back pain and constipation.
Description
Patients with advanced chronic kidney disease (eGFR < 30 mL/min/1.73 m 2 ), including dialysis-dependent patients, are at greater risk of severe hypocalcemia following denosumab products administration. Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported [see Warnings and Precautions ( 5.1 )] . The presence of chronic kidney disease-mineral bone disorder (CKD-MBD) markedly increases the risk of hypocalcemia in these patients [see Warnings and Precautions ( 5.1 )] . Prior to initiating Conexxence in patients with advanced chronic kidney disease, evaluate for the presence of CKD-MBD. Treatment with Conexxence in these patients should be supervised by a healthcare provider with expertise in the diagnosis and management of CKD-MBD [see Dosage and Administration ( 2.2 ) and Warnings and Precautions ( 5.1 )].
Section 42229-5
Patients with Advanced Chronic Kidney Disease
Patients with advanced chronic kidney disease [i.e., eGFR < 30 mL/min/1.73 m2] including dialysis-dependent patients are at greater risk for severe hypocalcemia following denosumab products administration. Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported. The presence of underlying chronic kidney disease-mineral bone disorder (CKD-MBD, renal osteodystrophy) markedly increases the risk of hypocalcemia. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk.
To minimize the risk of hypocalcemia in patients with advanced chronic kidney disease, evaluate for the presence of chronic kidney disease mineral and bone disorder with intact parathyroid hormone (iPTH), serum calcium, 25(OH) vitamin D, and 1,25(OH)2 vitamin D prior to decisions regarding Conexxence treatment. Consider also assessing bone turnover status (serum markers of bone turnover or bone biopsy) to evaluate the underlying bone disease that may be present. Monitor serum calcium weekly for the first month after Conexxence administration and monthly thereafter. Instruct all patients with advanced chronic kidney disease, including those who are dialysis-dependent, about the symptoms of hypocalcemia and the importance of maintaining serum calcium levels with adequate calcium and activated vitamin D supplementation. Treatment with Conexxence in these patients should be supervised by a healthcare provider who is experienced in diagnosis and management of CKD-MBD.
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Issued: 03/2025 |
|||
|
MEDICATION GUIDE Conexxence (kon ex' ens) (denosumab-bnht) Injection, for subcutaneous use |
||||
|
What is the most important information I should know about Conexxence?
|
||||
| Call your doctor right away if you have any of these side effects. | ||||
|
What is Conexxence?
Conexxence is a prescription medicine used to:
|
||||
| It is not known if Conexxence is safe and effective in children. Conexxence is not approved for use in children. | ||||
Do not take Conexxence if you:
|
||||
Before taking Conexxence, tell your doctor about all of your medical conditions, including if you:
|
||||
|
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of medicines with you to show to your doctor or pharmacist when you get a new medicine. |
||||
How will I receive Conexxence?
|
||||
|
What are the possible side effects of Conexxence?
Conexxence may cause serious side effects.
|
||||
| The most common side effects of Conexxence in women who are being treated for osteoporosis after menopause are: | ||||
|
|
|||
| The most common side effects of Conexxence in men with osteoporosis are: | ||||
|
|
|||
| The most common side effects of Conexxence in patients with glucocorticoid-induced osteoporosis are: | ||||
|
|
|||
| The most common side effects of Conexxence in patients receiving certain treatments for prostate or breast cancer are: | ||||
|
|
|||
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Conexxence. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store Conexxence if I need to pick it up from a pharmacy?
|
||||
| Keep Conexxence and all medicines out of the reach of children. | ||||
|
General information about the safe and effective use of Conexxence.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Conexxence for a condition for which it was not prescribed. Do not give Conexxence to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about Conexxence that is written for healthcare providers. |
||||
|
What are the ingredients in Conexxence?
|
Section 44425-7
Storage and Handling
Store Conexxence refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. Prior to administration, Conexxence may be allowed to reach room temperature up to 25°C (77°F) in the original container. Once removed from the refrigerator, Conexxence must not be exposed to temperatures above 25°C (77°F) and must be used within 14 days. Discard Conexxence if not used within 14 days. Do not use Conexxence after the expiry date printed on the label.
Protect Conexxence from direct light and heat.
Avoid vigorous shaking of Conexxence.
Section 51945-4
Principal Display Panel – 60 mg/mL Carton Label
NDC 65219-668-01 Rx only
Scan for additional information
Conexxence
denosumab-bnht
Injection
60 mg/mL
For Subcutaneous use only
Single-dose prefilled syringe.
discard unused portion.
Carton contains:
1 Single-dose prefilled syringe
1 Prescribing Information
1 Medication Guide
Dispense the enclosed Medication
Guide to each patient
Sterile Solution - No Preservative
Conexxence should be administered by a healthcare provider
Fresenius
Kabi
11 Description
Denosumab-bnht is a human IgG2 monoclonal antibody with affinity and specificity for human RANKL (receptor activator of nuclear factor kappa-B ligand). Denosumab-bnht has an approximate molecular weight of 147 kDa and is produced in genetically engineered mammalian (Chinese hamster ovary) cells.
Conexxence (denosumab-bnht) injection is a sterile, preservative-free, clear, colorless to pale yellow solution for subcutaneous use.
Each 1 mL single-dose prefilled syringe of Conexxence contains 60 mg denosumab-bnht (60 mg/mL solution), 0.23 mg glacial acetic acid, 0.1 mg polysorbate 20, 1.07 mg sodium acetate, 47 mg sorbitol, and Water for Injection (USP). The pH is 5.2.
8.4 Pediatric Use
The safety and effectiveness of Conexxence have not been established in pediatric patients.
In one multicenter, open-label study with denosumab conducted in 153 pediatric patients with osteogenesis imperfecta, aged 2 to 17 years, evaluating fracture risk reduction, efficacy was not demonstrated.
Hypercalcemia has been reported in pediatric patients with osteogenesis imperfecta treated with denosumab products. Some cases required hospitalization and were complicated by acute renal injury [see Warnings and Precautions (5.11)]. Clinical studies in pediatric patients with osteogenesis imperfecta were terminated early due to the occurrence of life-threatening events and hospitalizations due to hypercalcemia.
Based on results from animal studies, denosumab may negatively affect long-bone growth and dentition in pediatric patients below the age of 4 years.
8.5 Geriatric Use
Of the total number of patients in clinical studies of denosumab, 9943 patients (76%) were ≥ 65 years old, while 3576 (27%) were ≥ 75 years old. Of the patients in the osteoporosis study in men, 133 patients (55%) were ≥ 65 years old, while 39 patients (16%) were ≥ 75 years old. Of the patients in the glucocorticoid-induced osteoporosis study, 355 patients (47%) were ≥ 65 years old, while 132 patients (17%) were ≥ 75 years old. No overall differences in safety or efficacy were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of denosumab or of other denosumab products.
Using an electrochemiluminescent bridging immunoassay, less than 1% (55 out of 8113) of patients treated with denosumab for up to 5 years tested positive for binding antibodies (including pre-existing, transient, and developing antibodies). None of the patients tested positive for neutralizing antibodies, as was assessed using a chemiluminescent cell-based in vitro biological assay.
There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of denosumab.
4 Contraindications
Conexxence is contraindicated in:
- Patients with hypocalcemia: Pre-existing hypocalcemia must be corrected prior to initiating therapy with Conexxence [see Warnings and Precautions (5.1)].
- Pregnant women: Denosumab products may cause fetal harm when administered to a pregnant woman. In women of reproductive potential, pregnancy testing should be performed prior to initiating treatment with Conexxence [see Use in Specific Populations (8.1)].
- Patients with hypersensitivity to denosumab products: Conexxence is contraindicated in patients with a history of systemic hypersensitivity to any component of the product. Reactions have included anaphylaxis, facial swelling, and urticaria [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
6 Adverse Reactions
The following serious adverse reactions are discussed below and also elsewhere in the labeling:
- Severe Hypocalcemia and Mineral Metabolism Changes [see Warnings and Precautions (5.1)]
- Hypersensitivity [see Warnings and Precautions (5.3)]
- Osteonecrosis of the Jaw [see Warnings and Precautions (5.4)]
- Atypical Subtrochanteric and Diaphyseal Femoral Fractures [see Warnings and Precautions (5.5)]
- Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation of Treatment [see Warnings and Precautions (5.6)]
- Serious Infections [see Warnings and Precautions (5.7)]
- Dermatologic Adverse Reactions [see Warnings and Precautions (5.8)]
The most common adverse reactions reported with denosumab products in patients with postmenopausal osteoporosis are back pain, pain in extremity, musculoskeletal pain, hypercholesterolemia, and cystitis.
The most common adverse reactions reported with denosumab products in men with osteoporosis are back pain, arthralgia, and nasopharyngitis.
The most common adverse reactions reported with denosumab products in patients with glucocorticoid-induced osteoporosis are back pain, hypertension, bronchitis, and headache.
The most common (per patient incidence ≥ 10%) adverse reactions reported with denosumab products in patients with bone loss receiving androgen deprivation therapy for prostate cancer or adjuvant aromatase inhibitor therapy for breast cancer are arthralgia and back pain. Pain in extremity and musculoskeletal pain have also been reported in clinical trials.
