These Highlights Do Not Include All The Information Needed To Use Leflunomide Tablets Safely And Effectively. See Full Prescribing Information For Leflunomide Tablets.
fc044417-cd9a-42f6-8a3d-a14e7c2f81a2
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: EMBRYO-FETAL TOXICITY and HEPATOTOXICITY See full prescribing information for complete boxed warning. Embryo-Fetal Toxicity Teratogenicity and embryo-lethality occurred in animals administered leflunomide ( 5.1 , 8.1 ) Exclude pregnancy prior to initiating leflunomide therapy. ( 5.1 , 8.3 ) Advise use of effective contraception in females of reproductive potential during treatment and during a drug elimination procedure. ( 5.1 , 5.3 , 8.3 ) Stop leflunomide and use accelerated drug elimination procedure if the patient becomes pregnant. ( 5.1 , 5.3 , 8.1 ) Hepatotoxicity Severe liver injury and fatal liver failure have been reported. ( 5.2 ) Avoid leflunomide use in patients with pre-existing liver disease, or those with serum alanine aminotransferase (ALT) >2 × ULN. ( 5.2 , 8.6 ) Use caution when leflunomide is given with other potentially hepatotoxic drugs. ( 5.2 ) Monitor ALT levels. Interrupt leflunomide treatment if ALT elevation >3-fold ULN. If likely leflunomide-induced, start accelerated drug elimination procedure and monitor liver tests weekly until normalized. ( 5.2 , 5.3 )
Indications and Usage
Leflunomide Tablets, USP are indicated for the treatment of adults with active rheumatoid arthritis (RA).
Dosage and Administration
Loading dosage for patients at low risk for leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression: 100 mg daily for 3 days. ( 2.1 ) Maintenance dosage: 20 mg daily. ( 2.1 ) Maximum recommended daily dosage: 20 mg once daily. ( 2.1 ) If 20 mg once daily is not tolerated, may decrease dosage to 10 mg once daily. ( 2.1 ) Screen patients for active and latent tuberculosis, pregnancy test (females), blood pressure, and laboratory tests before starting leflunomide. ( 2.2 )
Warnings and Precautions
After stopping leflunomide, it is recommended that an accelerated drug elimination procedure be used to reduce the plasma concentrations of the active metabolite, teriflunomide. ( 5.3 ) Severe infections (including sepsis), pancytopenia, agranulocytosis, and thrombocytopenia: Stop leflunomide and use accelerated elimination procedure. Do not start leflunomide in patients with active infection. Monitor CBCs during treatment with leflunomide. ( 5.4 ) Stevens-Johnson syndrome and toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS): Stop leflunomide and use accelerated elimination procedure. ( 5.5 ) Skin ulcers: If skin ulcer is suspected, discontinue leflunomide treatment and consider an accelerated drug elimination procedure. ( 5.6 ) Peripheral neuropathy: If patient develops symptoms consistent with peripheral neuropathy, evaluate patient and consider discontinuing leflunomide. ( 5.8 ) Interstitial lung disease: May be fatal. New onset or worsening symptoms may necessitate discontinuation of leflunomide and initiation of accelerated elimination procedure. ( 5.9 ) Increased blood pressure: Monitor and treat. ( 5.11 )
Contraindications
Leflunomide is contraindicated in: Pregnant women. Leflunomide may cause fetal harm. If a woman becomes pregnant while taking this drug, stop leflunomide, apprise the patient of the potential hazard to the fetus, and begin a drug elimination procedure [see Warnings and Precautions (5.1 , 5.3) and Use in Specific Populations (8.1) ]. Patients with severe hepatic impairment [see Warnings and Precautions (5.2) ]. Patients with known hypersensitivity to leflunomide or any of the other components of leflunomide. Known reactions include anaphylaxis [see Adverse Reactions (6.1) ]. Patients being treated with teriflunomide [see Drug Interactions (7) ].
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Hepatotoxicity [see Warnings and Precautions (5.2) ] Immunosuppression [see Warnings and Precautions (5.4) ] Bone marrow suppression [see Warnings and Precautions (5.4) ] Serious infections [see Warnings and Precautions (5.4) ] Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug reactions with eosinophilia and systemic symptoms [see Warnings and Precautions (5.5) ] Skin ulcers [see Warnings and Precautions (5.6) ] Peripheral neuropathy [see Warnings and Precautions (5.8) ] Interstitial lung disease [see Warnings and Precautions (5.9) ]
Drug Interactions
Following oral administration, leflunomide is metabolized to an active metabolite, teriflunomide, which is responsible for essentially all of leflunomide's in vivo activity. Drug interaction studies have been conducted with both leflunomide and with its active metabolite, teriflunomide, where the metabolite was directly administered to the test subjects.
Storage and Handling
How Supplied Leflunomide Tablets, USP Strength Quantity NDC Number Description 10 mg 30 count bottle 35573-447-30 White to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side. 20 mg 30 count bottle 35573-448-30 White to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side.
How Supplied
How Supplied Leflunomide Tablets, USP Strength Quantity NDC Number Description 10 mg 30 count bottle 35573-447-30 White to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side. 20 mg 30 count bottle 35573-448-30 White to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side.
