These Highlights Do Not Include All The Information Needed To Use Omniscan Safely And Effectively. See Full Prescribing Information For Omniscan.
1e9a37e2-f28a-4373-bf0f-3e9b60f42d8a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS See full prescribing information for complete boxed warning. Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. OMNISCAN is not approved for intrathecal use ( 5.1 ). GBCAs increase the risk for nephrogenic systemic fibrosis among patients with impaired elimination of the drugs. Avoid use of OMNISCAN in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. Do not administer OMNISCAN to patients with: chronic, severe kidney disease (GFR < 30 mL/min/1.73m 2 ), or acute kidney injury ( 4 ). Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (e.g., age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing ( 5.2 ).
Indications and Usage
OMNISCAN is a gadolinium-based contrast agent for diagnostic magnetic resonance imaging (MRI) indicated for intravenous use to: Visualize lesions with abnormal vascularity in the brain, spine, and associated tissues ( 1.1 ) Facilitate the visualization of lesions with abnormal vascularity within the thoracic, abdominal, pelvic cavities, and the retroperitoneal space ( 1.2 )
Dosage and Administration
CNS – Adults and Pediatrics; 2 to 16 years of age: 0.2 mL/kg (0.1 mmol/kg) ( 2.1 , 2.4 ) Body – Adults and Pediatrics; 2 to 16 years of age: Kidney: 0.1 mL/kg (0.05 mmol/kg) Intrathoracic, intra-abdominal, and pelvic cavities: 0.2 mL/kg (0.1 mmol/kg) ( 2.2 , 2.4 )
Warnings and Precautions
Hypersensitivity Reactions: Anaphylactoid and other serious hypersensitivity reactions including fatal reactions have occurred particularly in patients with history of allergy or drug reactions. Monitor patients closely for need of emergency cardiorespiratory support ( 5.3 ). Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs ( 5.4 ). Acute Renal Failure: Acute renal failure has occurred in patients with preexisting renal insufficiency. Use the lowest necessary dose of OMNISCAN and evaluate renal function in these patients ( 5.5 ).
Contraindications
OMNISCAN is contraindicated in patients with: Chronic, severe kidney disease (glomerular filtration rate, GFR < 30 mL/min/1.73m 2 ) or acute kidney injury Prior hypersensitivity to OMNISCAN
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the label: Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2) ] Hypersensitivity reactions [see Warnings and Precautions (5.3) ] Acute Renal Failure [see Warnings and Precautions (5.5) ]
Drug Interactions
Specific drug interaction studies have not been conducted.
Storage and Handling
OMNISCAN (gadodiamide) Injection is a sterile, clear, colorless to slightly yellow, aqueous solution containing 287 mg/mL of gadodiamide in rubber stoppered vials. OMNISCAN is supplied in the following sizes: 10 mL vial, box of 10 (NDC 0407-0690-10) 15 mL fill in 20 mL vial, box of 10 (NDC 0407-0690-15) 20 mL vial, box of 10 (NDC 0407-0690-20)
How Supplied
OMNISCAN (gadodiamide) Injection is a sterile, clear, colorless to slightly yellow, aqueous solution containing 287 mg/mL of gadodiamide in rubber stoppered vials. OMNISCAN is supplied in the following sizes: 10 mL vial, box of 10 (NDC 0407-0690-10) 15 mL fill in 20 mL vial, box of 10 (NDC 0407-0690-15) 20 mL vial, box of 10 (NDC 0407-0690-20)
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: Anaphylactoid and other serious hypersensitivity reactions including fatal reactions have occurred particularly in patients with history of allergy or drug reactions. Monitor patients closely for need of emergency cardiorespiratory support ( 5.3 ). Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs ( 5.4 ). Acute Renal Failure: Acute renal failure has occurred in patients with preexisting renal insufficiency. Use the lowest necessary dose of OMNISCAN and evaluate renal function in these patients ( 5.5 ).
Indications and Usage
OMNISCAN is a gadolinium-based contrast agent for diagnostic magnetic resonance imaging (MRI) indicated for intravenous use to: Visualize lesions with abnormal vascularity in the brain, spine, and associated tissues ( 1.1 ) Facilitate the visualization of lesions with abnormal vascularity within the thoracic, abdominal, pelvic cavities, and the retroperitoneal space ( 1.2 )
Dosage and Administration
CNS – Adults and Pediatrics; 2 to 16 years of age: 0.2 mL/kg (0.1 mmol/kg) ( 2.1 , 2.4 ) Body – Adults and Pediatrics; 2 to 16 years of age: Kidney: 0.1 mL/kg (0.05 mmol/kg) Intrathoracic, intra-abdominal, and pelvic cavities: 0.2 mL/kg (0.1 mmol/kg) ( 2.2 , 2.4 )
Contraindications
OMNISCAN is contraindicated in patients with: Chronic, severe kidney disease (glomerular filtration rate, GFR < 30 mL/min/1.73m 2 ) or acute kidney injury Prior hypersensitivity to OMNISCAN
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the label: Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2) ] Hypersensitivity reactions [see Warnings and Precautions (5.3) ] Acute Renal Failure [see Warnings and Precautions (5.5) ]
Drug Interactions
Specific drug interaction studies have not been conducted.
Storage and Handling
OMNISCAN (gadodiamide) Injection is a sterile, clear, colorless to slightly yellow, aqueous solution containing 287 mg/mL of gadodiamide in rubber stoppered vials. OMNISCAN is supplied in the following sizes: 10 mL vial, box of 10 (NDC 0407-0690-10) 15 mL fill in 20 mL vial, box of 10 (NDC 0407-0690-15) 20 mL vial, box of 10 (NDC 0407-0690-20)
How Supplied
OMNISCAN (gadodiamide) Injection is a sterile, clear, colorless to slightly yellow, aqueous solution containing 287 mg/mL of gadodiamide in rubber stoppered vials. OMNISCAN is supplied in the following sizes: 10 mL vial, box of 10 (NDC 0407-0690-10) 15 mL fill in 20 mL vial, box of 10 (NDC 0407-0690-15) 20 mL vial, box of 10 (NDC 0407-0690-20)
Description
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS See full prescribing information for complete boxed warning. Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. OMNISCAN is not approved for intrathecal use ( 5.1 ). GBCAs increase the risk for nephrogenic systemic fibrosis among patients with impaired elimination of the drugs. Avoid use of OMNISCAN in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. Do not administer OMNISCAN to patients with: chronic, severe kidney disease (GFR < 30 mL/min/1.73m 2 ), or acute kidney injury ( 4 ). Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (e.g., age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing ( 5.2 ).