The most common adverse reactions leading to discontinuation of denosumab products in patients with postmenopausal osteoporosis are back pain and constipation.
5.3 Hypersensitivity
Clinically significant hypersensitivity including anaphylaxis has been reported with denosumab products. Symptoms have included hypotension, dyspnea, throat tightness, facial and upper airway edema, pruritus, and urticaria. If an anaphylactic or other clinically significant allergic reaction occurs, initiate appropriate therapy and discontinue further use of Conexxence [see Contraindications (4), Adverse Reactions (6.2)].
8.6 Renal Impairment
No dose adjustment is necessary in patients with renal impairment.
Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported postmarketing. In clinical studies, patients with advanced chronic kidney disease (i.e., eGFR < 30 mL/min/1.73 m2), including dialysis-dependent patients, were at greater risk of developing hypocalcemia. The presence of underlying chronic kidney disease-mineral bone disorder (CKD-MBD, renal osteodystrophy) markedly increases the risk of hypocalcemia. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk. Consider the benefits and risks to the patient when administering Conexxence to patients with advanced chronic kidney disease. Monitor calcium and mineral levels (phosphorus and magnesium). Adequate intake of calcium and vitamin D is important in patients with advanced chronic kidney disease including dialysis-dependent patients [see Dosage and Administration (2.2), Warnings and Precautions (5.1), Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In clinical studies, treatment with 60 mg of denosumab resulted in reduction in the bone resorption marker serum type 1 C-telopeptide (CTX) by approximately 85% by 3 days, with maximal reductions occurring by 1 month. CTX levels were below the limit of assay quantitation (0.049 ng/mL) in 39% to 68% of patients 1 to 3 months after dosing of denosumab. At the end of each dosing interval, CTX reductions were partially attenuated from a maximal reduction of ≥ 87% to ≥ 45% (range: 45% to 80%), as serum denosumab levels diminished, reflecting the reversibility of the effects of denosumab on bone remodeling. These effects were sustained with continued treatment. Upon reinitiation, the degree of inhibition of CTX by denosumab was similar to that observed in patients initiating denosumab treatment.
Consistent with the physiological coupling of bone formation and resorption in skeletal remodeling, subsequent reductions in bone formation markers (i.e., osteocalcin and procollagen type 1 N-terminal peptide [P1NP]) were observed starting 1 month after the first dose of denosumab. After discontinuation of denosumab therapy, markers of bone resorption increased to levels 40% to 60% above pretreatment values but returned to baseline levels within 12 months.
12.3 Pharmacokinetics
In a study conducted in healthy male and female volunteers (n = 73, age range: 18 to 64 years) following a single subcutaneously administered denosumab dose of 60 mg, the mean area-under-the-concentration-time curve up to 16 weeks (AUC0-16 weeks) of denosumab was 316 mcg⋅day/mL (standard deviation [SD] = 101 mcg⋅day/mL). The mean maximum denosumab concentration (Cmax) was 6.75 mcg/mL (SD = 1.89 mcg/mL). No accumulation or change in denosumab pharmacokinetics with time is observed with multiple dosing of 60 mg subcutaneously administered once every 6 months.
2.3 Recommended Dosage
Conexxence should be administered by a healthcare provider.
The recommended dose of Conexxence is 60 mg administered as a single subcutaneous injection once every 6 months. Administer Conexxence via subcutaneous injection in the upper arm, the upper thigh, or the abdomen. All patients should receive calcium 1000 mg daily and at least 400 IU vitamin D daily [see Warnings and Precautions (5.1)].
If a dose of Conexxence is missed, administer the injection as soon as the patient is available. Thereafter, schedule injections every 6 months from the date of the last injection.
5.7 Serious Infections
In a clinical trial of over 7800 women with postmenopausal osteoporosis, serious infections leading to hospitalization were reported more frequently in the denosumab group than in the placebo group [see Adverse Reactions (6.1)]. Serious skin infections, as well as infections of the abdomen, urinary tract, and ear, were more frequent in patients treated with denosumab. Endocarditis was also reported more frequently in denosumab-treated patients. The incidence of opportunistic infections was similar between placebo and denosumab groups, and the overall incidence of infections was similar between the treatment groups. Advise patients to seek prompt medical attention if they develop signs or symptoms of severe infection, including cellulitis.
Patients on concomitant immunosuppressant agents or with impaired immune systems may be at increased risk for serious infections. Consider the benefit-risk profile in such patients before treating with Conexxence. In patients who develop serious infections while on Conexxence, prescribers should assess the need for continued Conexxence therapy.
1 Indications and Usage
Conexxence is a RANK ligand (RANKL) inhibitor indicated for treatment:
- of postmenopausal women with osteoporosis at high risk for fracture (1.1)
- to increase bone mass in men with osteoporosis at high risk for fracture (1.2)
- of glucocorticoid-induced osteoporosis in men and women at high risk for fracture (1.3)
- to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy for nonmetastatic prostate cancer (1.4)
- to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer (1.5)
12.1 Mechanism of Action
Denosumab products bind to RANKL, a transmembrane or soluble protein essential for the formation, function, and survival of osteoclasts, the cells responsible for bone resorption. Denosumab products prevent RANKL from activating its receptor, RANK, on the surface of osteoclasts and their precursors. Prevention of the RANKL/RANK interaction inhibits osteoclast formation, function, and survival, thereby decreasing bone resorption and increasing bone mass and strength in both cortical and trabecular bone.
5.9 Musculoskeletal Pain
In postmarketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain has been reported in patients taking denosumab products [see Adverse Reactions (6.2)]. The time to onset of symptoms varied from one day to several months after starting denosumab products. Consider discontinuing use if severe symptoms develop [see Patient Counseling Information (17)].
5 Warnings and Precautions
- Hypocalcemia: Pre-existing hypocalcemia must be corrected before initiating Conexxence. May worsen, especially in patients with renal impairment. Adequately supplement all patients with calcium and vitamin D. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk. Evaluate for presence of chronic kidney disease mineral-bone disorder. Monitor serum calcium. (5.1)
- Same Active Ingredient: Patients receiving Conexxence should not receive other denosumab products concomitantly (5.2)
- Hypersensitivity including anaphylactic reactions may occur. Discontinue permanently if a clinically significant reaction occurs. (5.3)
- Osteonecrosis of the jaw: Has been reported with denosumab products. Monitor for symptoms. (5.4)
- Atypical femoral fractures: Have been reported. Evaluate patients with thigh or groin pain to rule out a femoral fracture. (5.5)
- Multiple vertebral fractures have been reported following treatment discontinuation. Patients should be transitioned to another antiresorptive agent if Conexxence is discontinued. (5.6)
- Serious infections including skin infections: May occur, including those leading to hospitalization. Advise patients to seek prompt medical attention if they develop signs or symptoms of infection, including cellulitis. (5.7)
- Dermatologic reactions: Dermatitis, rashes, and eczema have been reported. Consider discontinuing Conexxence if severe symptoms develop. (5.8)
- Severe bone, joint, muscle pain may occur. Discontinue use if severe symptoms develop. (5.9)
- Suppression of bone turnover: Significant suppression has been demonstrated. Monitor for consequences of bone over-suppression. (5.10)
2 Dosage and Administration
- Pregnancy must be ruled out prior to administration of Conexxence. (2.1)
- Before initiating Conexxence in patients with advanced chronic kidney disease, including dialysis patients, evaluate for the presence of chronic kidney disease mineral and bone disorder with intact parathyroid hormone, serum calcium, 25(OH) vitamin D, and 1,25(OH)2 vitamin D. (2.2, 5.1, 8.6)
- Conexxence should be administered by a healthcare provider. (2.3)
- Administer 60 mg every 6 months as a subcutaneous injection in the upper arm, upper thigh, or abdomen. (2.3)
- Instruct patients to take calcium 1000 mg daily and at least 400 IU vitamin D daily. (2.3)
3 Dosage Forms and Strengths
- Injection: 60 mg/mL clear, colorless to pale yellow solution in a single-dose prefilled syringe.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ), which can occur spontaneously, is generally associated with tooth extraction and/or local infection with delayed healing. ONJ has been reported in patients receiving denosumab products [see Adverse Reactions (6.1)]. A routine oral exam should be performed by the prescriber prior to initiation of Conexxence treatment. A dental examination with appropriate preventive dentistry is recommended prior to treatment with Conexxence in patients with risk factors for ONJ such as invasive dental procedures (e.g. tooth extraction, dental implants, oral surgery), diagnosis of cancer, concomitant therapies (e.g. chemotherapy, corticosteroids, angiogenesis inhibitors), poor oral hygiene, and comorbid disorders (e.g. periodontal and/or other pre-existing dental disease, anemia, coagulopathy, infection, ill-fitting dentures). Good oral hygiene practices should be maintained during treatment with Conexxence. Concomitant administration of drugs associated with ONJ may increase the risk of developing ONJ. The risk of ONJ may increase with duration of exposure to denosumab products.