Medication Information
Warnings and Precautions
After stopping leflunomide, it is recommended that an accelerated drug elimination procedure be used to reduce the plasma concentrations of the active metabolite, teriflunomide. ( 5.3 ) Severe infections (including sepsis), pancytopenia, agranulocytosis, and thrombocytopenia: Stop leflunomide and use accelerated elimination procedure. Do not start leflunomide in patients with active infection. Monitor CBCs during treatment with leflunomide. ( 5.4 ) Stevens-Johnson syndrome and toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS): Stop leflunomide and use accelerated elimination procedure. ( 5.5 ) Skin ulcers: If skin ulcer is suspected, discontinue leflunomide treatment and consider an accelerated drug elimination procedure. ( 5.6 ) Peripheral neuropathy: If patient develops symptoms consistent with peripheral neuropathy, evaluate patient and consider discontinuing leflunomide. ( 5.8 ) Interstitial lung disease: May be fatal. New onset or worsening symptoms may necessitate discontinuation of leflunomide and initiation of accelerated elimination procedure. ( 5.9 ) Increased blood pressure: Monitor and treat. ( 5.11 )
Indications and Usage
Leflunomide Tablets, USP are indicated for the treatment of adults with active rheumatoid arthritis (RA).
Dosage and Administration
Loading dosage for patients at low risk for leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression: 100 mg daily for 3 days. ( 2.1 ) Maintenance dosage: 20 mg daily. ( 2.1 ) Maximum recommended daily dosage: 20 mg once daily. ( 2.1 ) If 20 mg once daily is not tolerated, may decrease dosage to 10 mg once daily. ( 2.1 ) Screen patients for active and latent tuberculosis, pregnancy test (females), blood pressure, and laboratory tests before starting leflunomide. ( 2.2 )
Contraindications
Leflunomide is contraindicated in: Pregnant women. Leflunomide may cause fetal harm. If a woman becomes pregnant while taking this drug, stop leflunomide, apprise the patient of the potential hazard to the fetus, and begin a drug elimination procedure [see Warnings and Precautions (5.1 , 5.3) and Use in Specific Populations (8.1) ]. Patients with severe hepatic impairment [see Warnings and Precautions (5.2) ]. Patients with known hypersensitivity to leflunomide or any of the other components of leflunomide. Known reactions include anaphylaxis [see Adverse Reactions (6.1) ]. Patients being treated with teriflunomide [see Drug Interactions (7) ].
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Hepatotoxicity [see Warnings and Precautions (5.2) ] Immunosuppression [see Warnings and Precautions (5.4) ] Bone marrow suppression [see Warnings and Precautions (5.4) ] Serious infections [see Warnings and Precautions (5.4) ] Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug reactions with eosinophilia and systemic symptoms [see Warnings and Precautions (5.5) ] Skin ulcers [see Warnings and Precautions (5.6) ] Peripheral neuropathy [see Warnings and Precautions (5.8) ] Interstitial lung disease [see Warnings and Precautions (5.9) ]
Drug Interactions
Following oral administration, leflunomide is metabolized to an active metabolite, teriflunomide, which is responsible for essentially all of leflunomide's in vivo activity. Drug interaction studies have been conducted with both leflunomide and with its active metabolite, teriflunomide, where the metabolite was directly administered to the test subjects.
Storage and Handling
How Supplied Leflunomide Tablets, USP Strength Quantity NDC Number Description 10 mg 30 count bottle 35573-447-30 White to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side. 20 mg 30 count bottle 35573-448-30 White to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side.
How Supplied
How Supplied Leflunomide Tablets, USP Strength Quantity NDC Number Description 10 mg 30 count bottle 35573-447-30 White to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side. 20 mg 30 count bottle 35573-448-30 White to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side.
Description
WARNING: EMBRYO-FETAL TOXICITY and HEPATOTOXICITY See full prescribing information for complete boxed warning. Embryo-Fetal Toxicity Teratogenicity and embryo-lethality occurred in animals administered leflunomide ( 5.1 , 8.1 ) Exclude pregnancy prior to initiating leflunomide therapy. ( 5.1 , 8.3 ) Advise use of effective contraception in females of reproductive potential during treatment and during a drug elimination procedure. ( 5.1 , 5.3 , 8.3 ) Stop leflunomide and use accelerated drug elimination procedure if the patient becomes pregnant. ( 5.1 , 5.3 , 8.1 ) Hepatotoxicity Severe liver injury and fatal liver failure have been reported. ( 5.2 ) Avoid leflunomide use in patients with pre-existing liver disease, or those with serum alanine aminotransferase (ALT) >2 × ULN. ( 5.2 , 8.6 ) Use caution when leflunomide is given with other potentially hepatotoxic drugs. ( 5.2 ) Monitor ALT levels. Interrupt leflunomide treatment if ALT elevation >3-fold ULN. If likely leflunomide-induced, start accelerated drug elimination procedure and monitor liver tests weekly until normalized. ( 5.2 , 5.3 )
Section 42229-5
Embryo-Fetal Toxicity
Leflunomide is contraindicated for use in pregnant women because of the potential for fetal harm. Teratogenicity and embryo-lethality occurred in animals administered leflunomide at doses lower than the human exposure level. Exclude pregnancy before the start of treatment with leflunomide in females of reproductive potential. Advise females of reproductive potential to use effective contraception during leflunomide treatment and during an accelerated drug elimination procedure after leflunomide treatment. Stop leflunomide and use an accelerated drug elimination procedure if the patient becomes pregnant [see Contraindications (4), Warnings and Precautions (5.1, 5.3), Use in Specific Populations (8.1, 8.3), and Clinical Pharmacology (12.3)].
