These Highlights Do Not Include All The Information Needed To Use Kerendia Safely And Effectively. See Full Prescribing Information For Kerendia.
fc726765-5d5a-4d6e-b037-b847bda9fb7c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 7/2025 Dosage and Administration ( 2.3 ) 7/2025 Contraindications ( 4 ) 7/2025 Warnings and Precautions ( 5.2 ) 7/2025
Indications and Usage
Kerendia is indicated to reduce the risk of: sustained estimated glomerular filtration rate (eGFR) decline, end-stage kidney disease, cardiovascular death, non-fatal myocardial infarction, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) associated with type 2 diabetes (T2DM). cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in adult patients with heart failure with left ventricular ejection fraction (LVEF) ≥ 40%.
Dosage and Administration
The recommended starting dosage is 10 mg or 20 mg orally once daily based on eGFR and serum potassium thresholds. ( 2.1 ) Increase dosage after 4 weeks to the target dose of 20 mg once daily for CKD and T2DM based on eGFR and serum potassium thresholds. ( 2.3 ) Increase dosage after 4 weeks to the target dose of 20 mg or 40 mg once daily for HF with LVEF ≥ 40% based on eGFR and serum potassium thresholds. ( 2.3 ) Tablets may be taken with or without food ( 2.2 )
Warnings and Precautions
Hyperkalemia. Patients with decreased kidney function and higher baseline potassium levels are at increased risk. Monitor serum potassium levels and adjust dose as needed. ( 2.1 , 2.2 , 2.3 , 5.1 ) Worsening of Renal Function in Patients with Heart Failure. Measure eGFR and adjust dose as needed. ( 2.1 , 2.3 , 6.1 )
Contraindications
Kerendia is contraindicated in patients: Who are hypersensitive to any component of this product [see Adverse Reactions (6.2) ] . Who are receiving concomitant treatment with strong CYP3A4 inhibitors [see Drug Interactions (7.1) ]. With adrenal insufficiency.
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Hyperkalemia [see Warnings and Precautions (5.1) ]
Drug Interactions
Strong CYP3A4 Inhibitors: Use is contraindicated. ( 7.1 ) Grapefruit or grapefruit juice: Avoid concomitant use. ( 7.1 ) Moderate or weak CYP3A4 Inhibitors: Monitor serum potassium during drug initiation or dosage adjustment of either Kerendia or the moderate or weak CYP3A4 inhibitor, and adjust Kerendia dosage as appropriate ( 7.1 ) Strong or moderate CYP3A4 Inducers: Avoid concomitant use. ( 7.1 ) Sensitive CYP2C8 substrates at Kerendia 40 mg: Monitor more frequently for adverse reactions. ( 7.2 )
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions are permitted from 15°C to 30°C (59°F to 86°F ) [see USP Controlled Room Temperature].
How Supplied
Kerendia is available as a film-coated tablet in three strengths. The 10 mg is a pink oblong tablet with "FI" on one side of tablet and "10" on the other side of tablet. The 20 mg tablet is a yellow oblong tablet with "FI" on one side of tablet and "20" on the other side of tablet. The 40 mg tablet is a gray-orange oblong tablet with "FI" on one side of tablet and "40" on the other side of tablet. Kerendia Tablets 10 mg Bottle of 30 tablets NDC 50419-540-01 Bottle of 90 tablets NDC 50419-540-02 Hospital Blister Pack of 30 tablets NDC 50419-540-06 Kerendia Tablets 20 mg Bottle of 30 tablets NDC 50419-541-01 Bottle of 90 tablets NDC 50419-541-02 Hospital Blister Pack of 30 tablets NDC 50419-541-06 Kerendia Tablets 40 mg Bottle of 30 tablets NDC 50419-542-01 Bottle of 90 tablets NDC 50419-542-02 Hospital Blister Pack of 30 tablets NDC 50419-542-06
Medication Information
Warnings and Precautions
Hyperkalemia. Patients with decreased kidney function and higher baseline potassium levels are at increased risk. Monitor serum potassium levels and adjust dose as needed. ( 2.1 , 2.2 , 2.3 , 5.1 ) Worsening of Renal Function in Patients with Heart Failure. Measure eGFR and adjust dose as needed. ( 2.1 , 2.3 , 6.1 )
Indications and Usage
Kerendia is indicated to reduce the risk of: sustained estimated glomerular filtration rate (eGFR) decline, end-stage kidney disease, cardiovascular death, non-fatal myocardial infarction, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) associated with type 2 diabetes (T2DM). cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in adult patients with heart failure with left ventricular ejection fraction (LVEF) ≥ 40%.
Dosage and Administration
The recommended starting dosage is 10 mg or 20 mg orally once daily based on eGFR and serum potassium thresholds. ( 2.1 ) Increase dosage after 4 weeks to the target dose of 20 mg once daily for CKD and T2DM based on eGFR and serum potassium thresholds. ( 2.3 ) Increase dosage after 4 weeks to the target dose of 20 mg or 40 mg once daily for HF with LVEF ≥ 40% based on eGFR and serum potassium thresholds. ( 2.3 ) Tablets may be taken with or without food ( 2.2 )
Contraindications
Kerendia is contraindicated in patients: Who are hypersensitive to any component of this product [see Adverse Reactions (6.2) ] . Who are receiving concomitant treatment with strong CYP3A4 inhibitors [see Drug Interactions (7.1) ]. With adrenal insufficiency.
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Hyperkalemia [see Warnings and Precautions (5.1) ]
Drug Interactions
Strong CYP3A4 Inhibitors: Use is contraindicated. ( 7.1 ) Grapefruit or grapefruit juice: Avoid concomitant use. ( 7.1 ) Moderate or weak CYP3A4 Inhibitors: Monitor serum potassium during drug initiation or dosage adjustment of either Kerendia or the moderate or weak CYP3A4 inhibitor, and adjust Kerendia dosage as appropriate ( 7.1 ) Strong or moderate CYP3A4 Inducers: Avoid concomitant use. ( 7.1 ) Sensitive CYP2C8 substrates at Kerendia 40 mg: Monitor more frequently for adverse reactions. ( 7.2 )
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions are permitted from 15°C to 30°C (59°F to 86°F ) [see USP Controlled Room Temperature].
How Supplied
Kerendia is available as a film-coated tablet in three strengths. The 10 mg is a pink oblong tablet with "FI" on one side of tablet and "10" on the other side of tablet. The 20 mg tablet is a yellow oblong tablet with "FI" on one side of tablet and "20" on the other side of tablet. The 40 mg tablet is a gray-orange oblong tablet with "FI" on one side of tablet and "40" on the other side of tablet. Kerendia Tablets 10 mg Bottle of 30 tablets NDC 50419-540-01 Bottle of 90 tablets NDC 50419-540-02 Hospital Blister Pack of 30 tablets NDC 50419-540-06 Kerendia Tablets 20 mg Bottle of 30 tablets NDC 50419-541-01 Bottle of 90 tablets NDC 50419-541-02 Hospital Blister Pack of 30 tablets NDC 50419-541-06 Kerendia Tablets 40 mg Bottle of 30 tablets NDC 50419-542-01 Bottle of 90 tablets NDC 50419-542-02 Hospital Blister Pack of 30 tablets NDC 50419-542-06
Description
Indications and Usage ( 1 ) 7/2025 Dosage and Administration ( 2.3 ) 7/2025 Contraindications ( 4 ) 7/2025 Warnings and Precautions ( 5.2 ) 7/2025
Section 42229-5
CKD associated with T2DM
The target daily dose of Kerendia is 20 mg orally.
