pinrid
fc225b5b-0ce8-4394-a643-39dbc674d7cd
34390-5
HUMAN OTC DRUG LABEL
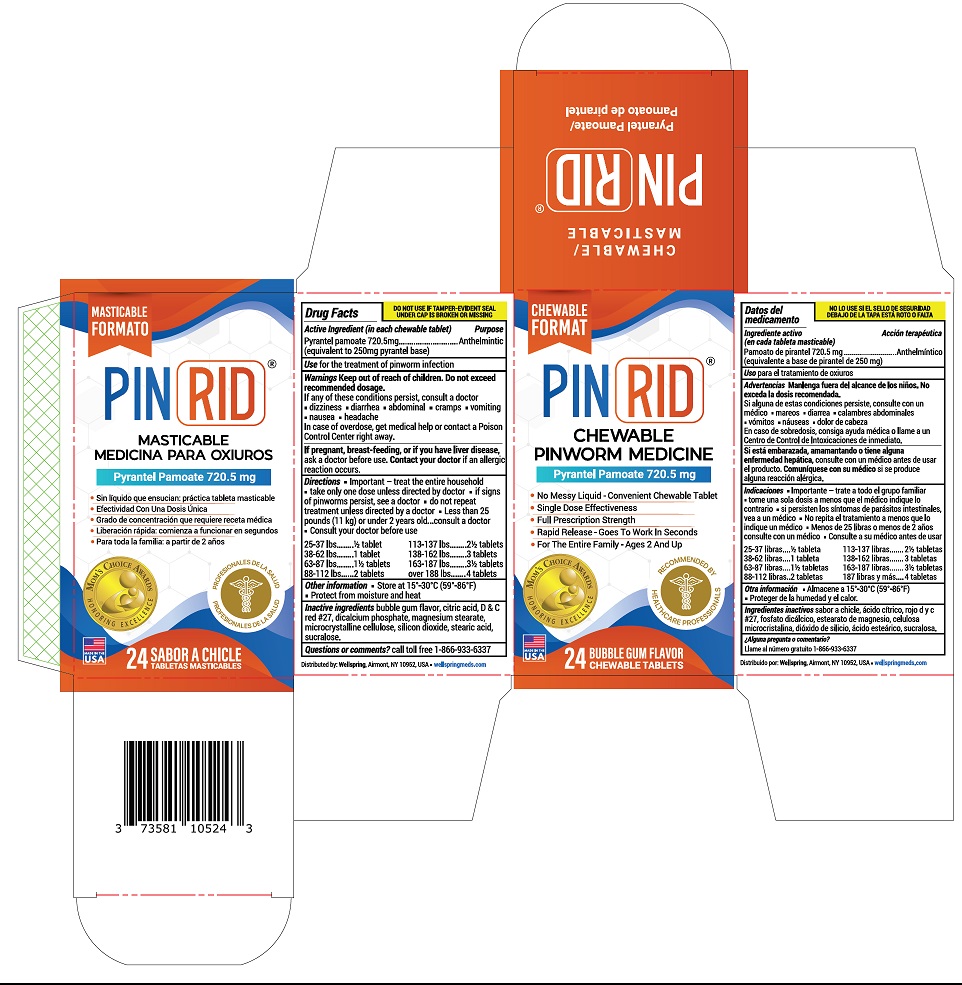
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Anthelmintic
Description
Drug Facts
Medication Information
Purpose
Anthelmintic
Directions
- Important- treat the entire household
- take only one dose unless directed by doctor
- if signs of pinworms persist, see a doctor
- do not repeat treatment unless directed by a doctor
- less than 25 pounds (11 kg) or under 2 years old, consult a doctor
- Consult your doctor before use
| 25-37 lb | ½ tablet |
| 38-62 lbs | 1 tablets |
| 63-87 lbs | 1½ tablets |
| 88-112 lbs | 2 tablets |
| 113-137 lbs | 2½ tablets |
| 138-162 lbs | 3 tablets |
| 163-187 lbs | 3½ tablets |
| over 188 lbs | 4 tablets |
Description
Drug Facts
Principal Display Panel
Use
for the treatment of pinworm infection
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. Do not exceed recommended dosage.
Section 50569-3
If any of these conditions persist, consult a doctor
• dizziness
• diarhea
• abdominal cramps
• vomiting
• nausea
• headache
In case of overdose, get medical help or contact a poison control center rightaway.
Section 53414-9
If pregnant, breast-feeding, or if you have liver disease, ask a doctor before use. Contact your doctorif an allergic reaction occurs.
Other Information:
- Store at 15°-30°C (59° - 86 °F)
- Protect from moisture and heat
Intactive Ingredients
bubble gum flavor, citric acid, D&C red#27, dicalcium phosphate, magnesium stearate, microcrystalline cellulose, silicon dioxide, stearic acid, sucralose.
Questions Or Comments:
call toll free 1-866-933-6337
Active Ingredient (in Each Chewable Tablet)
Pyrantel pamoate 720.5 mg
(equivalent to 250 mg pyrantel base)
Structured Label Content
Purpose
Anthelmintic
Directions
- Important- treat the entire household
- take only one dose unless directed by doctor
- if signs of pinworms persist, see a doctor
- do not repeat treatment unless directed by a doctor
- less than 25 pounds (11 kg) or under 2 years old, consult a doctor
- Consult your doctor before use
| 25-37 lb | ½ tablet |
| 38-62 lbs | 1 tablets |
| 63-87 lbs | 1½ tablets |
| 88-112 lbs | 2 tablets |
| 113-137 lbs | 2½ tablets |
| 138-162 lbs | 3 tablets |
| 163-187 lbs | 3½ tablets |
| over 188 lbs | 4 tablets |
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Use
for the treatment of pinworm infection
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. Do not exceed recommended dosage.
Section 50569-3 (50569-3)
If any of these conditions persist, consult a doctor
• dizziness
• diarhea
• abdominal cramps
• vomiting
• nausea
• headache
In case of overdose, get medical help or contact a poison control center rightaway.
Section 53414-9 (53414-9)
If pregnant, breast-feeding, or if you have liver disease, ask a doctor before use. Contact your doctorif an allergic reaction occurs.
Other Information: (Other information:)
- Store at 15°-30°C (59° - 86 °F)
- Protect from moisture and heat
Intactive Ingredients (Intactive ingredients)
bubble gum flavor, citric acid, D&C red#27, dicalcium phosphate, magnesium stearate, microcrystalline cellulose, silicon dioxide, stearic acid, sucralose.
Questions Or Comments: (Questions or comments:)
call toll free 1-866-933-6337
Active Ingredient (in Each Chewable Tablet) (Active Ingredient (in each chewable tablet))
Pyrantel pamoate 720.5 mg
(equivalent to 250 mg pyrantel base)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:27.626459 · Updated: 2026-03-14T23:09:13.832086