Section 42229-5
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. OMNISCAN is not approved for intrathecal use [see Warnings and Precautions (5.1)].
Section 42231-1
| MEDICATION GUIDE OMNISCAN™ (OMNI-scan) (gadodiamide) Injection, for intravenous use |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 3/2025 |
|
What is the most important information I should know about OMNISCAN?
|
|
What is OMNISCAN?
|
|
| Do not receive OMNISCAN if you have had a severe allergic reaction to OMNISCAN. | |
Before receiving OMNISCAN, tell your healthcare provider about all your medical conditions, including if you:
|
|
What are the possible side effects of OMNISCAN?
These are not all the possible side effects of OMNISCAN. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about the safe and effective use of OMNISCAN.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your healthcare provider for information about OMNISCAN that is written for health professionals. |
|
|
What are the ingredients in OMNISCAN?
Active ingredient: gadodiamide Inactive ingredients: caldiamide sodium, hydrochloric acid, sodium hydroxide © 2025 GE HealthCare Distributed by GE Healthcare Inc., Arlington Heights, IL 60004 U.S.A. GE is a trademark of General Electric Company used under trademark license. For more information, go to www.GEHealthCare.com or call 1-800-654-0118. |
Section 44425-7
Protect OMNISCAN from strong daylight and direct exposure to sunlight. Do not freeze. Freezing can cause small cracks in the vials, which would compromise the sterility of the product. Do not use if the product is inadvertently frozen.
Store OMNISCAN at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP].
10 Overdosage
Clinical consequences of overdose with OMNISCAN have not been reported. The minimum lethal dose of intravenously administered OMNISCAN in rats and mice is greater than 20 mmol/kg (200 times the recommended human dose of 0.1 mmol/kg; 67 times the cumulative 0.3 mmol/kg dose). OMNISCAN is dialyzable.
11 Description
OMNISCAN (gadodiamide) Injection is the formulation of the gadolinium complex of diethylenetriamine pentaacetic acid bismethylamide, and is an injectable, nonionic extracellular enhancing agent for magnetic resonance imaging. OMNISCAN is administered by intravenous injection.
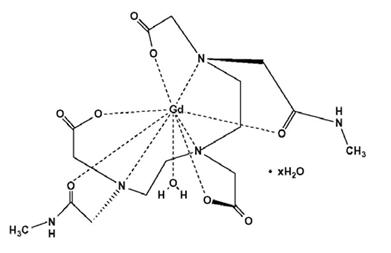
OMNISCAN is provided as a sterile, clear, colorless to slightly yellow, aqueous solution. Each 1 mL contains 287 mg gadodiamide and 12 mg caldiamide sodium in Water for Injection. The pH is adjusted between 5.5 and 7.0 with hydrochloric acid and/or sodium hydroxide. OMNISCAN contains no antimicrobial preservative. OMNISCAN is a 0.5 mol/L solution of aqua[5,8-bis(carboxymethyl)-11-[2-(methylamino)-2-oxoethyl]-3-oxo-2,5,8,11-tetraazatridecan-13-oato (3-)-N5, N8, N11, O3, O5, O8, O11, O13] gadolinium hydrate, with a molecular weight of 573.66 (anhydrous), an empirical formula of C16H28GdN5O9∙xH2O, and the following structural formula:
Pertinent physicochemical data for OMNISCAN are noted below:
| Osmolality (mOsmol/kg water) | @ 37°C | 789 |
| Viscosity (cP) | @ 20°C | 2 |
| @ 37°C | 1.4 | |
| Density (g/mL) | @ 25°C | 1.14 |
| Specific gravity | @ 25°C | 1.15 |
OMNISCAN has an osmolality approximately 2.8 times that of plasma at 37°C and is hypertonic under conditions of use.
2.3 Dosage Chart
| BODY WEIGHT | PEDIATRIC | ADULTS | |||
|---|---|---|---|---|---|
| 0.05 | 0.1 | 0.05 | 0.1 | ||
| kg | lb | (mmol/kg) | (mmol/kg) | ||
| VOLUME (mL) | VOLUME (mL) | ||||
| 12 | 26 | 1.2 | 2.4 | - | - |
| 14 | 31 | 1.4 | 2.8 | - | - |
| 16 | 35 | 1.6 | 3.2 | - | - |
| 18 | 40 | 1.8 | 3.6 | - | - |
| 20 | 44 | 2 | 4 | - | - |
| 22 | 48 | 2.2 | 4.4 | - | - |
| 24 | 53 | 2.4 | 4.8 | - | - |
| 26 | 57 | 2.6 | 5.2 | - | - |
| 28 | 62 | 2.8 | 5.6 | - | - |
| 30 | 66 | 3 | 6 | - | - |
| 40 | 88 | 4 | 8 | 4 | 8 |
| 50 | 110 | 5 | 10 | 5 | 10 |
| 60 | 132 | 6 | 12 | 6 | 12 |
| 70 | 154 | 7 | 14 | 7 | 14 |
| 80 | 176 | 8 | 16 | 8 | 16 |
| 90 | 198 | - | - | 9 | 18 |
| 100 | 220 | - | - | 10 | 20 |
| 110 | 242 | - | - | 11 | 22 |
| 120 | 264 | - | - | 12 | 24 |
| 130 The heaviest patient in clinical studies weighed 136 kg.
|
286 | - | - | 13 | 26 |
8.4 Pediatric Use
The safety and efficacy of OMNISCAN at a single dose of 0.05 to 0.1 mmol/kg have been established in pediatric patients over 2 years of age based on adequate and well controlled studies of OMNISCAN in adults, a pediatric CNS imaging study, and safety data in the scientific literature. However, the safety and efficacy of doses greater than 0.1 mmol/kg and of repeated doses have not been studied in pediatric patients.