For patients requiring invasive dental procedures, clinical judgment of the treating physician and/or oral surgeon should guide the management plan of each patient based on individual benefit-risk assessment.
Patients who are suspected of having or who develop ONJ while on Conexxence should receive care by a dentist or an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Discontinuation of Conexxence therapy should be considered based on individual benefit-risk assessment.
6.2 Postmarketing Experience
Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of denosumab products:
- Drug-related hypersensitivity reactions: anaphylaxis, rash, urticaria, facial swelling, and erythema
- Hypocalcemia: severe symptomatic hypocalcemia resulting in hospitalization, life-threatening events, and fatal cases
- Musculoskeletal pain, including severe cases
- Parathyroid hormone (PTH): Marked elevation in serum PTH in patients with severe renal impairment (creatinine clearance < 30 mL/min) or receiving dialysis
- Multiple vertebral fractures following treatment discontinuation
- Cutaneous and mucosal lichenoid drug eruptions (e.g. lichen planus-like reactions)
- Alopecia
- Vasculitis (e.g. ANCA positive vasculitis, leukocytoclastic vasculitis)
- Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome
8 Use in Specific Populations
- Pregnant women and females of reproductive potential: Denosumab products may cause fetal harm when administered to pregnant women. Advise females of reproductive potential to use effective contraception during therapy, and for at least 5 months after the last dose of Conexxence. (8.1, 8.3)
- Pediatric patients: Conexxence is not approved for use in pediatric patients. (8.4)
- Renal impairment: No dose adjustment is necessary in patients with renal impairment. Patients with advanced chronic kidney disease (eGFR < 30 mL/min/1.73 m2), including dialysis-dependent patients, are at greater risk of severe hypocalcemia. The presence of underlying chronic kidney disease-mineral bone disorder markedly increases the risk of hypocalcemia. (5.1, 8.6)
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.10 Suppression of Bone Turnover
In clinical trials in women with postmenopausal osteoporosis, treatment with denosumab resulted in significant suppression of bone remodeling as evidenced by markers of bone turnover and bone histomorphometry [see Clinical Pharmacology (12.2), Clinical Studies (14.1)]. The significance of these findings and the effect of long-term treatment with denosumab products are unknown. The long-term consequences of the degree of suppression of bone remodeling observed with denosumab may contribute to adverse outcomes such as osteonecrosis of the jaw, atypical fractures, and delayed fracture healing. Monitor patients for these consequences.
2.4 Preparation and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Conexxence is a clear, colorless to pale yellow solution that is free from visible particles. Do not use if the solution is discolored or cloudy or if the solution contains particles or foreign particulate matter.
Prior to administration, Conexxence may be removed from the refrigerator and brought to room temperature up to 25°C (77°F) by standing in the original container. This generally takes 15 to 30 minutes. Do not warm Conexxence in any other way [see How Supplied/Storage and Handling (16)]. Do not shake the prefilled syringe.
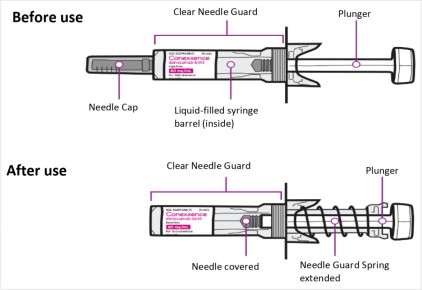
Instructions for Administration of Conexxence Prefilled Syringe with Needle Safety Guard
IMPORTANT:
In order to minimize accidental needlesticks, the Conexxence single-dose prefilled syringe has an automatic clear needle safety guard.
Conexxence prefilled syringe (Before & After Use). See figure below.
|
Step 1: Remove Needle Cap (see
Figure A
) Do not hold the prefilled syringe by the plunger rod. Do not twist or bend the needle cap. |
Figure A |
|
Step 2: Administer Subcutaneous Injection
Choose an injection site. The recommended injection sites for Conexxence include (see Figure B ): |
|
|
Figure B |
| Do not administer into muscle or blood vessel. Pinch the skin and insert the needle at a 45 to 90-degree angle. Push the plunger with a slow and constant pressure until you cannot press further and have injected all the liquid subcutaneously (see Figure C ). You may hear or feel a “click”. |
Figure C |
|
Step 3. Release Plunger
Keeping the prefilled syringe at the injection site, slowly release the plunger and lift syringe off the skin. The clear needle guard will automatically cover the needle (see Figure D ). |
|
|
Do not recap the needle. Immediately dispose of the used prefilled syringe into a sharps disposal container. |
Figure D |
5.8 Dermatologic Adverse Reactions
In a large clinical trial of over 7800 women with postmenopausal osteoporosis, epidermal and dermal adverse events such as dermatitis, eczema, and rashes occurred at a significantly higher rate in the denosumab group compared to the placebo group. Most of these events were not specific to the injection site [see Adverse Reactions (6.1)]. Consider discontinuing Conexxence if severe symptoms develop.
13.2 Animal Toxicology And/or Pharmacology
Denosumab products are inhibitors of osteoclastic bone resorption via inhibition of RANKL.
In ovariectomized monkeys, once-monthly treatment with denosumab suppressed bone turnover and increased BMD and strength of cancellous and cortical bone at doses 50-fold higher than the recommended human dose of 60 mg administered once every 6 months, based on body weight (mg/kg). Bone tissue was normal with no evidence of mineralization defects, accumulation of osteoid, or woven bone.
Because the biological activity of denosumab in animals is specific to nonhuman primates, evaluation of genetically engineered (“knockout”) mice or use of other biological inhibitors of the RANK/RANKL pathway, namely OPG-Fc, provided additional information on the pharmacodynamic properties of denosumab products. RANK/RANKL knockout mice exhibited absence of lymph node formation, as well as an absence of lactation due to inhibition of mammary gland maturation (lobulo-alveolar gland development during pregnancy). Neonatal RANK/RANKL knockout mice exhibited reduced bone growth and lack of tooth eruption. A corroborative study in 2-week-old rats given the RANKL inhibitor OPG-Fc also showed reduced bone growth, altered growth plates, and impaired tooth eruption. These changes were partially reversible in this model when dosing with the RANKL inhibitors was discontinued.
5.2 Drug Products With Same Active Ingredient
Patients receiving Conexxence should not receive other denosumab products concomitantly.
8.3 Females and Males of Reproductive Potential
Based on findings in animals, denosumab products can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
1.3 Treatment of Glucocorticoid Induced Osteoporosis
Conexxence is indicated for the treatment of glucocorticoid-induced osteoporosis in men and women at high risk of fracture who are either initiating or continuing systemic glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and expected to remain on glucocorticoids for at least 6 months. High risk of fracture is defined as a history of osteoporotic fracture, multiple risk factors for fracture, or patients who have failed or are intolerant to other available osteoporosis therapy [see Clinical Studies (14.3)].
14.3 Treatment of Glucocorticoid Induced Osteoporosis
The efficacy and safety of denosumab in the treatment of patients with glucocorticoid-induced osteoporosis was assessed in the 12-month primary analysis of a 2-year, randomized, multicenter, double-blind, parallel-group, active-controlled study (NCT 01575873) of 795 patients (70% women and 30% men) aged 20 to 94 years (mean age of 63 years) treated with greater than or equal to 7.5 mg/day oral prednisone (or equivalent) for < 3 months prior to study enrollment and planning to continue treatment for a total of at least 6 months (glucocorticoid-initiating subpopulation; n = 290) or ≥ 3 months prior to study enrollment and planning to continue treatment for a total of at least 6 months (glucocorticoid-continuing subpopulation, n = 505). Enrolled patients < 50 years of age were required to have a history of osteoporotic fracture. Enrolled patients ≥ 50 years of age who were in the glucocorticoid-continuing subpopulation were required to have a baseline BMD T-score of ≤ -2.0 at the lumbar spine, total hip, or femoral neck; or a BMD T-score ≤ -1.0 at the lumbar spine, total hip, or femoral neck and a history of osteoporotic fracture.
Patients were randomized (1:1) to receive either an oral daily bisphosphonate (active-control, risedronate 5 mg once daily) (n = 397) or denosumab 60 mg subcutaneously once every 6 months (n = 398) for one year. Randomization was stratified by gender within each subpopulation. Patients received at least 1000 mg calcium and 800 IU vitamin D supplementation daily.
5.1 Severe Hypocalcemia and Mineral Metabolism Changes
Denosumab products can cause severe hypocalcemia and fatal cases have been reported. Pre-existing hypocalcemia must be corrected prior to initiating therapy with Conexxence. Adequately supplement all patients with calcium and vitamin D [see Dosage and Administration (2.1), Contraindications (4), and Adverse Reactions (6.1)].
In patients without advanced chronic kidney disease who are predisposed to hypocalcemia and disturbances of mineral metabolism (e.g. history of hypoparathyroidism, thyroid surgery, parathyroid surgery, malabsorption syndromes, excision of small intestine, treatment with other calcium-lowering drugs), assess serum calcium and mineral levels (phosphorus and magnesium) 10 to14 days after Conexxence injection. In some postmarketing cases, hypocalcemia persisted for weeks or months and required frequent monitoring and intravenous and/or oral calcium replacement, with or without vitamin D.