Section 43683-2
Warnings and Precautions, Skin Ulcers (5.6)........................06/2024
Section 44425-7
TAMPER EVIDENT: Do not use if ring below the closure is broken or missing.
CHILD RESISTANT: This package features a child-resistant closure.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from light.
10 Overdosage
There have been reports of chronic overdose in patients taking leflunomide at daily dose up to five times the recommended daily dose and reports of acute overdose in adults and children. Adverse events were consistent with the safety profile for leflunomide [see Adverse Reactions (6)]. The most frequent adverse events observed were diarrhea, abdominal pain, leukopenia, anemia, and elevated liver function tests.
In the event of a significant overdose or toxicity, perform an accelerated drug elimination procedure to accelerate elimination [see Warnings and Precautions (5.3)].
Studies with both hemodialysis and CAPD (chronic ambulatory peritoneal dialysis) indicate that teriflunomide, the primary metabolite of leflunomide, is not dialyzable [see Clinical Pharmacology (12.3)].
11 Description
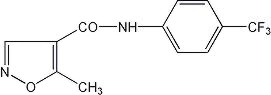
Leflunomide is a pyrimidine synthesis inhibitor. The chemical name for leflunomide is N-(4´-trifluoromethylphenyl)-5-methylisoxazole-4-carboxamide. It has an empirical formula C12H9F3N2O2, a molecular weight of 270.2 and the following structural formula:
Leflunomide is available for oral administration as tablets containing 10 or 20 mg of active drug. Combined with leflunomide are the following inactive ingredients: lactose monohydrate, hydroxypropyl cellulose, tartaric acid, sodium lauryl sulfate, magnesium stearate, opadry white, polyvinyl alcohol, titanium dioxide, talc, lecithin, and xanthan gum.
5.6 Skin Ulcers
Skin ulcers may occur in patients during therapy with leflunomide. If leflunomide-associated skin ulcer is suspected or if skin ulcers persist despite appropriate therapy, leflunomide discontinuation and an accelerated drug elimination procedure should be considered [see Warnings and Precautions (5.3)]. The decision to resume leflunomide following skin ulcers should be based on clinical judgment of adequate wound healing.
5.10 Vaccinations
No clinical data are available on the efficacy and safety of vaccinations during leflunomide treatment. Vaccination with live vaccines is, however, not recommended. The long half-life of the active metabolite of leflunomide should be considered when contemplating administration of a live vaccine after stopping leflunomide.
8.4 Pediatric Use
The safety and effectiveness of leflunomide in pediatric patients have not been established.
The safety and effectiveness of leflunomide in the treatment of polyarticular course juvenile idiopathic arthritis (JIA) was evaluated in a single multicenter, double-blind, active-controlled trial in 94 pediatric patients (1:1 randomization) with polyarticular course juvenile idiopathic arthritis (JIA) as defined by the American College of Rheumatology (ACR). In this population, leflunomide treatment was found not to be effective.
The safety of leflunomide was studied in 74 patients with polyarticular course JIA ranging in age from 3–17 years (47 patients from the active-controlled study and 27 from an open-label safety and pharmacokinetic study). The most common adverse events included abdominal pain, diarrhea, nausea, vomiting, oral ulcers, upper respiratory tract infections, alopecia, rash, headache, and dizziness. Less common adverse events included anemia, hypertension, and weight loss. Fourteen pediatric patients experienced ALT and/or AST elevations, nine between 1.2 and 3-fold the upper limit of normal, and five between 3 and 8-fold the upper limit of normal.
8.5 Geriatric Use
Of the total number of subjects in controlled clinical trials (Trials 1, 2, and 3) of leflunomide, 234 subjects were 65 years and over [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dosage adjustment is needed in patients over 65.
5.2 Hepatotoxicity
Severe liver injury, including fatal liver failure, has been reported in some patients treated with leflunomide. Patients with pre-existing acute or chronic liver disease, or those with serum alanine aminotransferase (ALT) of greater than twice the upper limits of normal (>2 × ULN) before initiating treatment, should not be treated with leflunomide. Use caution when leflunomide is given with other potentially hepatotoxic drugs. Monitoring of ALT levels is recommended at least monthly for six months after starting leflunomide, and thereafter every 6 to 8 weeks. If ALT elevation >3-fold ULN occurs, interrupt leflunomide therapy and investigate the cause. If likely leflunomide-induced, perform the accelerated drug elimination procedure and monitor liver tests weekly until normalized [see Warnings and Precautions (5.3)]. If leflunomide-induced liver injury is unlikely because some other cause has been found, resumption of leflunomide therapy may be considered.
