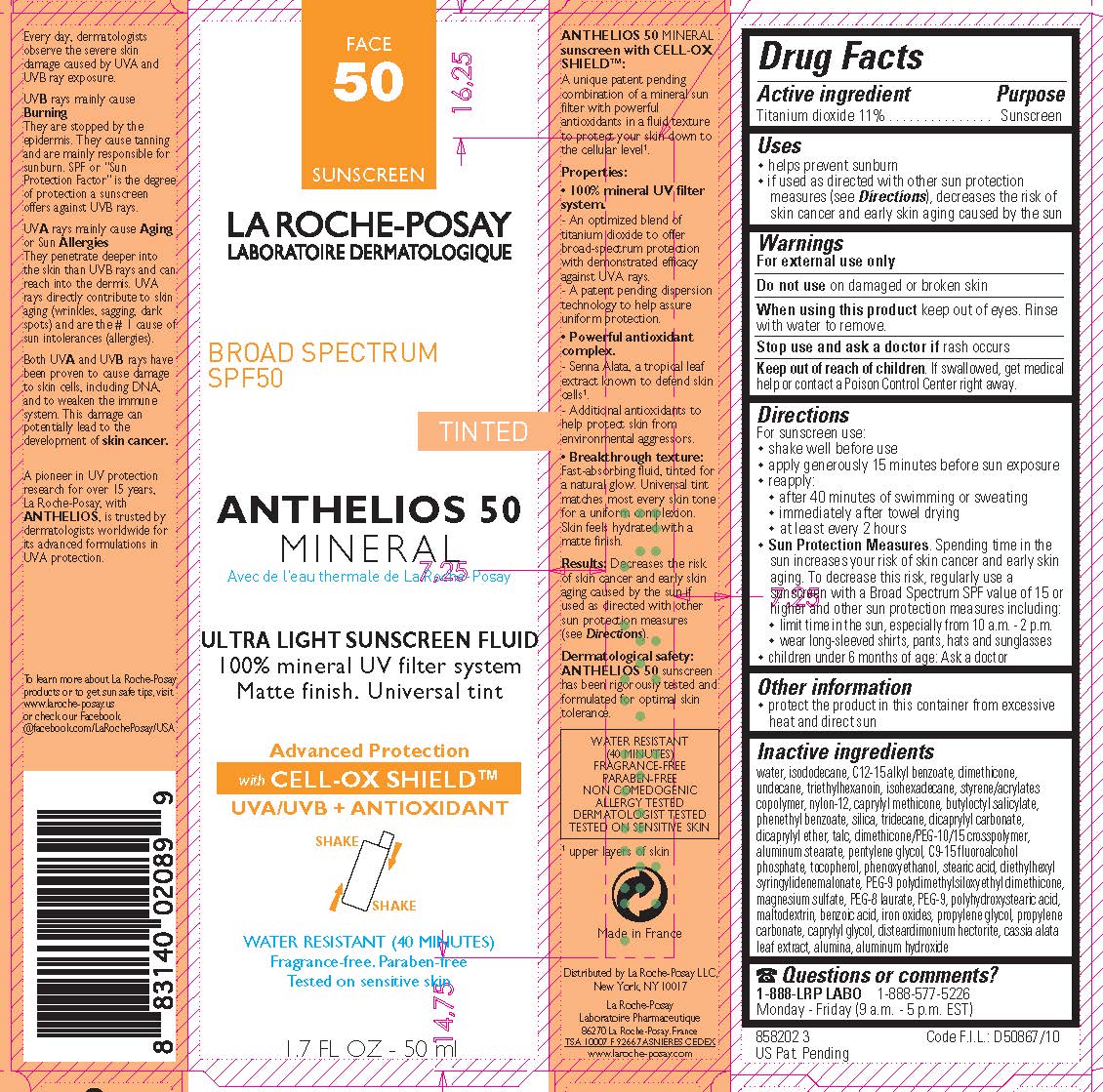
Drug Facts
fd2541e1-c409-40a8-9e3f-6ff69084ea66
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Titanium dioxide 11%
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
Titanium dioxide 11%
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Section 51945-4
Warnings
For external use only
Directions
For sunscreen use:
- shake well before use
- apply generously 15 minutes before sun exposure
- reapply:
- after 40 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection meausures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: Ask a doctor
Do Not Use
on damaged or broken skin
Active Ingredient
Titanium dioxide 11%
Other Information
protect the product in this container from excessive heat and direct sun
Inactive Ingredients
water, isododecane, C12-15 alkyl benzoate, dimethicone, undecane, triethylhexanoin, isohexadecane, styrene/acrylates copolymer, nylon-12, caprylyl methicone, butyloctyl salicylate, phenethyl benzoate, silica, tridecane, dicaprylyl carbonate, dicaprylyl ether, talc, dimethicone/PEG-10/15 crosspolymer, aluminum stearate, pentylene glycol, C9-15 fluoroalcohol phosphate, tocopherol, phenoxyethanol, stearic acid, diethylhexyl syringylidenemalonate, PEG-9 polydimethylsiloxyethyl dimethcone, magnesium sulfate, PEG-8 laurate, PEG-9, polyhydroxystearic acid, maltodextrin, benzoic acid, iron oxides, propylene glycol, propylene carbonate, caprylyl glycol, disteardimonim hectorite, cassia alata leaf extract, alumina, aluminum hydroxide
Questions Or Comments?
1-888-577-5226 1-888-LRP LABO
Monday - Friday (9 a.m. - 5 p.m. EST)
When Using This Product
keep out of eyes. Rinse with water to remove.
Stop Use and Ask A Doctor If
rash occurs
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Section 51945-4 (51945-4)
Purpose
Sunscreen
Warnings
For external use only
Directions
For sunscreen use:
- shake well before use
- apply generously 15 minutes before sun exposure
- reapply:
- after 40 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection meausures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: Ask a doctor
Do Not Use (Do not use)
on damaged or broken skin
Active Ingredient (Active ingredient)
Titanium dioxide 11%
Other Information (Other information)
protect the product in this container from excessive heat and direct sun
Inactive Ingredients (Inactive ingredients)
water, isododecane, C12-15 alkyl benzoate, dimethicone, undecane, triethylhexanoin, isohexadecane, styrene/acrylates copolymer, nylon-12, caprylyl methicone, butyloctyl salicylate, phenethyl benzoate, silica, tridecane, dicaprylyl carbonate, dicaprylyl ether, talc, dimethicone/PEG-10/15 crosspolymer, aluminum stearate, pentylene glycol, C9-15 fluoroalcohol phosphate, tocopherol, phenoxyethanol, stearic acid, diethylhexyl syringylidenemalonate, PEG-9 polydimethylsiloxyethyl dimethcone, magnesium sulfate, PEG-8 laurate, PEG-9, polyhydroxystearic acid, maltodextrin, benzoic acid, iron oxides, propylene glycol, propylene carbonate, caprylyl glycol, disteardimonim hectorite, cassia alata leaf extract, alumina, aluminum hydroxide
Questions Or Comments? (Questions or comments?)
1-888-577-5226 1-888-LRP LABO
Monday - Friday (9 a.m. - 5 p.m. EST)
When Using This Product (When using this product)
keep out of eyes. Rinse with water to remove.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
rash occurs
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:42.470188 · Updated: 2026-03-14T23:02:24.469183