Pharmacokinetics of OMNISCAN have not been studied in pediatrics. The glomerular filtration rate of neonates and infants is much lower than that of adults. The pharmacokinetics volume of distribution is also different. Therefore, the optimal dosing regimen and imaging times in patients under 2 years of age have not been established.
8.5 Geriatric Use
In clinical studies of OMNISCAN, 243 patients were between 65 and 80 years of age while 15 were over 80 years of age. No overall differences in safety or effectiveness were observed between these patients and younger patients. Other reported clinical experience has not identified differences in response between the elderly and younger patients, but greater sensitivity in the elderly cannot be ruled out. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
OMNISCAN is excreted by the kidney, and the risk of toxic reactions to OMNISCAN may be greater in patients with impaired renal function [see Warnings and Precautions (5.4)]. Because elderly patients are more likely to have decreased renal function, select dose carefully and consider assessment of renal function before OMNISCAN use.
4 Contraindications
OMNISCAN is contraindicated in patients with:
- Chronic, severe kidney disease (glomerular filtration rate, GFR < 30 mL/min/1.73m2) or acute kidney injury
- Prior hypersensitivity to OMNISCAN
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the label:
- Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2)]
- Hypersensitivity reactions [see Warnings and Precautions (5.3)]
- Acute Renal Failure [see Warnings and Precautions (5.5)]
7 Drug Interactions
Specific drug interaction studies have not been conducted.
12.2 Pharmacodynamics
In magnetic resonance imaging, visualization of normal and pathologic tissue depends in part on variations in the radiofrequency signal intensity. These variations occur due to: changes in proton density; alteration of the spin-lattice or longitudinal relaxation time (T1); and variation of the spin-spin or transverse relaxation time (T2). OMNISCAN is a paramagnetic agent with unpaired electron spins which generate a local magnetic field. As water protons move through this local magnetic field, the changes in magnetic field experienced by the protons reorient them with the main magnetic field more quickly than in the absence of a paramagnetic agent.
By increasing the relaxation rate, OMNISCAN decreases both the T1 and T2 relaxation times in tissues where it is distributed. At clinical doses, the effect is primarily on the T1 relaxation time, and produces an increase in signal intensity. Disruption of the blood-brain barrier or abnormal vascularity allows accumulation of OMNISCAN in lesions such as neoplasms, abscesses, and subacute infarcts. The pharmacokinetic parameters of OMNISCAN in various lesions are not known.
12.3 Pharmacokinetics
The pharmacokinetics of intravenously administered gadodiamide in normal subjects conforms to an open, two-compartment model with mean distribution and elimination half-lives (reported as mean ± SD) of 3.7 ± 2.7 minutes and 77.8 ± 16 minutes, respectively. Gadodiamide is eliminated primarily in the urine with 95.4 ± 5.5% (mean ± SD) of the administered dose eliminated by 24 hours. The renal and plasma clearance rates of gadodiamide are nearly identical (1.7 and 1.8 mL/min/kg, respectively), and are similar to that of substances excreted primarily by glomerular filtration. The volume of distribution of gadodiamide (200 ± 61 mL/kg) is equivalent to that of extracellular water. Gadodiamide does not bind to human serum proteins in vitro. Following GBCA administration, gadolinium is present for months or years in brain, bone, skin, and other organs [see Warnings and Precautions (5.4)]. Pharmacokinetic and pharmacodynamic studies have not been systematically conducted to determine the optimal dose and imaging time in patients with abnormal renal function or renal failure, in the elderly, or in pediatric patients with immature renal function.
2.4 Dosing Guidelines
Inspect OMNISCAN visually for particulate matter and discoloration before administration, whenever solution and container permit. Do not use the solution if it is discolored or particulate matter is present.
Draw OMNISCAN into the syringe and use immediately. Discard any unused portion of OMNISCAN Injection.
To ensure complete delivery of the desired volume of contrast medium, follow the injection of OMNISCAN with a 5 mL flush of 0.9% sodium chloride. Complete the imaging procedure within 1 hour of administration of OMNISCAN.
1 Indications and Usage
OMNISCAN is a gadolinium-based contrast agent for diagnostic magnetic resonance imaging (MRI) indicated for intravenous use to:
5.5 Acute Renal Failure
In patients with renal insufficiency, acute renal failure requiring dialysis or worsening renal function have occurred, mostly within 48 hours of OMNISCAN Injection. The risk of renal failure may increase with increasing dose of gadolinium contrast. Use the lowest necessary dose of contrast and evaluate renal function in patients with renal insufficiency. Acute renal failure was observed in < 1% of patients in OMNISCAN clinical studies [see Adverse Reactions (6)].
OMNISCAN is cleared by glomerular filtration. Hemodialysis also enhances OMNISCAN clearance [see Use in Specific Populations (8.5, 8.6)].
5.4 Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (e.g., brain, skin, kidney, liver, and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Dotarem (gadoterate meglumine), Gadavist (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2)]. There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2)].
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients [see Use in Specific Populations (8.1, 8.4)], and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies, particularly closely spaced studies, when possible.
5 Warnings and Precautions
- Hypersensitivity Reactions: Anaphylactoid and other serious hypersensitivity reactions including fatal reactions have occurred particularly in patients with history of allergy or drug reactions. Monitor patients closely for need of emergency cardiorespiratory support (5.3).
- Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs (5.4).
- Acute Renal Failure: Acute renal failure has occurred in patients with preexisting renal insufficiency. Use the lowest necessary dose of OMNISCAN and evaluate renal function in these patients (5.5).