14.4 Treatment of Bone Loss in Men With Prostate Cancer
The efficacy and safety of denosumab in the treatment of bone loss in men with nonmetastatic prostate cancer receiving androgen deprivation therapy (ADT) were demonstrated in a 3-year, randomized (1:1), double-blind, placebo-controlled, multinational study. Men less than 70 years of age had either a BMD T-score at the lumbar spine, total hip, or femoral neck between -1.0 and -4.0, or a history of an osteoporotic fracture. The mean baseline lumbar spine BMD T-score was -0.4, and 22% of men had a vertebral fracture at baseline. The 1468 men enrolled ranged in age from 48 to 97 years (median 76 years). Men were randomized to receive subcutaneous injections of either placebo (n = 734) or denosumab 60 mg (n = 734) once every 6 months for a total of 6 doses. Randomization was stratified by age (< 70 years vs. ≥ 70 years) and duration of ADT at trial entry (≤ 6 months vs. > 6 months). Seventy-nine percent of patients received ADT for more than 6 months at study entry. All men received at least 1000 mg calcium and 400 IU vitamin D supplementation daily.
14.5 Treatment of Bone Loss in Women With Breast Cancer
The efficacy and safety of denosumab in the treatment of bone loss in women receiving adjuvant aromatase inhibitor (AI) therapy for breast cancer was assessed in a 2-year, randomized (1:1), double-blind, placebo-controlled, multinational study. Women had baseline BMD T-scores between -1.0 to -2.5 at the lumbar spine, total hip, or femoral neck, and had not experienced fracture after age 25. The mean baseline lumbar spine BMD T-score was -1.1, and 2.0% of women had a vertebral fracture at baseline. The 252 women enrolled ranged in age from 35 to 84 years (median 59 years). Women were randomized to receive subcutaneous injections of either placebo (n = 125) or denosumab 60 mg (n = 127) once every 6 months for a total of 4 doses. Randomization was stratified by duration of adjuvant AI therapy at trial entry (≤ 6 months vs. > 6 months). Sixty-two percent of patients received adjuvant AI therapy for more than 6 months at study entry. All women received at least 1000 mg calcium and 400 IU vitamin D supplementation daily.
2.1 Pregnancy Testing Prior to Initiation of Conexxence
Pregnancy must be ruled out prior to administration of Conexxence. Perform pregnancy testing in all females of reproductive potential prior to administration of Conexxence. Based on findings in animals, denosumab products can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1, 8.3)].
14.1 Treatment of Postmenopausal Women With Osteoporosis
The efficacy and safety of denosumab in the treatment of postmenopausal osteoporosis was demonstrated in a 3-year, randomized, double-blind, placebo-controlled trial. Enrolled women had a baseline BMD T-score between -2.5 and -4.0 at either the lumbar spine or total hip. Women with other diseases (such as rheumatoid arthritis, osteogenesis imperfecta, and Paget's disease) or on therapies that affect bone were excluded from this study. The 7808 enrolled women were aged 60 to 91 years with a mean age of 72 years. Overall, the mean baseline lumbar spine BMD T-score was -2.8, and 23% of women had a vertebral fracture at baseline. Women were randomized to receive subcutaneous injections of either placebo (N = 3906) or denosumab 60 mg (N = 3902) once every 6 months. All women received at least 1000 mg calcium and 400 IU vitamin D supplementation daily.
The primary efficacy variable was the incidence of new morphometric (radiologically-diagnosed) vertebral fractures at 3 years. Vertebral fractures were diagnosed based on lateral spine radiographs (T4-L4) using a semiquantitative scoring method. Secondary efficacy variables included the incidence of hip fracture and nonvertebral fracture, assessed at 3 years.
1.2 Treatment to Increase Bone Mass in Men With Osteoporosis
Conexxence is indicated for treatment to increase bone mass in men with osteoporosis at high risk for fracture, defined as a history of osteoporotic fracture, or multiple risk factors for fracture; or patients who have failed or are intolerant to other available osteoporosis therapy [see Clinical Studies (14.2)].
14.2 Treatment to Increase Bone Mass in Men With Osteoporosis
The efficacy and safety of denosumab in the treatment to increase bone mass in men with osteoporosis was demonstrated in a 1-year, randomized, double-blind, placebo-controlled trial. Enrolled men had a baseline BMD T-score between -2.0 and -3.5 at the lumbar spine or femoral neck. Men with a BMD T-score between -1.0 and -3.5 at the lumbar spine or femoral neck were also enrolled if there was a history of prior fragility fracture. Men with other diseases (such as rheumatoid arthritis, osteogenesis imperfecta, and Paget's disease) or on therapies that may affect bone were excluded from this study. The 242 men enrolled in the study ranged in age from 31 to 84 years with a mean age of 65 years. Men were randomized to receive SC injections of either placebo (n = 121) or denosumab 60 mg (n = 121) once every 6 months. All men received at least 1000 mg calcium and at least 800 IU vitamin D supplementation daily.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical low energy or low trauma fractures of the shaft have been reported in patients receiving denosumab products [see Adverse Reactions (6.1)]. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with antiresorptive agents.
Atypical femoral fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral, and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
During Conexxence treatment, patients should be advised to report new or unusual thigh, hip, or groin pain. Any patient who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of Conexxence therapy should be considered, pending a benefit-risk assessment, on an individual basis.
5.11 Hypercalcemia in Pediatric Patients With Osteogenesis Imperfecta
Conexxence is not approved for use in pediatric patients. Hypercalcemia has been reported in pediatric patients with osteogenesis imperfecta treated with denosumab products. Some cases required hospitalization [see Use in Specific Populations (8.4)].
Warning: Severe Hypocalcemia in Patients With Advanced Kidney Disease
- Patients with advanced chronic kidney disease (eGFR < 30 mL/min/1.73 m 2 ), including dialysis-dependent patients, are at greater risk of severe hypocalcemia following denosumab products administration. Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported [see Warnings and Precautions (5.1)] .
- The presence of chronic kidney disease-mineral bone disorder (CKD-MBD) markedly increases the risk of hypocalcemia in these patients [see Warnings and Precautions (5.1)] .
- Prior to initiating Conexxence in patients with advanced chronic kidney disease, evaluate for the presence of CKD-MBD. Treatment with Conexxence in these patients should be supervised by a healthcare provider with expertise in the diagnosis and management of CKD-MBD [see Dosage and Administration (2.2) and Warnings and Precautions (5.1)].
5.6 Multiple Vertebral Fractures (mvf) Following Discontinuation of Treatment
Following discontinuation of denosumab treatment, fracture risk increases, including the risk of multiple vertebral fractures. Treatment with denosumab results in significant suppression of bone turnover and cessation of denosumab treatment results in increased bone turnover above pretreatment values 9 months after the last dose of denosumab. Bone turnover then returns to pretreatment values 24 months after the last dose of denosumab. In addition, bone mineral density (BMD) returns to pretreatment values within 18 months after the last injection [see Clinical Pharmacology (12.2), Clinical Studies (14.1)].
New vertebral fractures occurred as early as 7 months (on average 19 months) after the last dose of denosumab. Prior vertebral fracture was a predictor of multiple vertebral fractures after denosumab discontinuation. Evaluate an individual's benefit-risk before initiating treatment with Conexxence.
If Conexxence treatment is discontinued, patients should be transitioned to an alternative antiresorptive therapy [see Adverse Reactions (6.1)].
1.1 Treatment of Postmenopausal Women With Osteoporosis At High Risk for Fracture
Conexxence is indicated for the treatment of postmenopausal women with osteoporosis at high risk for fracture, defined as a history of osteoporotic fracture, or multiple risk factors for fracture; or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, denosumab reduces the incidence of vertebral, nonvertebral, and hip fractures [see Clinical Studies (14.1)].
1.4 Treatment of Bone Loss in Men Receiving Androgen Deprivation Therapy for Prostate Cancer
Conexxence is indicated as a treatment to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy (ADT) for nonmetastatic prostate cancer. In these patients denosumab also reduced the incidence of vertebral fractures [see Clinical Studies (14.4)].
1.5 Treatment of Bone Loss in Women Receiving Adjuvant Aromatase Inhibitor Therapy for Breast Cancer
Conexxence is indicated as a treatment to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer [see Clinical Studies (14.5)].
2.2 Laboratory Testing in Patients With Advanced Chronic Kidney Disease Prior to Initiation of Conexxence
In patients with advanced chronic kidney disease [i.e., estimated glomerular filtration rate (eGFR) < 30 mL/min/1.73 m2], including dialysis-dependent patients, evaluate for the presence of chronic kidney disease mineral and bone disorder (CKD-MBD) with intact parathyroid hormone (iPTH), serum calcium, 25(OH) vitamin D, and 1,25 (OH)2 vitamin D prior to decisions regarding Conexxence treatment. Consider also assessing bone turnover status (serum markers of bone turnover or bone biopsy) to evaluate the underlying bone disease that may be present [see Warnings and Precautions (5.1)].