Measure serum potassium 4 weeks after initiating treatment and adjust dose (see Table 2); if serum potassium levels are > 4.8 to 5.0 mEq/L, initiation of Kerendia treatment may be considered with additional serum potassium monitoring within the first 4 weeks based on clinical judgment and serum potassium levels [see Warnings and Precautions (5.1)]. Measure serum potassium 4 weeks after a dose adjustment and periodically throughout treatment, and adjust the dose as needed (see Table 2) [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
| Current Kerendia Dose | |||
|---|---|---|---|
| 10 mg once daily | 20 mg once daily | ||
| Current Serum Potassium (mEq/L) | ≤ 4.8 | Increase the dose to 20 mg once daily. If eGFR has decreased by more than 30% compared to previous measurement, maintain 10 mg dose.
|
Maintain 20 mg once daily. |
| > 4.8 – 5.5 | Maintain 10 mg once daily. | Maintain 20 mg once daily. | |
| > 5.5 | Withhold Kerendia. Consider restarting at 10 mg once daily when serum potassium ≤ 5.0 mEq/L. |
Withhold Kerendia. Restart at 10 mg once daily when serum potassium ≤ 5.0 mEq/L. |
Section 43683-2
10 Overdosage
In the event of suspected overdose, immediately interrupt Kerendia treatment. The most likely manifestation of overdose is hyperkalemia. If hyperkalemia develops, standard treatment should be initiated.
Finerenone is unlikely to be efficiently removed by hemodialysis given its fraction bound to plasma proteins of about 90%.
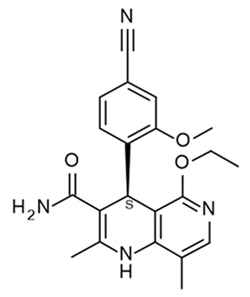
11 Description
Kerendia contains finerenone, a nonsteroidal mineralocorticoid receptor antagonist. Finerenone's chemical name is (4S)-4-(4-cyano-2- methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1,6-naphthyridine-3-carboxamide. The molecular formula is C21H22N4O3 and the molecular weight is 378.43 g/mol. The structural formula is:
Finerenone is a white to yellow crystalline powder. It is practically insoluble in water; and sparingly soluble in 0.1 M HCl, ethanol, and acetone.
Each Kerendia tablet contains 10 mg, 20 mg, or 40 mg of finerenone. The inactive ingredients of Kerendia are lactose monohydrate, cellulose microcrystalline, croscarmellose sodium, hypromellose, magnesium stearate, and sodium lauryl sulfate. The film coating contains hypromellose, titanium dioxide and talc, in addition to ferric oxide red (10 mg and 40 mg strength tablets) or ferric oxide yellow (20 mg and 40 mg strength tablets).
2.4 Missed Doses
Direct a patient to take a missed dose as soon as possible after it is noticed, but only on the same day. If this is not possible, the patient should skip the dose and continue with the next dose as prescribed.
5.1 Hyperkalemia
Kerendia can cause hyperkalemia [see Adverse Reactions (6.1)].
The risk for developing hyperkalemia increases with decreasing kidney function and is greater in patients with higher baseline potassium levels or other risk factors for hyperkalemia. Measure serum potassium and eGFR in all patients before initiation of treatment with Kerendia and dose accordingly [see Dosage and Administration (2.1)]. Do not initiate Kerendia if serum potassium is > 5.0 mEq/L.
Measure serum potassium periodically during treatment with Kerendia and adjust dose accordingly [see Dosage and Administration (2.3)]. More frequent monitoring may be necessary for patients at risk for hyperkalemia, including those on concomitant medications that impair potassium excretion or increase serum potassium [see Drug Interactions (7.1, 7.2)].
16.1 How Supplied
Kerendia is available as a film-coated tablet in three strengths. The 10 mg is a pink oblong tablet with "FI" on one side of tablet and "10" on the other side of tablet. The 20 mg tablet is a yellow oblong tablet with "FI" on one side of tablet and "20" on the other side of tablet. The 40 mg tablet is a gray-orange oblong tablet with "FI" on one side of tablet and "40" on the other side of tablet.
| Kerendia Tablets 10 mg | |
| Bottle of 30 tablets | NDC 50419-540-01 |
| Bottle of 90 tablets | NDC 50419-540-02 |
| Hospital Blister Pack of 30 tablets | NDC 50419-540-06 |
| Kerendia Tablets 20 mg | |
| Bottle of 30 tablets | NDC 50419-541-01 |
| Bottle of 90 tablets | NDC 50419-541-02 |
| Hospital Blister Pack of 30 tablets | NDC 50419-541-06 |
| Kerendia Tablets 40 mg | |
| Bottle of 30 tablets | NDC 50419-542-01 |
| Bottle of 90 tablets | NDC 50419-542-02 |
| Hospital Blister Pack of 30 tablets | NDC 50419-542-06 |
8.4 Pediatric Use
The safety and efficacy of Kerendia have not been established in patients below 18 years of age.
8.5 Geriatric Use
Of the 6510 patients who received Kerendia in the FIDELIO-DKD and FIGARO-DKD studies, 55% of patients were 65 years and older, and 14% were 75 years and older. Of the 2993 patients who received Kerendia in the FINEARTS study, 79% of patients were 65 years and older, and 43% were 75 years and older. No overall differences in safety or efficacy were observed between these patients and younger patients. No dose adjustment is required.
4 Contraindications
Kerendia is contraindicated in patients:
- Who are hypersensitive to any component of this product [see Adverse Reactions (6.2)].
- Who are receiving concomitant treatment with strong CYP3A4 inhibitors [see Drug Interactions (7.1)].
- With adrenal insufficiency.
6 Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling:
- Hyperkalemia [see Warnings and Precautions (5.1)]
7 Drug Interactions
- Strong CYP3A4 Inhibitors: Use is contraindicated. (7.1)
- Grapefruit or grapefruit juice: Avoid concomitant use. (7.1)
- Moderate or weak CYP3A4 Inhibitors: Monitor serum potassium during drug initiation or dosage adjustment of either Kerendia or the moderate or weak CYP3A4 inhibitor, and adjust Kerendia dosage as appropriate (7.1)
- Strong or moderate CYP3A4 Inducers: Avoid concomitant use. (7.1)
- Sensitive CYP2C8 substrates at Kerendia 40 mg: Monitor more frequently for adverse reactions. (7.2)
12.2 Pharmacodynamics
In FIDELIO-DKD and FIGARO-DKD, randomized, double-blind, placebo-controlled, multicenter studies in adult patients with chronic kidney disease associated with type 2 diabetes, the placebo-corrected relative reduction in urinary albumin-to-creatinine ratio (UACR) in patients randomized to finerenone was 31% (95% CI 29-34%), and 32% (95% CI 30-35%) respectively at Month 4 and remained stable for the duration of the trial.
In ARTS DN, a randomized, double-blind, placebo-controlled, multicenter phase IIb dose finding study in adults with CKD and T2DM, the placebo-corrected relative reduction in UACR at Day 90 was 25% and 38% in patients treated with finerenone 10 mg and 20 mg once daily, respectively. In patients treated with Kerendia, the mean systolic blood pressure decreased by 3 mmHg and the mean diastolic blood pressure decreased by 1-2 mmHg at month 1, remaining stable thereafter.
12.3 Pharmacokinetics
Finerenone exposure increased proportionally over a dose range of 1.25 to 80 mg (0.06 to 4 times the maximum approved recommended dosage). Steady state of finerenone was achieved after 2 days of dosing. The estimated steady-state geometric mean Cmax,md was 166 µg/L and steady-state geometric mean AUCτ,md was 718 µg.h/L following administration of finerenone 20 mg to patients.