2 Dosage and Administration
3 Dosage Forms and Strengths
Sterile aqueous solution for intravenous injection; 287 mg/mL.
5.7 Laboratory Test Findings
Asymptomatic, transitory changes in serum iron have been observed. The clinical significance is unknown.
OMNISCAN interferes with serum calcium measurements with some colorimetric (complexometric) methods commonly used in hospitals, resulting in serum calcium concentrations lower than the true values. In patients with normal renal function, this effect lasts for 12 to 24 hours. In patients with decreased renal function, the interference with calcium measurements is expected to last during the prolonged elimination of OMNISCAN. After patients receive OMNISCAN, careful attention should be used in selecting the type of method used to measure calcium.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of OMNISCAN or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute pancreatitis with onset within 48 hours after GBCA administration.
General Disorders: Nephrogenic Systemic Fibrosis (NSF)
Adverse reactions with variable onset and duration have been reported after GBCA administration. These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems.
Nervous System Disorders: Convulsions in patients with and without a history of convulsions or brain lesions.
Respiratory, Thoracic and Mediastinal Disorders: Acute respiratory distress syndrome, pulmonary edema
Renal and Urinary System Disorders: In patients with pre-existing renal insufficiency: acute renal failure, renal impairment, blood creatinine increased.
Skin: Gadolinium-associated plaques.
8.6 Renal/hepatic Impairment
Dose adjustments in renal or hepatic impairment have not been studied. Caution should be exercised in patients with impaired renal insufficiency [see Warnings and Precautions (5.2, 5.5)].
8 Use in Specific Populations
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed (8.1).
5.3 Hypersensitivity Reactions
Anaphylactoid and anaphylactic reactions, with cardiovascular, respiratory and/or cutaneous manifestations, resulting in death have occurred. Personnel trained in resuscitation techniques and resuscitation equipment should be present prior to OMNISCAN administration. If a hypersensitivity reaction occurs, stop OMNISCAN Injection and immediately begin appropriate therapy. Observe patients closely, particularly those with a history of drug reactions, asthma, allergy or other hypersensitivity disorders, during and up to several hours after OMNISCAN Injection.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.1 Cns (central Nervous System)
OMNISCAN is a gadolinium-based contrast agent indicated for intravenous use in MRI to visualize lesions with abnormal vascularity (or those thought to cause abnormalities in the blood-brain barrier) in the brain (intracranial lesions), spine, and associated tissues [see Clinical Studies (14.1)].
14.1 Cns (central Nervous System)
OMNISCAN (0.1 mmol/kg) contrast enhancement in CNS MRI was evident in a study of 439 adults. In a study of sequential dosing, 57 adults received OMNISCAN 0.1 mmol/kg followed by 0.2 mmol/kg within 20 minutes (for cumulative dose of 0.3 mmol/kg). The MRIs were compared blindly. In 54/56 (96%) patients, OMNISCAN contrast enhancement was evident with both the 0.1 mmol/kg and cumulative 0.3 mmol/kg OMNISCAN doses relative to non-contrast MRI.
In comparison to the non-contrast MRI, increased numbers of brain and spine lesions were noted in 42% of patients who received OMNISCAN at any dose. In comparisons of 0.1 mmol/kg versus 0.3 mmol/kg, the results were comparable in 25/56 (45%); in 1/56 (2%) OMNISCAN 0.1 mmol/kg dose provided more diagnostic value and in 30/56 (54%) the cumulative OMNISCAN 0.3 mmol/kg dose provided more diagnostic value. The usefulness of a single 0.3 mmol/kg bolus in comparison to the cumulative 0.3 mmol/kg (0.1 mmol/kg followed by 0.2 mmol/kg) has not been established.
OMNISCAN as a single 0.1 mmol/kg dose was evaluated in 97 pediatric patients with a mean age of 8.9 (2 to 18) years referred for CNS MRI. Postcontrast MRI provided added diagnostic information, diagnostic confidence, and new patient management information in 76%, 67%, and 52%, respectively, of pediatrics.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Patients receiving OMNISCAN should be instructed to inform their physician if they:
- are pregnant or breast feeding, or
- have a history of renal and/or liver disease, convulsions, asthma or allergic respiratory disorders, or recent administration of gadolinium-based contrast.
GBCAs increase the risk for NSF among patients with impaired elimination of the drugs. To counsel patients at risk for NSF:
- Describe the clinical manifestations of NSF
- Describe procedures to screen for the detection of renal impairment
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following OMNISCAN administration such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain deep in the hip bones or ribs; or muscle weakness.
5.2 Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of OMNISCAN among these patients unless the diagnostic information is essential and not available with non-contrast enhanced MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73m2) as well as patients with acute kidney injury. Do not administer OMNISCAN to these patients. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Report any diagnosis of NSF following OMNISCAN administration to GE HealthCare (1-800-654-0118) or FDA (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (e.g., age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and the degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. When administering a GBCA, do not exceed the recommended dose and allow a sufficient period of time for elimination of the agent prior to any readministration [see Boxed Warning, Contraindications (4), Clinical Pharmacology (12.2) and Dosage and Administration (2)].
16 How Supplied/storage and Handling
OMNISCAN (gadodiamide) Injection is a sterile, clear, colorless to slightly yellow, aqueous solution containing 287 mg/mL of gadodiamide in rubber stoppered vials. OMNISCAN is supplied in the following sizes:
10 mL vial, box of 10 (NDC 0407-0690-10)
15 mL fill in 20 mL vial, box of 10 (NDC 0407-0690-15)
20 mL vial, box of 10 (NDC 0407-0690-20)
5.1 Risk Associated With Intrathecal Use
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of OMNISCAN have not been established with intrathecal use. OMNISCAN is not approved for intrathecal use [see Dosage and Administration (2.1, 2.2)].