If leflunomide and methotrexate are given concomitantly, follow the American College of Rheumatology (ACR) guidelines for monitoring methotrexate liver toxicity with ALT, AST, and serum albumin testing.
14 Clinical Studies
The efficacy of leflunomide in the treatment of rheumatoid arthritis (RA) was demonstrated in three controlled trials showing reduction in signs and symptoms, and inhibition of structural damage. In two placebo-controlled trials, efficacy was demonstrated for improvement in physical function. In these trials, efficacy was evaluated by:
1. Reduction of signs and symptoms
Relief of signs and symptoms was assessed using the American College of Rheumatology (ACR) 20 Responder Index, a composite of clinical, laboratory, and functional measures in rheumatoid arthritis. An "ACR20 Responder" is a patient who had ≥20% improvement in both tender and swollen joint counts and in 3 of the following 5 criteria: physician global assessment, patient global assessment, functional ability measure (Modified Health Assessment Questionnaire [MHAQ]), visual analog pain scale, and erythrocyte sedimentation rate or C-reactive protein. An "ACR20 Responder at Endpoint" is a patient who completed the study and was an ACR20 Responder at the completion of the study.
2. Inhibition of structural damage
Inhibition of structural damage compared to control was assessed using the Sharp score, a composite score of x-ray erosions and joint space narrowing in hands/wrists and forefeet.
3. Improvement in physical function
Improvement in physical function was assessed using the Health Assessment Questionnaire (HAQ) and the Medical Outcomes Survey Short Form (SF-36).
In all leflunomide trials, participants of at least 18 years of age and in ARA functional class of I, II, or III received an initial loading dose of 100 mg leflunomide per day for three days, followed by 20 mg per day thereafter.
Exclusion criteria included patients with a history of hypersensitivity to the study medication; women who were pregnant or breastfeeding and men or women of child bearing age and potential who had not received contraceptives for at least 4 weeks before entering the study and to be maintained throughout the study and for at least 6 months after discontinuing treatment; patients with a history of inflammatory disease, impaired renal function or liver impairment, cardiac failure, congenital or acquired immunodeficiency, impaired coagulation, or a history of recent major traumatic injury; and patients taking intra-articular or systemic concomitant medications which could affect the safety and/or efficacy of the study medication.
4 Contraindications
Leflunomide is contraindicated in:
- Pregnant women. Leflunomide may cause fetal harm. If a woman becomes pregnant while taking this drug, stop leflunomide, apprise the patient of the potential hazard to the fetus, and begin a drug elimination procedure [see Warnings and Precautions (5.1, 5.3) and Use in Specific Populations (8.1)].
- Patients with severe hepatic impairment [see Warnings and Precautions (5.2)].
- Patients with known hypersensitivity to leflunomide or any of the other components of leflunomide. Known reactions include anaphylaxis [see Adverse Reactions (6.1)].
- Patients being treated with teriflunomide [see Drug Interactions (7)].
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Immunosuppression [see Warnings and Precautions (5.4)]
- Bone marrow suppression [see Warnings and Precautions (5.4)]
- Serious infections [see Warnings and Precautions (5.4)]
- Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug reactions with eosinophilia and systemic symptoms [see Warnings and Precautions (5.5)]
- Skin ulcers [see Warnings and Precautions (5.6)]
- Peripheral neuropathy [see Warnings and Precautions (5.8)]
- Interstitial lung disease [see Warnings and Precautions (5.9)]
7 Drug Interactions
Following oral administration, leflunomide is metabolized to an active metabolite, teriflunomide, which is responsible for essentially all of leflunomide's in vivo activity. Drug interaction studies have been conducted with both leflunomide and with its active metabolite, teriflunomide, where the metabolite was directly administered to the test subjects.
8.7 Renal Impairment
Dedicated studies of the effect of renal impairment on leflunomide pharmacokinetics have not been conducted. Given that the kidney plays an important role in drug elimination, caution should be used when leflunomide is administered to these patients.
12.3 Pharmacokinetics
Following oral administration, leflunomide is metabolized to an active metabolite, teriflunomide, which is responsible for essentially all of leflunomide's in vivo activity. Plasma concentrations of the parent drug, leflunomide, have been occasionally seen at very low concentrations. Studies of the pharmacokinetics of leflunomide have primarily examined the plasma concentrations of the active metabolite, teriflunomide.
2.1 Recommended Dosage
The recommended dosage of leflunomide is 20 mg once daily. Treatment may be initiated with or without a loading dose, depending upon the patient's risk of leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression. The loading dose provides steady-state concentrations more rapidly.
- For patients who are at low risk for leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression, the recommended leflunomide loading dose is 100 mg once daily for 3 days. Subsequently administer 20 mg once daily.
- For patients at high risk for leflunomide-associated hepatotoxicity (e.g., those taking concomitant methotrexate) or leflunomide-associated myelosuppression (e.g., patients taking concomitant immunosuppressants), the recommended leflunomide dose is 20 mg once daily without a loading dose [see Warnings and Precautions (5.2, 5.4)].
The maximum recommended daily dose is 20 mg once per day. Consider dosage reduction to 10 mg once daily for patients who are not able to tolerate 20 mg daily (i.e., for patients who experience any adverse events listed in Table 1).