Structured Label Content
Section 42229-5 (42229-5)
Patients with Advanced Chronic Kidney Disease
Patients with advanced chronic kidney disease [i.e., eGFR < 30 mL/min/1.73 m2] including dialysis-dependent patients are at greater risk for severe hypocalcemia following denosumab products administration. Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported. The presence of underlying chronic kidney disease-mineral bone disorder (CKD-MBD, renal osteodystrophy) markedly increases the risk of hypocalcemia. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk.
To minimize the risk of hypocalcemia in patients with advanced chronic kidney disease, evaluate for the presence of chronic kidney disease mineral and bone disorder with intact parathyroid hormone (iPTH), serum calcium, 25(OH) vitamin D, and 1,25(OH)2 vitamin D prior to decisions regarding Conexxence treatment. Consider also assessing bone turnover status (serum markers of bone turnover or bone biopsy) to evaluate the underlying bone disease that may be present. Monitor serum calcium weekly for the first month after Conexxence administration and monthly thereafter. Instruct all patients with advanced chronic kidney disease, including those who are dialysis-dependent, about the symptoms of hypocalcemia and the importance of maintaining serum calcium levels with adequate calcium and activated vitamin D supplementation. Treatment with Conexxence in these patients should be supervised by a healthcare provider who is experienced in diagnosis and management of CKD-MBD.
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Issued: 03/2025 |
|||
|
MEDICATION GUIDE Conexxence (kon ex' ens) (denosumab-bnht) Injection, for subcutaneous use |
||||
|
What is the most important information I should know about Conexxence?
|
||||
| Call your doctor right away if you have any of these side effects. | ||||
|
What is Conexxence?
Conexxence is a prescription medicine used to:
|
||||
| It is not known if Conexxence is safe and effective in children. Conexxence is not approved for use in children. | ||||
Do not take Conexxence if you:
|
||||
Before taking Conexxence, tell your doctor about all of your medical conditions, including if you:
|
||||
|
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of medicines with you to show to your doctor or pharmacist when you get a new medicine. |
||||
How will I receive Conexxence?
|
||||
|
What are the possible side effects of Conexxence?
Conexxence may cause serious side effects.
|
||||
| The most common side effects of Conexxence in women who are being treated for osteoporosis after menopause are: | ||||
|
|
|||
| The most common side effects of Conexxence in men with osteoporosis are: | ||||
|
|
|||
| The most common side effects of Conexxence in patients with glucocorticoid-induced osteoporosis are: | ||||
|
|
|||
| The most common side effects of Conexxence in patients receiving certain treatments for prostate or breast cancer are: | ||||
|
|
|||
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Conexxence. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store Conexxence if I need to pick it up from a pharmacy?
|
||||
| Keep Conexxence and all medicines out of the reach of children. | ||||
|
General information about the safe and effective use of Conexxence.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Conexxence for a condition for which it was not prescribed. Do not give Conexxence to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about Conexxence that is written for healthcare providers. |
||||
|
What are the ingredients in Conexxence?
|
Section 44425-7 (44425-7)
Storage and Handling
Store Conexxence refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. Prior to administration, Conexxence may be allowed to reach room temperature up to 25°C (77°F) in the original container. Once removed from the refrigerator, Conexxence must not be exposed to temperatures above 25°C (77°F) and must be used within 14 days. Discard Conexxence if not used within 14 days. Do not use Conexxence after the expiry date printed on the label.
Protect Conexxence from direct light and heat.
Avoid vigorous shaking of Conexxence.
Section 51945-4 (51945-4)
Principal Display Panel – 60 mg/mL Carton Label
NDC 65219-668-01 Rx only
Scan for additional information
Conexxence
denosumab-bnht
Injection
60 mg/mL
For Subcutaneous use only
Single-dose prefilled syringe.
discard unused portion.
Carton contains:
1 Single-dose prefilled syringe
1 Prescribing Information
1 Medication Guide
Dispense the enclosed Medication
Guide to each patient
Sterile Solution - No Preservative
Conexxence should be administered by a healthcare provider
Fresenius
Kabi
11 Description (11 DESCRIPTION)
Denosumab-bnht is a human IgG2 monoclonal antibody with affinity and specificity for human RANKL (receptor activator of nuclear factor kappa-B ligand). Denosumab-bnht has an approximate molecular weight of 147 kDa and is produced in genetically engineered mammalian (Chinese hamster ovary) cells.
Conexxence (denosumab-bnht) injection is a sterile, preservative-free, clear, colorless to pale yellow solution for subcutaneous use.
Each 1 mL single-dose prefilled syringe of Conexxence contains 60 mg denosumab-bnht (60 mg/mL solution), 0.23 mg glacial acetic acid, 0.1 mg polysorbate 20, 1.07 mg sodium acetate, 47 mg sorbitol, and Water for Injection (USP). The pH is 5.2.
8.4 Pediatric Use
The safety and effectiveness of Conexxence have not been established in pediatric patients.
In one multicenter, open-label study with denosumab conducted in 153 pediatric patients with osteogenesis imperfecta, aged 2 to 17 years, evaluating fracture risk reduction, efficacy was not demonstrated.
Hypercalcemia has been reported in pediatric patients with osteogenesis imperfecta treated with denosumab products. Some cases required hospitalization and were complicated by acute renal injury [see Warnings and Precautions (5.11)]. Clinical studies in pediatric patients with osteogenesis imperfecta were terminated early due to the occurrence of life-threatening events and hospitalizations due to hypercalcemia.
Based on results from animal studies, denosumab may negatively affect long-bone growth and dentition in pediatric patients below the age of 4 years.
8.5 Geriatric Use
Of the total number of patients in clinical studies of denosumab, 9943 patients (76%) were ≥ 65 years old, while 3576 (27%) were ≥ 75 years old. Of the patients in the osteoporosis study in men, 133 patients (55%) were ≥ 65 years old, while 39 patients (16%) were ≥ 75 years old. Of the patients in the glucocorticoid-induced osteoporosis study, 355 patients (47%) were ≥ 65 years old, while 132 patients (17%) were ≥ 75 years old. No overall differences in safety or efficacy were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of denosumab or of other denosumab products.
Using an electrochemiluminescent bridging immunoassay, less than 1% (55 out of 8113) of patients treated with denosumab for up to 5 years tested positive for binding antibodies (including pre-existing, transient, and developing antibodies). None of the patients tested positive for neutralizing antibodies, as was assessed using a chemiluminescent cell-based in vitro biological assay.
There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of denosumab.
4 Contraindications (4 CONTRAINDICATIONS)
Conexxence is contraindicated in:
- Patients with hypocalcemia: Pre-existing hypocalcemia must be corrected prior to initiating therapy with Conexxence [see Warnings and Precautions (5.1)].
- Pregnant women: Denosumab products may cause fetal harm when administered to a pregnant woman. In women of reproductive potential, pregnancy testing should be performed prior to initiating treatment with Conexxence [see Use in Specific Populations (8.1)].
- Patients with hypersensitivity to denosumab products: Conexxence is contraindicated in patients with a history of systemic hypersensitivity to any component of the product. Reactions have included anaphylaxis, facial swelling, and urticaria [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed below and also elsewhere in the labeling:
- Severe Hypocalcemia and Mineral Metabolism Changes [see Warnings and Precautions (5.1)]
- Hypersensitivity [see Warnings and Precautions (5.3)]
- Osteonecrosis of the Jaw [see Warnings and Precautions (5.4)]
- Atypical Subtrochanteric and Diaphyseal Femoral Fractures [see Warnings and Precautions (5.5)]
- Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation of Treatment [see Warnings and Precautions (5.6)]
- Serious Infections [see Warnings and Precautions (5.7)]
- Dermatologic Adverse Reactions [see Warnings and Precautions (5.8)]
The most common adverse reactions reported with denosumab products in patients with postmenopausal osteoporosis are back pain, pain in extremity, musculoskeletal pain, hypercholesterolemia, and cystitis.
The most common adverse reactions reported with denosumab products in men with osteoporosis are back pain, arthralgia, and nasopharyngitis.
The most common adverse reactions reported with denosumab products in patients with glucocorticoid-induced osteoporosis are back pain, hypertension, bronchitis, and headache.
The most common (per patient incidence ≥ 10%) adverse reactions reported with denosumab products in patients with bone loss receiving androgen deprivation therapy for prostate cancer or adjuvant aromatase inhibitor therapy for breast cancer are arthralgia and back pain. Pain in extremity and musculoskeletal pain have also been reported in clinical trials.
The most common adverse reactions leading to discontinuation of denosumab products in patients with postmenopausal osteoporosis are back pain and constipation.
5.3 Hypersensitivity
Clinically significant hypersensitivity including anaphylaxis has been reported with denosumab products. Symptoms have included hypotension, dyspnea, throat tightness, facial and upper airway edema, pruritus, and urticaria. If an anaphylactic or other clinically significant allergic reaction occurs, initiate appropriate therapy and discontinue further use of Conexxence [see Contraindications (4), Adverse Reactions (6.2)].
