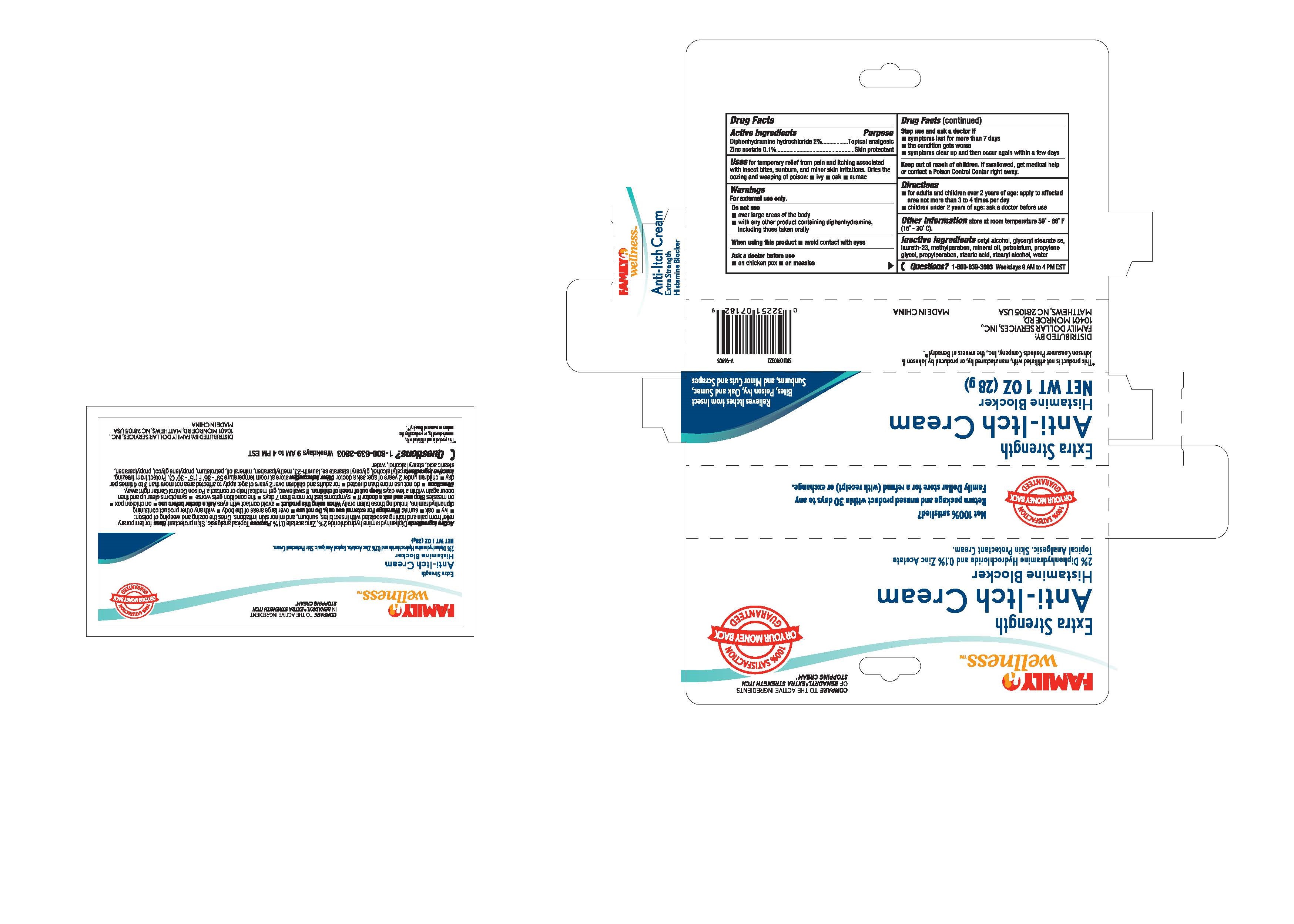
Family Wellness Anti-itch Cream, 28g
fde2069b-344e-46da-bb95-c5113e0f35d6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Diphenhydramine hydrochloride 2% Zinc acetate 0.1%
Medication Information
Warnings and Precautions
Warnings
. For external use only
Do not use
•avoid contact with eyes When using this product
Ask a doctor before use
•on chicken pox •on measles
Stop use and ask a doctor if
- over large areas of the body
- with any other product containing diphenhydramine including those taken orally
- symptoms last for more than 7 days
- the condition gets worse
- symptoms clear up and then occur again within a few days
Indications and Usage
for temprary relief from pain and itching associated Uses
with insect bites , sunburn and mior skin irritations. Dries the
oozing and weeping of poison: •ivy •oak •sumac
Dosage and Administration
Directions
•for adults and children over 2 years of age: apply to affected
area not more than 3 to 4 times daily
•children under 2 years of age: ask a doctor before use
Description
Active Ingredients Diphenhydramine hydrochloride 2% Zinc acetate 0.1%
Label
Section 50565-1
. If swallowed, get medical help Keep out of reach of children
or contact a Poison Control Center right away.
Section 51727-6
cetyl alcohol, glyceryl stearate se, Inactive ingredients
laureth-23, methylparaben, mineral oil, petrolatum, propylene
glycol, propylparaben, stearic acid, stearic alcohol, water
Section 53413-1
1-800-639-3803 Weekdays 9 AM to 4 PM EST Questions?
Section 55105-1
Purpose
Topical analgesic
Skin protectant
Section 55106-9
Active Ingredients
Diphenhydramine hydrochloride 2%
Zinc acetate 0.1%
Section 60561-8
store at room temperature 59° - 86° F Other information
(15° - 30° C).
Structured Label Content
Label (label)
Indications and Usage (34067-9)
for temprary relief from pain and itching associated Uses
with insect bites , sunburn and mior skin irritations. Dries the
oozing and weeping of poison: •ivy •oak •sumac
Dosage and Administration (34068-7)
Directions
•for adults and children over 2 years of age: apply to affected
area not more than 3 to 4 times daily
•children under 2 years of age: ask a doctor before use
Warnings and Precautions (34071-1)
Warnings
. For external use only
Do not use
•avoid contact with eyes When using this product
Ask a doctor before use
•on chicken pox •on measles
Stop use and ask a doctor if
- over large areas of the body
- with any other product containing diphenhydramine including those taken orally
- symptoms last for more than 7 days
- the condition gets worse
- symptoms clear up and then occur again within a few days
Section 50565-1 (50565-1)
. If swallowed, get medical help Keep out of reach of children
or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
cetyl alcohol, glyceryl stearate se, Inactive ingredients
laureth-23, methylparaben, mineral oil, petrolatum, propylene
glycol, propylparaben, stearic acid, stearic alcohol, water
Section 53413-1 (53413-1)
1-800-639-3803 Weekdays 9 AM to 4 PM EST Questions?
Section 55105-1 (55105-1)
Purpose
Topical analgesic
Skin protectant
Section 55106-9 (55106-9)
Active Ingredients
Diphenhydramine hydrochloride 2%
Zinc acetate 0.1%
Section 60561-8 (60561-8)
store at room temperature 59° - 86° F Other information
(15° - 30° C).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:09.301410 · Updated: 2026-03-14T23:01:33.532889