Monitor patients carefully after dosage reduction and after stopping therapy with leflunomide, since the active metabolite of leflunomide, teriflunomide, is slowly eliminated from the plasma [see Clinical Pharmacology (12.3)]. After stopping leflunomide treatment, an accelerated drug elimination procedure is recommended to reduce the plasma concentrations of the active metabolite, teriflunomide [see Warnings and Precautions (5.3)]. Without use of an accelerated drug elimination procedure, it may take up to 2 years to reach undetectable plasma teriflunomide concentrations after stopping leflunomide [see Clinical Pharmacology (12.3)].
8.6 Hepatic Impairment
Dedicated studies of the effect of hepatic impairment on leflunomide pharmacokinetics have not been conducted. Given the need to metabolize leflunomide into the active species, the role of the liver in drug elimination/recycling, and the possible risk of increased hepatic toxicity, the use of leflunomide in patients with hepatic impairment is not recommended.
1 Indications and Usage
Leflunomide Tablets, USP are indicated for the treatment of adults with active rheumatoid arthritis (RA).
12.1 Mechanism of Action
Leflunomide is an isoxazole immunomodulatory agent that inhibits dihydroorotate dehydrogenase (a mitochondrial enzyme involved in de novo pyrimidine synthesis) and has antiproliferative activity. Several in vivo and in vitro experimental models have demonstrated an anti-inflammatory effect.
5.1 Embryo Fetal Toxicity
Leflunomide may cause fetal harm when administered to a pregnant woman. Teratogenicity and embryo-lethality occurred in animal reproduction studies with leflunomide at doses lower than the human exposure level [see Use in Specific Populations (8.1)].
Leflunomide is contraindicated for use in pregnant women [see Contraindications (4)]. Exclude pregnancy before starting treatment with leflunomide in females of reproductive potential [see Dosage and Administration (2.2)]. Advise females of reproductive potential to use effective contraception during leflunomide treatment and during an accelerated drug elimination procedure after leflunomide treatment [see Use in Specific Populations (8.3)]. If a woman becomes pregnant while taking leflunomide, stop treatment with leflunomide, apprise the patient of the potential risk to a fetus, and perform an accelerated drug elimination procedure to achieve nondetectable plasma concentrations of teriflunomide, the active metabolite of leflunomide [see Warnings and Precautions (5.3)].
Upon discontinuing leflunomide, it is recommended that all females of reproductive potential undergo an accelerated drug elimination procedure. Women receiving leflunomide treatment who wish to become pregnant must discontinue leflunomide and undergo an accelerated drug elimination procedure, which includes verification that plasma concentrations of the active metabolite of leflunomide, teriflunomide, are less than 0.02 mg/L (0.02 mcg/mL). Based on animal data, human plasma concentrations of teriflunomide of less than 0.02 mg/L (0.02 mcg/mL) are expected to have minimal embryo-fetal risk [see Contraindications (4), Warnings and Precautions (5.3), and Use in Specific Populations (8.1)].
5.8 Peripheral Neuropathy
Cases of peripheral neuropathy have been reported in patients receiving leflunomide and in clinical studies with teriflunomide, the active metabolite of leflunomide. Most patients recovered after discontinuation of treatment, but some patients had persistent symptoms. Age older than 60 years, concomitant neurotoxic medications, and diabetes may increase the risk for peripheral neuropathy. If a patient taking leflunomide develops a peripheral neuropathy, consider discontinuing leflunomide therapy and performing an accelerated drug elimination procedure [see Warnings and Precautions (5.3)].
5 Warnings and Precautions
- After stopping leflunomide, it is recommended that an accelerated drug elimination procedure be used to reduce the plasma concentrations of the active metabolite, teriflunomide. (5.3)
- Severe infections (including sepsis), pancytopenia, agranulocytosis, and thrombocytopenia: Stop leflunomide and use accelerated elimination procedure. Do not start leflunomide in patients with active infection. Monitor CBCs during treatment with leflunomide. (5.4)
- Stevens-Johnson syndrome and toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS): Stop leflunomide and use accelerated elimination procedure. (5.5)
- Skin ulcers: If skin ulcer is suspected, discontinue leflunomide treatment and consider an accelerated drug elimination procedure. (5.6)
- Peripheral neuropathy: If patient develops symptoms consistent with peripheral neuropathy, evaluate patient and consider discontinuing leflunomide. (5.8)
- Interstitial lung disease: May be fatal. New onset or worsening symptoms may necessitate discontinuation of leflunomide and initiation of accelerated elimination procedure. (5.9)
- Increased blood pressure: Monitor and treat. (5.11)
2 Dosage and Administration
- Loading dosage for patients at low risk for leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression: 100 mg daily for 3 days. (2.1)
- Maintenance dosage: 20 mg daily. (2.1)
- Screen patients for active and latent tuberculosis, pregnancy test (females), blood pressure, and laboratory tests before starting leflunomide. (2.2)
3 Dosage Forms and Strengths
Leflunomide Tablets, USP are available in two strengths:
- Tablets: 10 mg, supplied as white to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side.