8.6 Renal Impairment
No dose adjustment is necessary in patients with renal impairment.
Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported postmarketing. In clinical studies, patients with advanced chronic kidney disease (i.e., eGFR < 30 mL/min/1.73 m2), including dialysis-dependent patients, were at greater risk of developing hypocalcemia. The presence of underlying chronic kidney disease-mineral bone disorder (CKD-MBD, renal osteodystrophy) markedly increases the risk of hypocalcemia. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk. Consider the benefits and risks to the patient when administering Conexxence to patients with advanced chronic kidney disease. Monitor calcium and mineral levels (phosphorus and magnesium). Adequate intake of calcium and vitamin D is important in patients with advanced chronic kidney disease including dialysis-dependent patients [see Dosage and Administration (2.2), Warnings and Precautions (5.1), Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In clinical studies, treatment with 60 mg of denosumab resulted in reduction in the bone resorption marker serum type 1 C-telopeptide (CTX) by approximately 85% by 3 days, with maximal reductions occurring by 1 month. CTX levels were below the limit of assay quantitation (0.049 ng/mL) in 39% to 68% of patients 1 to 3 months after dosing of denosumab. At the end of each dosing interval, CTX reductions were partially attenuated from a maximal reduction of ≥ 87% to ≥ 45% (range: 45% to 80%), as serum denosumab levels diminished, reflecting the reversibility of the effects of denosumab on bone remodeling. These effects were sustained with continued treatment. Upon reinitiation, the degree of inhibition of CTX by denosumab was similar to that observed in patients initiating denosumab treatment.
Consistent with the physiological coupling of bone formation and resorption in skeletal remodeling, subsequent reductions in bone formation markers (i.e., osteocalcin and procollagen type 1 N-terminal peptide [P1NP]) were observed starting 1 month after the first dose of denosumab. After discontinuation of denosumab therapy, markers of bone resorption increased to levels 40% to 60% above pretreatment values but returned to baseline levels within 12 months.
12.3 Pharmacokinetics
In a study conducted in healthy male and female volunteers (n = 73, age range: 18 to 64 years) following a single subcutaneously administered denosumab dose of 60 mg, the mean area-under-the-concentration-time curve up to 16 weeks (AUC0-16 weeks) of denosumab was 316 mcg⋅day/mL (standard deviation [SD] = 101 mcg⋅day/mL). The mean maximum denosumab concentration (Cmax) was 6.75 mcg/mL (SD = 1.89 mcg/mL). No accumulation or change in denosumab pharmacokinetics with time is observed with multiple dosing of 60 mg subcutaneously administered once every 6 months.
2.3 Recommended Dosage
Conexxence should be administered by a healthcare provider.
The recommended dose of Conexxence is 60 mg administered as a single subcutaneous injection once every 6 months. Administer Conexxence via subcutaneous injection in the upper arm, the upper thigh, or the abdomen. All patients should receive calcium 1000 mg daily and at least 400 IU vitamin D daily [see Warnings and Precautions (5.1)].
If a dose of Conexxence is missed, administer the injection as soon as the patient is available. Thereafter, schedule injections every 6 months from the date of the last injection.
5.7 Serious Infections
In a clinical trial of over 7800 women with postmenopausal osteoporosis, serious infections leading to hospitalization were reported more frequently in the denosumab group than in the placebo group [see Adverse Reactions (6.1)]. Serious skin infections, as well as infections of the abdomen, urinary tract, and ear, were more frequent in patients treated with denosumab. Endocarditis was also reported more frequently in denosumab-treated patients. The incidence of opportunistic infections was similar between placebo and denosumab groups, and the overall incidence of infections was similar between the treatment groups. Advise patients to seek prompt medical attention if they develop signs or symptoms of severe infection, including cellulitis.
Patients on concomitant immunosuppressant agents or with impaired immune systems may be at increased risk for serious infections. Consider the benefit-risk profile in such patients before treating with Conexxence. In patients who develop serious infections while on Conexxence, prescribers should assess the need for continued Conexxence therapy.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Conexxence is a RANK ligand (RANKL) inhibitor indicated for treatment:
- of postmenopausal women with osteoporosis at high risk for fracture (1.1)
- to increase bone mass in men with osteoporosis at high risk for fracture (1.2)
- of glucocorticoid-induced osteoporosis in men and women at high risk for fracture (1.3)
- to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy for nonmetastatic prostate cancer (1.4)
- to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer (1.5)
12.1 Mechanism of Action
Denosumab products bind to RANKL, a transmembrane or soluble protein essential for the formation, function, and survival of osteoclasts, the cells responsible for bone resorption. Denosumab products prevent RANKL from activating its receptor, RANK, on the surface of osteoclasts and their precursors. Prevention of the RANKL/RANK interaction inhibits osteoclast formation, function, and survival, thereby decreasing bone resorption and increasing bone mass and strength in both cortical and trabecular bone.
5.9 Musculoskeletal Pain
In postmarketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain has been reported in patients taking denosumab products [see Adverse Reactions (6.2)]. The time to onset of symptoms varied from one day to several months after starting denosumab products. Consider discontinuing use if severe symptoms develop [see Patient Counseling Information (17)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypocalcemia: Pre-existing hypocalcemia must be corrected before initiating Conexxence. May worsen, especially in patients with renal impairment. Adequately supplement all patients with calcium and vitamin D. Concomitant use of calcimimetic drugs may also worsen hypocalcemia risk. Evaluate for presence of chronic kidney disease mineral-bone disorder. Monitor serum calcium. (5.1)
- Same Active Ingredient: Patients receiving Conexxence should not receive other denosumab products concomitantly (5.2)
- Hypersensitivity including anaphylactic reactions may occur. Discontinue permanently if a clinically significant reaction occurs. (5.3)
- Osteonecrosis of the jaw: Has been reported with denosumab products. Monitor for symptoms. (5.4)
- Atypical femoral fractures: Have been reported. Evaluate patients with thigh or groin pain to rule out a femoral fracture. (5.5)
- Multiple vertebral fractures have been reported following treatment discontinuation. Patients should be transitioned to another antiresorptive agent if Conexxence is discontinued. (5.6)
- Serious infections including skin infections: May occur, including those leading to hospitalization. Advise patients to seek prompt medical attention if they develop signs or symptoms of infection, including cellulitis. (5.7)
- Dermatologic reactions: Dermatitis, rashes, and eczema have been reported. Consider discontinuing Conexxence if severe symptoms develop. (5.8)
- Severe bone, joint, muscle pain may occur. Discontinue use if severe symptoms develop. (5.9)
- Suppression of bone turnover: Significant suppression has been demonstrated. Monitor for consequences of bone over-suppression. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Pregnancy must be ruled out prior to administration of Conexxence. (2.1)
- Before initiating Conexxence in patients with advanced chronic kidney disease, including dialysis patients, evaluate for the presence of chronic kidney disease mineral and bone disorder with intact parathyroid hormone, serum calcium, 25(OH) vitamin D, and 1,25(OH)2 vitamin D. (2.2, 5.1, 8.6)
- Conexxence should be administered by a healthcare provider. (2.3)
- Administer 60 mg every 6 months as a subcutaneous injection in the upper arm, upper thigh, or abdomen. (2.3)
- Instruct patients to take calcium 1000 mg daily and at least 400 IU vitamin D daily. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Injection: 60 mg/mL clear, colorless to pale yellow solution in a single-dose prefilled syringe.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ), which can occur spontaneously, is generally associated with tooth extraction and/or local infection with delayed healing. ONJ has been reported in patients receiving denosumab products [see Adverse Reactions (6.1)]. A routine oral exam should be performed by the prescriber prior to initiation of Conexxence treatment. A dental examination with appropriate preventive dentistry is recommended prior to treatment with Conexxence in patients with risk factors for ONJ such as invasive dental procedures (e.g. tooth extraction, dental implants, oral surgery), diagnosis of cancer, concomitant therapies (e.g. chemotherapy, corticosteroids, angiogenesis inhibitors), poor oral hygiene, and comorbid disorders (e.g. periodontal and/or other pre-existing dental disease, anemia, coagulopathy, infection, ill-fitting dentures). Good oral hygiene practices should be maintained during treatment with Conexxence. Concomitant administration of drugs associated with ONJ may increase the risk of developing ONJ. The risk of ONJ may increase with duration of exposure to denosumab products.
For patients requiring invasive dental procedures, clinical judgment of the treating physician and/or oral surgeon should guide the management plan of each patient based on individual benefit-risk assessment.
Patients who are suspected of having or who develop ONJ while on Conexxence should receive care by a dentist or an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Discontinuation of Conexxence therapy should be considered based on individual benefit-risk assessment.
