Soothe Preservative Free Drug Facts
fef002ea-c4bd-4486-a42e-1bbd9d73d28d
34390-5
HUMAN OTC DRUG LABEL
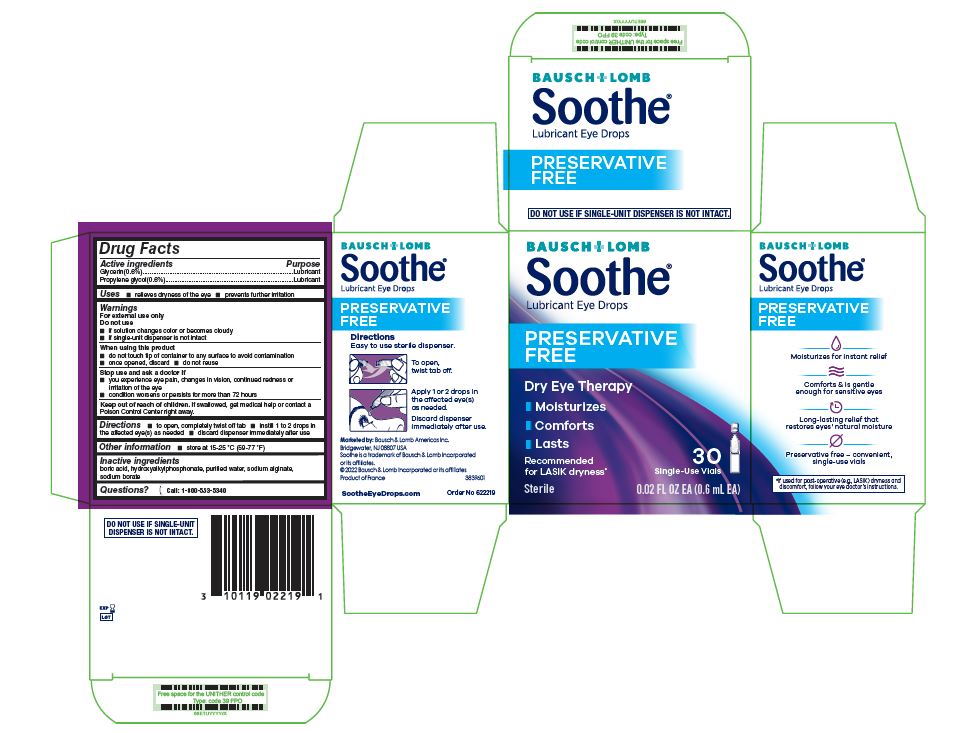
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Glycerin (0.6%) Propylene glycol (0.6%)
Purpose
Lubricant Lubricant
Medication Information
Purpose
Lubricant
Lubricant
Description
Glycerin (0.6%) Propylene glycol (0.6%)
Uses
- relieves dryness of the eye
- prevents further irritation
Warnings
For external use only
Do not use
- if solution changes color or becomes cloudy
- if single-unit dispenser is not intact
Directions
- to open, completely twist off tab
- instill 1 to 2 drops in the affected eye(s) as needed
- discard dispenser immediately after use
Questions?
[phone icon] Call: 1-800-553-5340
Marketed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Soothe is a trademark of|
Bausch & Lomb Incorporated
or its affiliates.
© 2022 Bausch & Lomb
Incorporated or its affiliates
Product of France
Other Information
- store at 15 – 25 °C (59 – 77 °F)
Active Ingredients
Glycerin (0.6%)
Propylene glycol (0.6%)
Inactive Ingredients
boric acid, hydroxyalkylphosphonate, purified water, sodium alginate, sodium borate
When Using This Product
- do not touch tip of container to any surface to avoid contamination
- once opened, discard
- do not reuse
Stop Use and Ask A Doctor If
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel
Bausch & Lomb
Soothe®
Lubricant Eye Drops
PRESERVATIVE FREE
Dry Eye Therapy
- Moisturizes
- Comforts
- Lasts
- Recommended
for LASIK dryness* - 30 Single-Use Vials
Sterile 0.02 FL OZ EA (0.6 mL EA)
3839601
SootheEyeDrops.com
Order No. 622219
Structured Label Content
Uses
- relieves dryness of the eye
- prevents further irritation
Purpose
Lubricant
Lubricant
Warnings
For external use only
Do not use
- if solution changes color or becomes cloudy
- if single-unit dispenser is not intact
Directions
- to open, completely twist off tab
- instill 1 to 2 drops in the affected eye(s) as needed
- discard dispenser immediately after use
Questions?
[phone icon] Call: 1-800-553-5340
Marketed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Soothe is a trademark of|
Bausch & Lomb Incorporated
or its affiliates.
© 2022 Bausch & Lomb
Incorporated or its affiliates
Product of France
Other Information (Other information)
- store at 15 – 25 °C (59 – 77 °F)
Active Ingredients (Active ingredients)
Glycerin (0.6%)
Propylene glycol (0.6%)
Inactive Ingredients (Inactive ingredients)
boric acid, hydroxyalkylphosphonate, purified water, sodium alginate, sodium borate
When Using This Product (When using this product)
- do not touch tip of container to any surface to avoid contamination
- once opened, discard
- do not reuse
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel (Package/Label Principal Display Panel)
Bausch & Lomb
Soothe®
Lubricant Eye Drops
PRESERVATIVE FREE
Dry Eye Therapy
- Moisturizes
- Comforts
- Lasts
- Recommended
for LASIK dryness* - 30 Single-Use Vials
Sterile 0.02 FL OZ EA (0.6 mL EA)
3839601
SootheEyeDrops.com
Order No. 622219
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:36.601010 · Updated: 2026-03-14T23:03:47.044278