8.6 Hepatic Impairment
Avoid use of Kerendia in patients with severe hepatic impairment (Child Pugh C).
No dosage adjustment is recommended in patients with mild or moderate hepatic impairment (Child Pugh A or B).
Consider additional serum potassium monitoring in patients with moderate hepatic impairment (Child Pugh B) [see Dosing and Administration (2.3) and Clinical Pharmacology (12.3)].
1 Indications and Usage
Kerendia is indicated to reduce the risk of:
- sustained estimated glomerular filtration rate (eGFR) decline, end-stage kidney disease, cardiovascular death, non-fatal myocardial infarction, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) associated with type 2 diabetes (T2DM).
- cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in adult patients with heart failure with left ventricular ejection fraction (LVEF) ≥ 40%.
12.1 Mechanism of Action
Finerenone is a nonsteroidal, selective antagonist of the mineralocorticoid receptor (MR), which is activated by aldosterone and cortisol and regulates gene transcription. Finerenone blocks MR mediated sodium reabsorption and MR overactivation in both epithelial (e.g., kidney) and nonepithelial (e.g., heart, and blood vessels) tissues. MR overactivation is thought to contribute to fibrosis and inflammation. Finerenone has a high potency and selectivity for the MR and has no relevant affinity for androgen, progesterone, estrogen, and glucocorticoid receptors.
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions are permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5 Warnings and Precautions
- Hyperkalemia. Patients with decreased kidney function and higher baseline potassium levels are at increased risk. Monitor serum potassium levels and adjust dose as needed. (2.1, 2.2, 2.3, 5.1)
- Worsening of Renal Function in Patients with Heart Failure. Measure eGFR and adjust dose as needed. (2.1, 2.3, 6.1)
2 Dosage and Administration
- The recommended starting dosage is 10 mg or 20 mg orally once daily based on eGFR and serum potassium thresholds. (2.1)
- Increase dosage after 4 weeks to the target dose of 20 mg once daily for CKD and T2DM based on eGFR and serum potassium thresholds. (2.3)
- Increase dosage after 4 weeks to the target dose of 20 mg or 40 mg once daily for HF with LVEF ≥ 40% based on eGFR and serum potassium thresholds. (2.3)
- Tablets may be taken with or without food (2.2)
3 Dosage Forms and Strengths
Kerendia is available as film-coated, oblong tablets in three strengths.
- 10 mg: pink, with "FI" on one side, "10" on the other side.
- 20 mg: yellow, with "FI" on one side, "20" on the other side.
- 40 mg: gray-orange, with "FI" on one side, "40" on the other side.
6.2 Postmarketing Experience
The following additional adverse reactions have been reported in postmarketing experience with finerenone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure:
Hypersensitivity: Angioedema, Rash and Urticaria
14.1 Ckd Associated With T2dm
FIDELIO-DKD (NCT: 02540993) and FIGARO-DKD (NCT: 02545049) studies were randomized, double-blind, placebo-controlled, multicenter studies in adult patients with chronic kidney disease (CKD) associated with type 2 diabetes (T2DM). In FIDELIO-DKD, patients needed to either have an UACR of 30 to < 300 mg/g, eGFR 25 to < 60 mL/min/1.73 m2 and diabetic retinopathy, or an UACR of ≥ 300 mg/g and an eGFR of 25 to < 75 mL/min/1.73 m2 to qualify for enrollment. In FIGARO-DKD, patients needed to have an UACR of 30 mg/g to < 300 mg/g and an eGFR of 25 to 90 mL/min/1.73m2, or an UACR ≥ 300 mg/g and an eGFR ≥ 60 mL/min/1.73m2.
Both trials excluded patients with known significant non-diabetic kidney disease. All patients were to have a serum potassium ≤ 4.8 mEq/L at screening and be receiving standard of care background therapy, including a maximum tolerated labeled dose of an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB). Patients with a clinical diagnosis of chronic heart failure with reduced ejection fraction and persistent symptoms (New York Heart Association class II to IV) were excluded. The starting dose of Kerendia was based on screening eGFR (10 mg once daily in patients with an eGFR of 25 to < 60 mL/min/1.73 m2 and 20 mg once daily in patients with an eGFR ≥ 60 mL/min/1.73 m2). The dose of Kerendia could be titrated during the study, with a target dose of 20 mg daily.
The primary objective of the FIDELIO-DKD study was to determine whether Kerendia reduced the incidence of a sustained decline in eGFR of ≥ 40%, kidney failure (defined as chronic dialysis, kidney transplantation, or a sustained decrease in eGFR to < 15 mL/min/1.73 m2), or renal death. The secondary outcome was a composite of time to first occurrence of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure. The primary objective of the FIGARO-DKD study was to determine whether Kerendia reduced the time to first occurrence of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure. The secondary outcome was a composite of time to kidney failure, a sustained decline in eGFR of 40% or more compared to baseline over at least 4 weeks, or renal death.
In FIDELIO-DKD, a total of 5674 patients were randomized to receive Kerendia (N=2833) or placebo (N=2841) and were followed for a median of 2.6 years. The mean age of the study population was 66 years, and 70% of patients were male. This global trial population was 63% White, 25% Asian, and 5% Black. At baseline, the mean eGFR was 44 mL/min/1.73m2, with 55% of patients having an eGFR < 45 mL/min/1.73m2. Median urine albumin-to-creatinine ratio (UACR) was 852 mg/g, mean glycated hemoglobin A1c (HbA1c) was 7.7%, and the mean blood pressure was 138/76 mmHg. Approximately 46% of patients had a history of atherosclerotic cardiovascular disease and 8% had a history of heart failure. At baseline, 99.8% of patients were treated with an ACEi or ARB. Approximately 97% were on an antidiabetic agent (insulin [64.1%], biguanides [44%], glucagon-like peptide-1 [GLP-1] receptor agonists [7%], sodium-glucose cotransporter 2 [SGLT2] inhibitors [5%]), 74% were on a statin, and 57% were on an antiplatelet agent.
In FIGARO-DKD, a total of 7352 patients were randomized to receive Kerendia (N=3686) or placebo (N=3666) and were followed for 3.4 years. As compared to FIDELIO-DKD, baseline eGFR was higher in FIGARO-DKD (mean eGFR 68, with 62% of patients having an eGFR ≥ 60 mL/min/1.73 m2) and median UACR was lower (308 mg/g). Otherwise, baseline patient characteristics and background therapies were similar in the two trials.
In FIDELIO-DKD, Kerendia reduced the incidence of the primary composite endpoint of a sustained decline in eGFR of ≥ 40%, kidney failure, or renal death (HR 0.82, 95% CI 0.73-0.93, p=0.001) as shown in Table 5 and Figure 1. The treatment effect reflected a reduction in a sustained decline in eGFR of ≥ 40% and progression to kidney failure. There were few renal deaths during the trial. Kerendia also reduced the incidence of the secondary composite endpoint of cardiovascular (CV) death, non-fatal myocardial infarction (MI), non-fatal stroke or hospitalization for heart failure (HR 0.86, 95% CI 0.75-0.99, p=0.034) as shown in Table 5 and Figure 3. The treatment effect reflected a reduction in CV death, non-fatal MI, and hospitalization for heart failure. The treatment effect on the primary and secondary composite endpoints was generally consistent across subgroups.