- Tablets: 20 mg, supplied as white to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post approval use of leflunomide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System: agranulocytosis, leukopenia, neutropenia, pancytopenia
Infection: opportunistic infections, severe infections including sepsis
Gastrointestinal: acute hepatic necrosis, colitis, including microscopic colitis, hepatitis, jaundice/cholestasis, pancreatitis, severe liver injury such as hepatic failure
Immune System: angioedema
Nervous system: peripheral neuropathy
Respiratory: interstitial lung disease, including interstitial pneumonitis and pulmonary fibrosis, which may be fatal, pulmonary hypertension
Skin and Appendages: erythema multiforme, vasculitis including cutaneous necrotizing vasculitis, cutaneous lupus erythematosus, pustular psoriasis or worsening psoriasis
5.9 Interstitial Lung Disease
Interstitial lung disease and worsening of pre-existing interstitial lung disease have been reported during treatment with leflunomide and has been associated with fatal outcomes [see Adverse Reactions (6.2)]. The risk of leflunomide-associated interstitial lung disease is increased in patients with a history of interstitial lung disease. Interstitial lung disease is a potentially fatal disorder that may occur acutely at any time during therapy and has a variable clinical presentation. New onset or worsening pulmonary symptoms, such as cough and dyspnea, with or without associated fever, may be a reason for discontinuation of leflunomide therapy and for further investigation as appropriate. If discontinuation of leflunomide is necessary, consider performing an accelerated drug elimination procedure [see Warnings and Precautions (5.3)].
8 Use in Specific Populations
5.11 Blood Pressure Monitoring
In placebo-controlled studies with the active metabolite of leflunomide, teriflunomide, elevations in blood pressure were observed in some subjects. Blood pressure should be checked before starting treatment with leflunomide and monitored periodically thereafter [see Adverse Reactions (6.1)].
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
In clinical studies (Trials 1, 2, and 3), 1,865 patients were treated with leflunomide administered as either monotherapy or in combination with methotrexate or sulfasalazine. Patients ranged in age from 19 to 85 years, with an overall median age of 58 years. The mean duration of RA was 6 years ranging from 0 to 45 years.
16 How Supplied/storage and Handling
How Supplied
Leflunomide Tablets, USP
| Strength | Quantity | NDC Number | Description |
| 10 mg | 30 count bottle | 35573-447-30 | White to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side. |
| 20 mg | 30 count bottle | 35573-448-30 | White to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side. |
Principal Display Panel 10 Mg Carton
NDC 35573-447-30
burelpharma
Leflunomide
Tablets, USP
10mg
30 Tablets
Rx only
Principal Display Panel 20 Mg Carton
NDC 35573-448-30
burelpharma
Leflunomide
Tablets, USP
20mg
30 Tablets
Rx only
8.3 Females and Males of Reproductive Potential
Leflunomide may cause fetal harm if administered during pregnancy [see Use in Specific Populations (8.1)].
5.7 Malignancy and Lymphoproliferative Disorders
The risk of malignancy, particularly lymphoproliferative disorders, is increased with the use of some immunosuppression medications. There is a potential for immunosuppression with leflunomide. No apparent increase in the incidence of malignancies and lymphoproliferative disorders was reported in the clinical trials of leflunomide, but larger dosages and longer-term studies would be needed to determine whether there is an increased risk of malignancy or lymphoproliferative disorders with leflunomide.
Warning: Embryo Fetal Toxicity and Hepatotoxicity
WARNING: EMBRYO-FETAL TOXICITY and HEPATOTOXICITY
See full prescribing information for complete boxed warning.
Embryo-Fetal Toxicity
- Teratogenicity and embryo-lethality occurred in animals administered leflunomide (5.1, 8.1)
- Exclude pregnancy prior to initiating leflunomide therapy. (5.1, 8.3)
- Advise use of effective contraception in females of reproductive potential during treatment and during a drug elimination procedure. (5.1, 5.3, 8.3)
- Stop leflunomide and use accelerated drug elimination procedure if the patient becomes pregnant. (5.1, 5.3, 8.1)
Hepatotoxicity
- Severe liver injury and fatal liver failure have been reported. (5.2)
- Avoid leflunomide use in patients with pre-existing liver disease, or those with serum alanine aminotransferase (ALT) >2 × ULN. (5.2, 8.6)
- Use caution when leflunomide is given with other potentially hepatotoxic drugs. (5.2)
- Monitor ALT levels. Interrupt leflunomide treatment if ALT elevation >3-fold ULN. If likely leflunomide-induced, start accelerated drug elimination procedure and monitor liver tests weekly until normalized. (5.2, 5.3)
2.2 Evaluation and Testing Prior to Starting Leflunomide
Prior to starting leflunomide treatment, the following evaluations and tests are recommended:
- Evaluate patients for active tuberculosis and screen patients for latent tuberculosis infection [see Warnings and Precautions (5.4)]
- Laboratory tests including serum alanine aminotransferase (ALT); and white blood cell, hemoglobin or hematocrit, and platelet counts [see Warnings and Precautions (5.2, 5.4)]
- For females of reproductive potential, pregnancy testing [see Warnings and Precautions (5.1)]
- Check blood pressure [see Warnings and Precautions (5.11)]
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenicity was observed in a 2-year bioassay in rats at oral doses of leflunomide up to the maximally tolerated dose of 6 mg/kg (approximately 1/40 the maximum human teriflunomide systemic exposure based on AUC). However, male mice in a 2-year bioassay exhibited an increased incidence in lymphoma at an oral dose of 15 mg/kg, the highest dose studied (1.7 times the human teriflunomide exposure based on AUC). Female mice, in the same study, exhibited a dose-related increased incidence of bronchoalveolar adenomas and carcinomas combined beginning at 1.5 mg/kg (approximately 1/10 the human teriflunomide exposure based on AUC). The significance of the findings in mice relative to the clinical use of leflunomide is not known.