Principal Display Panel 10 Ml Vial Label
NDC 0407-0690-10
OMNISCAN™
(gadodiamide) Injection
10 mL 287 mg/mL
Single-Dose Vial
Sterile Aqueous Injection
Dispense the accompanying
Medication Guide to each patient
J-100
Contains
10 x 10 mL
Vials
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term animal studies have not been performed to evaluate the carcinogenic potential of gadodiamide. The results of the following genotoxicity assays were negative: in vitro bacterial reverse mutation assay, in vitro Chinese Hamster Ovary (CHO)/Hypoxanthine Guanine Phosphoribosyl Transferase (HGPT) forward mutation assay, in vitro CHO chromosome aberration assay, and the in vivo mouse micronucleus assay at intravenous doses of 27 mmol/kg (approximately 7 times the maximum human dose based on a body surface area comparison). Impairment of male or female fertility was not observed in rats after intravenous administration three times per week at the maximum dose tested of 1.0 mmol/kg (approximately 0.5 times the maximum human dose based on a body surface area comparison).
5.6 Impaired Visualization of Lesions Detectable With Non Contrast Mri
Paramagnetic contrast agents such as OMNISCAN might impair the visualization of lesions which are seen on the non-contrast MRI. This may be due to effects of the paramagnetic contrast agent, or imaging parameters. Exercise caution when OMNISCAN MRI scans are interpreted in the absence of a companion non-contrast MRI.
Warning: Risk Associated With Intrathecal Use and Nephrogenic Systemic Fibrosis
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS
See full prescribing information for complete boxed warning.
- Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. OMNISCAN is not approved for intrathecal use ( 5.1 ).
-
GBCAs increase the risk for nephrogenic systemic fibrosis among patients with impaired elimination of the drugs. Avoid use of OMNISCAN in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. Do not administer OMNISCAN to patients with:
- chronic, severe kidney disease (GFR < 30 mL/min/1.73m2), or
- acute kidney injury ( 4 ).
Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (e.g., age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing ( 5.2 ).
1.2 Body (intrathoracic [noncardiac], Intra Abdominal, Pelvic and Retroperitoneal Regions)
OMNISCAN is a gadolinium-based contrast agent indicated for intravenous use in MRI to facilitate the visualization of lesions with abnormal vascularity within the thoracic (noncardiac), abdominal, pelvic cavities, and the retroperitoneal space [see Clinical Studies (14.2)].
14.2 Body (intrathoracic [noncardiac], Intra Abdominal, Pelvic and Retroperitoneal Regions)
OMNISCAN was evaluated in a controlled trial of 276 patients referred for body MRI. These patients had a mean age of 57 (9 to 88) years. Patients received 0.1 mmol/kg OMNISCAN for imaging the thorax (noncardiac), abdomen, and pelvic organs, or a dose of 0.05 mmol/kg for imaging the kidney. Pre- and post-OMNISCAN images were evaluated blindly for the degree of diagnostic value rated on a scale of "remarkably improved, improved, no change, worse, and cannot be determined." The postcontrast results showed "remarkably improved" or "improved" diagnostic value in 90% of the thorax, liver, and pelvis patients, and in 95% of the kidney patients. In a dose ranging study 258 patients referred for body MRI received OMNISCAN 0.025, 0.05, 0.1 mmol/kg. The lowest effective dose of OMNISCAN for the kidney was 0.05 mmol/kg.
Structured Label Content
Section 42229-5 (42229-5)
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. OMNISCAN is not approved for intrathecal use [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE OMNISCAN™ (OMNI-scan) (gadodiamide) Injection, for intravenous use |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 3/2025 |
|
What is the most important information I should know about OMNISCAN?
|
|
What is OMNISCAN?
|
|
| Do not receive OMNISCAN if you have had a severe allergic reaction to OMNISCAN. | |
Before receiving OMNISCAN, tell your healthcare provider about all your medical conditions, including if you:
|
|
What are the possible side effects of OMNISCAN?
These are not all the possible side effects of OMNISCAN. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about the safe and effective use of OMNISCAN.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your healthcare provider for information about OMNISCAN that is written for health professionals. |
|
|
What are the ingredients in OMNISCAN?
Active ingredient: gadodiamide Inactive ingredients: caldiamide sodium, hydrochloric acid, sodium hydroxide © 2025 GE HealthCare Distributed by GE Healthcare Inc., Arlington Heights, IL 60004 U.S.A. GE is a trademark of General Electric Company used under trademark license. For more information, go to www.GEHealthCare.com or call 1-800-654-0118. |
Section 44425-7 (44425-7)
Protect OMNISCAN from strong daylight and direct exposure to sunlight. Do not freeze. Freezing can cause small cracks in the vials, which would compromise the sterility of the product. Do not use if the product is inadvertently frozen.
Store OMNISCAN at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP].
10 Overdosage (10 OVERDOSAGE)
Clinical consequences of overdose with OMNISCAN have not been reported. The minimum lethal dose of intravenously administered OMNISCAN in rats and mice is greater than 20 mmol/kg (200 times the recommended human dose of 0.1 mmol/kg; 67 times the cumulative 0.3 mmol/kg dose). OMNISCAN is dialyzable.
11 Description (11 DESCRIPTION)
OMNISCAN (gadodiamide) Injection is the formulation of the gadolinium complex of diethylenetriamine pentaacetic acid bismethylamide, and is an injectable, nonionic extracellular enhancing agent for magnetic resonance imaging. OMNISCAN is administered by intravenous injection.
