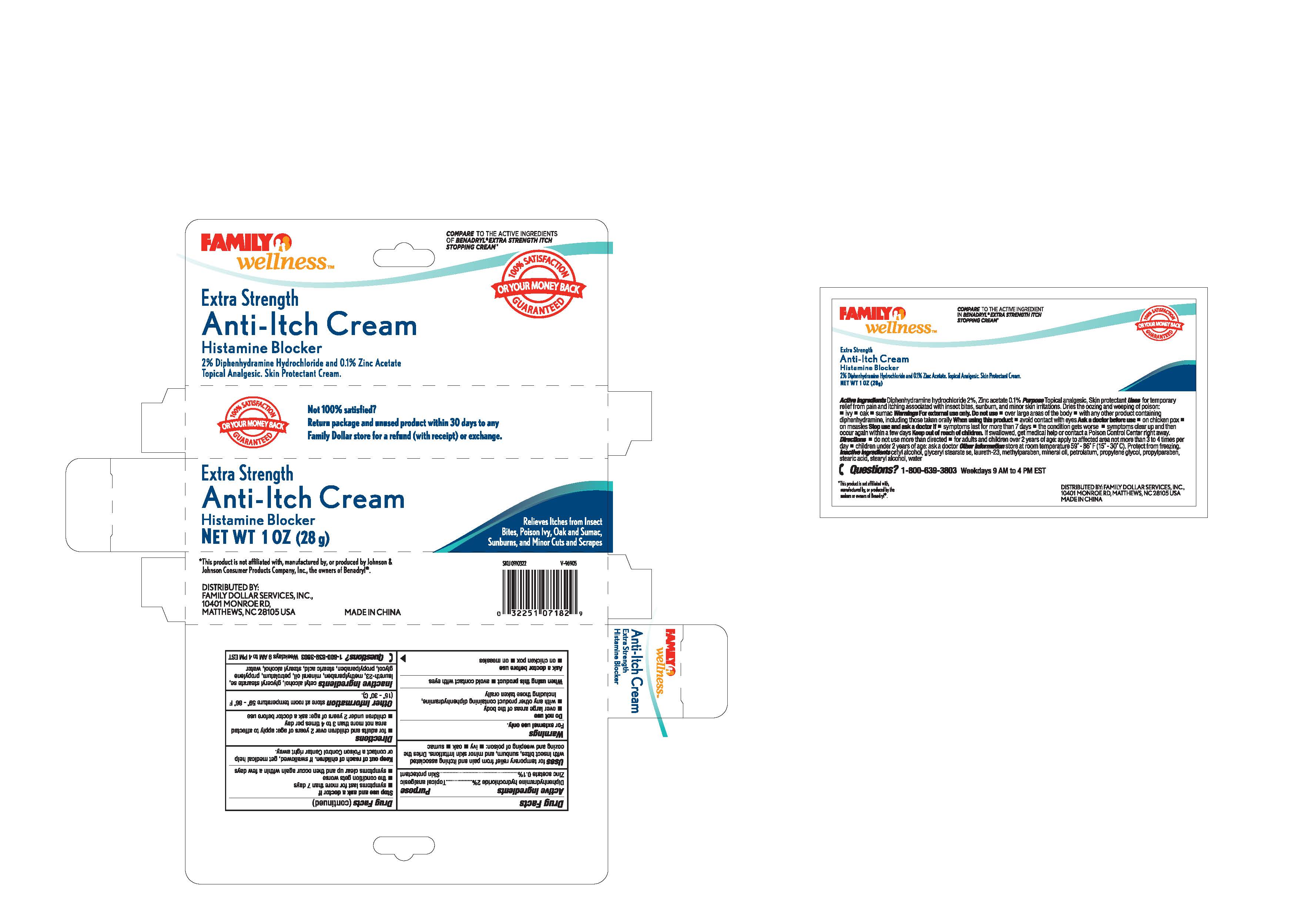
Drug Facts
fefbe351-1cd9-4a2f-a43d-51e2c4eb180b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Inactive Ingredients - cetyl alcohol, glycerol stearate, laureth-23, methylparaben, mineral oil, petrolatum, propylene glycol, propylparaben, stearic acid, stearyl alcohol, water
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Uses for temporary relief from pain and itching associated
with insect bites, sunburn and minor skin irritations. Dries the
oozing and weeping of poison: -ivy -oak - sumac
Dosage and Administration
Directions
-for adults and children over 2 years of age: apply to affected area
not more than 3 or 4 times per day
-children under 2 years of age: ask a doctor before use
Description
Inactive Ingredients - cetyl alcohol, glycerol stearate, laureth-23, methylparaben, mineral oil, petrolatum, propylene glycol, propylparaben, stearic acid, stearyl alcohol, water
Section 44425-7
Other information - store at room temperature 59 - 86 F (15 - 30 C)
Section 50565-1
Keep out of reach of children. If swallowed, get medical help
or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if:
-symptoms last for more than 7 days
-the condition gets worse
-symptoms clear up and then occur again within a few days
Section 50567-7
When using this product - avoid contact with eyes
Section 50569-3
Ask a doctor before use
-on chicken pox
-on measles
Section 50570-1
Do not use
-over large areas of the body
-with any other product containing diphenhydramine,
including those taken orally
Section 51727-6
Inactive Ingredients - cetyl alcohol, glycerol stearate,
laureth-23, methylparaben, mineral oil, petrolatum, propylene
glycol, propylparaben, stearic acid, stearyl alcohol, water
Section 51945-4
Section 53413-1
Questions? 1-800-639-3803 Weekdays 9 AM to 4 PM EST
Section 55105-1
Purpose
---Topical analgesic
---Skin protectant
Section 55106-9
Active Ingredients
Diphenhydramine hydrochloride 2 %
Zinc acetate 0.1 %
Structured Label Content
Indications and Usage (34067-9)
Uses for temporary relief from pain and itching associated
with insect bites, sunburn and minor skin irritations. Dries the
oozing and weeping of poison: -ivy -oak - sumac
Dosage and Administration (34068-7)
Directions
-for adults and children over 2 years of age: apply to affected area
not more than 3 or 4 times per day
-children under 2 years of age: ask a doctor before use
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 44425-7 (44425-7)
Other information - store at room temperature 59 - 86 F (15 - 30 C)
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help
or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if:
-symptoms last for more than 7 days
-the condition gets worse
-symptoms clear up and then occur again within a few days
Section 50567-7 (50567-7)
When using this product - avoid contact with eyes
Section 50569-3 (50569-3)
Ask a doctor before use
-on chicken pox
-on measles
Section 50570-1 (50570-1)
Do not use
-over large areas of the body
-with any other product containing diphenhydramine,
including those taken orally
Section 51727-6 (51727-6)
Inactive Ingredients - cetyl alcohol, glycerol stearate,
laureth-23, methylparaben, mineral oil, petrolatum, propylene
glycol, propylparaben, stearic acid, stearyl alcohol, water
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions? 1-800-639-3803 Weekdays 9 AM to 4 PM EST
Section 55105-1 (55105-1)
Purpose
---Topical analgesic
---Skin protectant
Section 55106-9 (55106-9)
Active Ingredients
Diphenhydramine hydrochloride 2 %
Zinc acetate 0.1 %
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:01:42.645748 · Updated: 2026-03-14T23:01:30.191020