6.2 Postmarketing Experience
Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of denosumab products:
- Drug-related hypersensitivity reactions: anaphylaxis, rash, urticaria, facial swelling, and erythema
- Hypocalcemia: severe symptomatic hypocalcemia resulting in hospitalization, life-threatening events, and fatal cases
- Musculoskeletal pain, including severe cases
- Parathyroid hormone (PTH): Marked elevation in serum PTH in patients with severe renal impairment (creatinine clearance < 30 mL/min) or receiving dialysis
- Multiple vertebral fractures following treatment discontinuation
- Cutaneous and mucosal lichenoid drug eruptions (e.g. lichen planus-like reactions)
- Alopecia
- Vasculitis (e.g. ANCA positive vasculitis, leukocytoclastic vasculitis)
- Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnant women and females of reproductive potential: Denosumab products may cause fetal harm when administered to pregnant women. Advise females of reproductive potential to use effective contraception during therapy, and for at least 5 months after the last dose of Conexxence. (8.1, 8.3)
- Pediatric patients: Conexxence is not approved for use in pediatric patients. (8.4)
- Renal impairment: No dose adjustment is necessary in patients with renal impairment. Patients with advanced chronic kidney disease (eGFR < 30 mL/min/1.73 m2), including dialysis-dependent patients, are at greater risk of severe hypocalcemia. The presence of underlying chronic kidney disease-mineral bone disorder markedly increases the risk of hypocalcemia. (5.1, 8.6)
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.10 Suppression of Bone Turnover
In clinical trials in women with postmenopausal osteoporosis, treatment with denosumab resulted in significant suppression of bone remodeling as evidenced by markers of bone turnover and bone histomorphometry [see Clinical Pharmacology (12.2), Clinical Studies (14.1)]. The significance of these findings and the effect of long-term treatment with denosumab products are unknown. The long-term consequences of the degree of suppression of bone remodeling observed with denosumab may contribute to adverse outcomes such as osteonecrosis of the jaw, atypical fractures, and delayed fracture healing. Monitor patients for these consequences.
2.4 Preparation and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Conexxence is a clear, colorless to pale yellow solution that is free from visible particles. Do not use if the solution is discolored or cloudy or if the solution contains particles or foreign particulate matter.
Prior to administration, Conexxence may be removed from the refrigerator and brought to room temperature up to 25°C (77°F) by standing in the original container. This generally takes 15 to 30 minutes. Do not warm Conexxence in any other way [see How Supplied/Storage and Handling (16)]. Do not shake the prefilled syringe.
Instructions for Administration of Conexxence Prefilled Syringe with Needle Safety Guard
IMPORTANT:
In order to minimize accidental needlesticks, the Conexxence single-dose prefilled syringe has an automatic clear needle safety guard.
Conexxence prefilled syringe (Before & After Use). See figure below.
|
Step 1: Remove Needle Cap (see
Figure A
) Do not hold the prefilled syringe by the plunger rod. Do not twist or bend the needle cap. |
Figure A |
|
Step 2: Administer Subcutaneous Injection
Choose an injection site. The recommended injection sites for Conexxence include (see Figure B ): |
|
|
Figure B |
| Do not administer into muscle or blood vessel. Pinch the skin and insert the needle at a 45 to 90-degree angle. Push the plunger with a slow and constant pressure until you cannot press further and have injected all the liquid subcutaneously (see Figure C ). You may hear or feel a “click”. |
Figure C |
|
Step 3. Release Plunger
Keeping the prefilled syringe at the injection site, slowly release the plunger and lift syringe off the skin. The clear needle guard will automatically cover the needle (see Figure D ). |
|
|
Do not recap the needle. Immediately dispose of the used prefilled syringe into a sharps disposal container. |
Figure D |
5.8 Dermatologic Adverse Reactions
In a large clinical trial of over 7800 women with postmenopausal osteoporosis, epidermal and dermal adverse events such as dermatitis, eczema, and rashes occurred at a significantly higher rate in the denosumab group compared to the placebo group. Most of these events were not specific to the injection site [see Adverse Reactions (6.1)]. Consider discontinuing Conexxence if severe symptoms develop.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Denosumab products are inhibitors of osteoclastic bone resorption via inhibition of RANKL.
In ovariectomized monkeys, once-monthly treatment with denosumab suppressed bone turnover and increased BMD and strength of cancellous and cortical bone at doses 50-fold higher than the recommended human dose of 60 mg administered once every 6 months, based on body weight (mg/kg). Bone tissue was normal with no evidence of mineralization defects, accumulation of osteoid, or woven bone.
Because the biological activity of denosumab in animals is specific to nonhuman primates, evaluation of genetically engineered (“knockout”) mice or use of other biological inhibitors of the RANK/RANKL pathway, namely OPG-Fc, provided additional information on the pharmacodynamic properties of denosumab products. RANK/RANKL knockout mice exhibited absence of lymph node formation, as well as an absence of lactation due to inhibition of mammary gland maturation (lobulo-alveolar gland development during pregnancy). Neonatal RANK/RANKL knockout mice exhibited reduced bone growth and lack of tooth eruption. A corroborative study in 2-week-old rats given the RANKL inhibitor OPG-Fc also showed reduced bone growth, altered growth plates, and impaired tooth eruption. These changes were partially reversible in this model when dosing with the RANKL inhibitors was discontinued.
5.2 Drug Products With Same Active Ingredient (5.2 Drug Products with Same Active Ingredient)
Patients receiving Conexxence should not receive other denosumab products concomitantly.
8.3 Females and Males of Reproductive Potential
Based on findings in animals, denosumab products can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
1.3 Treatment of Glucocorticoid Induced Osteoporosis (1.3 Treatment of Glucocorticoid-Induced Osteoporosis)
Conexxence is indicated for the treatment of glucocorticoid-induced osteoporosis in men and women at high risk of fracture who are either initiating or continuing systemic glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and expected to remain on glucocorticoids for at least 6 months. High risk of fracture is defined as a history of osteoporotic fracture, multiple risk factors for fracture, or patients who have failed or are intolerant to other available osteoporosis therapy [see Clinical Studies (14.3)].
14.3 Treatment of Glucocorticoid Induced Osteoporosis (14.3 Treatment of Glucocorticoid-Induced Osteoporosis)
The efficacy and safety of denosumab in the treatment of patients with glucocorticoid-induced osteoporosis was assessed in the 12-month primary analysis of a 2-year, randomized, multicenter, double-blind, parallel-group, active-controlled study (NCT 01575873) of 795 patients (70% women and 30% men) aged 20 to 94 years (mean age of 63 years) treated with greater than or equal to 7.5 mg/day oral prednisone (or equivalent) for < 3 months prior to study enrollment and planning to continue treatment for a total of at least 6 months (glucocorticoid-initiating subpopulation; n = 290) or ≥ 3 months prior to study enrollment and planning to continue treatment for a total of at least 6 months (glucocorticoid-continuing subpopulation, n = 505). Enrolled patients < 50 years of age were required to have a history of osteoporotic fracture. Enrolled patients ≥ 50 years of age who were in the glucocorticoid-continuing subpopulation were required to have a baseline BMD T-score of ≤ -2.0 at the lumbar spine, total hip, or femoral neck; or a BMD T-score ≤ -1.0 at the lumbar spine, total hip, or femoral neck and a history of osteoporotic fracture.
Patients were randomized (1:1) to receive either an oral daily bisphosphonate (active-control, risedronate 5 mg once daily) (n = 397) or denosumab 60 mg subcutaneously once every 6 months (n = 398) for one year. Randomization was stratified by gender within each subpopulation. Patients received at least 1000 mg calcium and 800 IU vitamin D supplementation daily.
5.1 Severe Hypocalcemia and Mineral Metabolism Changes
Denosumab products can cause severe hypocalcemia and fatal cases have been reported. Pre-existing hypocalcemia must be corrected prior to initiating therapy with Conexxence. Adequately supplement all patients with calcium and vitamin D [see Dosage and Administration (2.1), Contraindications (4), and Adverse Reactions (6.1)].
In patients without advanced chronic kidney disease who are predisposed to hypocalcemia and disturbances of mineral metabolism (e.g. history of hypoparathyroidism, thyroid surgery, parathyroid surgery, malabsorption syndromes, excision of small intestine, treatment with other calcium-lowering drugs), assess serum calcium and mineral levels (phosphorus and magnesium) 10 to14 days after Conexxence injection. In some postmarketing cases, hypocalcemia persisted for weeks or months and required frequent monitoring and intravenous and/or oral calcium replacement, with or without vitamin D.
14.4 Treatment of Bone Loss in Men With Prostate Cancer (14.4 Treatment of Bone Loss in Men with Prostate Cancer)
The efficacy and safety of denosumab in the treatment of bone loss in men with nonmetastatic prostate cancer receiving androgen deprivation therapy (ADT) were demonstrated in a 3-year, randomized (1:1), double-blind, placebo-controlled, multinational study. Men less than 70 years of age had either a BMD T-score at the lumbar spine, total hip, or femoral neck between -1.0 and -4.0, or a history of an osteoporotic fracture. The mean baseline lumbar spine BMD T-score was -0.4, and 22% of men had a vertebral fracture at baseline. The 1468 men enrolled ranged in age from 48 to 97 years (median 76 years). Men were randomized to receive subcutaneous injections of either placebo (n = 734) or denosumab 60 mg (n = 734) once every 6 months for a total of 6 doses. Randomization was stratified by age (< 70 years vs. ≥ 70 years) and duration of ADT at trial entry (≤ 6 months vs. > 6 months). Seventy-nine percent of patients received ADT for more than 6 months at study entry. All men received at least 1000 mg calcium and 400 IU vitamin D supplementation daily.
