allergy relief
fdd3f9a0-b4be-498a-b8e0-1a9740399736
34390-5
HUMAN OTC DRUG LABEL
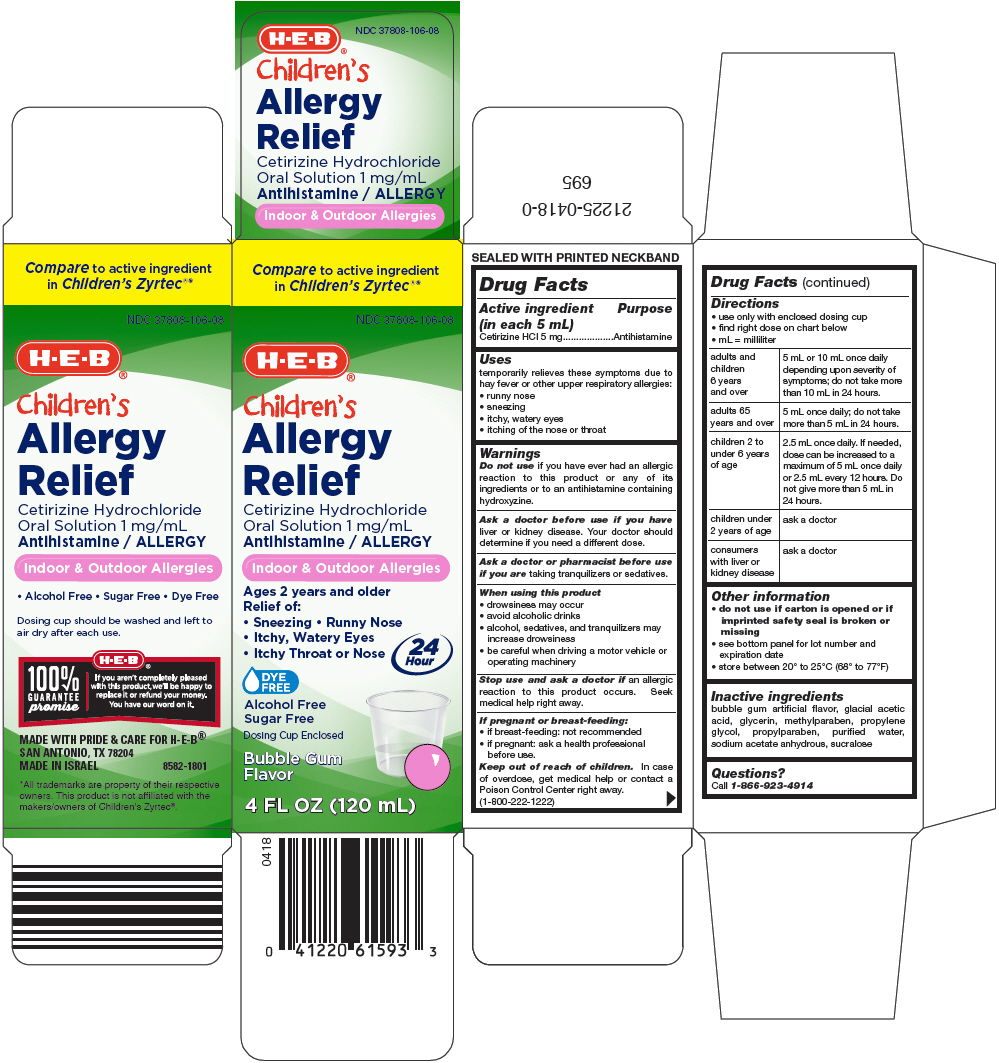
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antihistamine
Description
Drug Facts
Medication Information
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Purpose
Antihistamine
Directions
- use only with enclosed dosing cup
- find right dose on chart below
- mL = milliliter
| adults and children 6 years and over | 5 mL or 10 mL once daily depending upon severity of symptoms; do not take more than 10 mL in 24 hours. |
| adults 65 years and over | 5 mL once daily; do not take more than 5 mL in 24 hours. |
| children 2 to under 6 years of age | 2.5 mL once daily. If needed, dose can be increased to a maximum of 5 mL once daily or 2.5 mL every 12 hours. Do not give more than 5 mL in 24 hours. |
| children under 2 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information
- do not use if carton is opened or if imprinted safety seal is broken or missing
- see bottom panel for lot number and expiration date
- store between 20° to 25°C (68° to 77°F)
Inactive Ingredients
bubble gum artificial flavor, glacial acetic acid, glycerin, methylparaben, propylene glycol, propylparaben, purified water, sodium acetate anhydrous, sucralose
Description
Drug Facts
Questions?
Call 1-866-923-4914
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Section 50566-9
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Section 50568-5
Ask a doctor or pharmacist before use if you are taking tranquilizers or sedatives.
Section 50569-3
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 50570-1
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
When Using This Product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
If Pregnant Or Breast Feeding
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Active Ingredient (in Each 5 Ml)
Cetirizine HCl 5 mg
Principal Display Panel 120 Ml Bottle Carton
Compare to active ingredient
in Children's Zyrtec
®
*
NDC 37808-106-08
H-E-B ®
Children's
Allergy
Relief
Cetirizine Hydrochloride
Oral Solution 1 mg/mL
Antihistamine / ALLERGY
Indoor & Outdoor Allergies
Ages 2 years and older
Relief of:
- Sneezing • Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
24
Hour
DYE
FREE
Alcohol Free
Sugar Free
Dosing Cup Enclosed
Bubble Gum
Flavor
4 FL OZ (120 mL)
Structured Label Content
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Purpose
Antihistamine
Directions
- use only with enclosed dosing cup
- find right dose on chart below
- mL = milliliter
| adults and children 6 years and over | 5 mL or 10 mL once daily depending upon severity of symptoms; do not take more than 10 mL in 24 hours. |
| adults 65 years and over | 5 mL once daily; do not take more than 5 mL in 24 hours. |
| children 2 to under 6 years of age | 2.5 mL once daily. If needed, dose can be increased to a maximum of 5 mL once daily or 2.5 mL every 12 hours. Do not give more than 5 mL in 24 hours. |
| children under 2 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information (Other information)
- do not use if carton is opened or if imprinted safety seal is broken or missing
- see bottom panel for lot number and expiration date
- store between 20° to 25°C (68° to 77°F)
Inactive Ingredients (Inactive ingredients)
bubble gum artificial flavor, glacial acetic acid, glycerin, methylparaben, propylene glycol, propylparaben, purified water, sodium acetate anhydrous, sucralose
Questions?
Call 1-866-923-4914
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Section 50566-9 (50566-9)
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking tranquilizers or sedatives.
Section 50569-3 (50569-3)
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 50570-1 (50570-1)
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
When Using This Product (When using this product)
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Active Ingredient (in Each 5 Ml) (Active ingredient (in each 5 mL))
Cetirizine HCl 5 mg
Principal Display Panel 120 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 120 mL Bottle Carton)
Compare to active ingredient
in Children's Zyrtec
®
*
NDC 37808-106-08
H-E-B ®
Children's
Allergy
Relief
Cetirizine Hydrochloride
Oral Solution 1 mg/mL
Antihistamine / ALLERGY
Indoor & Outdoor Allergies
Ages 2 years and older
Relief of:
- Sneezing • Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
24
Hour
DYE
FREE
Alcohol Free
Sugar Free
Dosing Cup Enclosed
Bubble Gum
Flavor
4 FL OZ (120 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:34.178583 · Updated: 2026-03-14T22:55:07.187033