OMNISCAN is provided as a sterile, clear, colorless to slightly yellow, aqueous solution. Each 1 mL contains 287 mg gadodiamide and 12 mg caldiamide sodium in Water for Injection. The pH is adjusted between 5.5 and 7.0 with hydrochloric acid and/or sodium hydroxide. OMNISCAN contains no antimicrobial preservative. OMNISCAN is a 0.5 mol/L solution of aqua[5,8-bis(carboxymethyl)-11-[2-(methylamino)-2-oxoethyl]-3-oxo-2,5,8,11-tetraazatridecan-13-oato (3-)-N5, N8, N11, O3, O5, O8, O11, O13] gadolinium hydrate, with a molecular weight of 573.66 (anhydrous), an empirical formula of C16H28GdN5O9∙xH2O, and the following structural formula:
Pertinent physicochemical data for OMNISCAN are noted below:
| Osmolality (mOsmol/kg water) | @ 37°C | 789 |
| Viscosity (cP) | @ 20°C | 2 |
| @ 37°C | 1.4 | |
| Density (g/mL) | @ 25°C | 1.14 |
| Specific gravity | @ 25°C | 1.15 |
OMNISCAN has an osmolality approximately 2.8 times that of plasma at 37°C and is hypertonic under conditions of use.
2.3 Dosage Chart
| BODY WEIGHT | PEDIATRIC | ADULTS | |||
|---|---|---|---|---|---|
| 0.05 | 0.1 | 0.05 | 0.1 | ||
| kg | lb | (mmol/kg) | (mmol/kg) | ||
| VOLUME (mL) | VOLUME (mL) | ||||
| 12 | 26 | 1.2 | 2.4 | - | - |
| 14 | 31 | 1.4 | 2.8 | - | - |
| 16 | 35 | 1.6 | 3.2 | - | - |
| 18 | 40 | 1.8 | 3.6 | - | - |
| 20 | 44 | 2 | 4 | - | - |
| 22 | 48 | 2.2 | 4.4 | - | - |
| 24 | 53 | 2.4 | 4.8 | - | - |
| 26 | 57 | 2.6 | 5.2 | - | - |
| 28 | 62 | 2.8 | 5.6 | - | - |
| 30 | 66 | 3 | 6 | - | - |
| 40 | 88 | 4 | 8 | 4 | 8 |
| 50 | 110 | 5 | 10 | 5 | 10 |
| 60 | 132 | 6 | 12 | 6 | 12 |
| 70 | 154 | 7 | 14 | 7 | 14 |
| 80 | 176 | 8 | 16 | 8 | 16 |
| 90 | 198 | - | - | 9 | 18 |
| 100 | 220 | - | - | 10 | 20 |
| 110 | 242 | - | - | 11 | 22 |
| 120 | 264 | - | - | 12 | 24 |
| 130 The heaviest patient in clinical studies weighed 136 kg.
|
286 | - | - | 13 | 26 |
8.4 Pediatric Use
The safety and efficacy of OMNISCAN at a single dose of 0.05 to 0.1 mmol/kg have been established in pediatric patients over 2 years of age based on adequate and well controlled studies of OMNISCAN in adults, a pediatric CNS imaging study, and safety data in the scientific literature. However, the safety and efficacy of doses greater than 0.1 mmol/kg and of repeated doses have not been studied in pediatric patients.
Pharmacokinetics of OMNISCAN have not been studied in pediatrics. The glomerular filtration rate of neonates and infants is much lower than that of adults. The pharmacokinetics volume of distribution is also different. Therefore, the optimal dosing regimen and imaging times in patients under 2 years of age have not been established.
8.5 Geriatric Use
In clinical studies of OMNISCAN, 243 patients were between 65 and 80 years of age while 15 were over 80 years of age. No overall differences in safety or effectiveness were observed between these patients and younger patients. Other reported clinical experience has not identified differences in response between the elderly and younger patients, but greater sensitivity in the elderly cannot be ruled out. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
OMNISCAN is excreted by the kidney, and the risk of toxic reactions to OMNISCAN may be greater in patients with impaired renal function [see Warnings and Precautions (5.4)]. Because elderly patients are more likely to have decreased renal function, select dose carefully and consider assessment of renal function before OMNISCAN use.
4 Contraindications (4 CONTRAINDICATIONS)
OMNISCAN is contraindicated in patients with:
- Chronic, severe kidney disease (glomerular filtration rate, GFR < 30 mL/min/1.73m2) or acute kidney injury
- Prior hypersensitivity to OMNISCAN
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the label:
- Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2)]
- Hypersensitivity reactions [see Warnings and Precautions (5.3)]
- Acute Renal Failure [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Specific drug interaction studies have not been conducted.
12.2 Pharmacodynamics
In magnetic resonance imaging, visualization of normal and pathologic tissue depends in part on variations in the radiofrequency signal intensity. These variations occur due to: changes in proton density; alteration of the spin-lattice or longitudinal relaxation time (T1); and variation of the spin-spin or transverse relaxation time (T2). OMNISCAN is a paramagnetic agent with unpaired electron spins which generate a local magnetic field. As water protons move through this local magnetic field, the changes in magnetic field experienced by the protons reorient them with the main magnetic field more quickly than in the absence of a paramagnetic agent.
By increasing the relaxation rate, OMNISCAN decreases both the T1 and T2 relaxation times in tissues where it is distributed. At clinical doses, the effect is primarily on the T1 relaxation time, and produces an increase in signal intensity. Disruption of the blood-brain barrier or abnormal vascularity allows accumulation of OMNISCAN in lesions such as neoplasms, abscesses, and subacute infarcts. The pharmacokinetic parameters of OMNISCAN in various lesions are not known.
12.3 Pharmacokinetics
The pharmacokinetics of intravenously administered gadodiamide in normal subjects conforms to an open, two-compartment model with mean distribution and elimination half-lives (reported as mean ± SD) of 3.7 ± 2.7 minutes and 77.8 ± 16 minutes, respectively. Gadodiamide is eliminated primarily in the urine with 95.4 ± 5.5% (mean ± SD) of the administered dose eliminated by 24 hours. The renal and plasma clearance rates of gadodiamide are nearly identical (1.7 and 1.8 mL/min/kg, respectively), and are similar to that of substances excreted primarily by glomerular filtration. The volume of distribution of gadodiamide (200 ± 61 mL/kg) is equivalent to that of extracellular water. Gadodiamide does not bind to human serum proteins in vitro. Following GBCA administration, gadolinium is present for months or years in brain, bone, skin, and other organs [see Warnings and Precautions (5.4)]. Pharmacokinetic and pharmacodynamic studies have not been systematically conducted to determine the optimal dose and imaging time in patients with abnormal renal function or renal failure, in the elderly, or in pediatric patients with immature renal function.