In FIGARO-DKD, Kerendia reduced the incidence of the primary composite endpoint of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure (HR 0.87, 95% CI 0.76-0.98, p = 0.026) as shown in Table 5 and Figure 4. The treatment effect was mainly driven by an effect on hospitalization for heart failure, though CV death also contributed to the treatment effect. The treatment effect on the primary composite endpoint was generally consistent across subgroups, including patients with and without pre-existing cardiovascular disease. The findings for the renal composite endpoint are shown in Table 5 and Figure 2.
| FIDELIO-DKD | FIGARO-DKD | |||||||
|---|---|---|---|---|---|---|---|---|
| Kerendia N=2833 |
Placebo N=2841 |
Treatment Effect Kerendia / Placebo |
Kerendia N=3686 |
Placebo N=3666 |
Treatment Effect Kerendia / Placebo |
|||
| Time-to-event Endpoints: | Event Rate (100 pt-yr) | Event Rate (100 pt-yr) | Hazard Ratio (95% CI) |
p-value | Event Rate (100 pt-yr) | Event Rate (100 pt-yr) | Hazard Ratio (95% CI) |
p-value |
| p-value: two-sided p-value from stratified logrank test CI = confidence interval, CV = cardiovascular, eGFR = estimated glomerular filtration rate, MI = myocardial infarction, N = number of subjects, n = number of subjects with event, pt-yr = patient year. NOTE: Time to first event was analyzed in a Cox proportional hazards model. For patients with multiple events, only the first event contributed to the composite endpoint. Sums of the numbers of first events for the single components do not add up to the numbers of events in the composite endpoint. |
||||||||
| Composite of kidney failure, sustained eGFR decline ≥40% or renal death | 7.6 | 9.1 | 0.82 [0.73; 0.93] |
0.001 | 3.2 | 3.6 | 0.87 [0.76; 1.01] |
- |
| Kidney failure | 3.0 | 3.4 | 0.87 [0.72; 1.05] |
- | 0.4 | 0.5 | 0.72 [0.49;1.05] |
- |
| Sustained eGFR decline ≥40% | 7.2 | 8.7 | 0.81 [0.72; 0.92] |
- | 3.1 | 3.5 | 0.87 [0.75;>1.00] |
- |
| Renal death | - | - | - | - | - | - | - | - |
| Composite of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure | 5.1 | 5.9 | 0.86 [0.75; 0.99] |
0.034 | 3.9 | 4.5 | 0.87 [0.76; 0.98] |
0.026 |
| CV death | 1.7 | 2.0 | 0.86 [0.68;1.08] |
- | 1.6 | 1.8 | 0.90 [0.73; 1.08] |
- |
| Non-fatal MI | 0.9 | 1.2 | 0.80 [0.58;1.09] |
- | 0.9 | 0.8 | 0.99 [0.76; 1.32] |
- |
| Non-fatal stroke | 1.2 | 1.2 | 1.03 [0.76;1.38] |
- | 0.9 | 0.9 | 0.97 [0.74; 1.26] |
- |
| Hospitalization for heart failure | 1.9 | 2.2 | 0.86 [0.68;1.08] |
- | 1.0 | 1.4 | 0.71 [0.56; 0.90] |
- |
Figure 1: Time to first occurrence of kidney failure, sustained decline in eGFR ≥ 40% from baseline, or renal death in the FIDELIO-DKD study
Figure 2: Time to first occurrence of kidney failure, sustained decline in eGFR ≥ 40% from baseline, or renal death in the FIGARO-DKD study
Figure 3: Time to first occurrence of CV death, non-fatal myocardial infarction, non-fatal stroke or hospitalization for heart failure in the FIDELIO-DKD study
Figure 4: Time to first occurrence of CV death, non-fatal myocardial infarction, non-fatal stroke or hospitalization for heart failure in the FIGARO-DKD study
8 Use in Specific Populations
Lactation: Breastfeeding not recommended (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.2 Recommended Starting Dosage
The recommended starting dose of Kerendia is based on eGFR and is presented in Table 1.
| eGFR (mL/min/1.73m2) | Starting Dose |
|---|---|
| ≥ 60 | 20 mg orally once daily |
| ≥ 25 to < 60 | 10 mg orally once daily |
| < 25 | Initiation is not recommended |
For patients who are unable to swallow whole tablets, Kerendia may be crushed and mixed with water or soft foods such as applesauce immediately prior to use and administered orally [see Clinical Pharmacology (12.3)].
14.2 Heart Failure (lvef ≥40%)
FINEARTS-HF (NCT: 04435626) was a randomized, double-blind, placebo-controlled, multicenter study in adult patients with heart failure (New York Heart Association [NYHA] class II–IV) with documented left ventricular ejection fraction (LVEF) ≥40%. Patients were required to have an eGFR ≥25 mL/min/1.73m2 and serum potassium ≤5.0 mEq/L at screening and randomization and were receiving background heart failure medical treatment, including diuretics. The primary endpoint was the composite of cardiovascular (CV) death and total (first and recurrent) heart failure events comprised of hospitalization for heart failure and urgent heart failure visits.
In FINEARTS-HF, 6001 patients were analyzed; 3003 were randomized to Kerendia and 2998 were randomized to placebo and were followed for a median of 2.7 years. The study included 3247 (54%) patients with a heart failure event in the past 3 months, including 1219 (20%) patients randomized during the hospitalization or within 7 days from the heart failure event. Patients with eGFR ≥ 25 to <60 mL/min/1.73m2 initiated Kerendia 10 mg and patients with eGFR ≥60 mL/min/1.73m2 initiated Kerendia 20 mg, and both groups were titrated up to a target dose of Kerendia 20 mg and 40 mg, respectively. At month 6, approximately 65% of the patients reached their target dose. The trial population was 79% White, 17% Asian, and 1.5% Black. The mean age at enrollment was 72 years and 46% of patients were female. At baseline, the mean LVEF was 53%, with 64% of patients having an LVEF ≥50%, and 69% and 30% of patients were NYHA class II and III, respectively. Mean blood pressure was 129/75 mmHg and mean body mass index (BMI) was 30 kg/m2. The median NT-proBNP was 1041 pg/mL, the mean eGFR was 62 mL/min/1.73m2 with 48% of patients having an eGFR <60 mL/min/1.73m2. Atrial fibrillation was present on screening/baseline ECG for 38% of patients and 41% had diabetes mellitus. The majority of patients were on loop diuretics (87%), an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB) (79%), or an angiotensin receptor neprilysin inhibitor (ARNI) (9%), and 14% were on sodium-glucose cotransporter 2 (SGLT2) inhibitors.
Kerendia reduced the risk of the primary composite endpoint compared to placebo (Relative Risk [RR] 0.84, 95% CI 0.74-0.95, p=0.007). See Table 6 and Figure 5 below. The Kerendia and placebo event curves separated early and continued to diverge over the study period, see Figure 5 below. Kerendia reduced the risk of the secondary endpoint of total heart failure events (hospitalization for HF or urgent HF visit) compared to placebo (RR 0.82, 95% CI 0.71-0.94, p=0.006). The treatment effect for the primary endpoint was consistent across all pre-specified subgroups, including sex, LVEF, NYHA class, eGFR, time since latest heart failure event, SGLT2 inhibitor therapy, and diabetes mellitus status.
| FINEARTS-HF | ||||||
|---|---|---|---|---|---|---|
| Kerendia 10 or 20 or 40 mg OD N=3003 |
Placebo N=2998 |
Treatment Effect Kerendia vs Placebo |
||||
| Primary Efficacy Outcome and Components: | Value | Event Rate (100 pt–yr) |
Value | Event Rate (100 pt–yr) | Ratio or difference (95% CI) | p-value |
| E = total number of events; n = number of patients with an event ; RR = rate ratio; HR = hazard ratio; HF = Heart Failure; CV = Cardiovascular. | ||||||
| Primary composite of CV death and total HF events – E (n; %) | 1083 (624; 20.8%) |
14.9 | 1283 (719; 24.0%) |
17.7 | RR 0.84 (0.74, 0.95) |
0.007 |
| Total HF events – E (n; %) Total HF events (hospitalization for HF or urgent HF visit) was a prespecified secondary endpoint.
|
842 (479; 16.0%) |
11.6 | 1024 (573; 19.1%) |
14.1 | RR 0.82 (0.71, 0.94) |
0.006 |
| CV death – n (%) | 242 (8.1%) |
3.3 | 260 (8.7%) |
3.6 | HR 0.93 (0.78, 1.11) |
- |
Figure 5: Mean cumulative functions for primary composite endpoint of CV death and total HF events in Phase 3 study FINEARTS-HF
17 Patient Counseling Information
Advise patients of the need for periodic monitoring of serum potassium levels. Advise patients receiving Kerendia to consult with their physician before using potassium supplements or salt substitutes containing potassium [see Warnings and Precautions (5.1)].