Leflunomide was not mutagenic in the Ames assay, the unscheduled DNA synthesis assay, or in the HGPRT gene mutation assay. In addition, leflunomide was not clastogenic in the in vivo mouse micronucleus assay or in the in vivo Chinese hamster bone marrow cell cytogenic test. However, 4-trifluoromethylaniline (TFMA), a minor metabolite of leflunomide, was mutagenic in the Ames assay and in the HGPRT gene mutation assay, and was clastogenic in the in vitro Chinese hamster cell chromosomal aberration assay. TFMA was not clastogenic in the in vivo mouse micronucleus assay or in the in vivo Chinese hamster bone marrow cell cytogenic test.
Leflunomide had no effect on fertility or reproductive performance in either male or female rats at oral doses up to 4.0 mg/kg (approximately 1/30 the human teriflunomide exposure based on AUC) [see Use in Specific Populations (8.1, 8.6)].
5.4 Immunosuppression, Bone Marrow Suppression, and Risk of Serious Infections
Leflunomide is not recommended for patients with severe immunodeficiency, bone marrow dysplasia, or severe, uncontrolled infections. If a serious infection occurs, consider interrupting leflunomide therapy and initiating the accelerated drug elimination procedure [see Warnings and Precautions (5.3)]. Medications like leflunomide that have immunosuppression potential may cause patients to be more susceptible to infections, including opportunistic infections, especially Pneumocystis jirovecii pneumonia, tuberculosis (including extra-pulmonary tuberculosis), and aspergillosis. Severe infections including sepsis, which may be fatal, have been reported in patients receiving leflunomide, especially Pneumocystis jirovecii pneumonia and aspergillosis. Most of the reports were confounded by concomitant immunosuppressant therapy and/or comorbid illness which, in addition to rheumatoid arthritis, may predispose patients to infection.
Cases of tuberculosis were observed in clinical studies with teriflunomide, the metabolite of leflunomide. Prior to initiating leflunomide, all patients should be screened for active and inactive ("latent") tuberculosis infection as per commonly used diagnostic tests. Leflunomide has not been studied in patients with a positive tuberculosis screen, and the safety of leflunomide in individuals with latent tuberculosis infection is unknown. Patients testing positive in tuberculosis screening should be treated by standard medical practice prior to therapy with leflunomide and monitored carefully during leflunomide treatment for possible reactivation of the infection.
Pancytopenia, agranulocytosis and thrombocytopenia have been reported in patients receiving leflunomide alone. These events have been reported most frequently in patients who received concomitant treatment with methotrexate or other immunosuppressive agents, or who had recently discontinued these therapies; in some cases, patients had a prior history of a significant hematologic abnormality.
Patients taking leflunomide should have platelet, white blood cell count and hemoglobin or hematocrit monitored at baseline and monthly for six months following initiation of therapy and every 6 to 8 weeks thereafter. If used with concomitant methotrexate and/or other potential immunosuppressive agents, chronic monitoring should be monthly. If evidence of bone marrow suppression occurs in a patient taking leflunomide, stop treatment with leflunomide and perform an accelerated drug elimination procedure to reduce the plasma concentration of the leflunomide active metabolite, teriflunomide [see Warnings and Precautions (5.3)].
In any situation in which the decision is made to switch from leflunomide to another antirheumatic agent with a known potential for hematologic suppression, it would be prudent to monitor for hematologic toxicity, because there will be overlap of systemic exposure to both compounds.
5.3 Procedure for Accelerated Elimination of Leflunomide and Its Active Metabolite
The active metabolite of leflunomide, teriflunomide, is eliminated slowly from the plasma [see Clinical Pharmacology (12.3)].
Use of an accelerated drug elimination procedure will rapidly reduce plasma concentrations of leflunomide and its active metabolite, teriflunomide. Therefore, an accelerated elimination procedure should be considered at any time after discontinuation of leflunomide, and in particular, when a patient has experienced a severe adverse reaction (e.g., hepatotoxicity, serious infection, bone marrow suppression, Stevens-Johnson Syndrome, toxic epidermal necrolysis, peripheral neuropathy, interstitial lung disease), suspected hypersensitivity, or has become pregnant. It is recommended that all women of childbearing potential undergo an accelerated elimination procedure after stopping leflunomide treatment.
Without use of an accelerated drug elimination procedure, it may take up to 2 years to reach plasma teriflunomide concentrations of less than 0.02 mg/L, the plasma concentration not associated with embryo-fetal toxicity in animals.