14.5 Treatment of Bone Loss in Women With Breast Cancer (14.5 Treatment of Bone Loss in Women with Breast Cancer)
The efficacy and safety of denosumab in the treatment of bone loss in women receiving adjuvant aromatase inhibitor (AI) therapy for breast cancer was assessed in a 2-year, randomized (1:1), double-blind, placebo-controlled, multinational study. Women had baseline BMD T-scores between -1.0 to -2.5 at the lumbar spine, total hip, or femoral neck, and had not experienced fracture after age 25. The mean baseline lumbar spine BMD T-score was -1.1, and 2.0% of women had a vertebral fracture at baseline. The 252 women enrolled ranged in age from 35 to 84 years (median 59 years). Women were randomized to receive subcutaneous injections of either placebo (n = 125) or denosumab 60 mg (n = 127) once every 6 months for a total of 4 doses. Randomization was stratified by duration of adjuvant AI therapy at trial entry (≤ 6 months vs. > 6 months). Sixty-two percent of patients received adjuvant AI therapy for more than 6 months at study entry. All women received at least 1000 mg calcium and 400 IU vitamin D supplementation daily.
2.1 Pregnancy Testing Prior to Initiation of Conexxence
Pregnancy must be ruled out prior to administration of Conexxence. Perform pregnancy testing in all females of reproductive potential prior to administration of Conexxence. Based on findings in animals, denosumab products can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1, 8.3)].
14.1 Treatment of Postmenopausal Women With Osteoporosis (14.1 Treatment of Postmenopausal Women with Osteoporosis)
The efficacy and safety of denosumab in the treatment of postmenopausal osteoporosis was demonstrated in a 3-year, randomized, double-blind, placebo-controlled trial. Enrolled women had a baseline BMD T-score between -2.5 and -4.0 at either the lumbar spine or total hip. Women with other diseases (such as rheumatoid arthritis, osteogenesis imperfecta, and Paget's disease) or on therapies that affect bone were excluded from this study. The 7808 enrolled women were aged 60 to 91 years with a mean age of 72 years. Overall, the mean baseline lumbar spine BMD T-score was -2.8, and 23% of women had a vertebral fracture at baseline. Women were randomized to receive subcutaneous injections of either placebo (N = 3906) or denosumab 60 mg (N = 3902) once every 6 months. All women received at least 1000 mg calcium and 400 IU vitamin D supplementation daily.
The primary efficacy variable was the incidence of new morphometric (radiologically-diagnosed) vertebral fractures at 3 years. Vertebral fractures were diagnosed based on lateral spine radiographs (T4-L4) using a semiquantitative scoring method. Secondary efficacy variables included the incidence of hip fracture and nonvertebral fracture, assessed at 3 years.
1.2 Treatment to Increase Bone Mass in Men With Osteoporosis (1.2 Treatment to Increase Bone Mass in Men with Osteoporosis)
Conexxence is indicated for treatment to increase bone mass in men with osteoporosis at high risk for fracture, defined as a history of osteoporotic fracture, or multiple risk factors for fracture; or patients who have failed or are intolerant to other available osteoporosis therapy [see Clinical Studies (14.2)].
14.2 Treatment to Increase Bone Mass in Men With Osteoporosis (14.2 Treatment to Increase Bone Mass in Men with Osteoporosis)
The efficacy and safety of denosumab in the treatment to increase bone mass in men with osteoporosis was demonstrated in a 1-year, randomized, double-blind, placebo-controlled trial. Enrolled men had a baseline BMD T-score between -2.0 and -3.5 at the lumbar spine or femoral neck. Men with a BMD T-score between -1.0 and -3.5 at the lumbar spine or femoral neck were also enrolled if there was a history of prior fragility fracture. Men with other diseases (such as rheumatoid arthritis, osteogenesis imperfecta, and Paget's disease) or on therapies that may affect bone were excluded from this study. The 242 men enrolled in the study ranged in age from 31 to 84 years with a mean age of 65 years. Men were randomized to receive SC injections of either placebo (n = 121) or denosumab 60 mg (n = 121) once every 6 months. All men received at least 1000 mg calcium and at least 800 IU vitamin D supplementation daily.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical low energy or low trauma fractures of the shaft have been reported in patients receiving denosumab products [see Adverse Reactions (6.1)]. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with antiresorptive agents.
Atypical femoral fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral, and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
During Conexxence treatment, patients should be advised to report new or unusual thigh, hip, or groin pain. Any patient who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of Conexxence therapy should be considered, pending a benefit-risk assessment, on an individual basis.
5.11 Hypercalcemia in Pediatric Patients With Osteogenesis Imperfecta (5.11 Hypercalcemia in Pediatric Patients with Osteogenesis Imperfecta)
Conexxence is not approved for use in pediatric patients. Hypercalcemia has been reported in pediatric patients with osteogenesis imperfecta treated with denosumab products. Some cases required hospitalization [see Use in Specific Populations (8.4)].
Warning: Severe Hypocalcemia in Patients With Advanced Kidney Disease (WARNING: SEVERE HYPOCALCEMIA IN PATIENTS WITH ADVANCED KIDNEY DISEASE)
- Patients with advanced chronic kidney disease (eGFR < 30 mL/min/1.73 m 2 ), including dialysis-dependent patients, are at greater risk of severe hypocalcemia following denosumab products administration. Severe hypocalcemia resulting in hospitalization, life-threatening events and fatal cases have been reported [see Warnings and Precautions (5.1)] .
- The presence of chronic kidney disease-mineral bone disorder (CKD-MBD) markedly increases the risk of hypocalcemia in these patients [see Warnings and Precautions (5.1)] .
- Prior to initiating Conexxence in patients with advanced chronic kidney disease, evaluate for the presence of CKD-MBD. Treatment with Conexxence in these patients should be supervised by a healthcare provider with expertise in the diagnosis and management of CKD-MBD [see Dosage and Administration (2.2) and Warnings and Precautions (5.1)].
5.6 Multiple Vertebral Fractures (mvf) Following Discontinuation of Treatment (5.6 Multiple Vertebral Fractures (MVF) Following Discontinuation of Treatment)
Following discontinuation of denosumab treatment, fracture risk increases, including the risk of multiple vertebral fractures. Treatment with denosumab results in significant suppression of bone turnover and cessation of denosumab treatment results in increased bone turnover above pretreatment values 9 months after the last dose of denosumab. Bone turnover then returns to pretreatment values 24 months after the last dose of denosumab. In addition, bone mineral density (BMD) returns to pretreatment values within 18 months after the last injection [see Clinical Pharmacology (12.2), Clinical Studies (14.1)].
New vertebral fractures occurred as early as 7 months (on average 19 months) after the last dose of denosumab. Prior vertebral fracture was a predictor of multiple vertebral fractures after denosumab discontinuation. Evaluate an individual's benefit-risk before initiating treatment with Conexxence.
If Conexxence treatment is discontinued, patients should be transitioned to an alternative antiresorptive therapy [see Adverse Reactions (6.1)].
1.1 Treatment of Postmenopausal Women With Osteoporosis At High Risk for Fracture (1.1 Treatment of Postmenopausal Women with Osteoporosis at High Risk for Fracture)
Conexxence is indicated for the treatment of postmenopausal women with osteoporosis at high risk for fracture, defined as a history of osteoporotic fracture, or multiple risk factors for fracture; or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, denosumab reduces the incidence of vertebral, nonvertebral, and hip fractures [see Clinical Studies (14.1)].
1.4 Treatment of Bone Loss in Men Receiving Androgen Deprivation Therapy for Prostate Cancer
Conexxence is indicated as a treatment to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy (ADT) for nonmetastatic prostate cancer. In these patients denosumab also reduced the incidence of vertebral fractures [see Clinical Studies (14.4)].
1.5 Treatment of Bone Loss in Women Receiving Adjuvant Aromatase Inhibitor Therapy for Breast Cancer
Conexxence is indicated as a treatment to increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer [see Clinical Studies (14.5)].
2.2 Laboratory Testing in Patients With Advanced Chronic Kidney Disease Prior to Initiation of Conexxence (2.2 Laboratory Testing in Patients with Advanced Chronic Kidney Disease Prior to Initiation of Conexxence)
In patients with advanced chronic kidney disease [i.e., estimated glomerular filtration rate (eGFR) < 30 mL/min/1.73 m2], including dialysis-dependent patients, evaluate for the presence of chronic kidney disease mineral and bone disorder (CKD-MBD) with intact parathyroid hormone (iPTH), serum calcium, 25(OH) vitamin D, and 1,25 (OH)2 vitamin D prior to decisions regarding Conexxence treatment. Consider also assessing bone turnover status (serum markers of bone turnover or bone biopsy) to evaluate the underlying bone disease that may be present [see Warnings and Precautions (5.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:19.642602 · Updated: 2026-03-14T22:40:03.259578