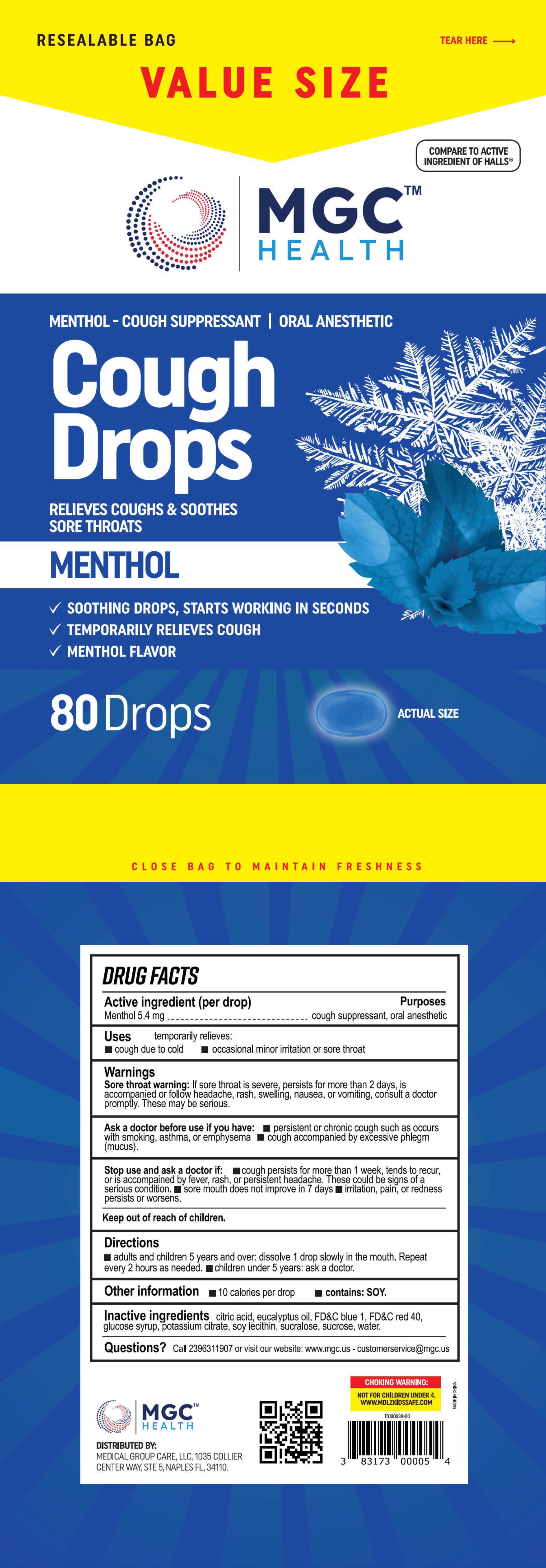
Cough Drops
ff3fa2fa-6741-fa43-e053-6294a90a5d5b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cough Drops
Medication Information
Warnings and Precautions
Sore throat warning: lf sore throat is severe, persists for more than 2 days, isaccompanied or follow headache, rash, swelling, nausea, or vomiting, consult a doctorprompfly. These may be serious.
Indications and Usage
temporarily relieves: cough due to cold, occasional minor irritation or sore throat
Dosage and Administration
adults and children 5 years and over: dissolve 1 drop slowly in the mouth. Repeatevery 2 hours as needed.
children under 5 years: ask a doctor.
Description
Cough Drops
Section 42229-5
Cough Drops
Section 43683-2
Section 44425-7
Dry, avoid sunlight
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor if: cough persists for more than 1 week, tends to recur,
or is accompained by fever, rash, or persistent headache. These could be signs of a
serious condition. sore mouth does not improve in 7 days irritation, pain, or redness
persists or worsens.
Section 50567-7
adults and children 5 years and over: dissolve 1 drop slowly in the mouth. Repeatevery 2 hours as needed.
children under 5 years: ask a doctor.
Section 50570-1
Ask a doctor before use if you have:persistent or chronic cough such as occurs with smoking, asthma, or emphysema
cough accompanied by excessive phlegm (mucus).
Stop use and ask a doctor if: cough persists for more than 1 week, tends to recur,
or is accompained by fever, rash, or persistent headache. These could be signs of a
serious condition. sore mouth does not improve in 7 days
irritation, pain, or redness persists or worsens.
Section 51727-6
EUCALYPTUS OIL
FD&C BLUE NO. 1
FD&C RED NO. 40
POTASSIUM CITRATE
SOY LECITHIN
SUCRALOSE
SUCROSE
GLUCOSE SYRUP
CITRIC ACID
Water
Section 51945-4
Section 55105-1
cough suppressant, oral anesthetic
Section 55106-9
Menthol
Structured Label Content
Indications and Usage (34067-9)
temporarily relieves: cough due to cold, occasional minor irritation or sore throat
Dosage and Administration (34068-7)
adults and children 5 years and over: dissolve 1 drop slowly in the mouth. Repeatevery 2 hours as needed.
children under 5 years: ask a doctor.
Warnings and Precautions (34071-1)
Sore throat warning: lf sore throat is severe, persists for more than 2 days, isaccompanied or follow headache, rash, swelling, nausea, or vomiting, consult a doctorprompfly. These may be serious.
Section 42229-5 (42229-5)
Cough Drops
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Dry, avoid sunlight
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor if: cough persists for more than 1 week, tends to recur,
or is accompained by fever, rash, or persistent headache. These could be signs of a
serious condition. sore mouth does not improve in 7 days irritation, pain, or redness
persists or worsens.
Section 50567-7 (50567-7)
adults and children 5 years and over: dissolve 1 drop slowly in the mouth. Repeatevery 2 hours as needed.
children under 5 years: ask a doctor.
Section 50570-1 (50570-1)
Ask a doctor before use if you have:persistent or chronic cough such as occurs with smoking, asthma, or emphysema
cough accompanied by excessive phlegm (mucus).
Stop use and ask a doctor if: cough persists for more than 1 week, tends to recur,
or is accompained by fever, rash, or persistent headache. These could be signs of a
serious condition. sore mouth does not improve in 7 days
irritation, pain, or redness persists or worsens.
Section 51727-6 (51727-6)
EUCALYPTUS OIL
FD&C BLUE NO. 1
FD&C RED NO. 40
POTASSIUM CITRATE
SOY LECITHIN
SUCRALOSE
SUCROSE
GLUCOSE SYRUP
CITRIC ACID
Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
cough suppressant, oral anesthetic
Section 55106-9 (55106-9)
Menthol
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:01:42.648225 · Updated: 2026-03-14T23:01:30.191020