Advise patients to avoid strong or moderate CYP3A4 inducers and to find alternative medicinal products with no or weak potential to induce CYP3A4 [see Drug Interactions (7.1)]
Avoid concomitant intake of grapefruit or grapefruit juice as it is expected to increase the plasma concentration of finerenone [see Drug Interactions (7.1)].
Advise women that breastfeeding is not recommended at the time of treatment with KERENDIA and for 1 day after treatment [see Use in Specific Populations (8.2)].
2.1 Prior to Initiation of Kerendia
Measure serum potassium levels and eGFR before initiation. Do not initiate treatment if serum potassium is > 5.0 mEq/L [see Warnings and Precautions (5.1)].
7.3 Drugs That Affect Serum Potassium
More frequent serum potassium monitoring is warranted in patients receiving concomitant therapy with drugs or supplements that increase serum potassium. [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
Principal Display Panel 10 Mg Tablet Carton
NDC 50419-540-01
Rx only
Kerendia®
(finerenone) tablets
10 mg
30 film-coated tablets
BAYER
Principal Display Panel 20 Mg Tablet Carton
NDC 50419-541-01
Rx only
Kerendia®
(finerenone) tablets
20 mg
30 film-coated tablets
BAYER
Principal Display Panel 40 Mg Tablet Carton
NDC 50419-542-01
Rx only
Kerendia®
(finerenone) tablets
40 mg
30 film-coated tablets
BAYER
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Finerenone was non-genotoxic in an in vitro bacterial reverse mutation (Ames) assay, the in vitro chromosomal aberration assay in cultured Chinese hamster V79 cells, or the in vivo micronucleus assay in mice.
In 2-year carcinogenicity studies, finerenone did not show a statistically significant increase in tumor response in Wistar rats or in CD1 mice. In male mice, Leydig cell adenoma was numerically increased at a dose representing 10 times the AUCunbound in humans and is not considered clinically relevant. Finerenone did not impair fertility in male rats but impaired fertility in female rats at 9 times AUC to the maximum human exposure.
5.2 Worsening of Renal Function in Patients With Heart Failure
Kerendia can cause worsening of renal function in patients with heart failure. Rarely, severe events associated with worsening renal function, including events requiring hospitalization, have been observed [see Adverse Reactions (6.1)].
Measure eGFR in all patients before initiation of treatment or with dose titration of Kerendia and dose accordingly [see Dosage and Administration (2.1, 2.3)]. Initiation of Kerendia in patients with heart failure and an eGFR <25 mL/min/1.73m2 is not recommended.
Measure eGFR periodically during maintenance treatment with Kerendia in patients with heart failure. Consider delaying up-titration or interrupting treatment with Kerendia in patients who develop clinically significant worsening of renal function.
Structured Label Content
Section 42229-5 (42229-5)
CKD associated with T2DM
The target daily dose of Kerendia is 20 mg orally.
Measure serum potassium 4 weeks after initiating treatment and adjust dose (see Table 2); if serum potassium levels are > 4.8 to 5.0 mEq/L, initiation of Kerendia treatment may be considered with additional serum potassium monitoring within the first 4 weeks based on clinical judgment and serum potassium levels [see Warnings and Precautions (5.1)]. Measure serum potassium 4 weeks after a dose adjustment and periodically throughout treatment, and adjust the dose as needed (see Table 2) [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
| Current Kerendia Dose | |||
|---|---|---|---|
| 10 mg once daily | 20 mg once daily | ||
| Current Serum Potassium (mEq/L) | ≤ 4.8 | Increase the dose to 20 mg once daily. If eGFR has decreased by more than 30% compared to previous measurement, maintain 10 mg dose.
|
Maintain 20 mg once daily. |
| > 4.8 – 5.5 | Maintain 10 mg once daily. | Maintain 20 mg once daily. | |
| > 5.5 | Withhold Kerendia. Consider restarting at 10 mg once daily when serum potassium ≤ 5.0 mEq/L. |
Withhold Kerendia. Restart at 10 mg once daily when serum potassium ≤ 5.0 mEq/L. |
Section 43683-2 (43683-2)
10 Overdosage (10 OVERDOSAGE)
In the event of suspected overdose, immediately interrupt Kerendia treatment. The most likely manifestation of overdose is hyperkalemia. If hyperkalemia develops, standard treatment should be initiated.
Finerenone is unlikely to be efficiently removed by hemodialysis given its fraction bound to plasma proteins of about 90%.
11 Description (11 DESCRIPTION)
Kerendia contains finerenone, a nonsteroidal mineralocorticoid receptor antagonist. Finerenone's chemical name is (4S)-4-(4-cyano-2- methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1,6-naphthyridine-3-carboxamide. The molecular formula is C21H22N4O3 and the molecular weight is 378.43 g/mol. The structural formula is:
Finerenone is a white to yellow crystalline powder. It is practically insoluble in water; and sparingly soluble in 0.1 M HCl, ethanol, and acetone.
Each Kerendia tablet contains 10 mg, 20 mg, or 40 mg of finerenone. The inactive ingredients of Kerendia are lactose monohydrate, cellulose microcrystalline, croscarmellose sodium, hypromellose, magnesium stearate, and sodium lauryl sulfate. The film coating contains hypromellose, titanium dioxide and talc, in addition to ferric oxide red (10 mg and 40 mg strength tablets) or ferric oxide yellow (20 mg and 40 mg strength tablets).
2.4 Missed Doses
Direct a patient to take a missed dose as soon as possible after it is noticed, but only on the same day. If this is not possible, the patient should skip the dose and continue with the next dose as prescribed.
5.1 Hyperkalemia
Kerendia can cause hyperkalemia [see Adverse Reactions (6.1)].
The risk for developing hyperkalemia increases with decreasing kidney function and is greater in patients with higher baseline potassium levels or other risk factors for hyperkalemia. Measure serum potassium and eGFR in all patients before initiation of treatment with Kerendia and dose accordingly [see Dosage and Administration (2.1)]. Do not initiate Kerendia if serum potassium is > 5.0 mEq/L.
Measure serum potassium periodically during treatment with Kerendia and adjust dose accordingly [see Dosage and Administration (2.3)]. More frequent monitoring may be necessary for patients at risk for hyperkalemia, including those on concomitant medications that impair potassium excretion or increase serum potassium [see Drug Interactions (7.1, 7.2)].