2.4 Dosing Guidelines
Inspect OMNISCAN visually for particulate matter and discoloration before administration, whenever solution and container permit. Do not use the solution if it is discolored or particulate matter is present.
Draw OMNISCAN into the syringe and use immediately. Discard any unused portion of OMNISCAN Injection.
To ensure complete delivery of the desired volume of contrast medium, follow the injection of OMNISCAN with a 5 mL flush of 0.9% sodium chloride. Complete the imaging procedure within 1 hour of administration of OMNISCAN.
1 Indications and Usage (1 INDICATIONS AND USAGE)
OMNISCAN is a gadolinium-based contrast agent for diagnostic magnetic resonance imaging (MRI) indicated for intravenous use to:
5.5 Acute Renal Failure
In patients with renal insufficiency, acute renal failure requiring dialysis or worsening renal function have occurred, mostly within 48 hours of OMNISCAN Injection. The risk of renal failure may increase with increasing dose of gadolinium contrast. Use the lowest necessary dose of contrast and evaluate renal function in patients with renal insufficiency. Acute renal failure was observed in < 1% of patients in OMNISCAN clinical studies [see Adverse Reactions (6)].
OMNISCAN is cleared by glomerular filtration. Hemodialysis also enhances OMNISCAN clearance [see Use in Specific Populations (8.5, 8.6)].
5.4 Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (e.g., brain, skin, kidney, liver, and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Dotarem (gadoterate meglumine), Gadavist (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2)]. There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2)].
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients [see Use in Specific Populations (8.1, 8.4)], and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies, particularly closely spaced studies, when possible.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Anaphylactoid and other serious hypersensitivity reactions including fatal reactions have occurred particularly in patients with history of allergy or drug reactions. Monitor patients closely for need of emergency cardiorespiratory support (5.3).
- Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs (5.4).
- Acute Renal Failure: Acute renal failure has occurred in patients with preexisting renal insufficiency. Use the lowest necessary dose of OMNISCAN and evaluate renal function in these patients (5.5).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Sterile aqueous solution for intravenous injection; 287 mg/mL.
5.7 Laboratory Test Findings
Asymptomatic, transitory changes in serum iron have been observed. The clinical significance is unknown.
OMNISCAN interferes with serum calcium measurements with some colorimetric (complexometric) methods commonly used in hospitals, resulting in serum calcium concentrations lower than the true values. In patients with normal renal function, this effect lasts for 12 to 24 hours. In patients with decreased renal function, the interference with calcium measurements is expected to last during the prolonged elimination of OMNISCAN. After patients receive OMNISCAN, careful attention should be used in selecting the type of method used to measure calcium.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of OMNISCAN or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute pancreatitis with onset within 48 hours after GBCA administration.
General Disorders: Nephrogenic Systemic Fibrosis (NSF)
Adverse reactions with variable onset and duration have been reported after GBCA administration. These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems.
Nervous System Disorders: Convulsions in patients with and without a history of convulsions or brain lesions.
Respiratory, Thoracic and Mediastinal Disorders: Acute respiratory distress syndrome, pulmonary edema
Renal and Urinary System Disorders: In patients with pre-existing renal insufficiency: acute renal failure, renal impairment, blood creatinine increased.
Skin: Gadolinium-associated plaques.
8.6 Renal/hepatic Impairment (8.6 Renal/Hepatic Impairment)
Dose adjustments in renal or hepatic impairment have not been studied. Caution should be exercised in patients with impaired renal insufficiency [see Warnings and Precautions (5.2, 5.5)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed (8.1).
5.3 Hypersensitivity Reactions
Anaphylactoid and anaphylactic reactions, with cardiovascular, respiratory and/or cutaneous manifestations, resulting in death have occurred. Personnel trained in resuscitation techniques and resuscitation equipment should be present prior to OMNISCAN administration. If a hypersensitivity reaction occurs, stop OMNISCAN Injection and immediately begin appropriate therapy. Observe patients closely, particularly those with a history of drug reactions, asthma, allergy or other hypersensitivity disorders, during and up to several hours after OMNISCAN Injection.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.1 Cns (central Nervous System) (1.1 CNS (Central Nervous System))
OMNISCAN is a gadolinium-based contrast agent indicated for intravenous use in MRI to visualize lesions with abnormal vascularity (or those thought to cause abnormalities in the blood-brain barrier) in the brain (intracranial lesions), spine, and associated tissues [see Clinical Studies (14.1)].
14.1 Cns (central Nervous System) (14.1 CNS (Central Nervous System))
OMNISCAN (0.1 mmol/kg) contrast enhancement in CNS MRI was evident in a study of 439 adults. In a study of sequential dosing, 57 adults received OMNISCAN 0.1 mmol/kg followed by 0.2 mmol/kg within 20 minutes (for cumulative dose of 0.3 mmol/kg). The MRIs were compared blindly. In 54/56 (96%) patients, OMNISCAN contrast enhancement was evident with both the 0.1 mmol/kg and cumulative 0.3 mmol/kg OMNISCAN doses relative to non-contrast MRI.
In comparison to the non-contrast MRI, increased numbers of brain and spine lesions were noted in 42% of patients who received OMNISCAN at any dose. In comparisons of 0.1 mmol/kg versus 0.3 mmol/kg, the results were comparable in 25/56 (45%); in 1/56 (2%) OMNISCAN 0.1 mmol/kg dose provided more diagnostic value and in 30/56 (54%) the cumulative OMNISCAN 0.3 mmol/kg dose provided more diagnostic value. The usefulness of a single 0.3 mmol/kg bolus in comparison to the cumulative 0.3 mmol/kg (0.1 mmol/kg followed by 0.2 mmol/kg) has not been established.
