ff4df6c5-c85f-406a-89d7-8c2853fd5e10
34390-5
HUMAN OTC DRUG LABEL
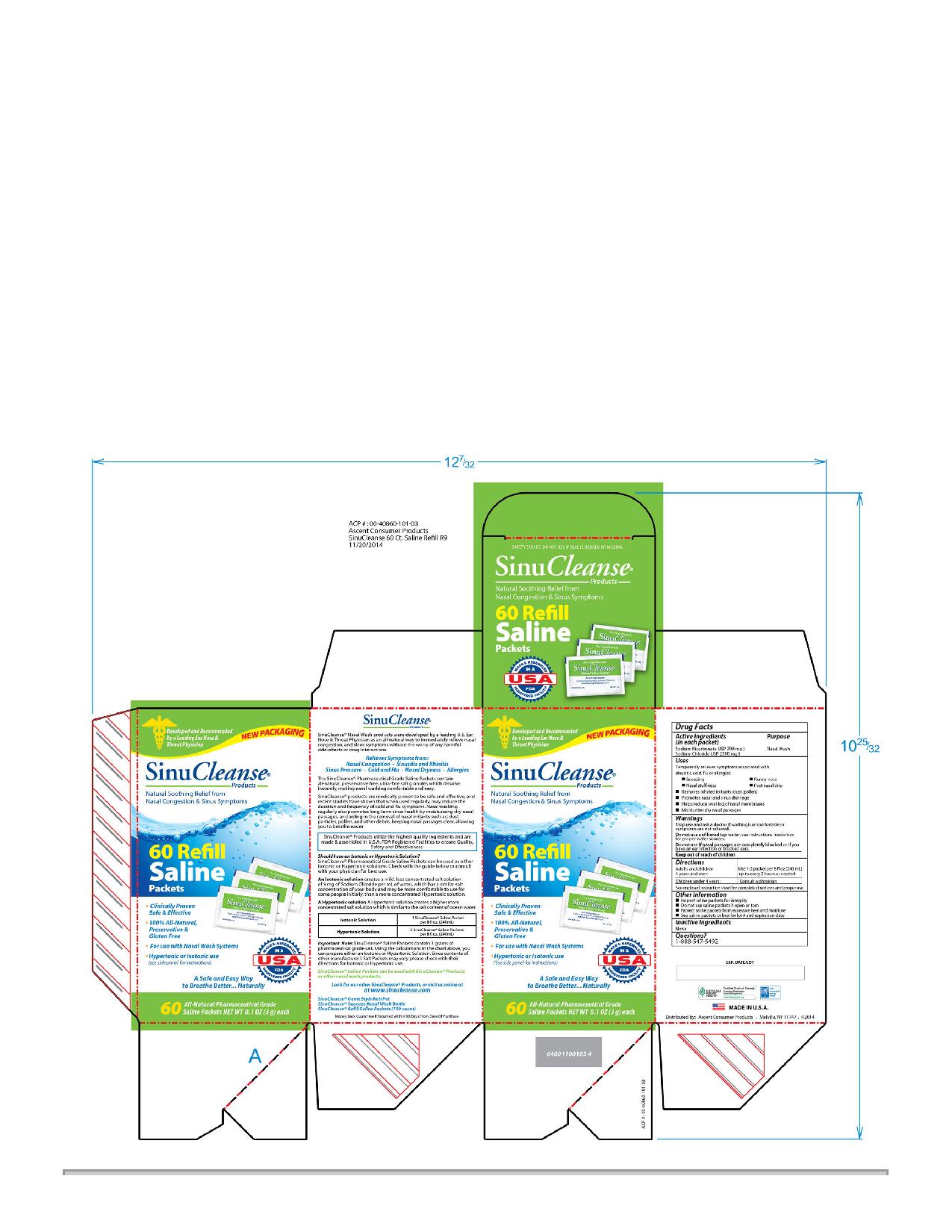
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients (in each packet) Sodium Bicarbonate USP 700 mg Sodium Chloride USP 2300 mg
Medication Information
Warnings and Precautions
Warnings
Stop use and ask a doctor if washing is uncomfortable or symptoms
are not relieved.
Do not use unfiltered tap water, see instructions inside box
for proper water sources.
Do not use if nasal passages are completely blocked or if you have
an ear infection or blocked ears.
Indications and Usage
Uses
Temporarily relieves symptoms associated with
sinusitis, cold, flu or allergies:
-Sneezing
-Runny nose
-Nasal stuffiness
-Post nasal drip
-Removes inhaled irritants (dust, pollen)
-Promotes nasal and sinus drainage
-Helps reduce swelling of nasal membranes
-Moisturizes dry nasal passages
Dosage and Administration
Directions
Adults and children 4 years and over: use 1/2 - 1 packet per 4 fl oz (120 mL)up to every two hours as needed
Children under 4 years: Consult a physician
See enclosed instruction sheet for complete directions and proper use
Description
Active Ingredients (in each packet) Sodium Bicarbonate USP 700 mg Sodium Chloride USP 2300 mg
Section 50565-1
Keep out of reach of children
Section 50567-7
When using this product:
-Use by only one person
-Wash with soap and water after each use
-Top rack of dishwasher safe
-Do not heat in microwave
Section 51727-6
Inactive Ingredients
None
Section 51945-4
Section 53413-1
Questions?
1-888-547-5492
Section 55105-1
Purpose
Nasal Wash
Section 55106-9
Active Ingredients
(in each packet)
Sodium Bicarbonate USP 700 mg
Sodium Chloride USP 2300 mg
Section 60561-8
Other Information
-Inspect saline packets for integrity
-Do not use saline packets if open or torn
-Protect saline packets from excessive heat and moisture
-See saline packets or box for lot # and expiration date
Structured Label Content
Indications and Usage (34067-9)
Uses
Temporarily relieves symptoms associated with
sinusitis, cold, flu or allergies:
-Sneezing
-Runny nose
-Nasal stuffiness
-Post nasal drip
-Removes inhaled irritants (dust, pollen)
-Promotes nasal and sinus drainage
-Helps reduce swelling of nasal membranes
-Moisturizes dry nasal passages
Dosage and Administration (34068-7)
Directions
Adults and children 4 years and over: use 1/2 - 1 packet per 4 fl oz (120 mL)up to every two hours as needed
Children under 4 years: Consult a physician
See enclosed instruction sheet for complete directions and proper use
Warnings and Precautions (34071-1)
Warnings
Stop use and ask a doctor if washing is uncomfortable or symptoms
are not relieved.
Do not use unfiltered tap water, see instructions inside box
for proper water sources.
Do not use if nasal passages are completely blocked or if you have
an ear infection or blocked ears.
Section 50565-1 (50565-1)
Keep out of reach of children
Section 50567-7 (50567-7)
When using this product:
-Use by only one person
-Wash with soap and water after each use
-Top rack of dishwasher safe
-Do not heat in microwave
Section 51727-6 (51727-6)
Inactive Ingredients
None
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions?
1-888-547-5492
Section 55105-1 (55105-1)
Purpose
Nasal Wash
Section 55106-9 (55106-9)
Active Ingredients
(in each packet)
Sodium Bicarbonate USP 700 mg
Sodium Chloride USP 2300 mg
Section 60561-8 (60561-8)
Other Information
-Inspect saline packets for integrity
-Do not use saline packets if open or torn
-Protect saline packets from excessive heat and moisture
-See saline packets or box for lot # and expiration date
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:27.645544 · Updated: 2026-03-14T23:07:19.548615