16.1 How Supplied
Kerendia is available as a film-coated tablet in three strengths. The 10 mg is a pink oblong tablet with "FI" on one side of tablet and "10" on the other side of tablet. The 20 mg tablet is a yellow oblong tablet with "FI" on one side of tablet and "20" on the other side of tablet. The 40 mg tablet is a gray-orange oblong tablet with "FI" on one side of tablet and "40" on the other side of tablet.
| Kerendia Tablets 10 mg | |
| Bottle of 30 tablets | NDC 50419-540-01 |
| Bottle of 90 tablets | NDC 50419-540-02 |
| Hospital Blister Pack of 30 tablets | NDC 50419-540-06 |
| Kerendia Tablets 20 mg | |
| Bottle of 30 tablets | NDC 50419-541-01 |
| Bottle of 90 tablets | NDC 50419-541-02 |
| Hospital Blister Pack of 30 tablets | NDC 50419-541-06 |
| Kerendia Tablets 40 mg | |
| Bottle of 30 tablets | NDC 50419-542-01 |
| Bottle of 90 tablets | NDC 50419-542-02 |
| Hospital Blister Pack of 30 tablets | NDC 50419-542-06 |
8.4 Pediatric Use
The safety and efficacy of Kerendia have not been established in patients below 18 years of age.
8.5 Geriatric Use
Of the 6510 patients who received Kerendia in the FIDELIO-DKD and FIGARO-DKD studies, 55% of patients were 65 years and older, and 14% were 75 years and older. Of the 2993 patients who received Kerendia in the FINEARTS study, 79% of patients were 65 years and older, and 43% were 75 years and older. No overall differences in safety or efficacy were observed between these patients and younger patients. No dose adjustment is required.
4 Contraindications (4 CONTRAINDICATIONS)
Kerendia is contraindicated in patients:
- Who are hypersensitive to any component of this product [see Adverse Reactions (6.2)].
- Who are receiving concomitant treatment with strong CYP3A4 inhibitors [see Drug Interactions (7.1)].
- With adrenal insufficiency.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed elsewhere in the labeling:
- Hyperkalemia [see Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Strong CYP3A4 Inhibitors: Use is contraindicated. (7.1)
- Grapefruit or grapefruit juice: Avoid concomitant use. (7.1)
- Moderate or weak CYP3A4 Inhibitors: Monitor serum potassium during drug initiation or dosage adjustment of either Kerendia or the moderate or weak CYP3A4 inhibitor, and adjust Kerendia dosage as appropriate (7.1)
- Strong or moderate CYP3A4 Inducers: Avoid concomitant use. (7.1)
- Sensitive CYP2C8 substrates at Kerendia 40 mg: Monitor more frequently for adverse reactions. (7.2)
12.2 Pharmacodynamics
In FIDELIO-DKD and FIGARO-DKD, randomized, double-blind, placebo-controlled, multicenter studies in adult patients with chronic kidney disease associated with type 2 diabetes, the placebo-corrected relative reduction in urinary albumin-to-creatinine ratio (UACR) in patients randomized to finerenone was 31% (95% CI 29-34%), and 32% (95% CI 30-35%) respectively at Month 4 and remained stable for the duration of the trial.
In ARTS DN, a randomized, double-blind, placebo-controlled, multicenter phase IIb dose finding study in adults with CKD and T2DM, the placebo-corrected relative reduction in UACR at Day 90 was 25% and 38% in patients treated with finerenone 10 mg and 20 mg once daily, respectively. In patients treated with Kerendia, the mean systolic blood pressure decreased by 3 mmHg and the mean diastolic blood pressure decreased by 1-2 mmHg at month 1, remaining stable thereafter.
12.3 Pharmacokinetics
Finerenone exposure increased proportionally over a dose range of 1.25 to 80 mg (0.06 to 4 times the maximum approved recommended dosage). Steady state of finerenone was achieved after 2 days of dosing. The estimated steady-state geometric mean Cmax,md was 166 µg/L and steady-state geometric mean AUCτ,md was 718 µg.h/L following administration of finerenone 20 mg to patients.
8.6 Hepatic Impairment
Avoid use of Kerendia in patients with severe hepatic impairment (Child Pugh C).
No dosage adjustment is recommended in patients with mild or moderate hepatic impairment (Child Pugh A or B).
Consider additional serum potassium monitoring in patients with moderate hepatic impairment (Child Pugh B) [see Dosing and Administration (2.3) and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Kerendia is indicated to reduce the risk of:
- sustained estimated glomerular filtration rate (eGFR) decline, end-stage kidney disease, cardiovascular death, non-fatal myocardial infarction, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) associated with type 2 diabetes (T2DM).
- cardiovascular death, hospitalization for heart failure, and urgent heart failure visits in adult patients with heart failure with left ventricular ejection fraction (LVEF) ≥ 40%.
12.1 Mechanism of Action
Finerenone is a nonsteroidal, selective antagonist of the mineralocorticoid receptor (MR), which is activated by aldosterone and cortisol and regulates gene transcription. Finerenone blocks MR mediated sodium reabsorption and MR overactivation in both epithelial (e.g., kidney) and nonepithelial (e.g., heart, and blood vessels) tissues. MR overactivation is thought to contribute to fibrosis and inflammation. Finerenone has a high potency and selectivity for the MR and has no relevant affinity for androgen, progesterone, estrogen, and glucocorticoid receptors.
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions are permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hyperkalemia. Patients with decreased kidney function and higher baseline potassium levels are at increased risk. Monitor serum potassium levels and adjust dose as needed. (2.1, 2.2, 2.3, 5.1)
- Worsening of Renal Function in Patients with Heart Failure. Measure eGFR and adjust dose as needed. (2.1, 2.3, 6.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended starting dosage is 10 mg or 20 mg orally once daily based on eGFR and serum potassium thresholds. (2.1)
- Increase dosage after 4 weeks to the target dose of 20 mg once daily for CKD and T2DM based on eGFR and serum potassium thresholds. (2.3)
- Increase dosage after 4 weeks to the target dose of 20 mg or 40 mg once daily for HF with LVEF ≥ 40% based on eGFR and serum potassium thresholds. (2.3)
- Tablets may be taken with or without food (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Kerendia is available as film-coated, oblong tablets in three strengths.
- 10 mg: pink, with "FI" on one side, "10" on the other side.
- 20 mg: yellow, with "FI" on one side, "20" on the other side.
- 40 mg: gray-orange, with "FI" on one side, "40" on the other side.
6.2 Postmarketing Experience
The following additional adverse reactions have been reported in postmarketing experience with finerenone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure:
Hypersensitivity: Angioedema, Rash and Urticaria
14.1 Ckd Associated With T2dm (14.1 CKD associated with T2DM)
FIDELIO-DKD (NCT: 02540993) and FIGARO-DKD (NCT: 02545049) studies were randomized, double-blind, placebo-controlled, multicenter studies in adult patients with chronic kidney disease (CKD) associated with type 2 diabetes (T2DM). In FIDELIO-DKD, patients needed to either have an UACR of 30 to < 300 mg/g, eGFR 25 to < 60 mL/min/1.73 m2 and diabetic retinopathy, or an UACR of ≥ 300 mg/g and an eGFR of 25 to < 75 mL/min/1.73 m2 to qualify for enrollment. In FIGARO-DKD, patients needed to have an UACR of 30 mg/g to < 300 mg/g and an eGFR of 25 to 90 mL/min/1.73m2, or an UACR ≥ 300 mg/g and an eGFR ≥ 60 mL/min/1.73m2.
Both trials excluded patients with known significant non-diabetic kidney disease. All patients were to have a serum potassium ≤ 4.8 mEq/L at screening and be receiving standard of care background therapy, including a maximum tolerated labeled dose of an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB). Patients with a clinical diagnosis of chronic heart failure with reduced ejection fraction and persistent symptoms (New York Heart Association class II to IV) were excluded. The starting dose of Kerendia was based on screening eGFR (10 mg once daily in patients with an eGFR of 25 to < 60 mL/min/1.73 m2 and 20 mg once daily in patients with an eGFR ≥ 60 mL/min/1.73 m2). The dose of Kerendia could be titrated during the study, with a target dose of 20 mg daily.