OMNISCAN as a single 0.1 mmol/kg dose was evaluated in 97 pediatric patients with a mean age of 8.9 (2 to 18) years referred for CNS MRI. Postcontrast MRI provided added diagnostic information, diagnostic confidence, and new patient management information in 76%, 67%, and 52%, respectively, of pediatrics.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Patients receiving OMNISCAN should be instructed to inform their physician if they:
- are pregnant or breast feeding, or
- have a history of renal and/or liver disease, convulsions, asthma or allergic respiratory disorders, or recent administration of gadolinium-based contrast.
GBCAs increase the risk for NSF among patients with impaired elimination of the drugs. To counsel patients at risk for NSF:
- Describe the clinical manifestations of NSF
- Describe procedures to screen for the detection of renal impairment
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following OMNISCAN administration such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain deep in the hip bones or ribs; or muscle weakness.
5.2 Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of OMNISCAN among these patients unless the diagnostic information is essential and not available with non-contrast enhanced MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73m2) as well as patients with acute kidney injury. Do not administer OMNISCAN to these patients. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Report any diagnosis of NSF following OMNISCAN administration to GE HealthCare (1-800-654-0118) or FDA (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (e.g., age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and the degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. When administering a GBCA, do not exceed the recommended dose and allow a sufficient period of time for elimination of the agent prior to any readministration [see Boxed Warning, Contraindications (4), Clinical Pharmacology (12.2) and Dosage and Administration (2)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
OMNISCAN (gadodiamide) Injection is a sterile, clear, colorless to slightly yellow, aqueous solution containing 287 mg/mL of gadodiamide in rubber stoppered vials. OMNISCAN is supplied in the following sizes:
10 mL vial, box of 10 (NDC 0407-0690-10)
15 mL fill in 20 mL vial, box of 10 (NDC 0407-0690-15)
20 mL vial, box of 10 (NDC 0407-0690-20)
5.1 Risk Associated With Intrathecal Use (5.1 Risk Associated with Intrathecal Use)
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of OMNISCAN have not been established with intrathecal use. OMNISCAN is not approved for intrathecal use [see Dosage and Administration (2.1, 2.2)].
Principal Display Panel 10 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 10 mL Vial Label)
NDC 0407-0690-10
OMNISCAN™
(gadodiamide) Injection
10 mL 287 mg/mL
Single-Dose Vial
Sterile Aqueous Injection
Dispense the accompanying
Medication Guide to each patient
J-100
Contains
10 x 10 mL
Vials
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term animal studies have not been performed to evaluate the carcinogenic potential of gadodiamide. The results of the following genotoxicity assays were negative: in vitro bacterial reverse mutation assay, in vitro Chinese Hamster Ovary (CHO)/Hypoxanthine Guanine Phosphoribosyl Transferase (HGPT) forward mutation assay, in vitro CHO chromosome aberration assay, and the in vivo mouse micronucleus assay at intravenous doses of 27 mmol/kg (approximately 7 times the maximum human dose based on a body surface area comparison). Impairment of male or female fertility was not observed in rats after intravenous administration three times per week at the maximum dose tested of 1.0 mmol/kg (approximately 0.5 times the maximum human dose based on a body surface area comparison).
5.6 Impaired Visualization of Lesions Detectable With Non Contrast Mri (5.6 Impaired Visualization of Lesions Detectable with Non-contrast MRI)
Paramagnetic contrast agents such as OMNISCAN might impair the visualization of lesions which are seen on the non-contrast MRI. This may be due to effects of the paramagnetic contrast agent, or imaging parameters. Exercise caution when OMNISCAN MRI scans are interpreted in the absence of a companion non-contrast MRI.
Warning: Risk Associated With Intrathecal Use and Nephrogenic Systemic Fibrosis (WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS)
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS
See full prescribing information for complete boxed warning.
- Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. OMNISCAN is not approved for intrathecal use ( 5.1 ).
-
GBCAs increase the risk for nephrogenic systemic fibrosis among patients with impaired elimination of the drugs. Avoid use of OMNISCAN in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. Do not administer OMNISCAN to patients with:
- chronic, severe kidney disease (GFR < 30 mL/min/1.73m2), or
- acute kidney injury ( 4 ).
Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (e.g., age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing ( 5.2 ).
1.2 Body (intrathoracic [noncardiac], Intra Abdominal, Pelvic and Retroperitoneal Regions) (1.2 Body (Intrathoracic [noncardiac], Intra-abdominal, Pelvic and Retroperitoneal Regions))
OMNISCAN is a gadolinium-based contrast agent indicated for intravenous use in MRI to facilitate the visualization of lesions with abnormal vascularity within the thoracic (noncardiac), abdominal, pelvic cavities, and the retroperitoneal space [see Clinical Studies (14.2)].
14.2 Body (intrathoracic [noncardiac], Intra Abdominal, Pelvic and Retroperitoneal Regions) (14.2 Body (Intrathoracic [noncardiac], Intra-abdominal, Pelvic and Retroperitoneal Regions))
OMNISCAN was evaluated in a controlled trial of 276 patients referred for body MRI. These patients had a mean age of 57 (9 to 88) years. Patients received 0.1 mmol/kg OMNISCAN for imaging the thorax (noncardiac), abdomen, and pelvic organs, or a dose of 0.05 mmol/kg for imaging the kidney. Pre- and post-OMNISCAN images were evaluated blindly for the degree of diagnostic value rated on a scale of "remarkably improved, improved, no change, worse, and cannot be determined." The postcontrast results showed "remarkably improved" or "improved" diagnostic value in 90% of the thorax, liver, and pelvis patients, and in 95% of the kidney patients. In a dose ranging study 258 patients referred for body MRI received OMNISCAN 0.025, 0.05, 0.1 mmol/kg. The lowest effective dose of OMNISCAN for the kidney was 0.05 mmol/kg.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:49.483703 · Updated: 2026-03-14T22:25:41.517877