The primary objective of the FIDELIO-DKD study was to determine whether Kerendia reduced the incidence of a sustained decline in eGFR of ≥ 40%, kidney failure (defined as chronic dialysis, kidney transplantation, or a sustained decrease in eGFR to < 15 mL/min/1.73 m2), or renal death. The secondary outcome was a composite of time to first occurrence of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure. The primary objective of the FIGARO-DKD study was to determine whether Kerendia reduced the time to first occurrence of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure. The secondary outcome was a composite of time to kidney failure, a sustained decline in eGFR of 40% or more compared to baseline over at least 4 weeks, or renal death.
In FIDELIO-DKD, a total of 5674 patients were randomized to receive Kerendia (N=2833) or placebo (N=2841) and were followed for a median of 2.6 years. The mean age of the study population was 66 years, and 70% of patients were male. This global trial population was 63% White, 25% Asian, and 5% Black. At baseline, the mean eGFR was 44 mL/min/1.73m2, with 55% of patients having an eGFR < 45 mL/min/1.73m2. Median urine albumin-to-creatinine ratio (UACR) was 852 mg/g, mean glycated hemoglobin A1c (HbA1c) was 7.7%, and the mean blood pressure was 138/76 mmHg. Approximately 46% of patients had a history of atherosclerotic cardiovascular disease and 8% had a history of heart failure. At baseline, 99.8% of patients were treated with an ACEi or ARB. Approximately 97% were on an antidiabetic agent (insulin [64.1%], biguanides [44%], glucagon-like peptide-1 [GLP-1] receptor agonists [7%], sodium-glucose cotransporter 2 [SGLT2] inhibitors [5%]), 74% were on a statin, and 57% were on an antiplatelet agent.
In FIGARO-DKD, a total of 7352 patients were randomized to receive Kerendia (N=3686) or placebo (N=3666) and were followed for 3.4 years. As compared to FIDELIO-DKD, baseline eGFR was higher in FIGARO-DKD (mean eGFR 68, with 62% of patients having an eGFR ≥ 60 mL/min/1.73 m2) and median UACR was lower (308 mg/g). Otherwise, baseline patient characteristics and background therapies were similar in the two trials.
In FIDELIO-DKD, Kerendia reduced the incidence of the primary composite endpoint of a sustained decline in eGFR of ≥ 40%, kidney failure, or renal death (HR 0.82, 95% CI 0.73-0.93, p=0.001) as shown in Table 5 and Figure 1. The treatment effect reflected a reduction in a sustained decline in eGFR of ≥ 40% and progression to kidney failure. There were few renal deaths during the trial. Kerendia also reduced the incidence of the secondary composite endpoint of cardiovascular (CV) death, non-fatal myocardial infarction (MI), non-fatal stroke or hospitalization for heart failure (HR 0.86, 95% CI 0.75-0.99, p=0.034) as shown in Table 5 and Figure 3. The treatment effect reflected a reduction in CV death, non-fatal MI, and hospitalization for heart failure. The treatment effect on the primary and secondary composite endpoints was generally consistent across subgroups.
In FIGARO-DKD, Kerendia reduced the incidence of the primary composite endpoint of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure (HR 0.87, 95% CI 0.76-0.98, p = 0.026) as shown in Table 5 and Figure 4. The treatment effect was mainly driven by an effect on hospitalization for heart failure, though CV death also contributed to the treatment effect. The treatment effect on the primary composite endpoint was generally consistent across subgroups, including patients with and without pre-existing cardiovascular disease. The findings for the renal composite endpoint are shown in Table 5 and Figure 2.
| FIDELIO-DKD | FIGARO-DKD | |||||||
|---|---|---|---|---|---|---|---|---|
| Kerendia N=2833 |
Placebo N=2841 |
Treatment Effect Kerendia / Placebo |
Kerendia N=3686 |
Placebo N=3666 |
Treatment Effect Kerendia / Placebo |
|||
| Time-to-event Endpoints: | Event Rate (100 pt-yr) | Event Rate (100 pt-yr) | Hazard Ratio (95% CI) |
p-value | Event Rate (100 pt-yr) | Event Rate (100 pt-yr) | Hazard Ratio (95% CI) |
p-value |
| p-value: two-sided p-value from stratified logrank test CI = confidence interval, CV = cardiovascular, eGFR = estimated glomerular filtration rate, MI = myocardial infarction, N = number of subjects, n = number of subjects with event, pt-yr = patient year. NOTE: Time to first event was analyzed in a Cox proportional hazards model. For patients with multiple events, only the first event contributed to the composite endpoint. Sums of the numbers of first events for the single components do not add up to the numbers of events in the composite endpoint. |
||||||||
| Composite of kidney failure, sustained eGFR decline ≥40% or renal death | 7.6 | 9.1 | 0.82 [0.73; 0.93] |
0.001 | 3.2 | 3.6 | 0.87 [0.76; 1.01] |
- |
| Kidney failure | 3.0 | 3.4 | 0.87 [0.72; 1.05] |
- | 0.4 | 0.5 | 0.72 [0.49;1.05] |
- |
| Sustained eGFR decline ≥40% | 7.2 | 8.7 | 0.81 [0.72; 0.92] |
- | 3.1 | 3.5 | 0.87 [0.75;>1.00] |
- |
| Renal death | - | - | - | - | - | - | - | - |
| Composite of CV death, non-fatal MI, non-fatal stroke or hospitalization for heart failure | 5.1 | 5.9 | 0.86 [0.75; 0.99] |
0.034 | 3.9 | 4.5 | 0.87 [0.76; 0.98] |
0.026 |
| CV death | 1.7 | 2.0 | 0.86 [0.68;1.08] |
- | 1.6 | 1.8 | 0.90 [0.73; 1.08] |
- |
| Non-fatal MI | 0.9 | 1.2 | 0.80 [0.58;1.09] |
- | 0.9 | 0.8 | 0.99 [0.76; 1.32] |
- |
| Non-fatal stroke | 1.2 | 1.2 | 1.03 [0.76;1.38] |
- | 0.9 | 0.9 | 0.97 [0.74; 1.26] |
- |
| Hospitalization for heart failure | 1.9 | 2.2 | 0.86 [0.68;1.08] |
- | 1.0 | 1.4 | 0.71 [0.56; 0.90] |
- |
Figure 1: Time to first occurrence of kidney failure, sustained decline in eGFR ≥ 40% from baseline, or renal death in the FIDELIO-DKD study
Figure 2: Time to first occurrence of kidney failure, sustained decline in eGFR ≥ 40% from baseline, or renal death in the FIGARO-DKD study
Figure 3: Time to first occurrence of CV death, non-fatal myocardial infarction, non-fatal stroke or hospitalization for heart failure in the FIDELIO-DKD study
Figure 4: Time to first occurrence of CV death, non-fatal myocardial infarction, non-fatal stroke or hospitalization for heart failure in the FIGARO-DKD study
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Breastfeeding not recommended (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.2 Recommended Starting Dosage
The recommended starting dose of Kerendia is based on eGFR and is presented in Table 1.
| eGFR (mL/min/1.73m2) | Starting Dose |
|---|---|
| ≥ 60 | 20 mg orally once daily |
| ≥ 25 to < 60 | 10 mg orally once daily |
| < 25 | Initiation is not recommended |
For patients who are unable to swallow whole tablets, Kerendia may be crushed and mixed with water or soft foods such as applesauce immediately prior to use and administered orally [see Clinical Pharmacology (12.3)].
14.2 Heart Failure (lvef ≥40%) (14.2 Heart Failure (LVEF ≥40%))
FINEARTS-HF (NCT: 04435626) was a randomized, double-blind, placebo-controlled, multicenter study in adult patients with heart failure (New York Heart Association [NYHA] class II–IV) with documented left ventricular ejection fraction (LVEF) ≥40%. Patients were required to have an eGFR ≥25 mL/min/1.73m2 and serum potassium ≤5.0 mEq/L at screening and randomization and were receiving background heart failure medical treatment, including diuretics. The primary endpoint was the composite of cardiovascular (CV) death and total (first and recurrent) heart failure events comprised of hospitalization for heart failure and urgent heart failure visits.
In FINEARTS-HF, 6001 patients were analyzed; 3003 were randomized to Kerendia and 2998 were randomized to placebo and were followed for a median of 2.7 years. The study included 3247 (54%) patients with a heart failure event in the past 3 months, including 1219 (20%) patients randomized during the hospitalization or within 7 days from the heart failure event. Patients with eGFR ≥ 25 to <60 mL/min/1.73m2 initiated Kerendia 10 mg and patients with eGFR ≥60 mL/min/1.73m2 initiated Kerendia 20 mg, and both groups were titrated up to a target dose of Kerendia 20 mg and 40 mg, respectively. At month 6, approximately 65% of the patients reached their target dose. The trial population was 79% White, 17% Asian, and 1.5% Black. The mean age at enrollment was 72 years and 46% of patients were female. At baseline, the mean LVEF was 53%, with 64% of patients having an LVEF ≥50%, and 69% and 30% of patients were NYHA class II and III, respectively. Mean blood pressure was 129/75 mmHg and mean body mass index (BMI) was 30 kg/m2. The median NT-proBNP was 1041 pg/mL, the mean eGFR was 62 mL/min/1.73m2 with 48% of patients having an eGFR <60 mL/min/1.73m2. Atrial fibrillation was present on screening/baseline ECG for 38% of patients and 41% had diabetes mellitus. The majority of patients were on loop diuretics (87%), an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB) (79%), or an angiotensin receptor neprilysin inhibitor (ARNI) (9%), and 14% were on sodium-glucose cotransporter 2 (SGLT2) inhibitors.
Kerendia reduced the risk of the primary composite endpoint compared to placebo (Relative Risk [RR] 0.84, 95% CI 0.74-0.95, p=0.007). See Table 6 and Figure 5 below. The Kerendia and placebo event curves separated early and continued to diverge over the study period, see Figure 5 below. Kerendia reduced the risk of the secondary endpoint of total heart failure events (hospitalization for HF or urgent HF visit) compared to placebo (RR 0.82, 95% CI 0.71-0.94, p=0.006). The treatment effect for the primary endpoint was consistent across all pre-specified subgroups, including sex, LVEF, NYHA class, eGFR, time since latest heart failure event, SGLT2 inhibitor therapy, and diabetes mellitus status.
| FINEARTS-HF | ||||||
|---|---|---|---|---|---|---|
| Kerendia 10 or 20 or 40 mg OD N=3003 |
Placebo N=2998 |
Treatment Effect Kerendia vs Placebo |
||||
| Primary Efficacy Outcome and Components: | Value | Event Rate (100 pt–yr) |
Value | Event Rate (100 pt–yr) | Ratio or difference (95% CI) | p-value |
| E = total number of events; n = number of patients with an event ; RR = rate ratio; HR = hazard ratio; HF = Heart Failure; CV = Cardiovascular. | ||||||
| Primary composite of CV death and total HF events – E (n; %) | 1083 (624; 20.8%) |
14.9 | 1283 (719; 24.0%) |
17.7 | RR 0.84 (0.74, 0.95) |
0.007 |
| Total HF events – E (n; %) Total HF events (hospitalization for HF or urgent HF visit) was a prespecified secondary endpoint.
|
842 (479; 16.0%) |
11.6 | 1024 (573; 19.1%) |
14.1 | RR 0.82 (0.71, 0.94) |
0.006 |
| CV death – n (%) | 242 (8.1%) |
3.3 | 260 (8.7%) |
3.6 | HR 0.93 (0.78, 1.11) |
- |
Figure 5: Mean cumulative functions for primary composite endpoint of CV death and total HF events in Phase 3 study FINEARTS-HF
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients of the need for periodic monitoring of serum potassium levels. Advise patients receiving Kerendia to consult with their physician before using potassium supplements or salt substitutes containing potassium [see Warnings and Precautions (5.1)].
Advise patients to avoid strong or moderate CYP3A4 inducers and to find alternative medicinal products with no or weak potential to induce CYP3A4 [see Drug Interactions (7.1)]
Avoid concomitant intake of grapefruit or grapefruit juice as it is expected to increase the plasma concentration of finerenone [see Drug Interactions (7.1)].
Advise women that breastfeeding is not recommended at the time of treatment with KERENDIA and for 1 day after treatment [see Use in Specific Populations (8.2)].
2.1 Prior to Initiation of Kerendia
Measure serum potassium levels and eGFR before initiation. Do not initiate treatment if serum potassium is > 5.0 mEq/L [see Warnings and Precautions (5.1)].
7.3 Drugs That Affect Serum Potassium
More frequent serum potassium monitoring is warranted in patients receiving concomitant therapy with drugs or supplements that increase serum potassium. [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
Principal Display Panel 10 Mg Tablet Carton (PRINCIPAL DISPLAY PANEL- 10 mg Tablet Carton)
NDC 50419-540-01
Rx only
Kerendia®
(finerenone) tablets
10 mg
30 film-coated tablets
BAYER
Principal Display Panel 20 Mg Tablet Carton (PRINCIPAL DISPLAY PANEL- 20 mg Tablet Carton)
NDC 50419-541-01
Rx only
Kerendia®
(finerenone) tablets
20 mg
30 film-coated tablets
BAYER
Principal Display Panel 40 Mg Tablet Carton (PRINCIPAL DISPLAY PANEL- 40 mg Tablet Carton)
NDC 50419-542-01
Rx only
Kerendia®
(finerenone) tablets
40 mg
30 film-coated tablets
BAYER
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Finerenone was non-genotoxic in an in vitro bacterial reverse mutation (Ames) assay, the in vitro chromosomal aberration assay in cultured Chinese hamster V79 cells, or the in vivo micronucleus assay in mice.
In 2-year carcinogenicity studies, finerenone did not show a statistically significant increase in tumor response in Wistar rats or in CD1 mice. In male mice, Leydig cell adenoma was numerically increased at a dose representing 10 times the AUCunbound in humans and is not considered clinically relevant. Finerenone did not impair fertility in male rats but impaired fertility in female rats at 9 times AUC to the maximum human exposure.
5.2 Worsening of Renal Function in Patients With Heart Failure (5.2 Worsening of Renal Function in Patients with Heart Failure)
Kerendia can cause worsening of renal function in patients with heart failure. Rarely, severe events associated with worsening renal function, including events requiring hospitalization, have been observed [see Adverse Reactions (6.1)].
Measure eGFR in all patients before initiation of treatment or with dose titration of Kerendia and dose accordingly [see Dosage and Administration (2.1, 2.3)]. Initiation of Kerendia in patients with heart failure and an eGFR <25 mL/min/1.73m2 is not recommended.
Measure eGFR periodically during maintenance treatment with Kerendia in patients with heart failure. Consider delaying up-titration or interrupting treatment with Kerendia in patients who develop clinically significant worsening of renal function.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:12.162972 · Updated: 2026-03-14T22:32:22.096833