Elimination can be accelerated by the following procedures:
- Administer cholestyramine 8 grams orally 3 times daily for 11 days.
- Alternatively, administer 50 grams of activated charcoal powder (made into a suspension) orally every 12 hours for 11 days.
Verify plasma teriflunomide concentrations of less than 0.02 mg/L (0.02 µg/mL) by two separate tests at least 14 days apart. If plasma teriflunomide concentrations are higher than 0.02 mg/L, repeat cholestyramine and/or activated charcoal treatment.
The duration of accelerated drug elimination treatment may be modified based on the clinical status and tolerability of the elimination procedure. The procedure may be repeated as needed, based on teriflunomide concentrations and clinical status.
Use of the accelerated drug elimination procedure may potentially result in return of disease activity if the patient had been responding to leflunomide treatment.
5.5 Stevens Johnson Syndrome, Toxic Epidermal Necrolysis, and Drug Reactions With Eosinophilia and Systemic Symptoms
Rare cases of Stevens-Johnson syndrome and toxic epidermal necrolysis, and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported in patients receiving leflunomide. If a patient taking leflunomide develops any of these conditions, stop leflunomide treatment and perform an accelerated drug elimination procedure [see Warnings and Precautions (5.3)].
Structured Label Content
Section 42229-5 (42229-5)
Embryo-Fetal Toxicity
Leflunomide is contraindicated for use in pregnant women because of the potential for fetal harm. Teratogenicity and embryo-lethality occurred in animals administered leflunomide at doses lower than the human exposure level. Exclude pregnancy before the start of treatment with leflunomide in females of reproductive potential. Advise females of reproductive potential to use effective contraception during leflunomide treatment and during an accelerated drug elimination procedure after leflunomide treatment. Stop leflunomide and use an accelerated drug elimination procedure if the patient becomes pregnant [see Contraindications (4), Warnings and Precautions (5.1, 5.3), Use in Specific Populations (8.1, 8.3), and Clinical Pharmacology (12.3)].
Section 43683-2 (43683-2)
Warnings and Precautions, Skin Ulcers (5.6)........................06/2024
Section 44425-7 (44425-7)
TAMPER EVIDENT: Do not use if ring below the closure is broken or missing.
CHILD RESISTANT: This package features a child-resistant closure.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from light.
10 Overdosage (10 OVERDOSAGE)
There have been reports of chronic overdose in patients taking leflunomide at daily dose up to five times the recommended daily dose and reports of acute overdose in adults and children. Adverse events were consistent with the safety profile for leflunomide [see Adverse Reactions (6)]. The most frequent adverse events observed were diarrhea, abdominal pain, leukopenia, anemia, and elevated liver function tests.
In the event of a significant overdose or toxicity, perform an accelerated drug elimination procedure to accelerate elimination [see Warnings and Precautions (5.3)].
Studies with both hemodialysis and CAPD (chronic ambulatory peritoneal dialysis) indicate that teriflunomide, the primary metabolite of leflunomide, is not dialyzable [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Leflunomide is a pyrimidine synthesis inhibitor. The chemical name for leflunomide is N-(4´-trifluoromethylphenyl)-5-methylisoxazole-4-carboxamide. It has an empirical formula C12H9F3N2O2, a molecular weight of 270.2 and the following structural formula:
Leflunomide is available for oral administration as tablets containing 10 or 20 mg of active drug. Combined with leflunomide are the following inactive ingredients: lactose monohydrate, hydroxypropyl cellulose, tartaric acid, sodium lauryl sulfate, magnesium stearate, opadry white, polyvinyl alcohol, titanium dioxide, talc, lecithin, and xanthan gum.
5.6 Skin Ulcers
Skin ulcers may occur in patients during therapy with leflunomide. If leflunomide-associated skin ulcer is suspected or if skin ulcers persist despite appropriate therapy, leflunomide discontinuation and an accelerated drug elimination procedure should be considered [see Warnings and Precautions (5.3)]. The decision to resume leflunomide following skin ulcers should be based on clinical judgment of adequate wound healing.
5.10 Vaccinations
No clinical data are available on the efficacy and safety of vaccinations during leflunomide treatment. Vaccination with live vaccines is, however, not recommended. The long half-life of the active metabolite of leflunomide should be considered when contemplating administration of a live vaccine after stopping leflunomide.
8.4 Pediatric Use
The safety and effectiveness of leflunomide in pediatric patients have not been established.
The safety and effectiveness of leflunomide in the treatment of polyarticular course juvenile idiopathic arthritis (JIA) was evaluated in a single multicenter, double-blind, active-controlled trial in 94 pediatric patients (1:1 randomization) with polyarticular course juvenile idiopathic arthritis (JIA) as defined by the American College of Rheumatology (ACR). In this population, leflunomide treatment was found not to be effective.
The safety of leflunomide was studied in 74 patients with polyarticular course JIA ranging in age from 3–17 years (47 patients from the active-controlled study and 27 from an open-label safety and pharmacokinetic study). The most common adverse events included abdominal pain, diarrhea, nausea, vomiting, oral ulcers, upper respiratory tract infections, alopecia, rash, headache, and dizziness. Less common adverse events included anemia, hypertension, and weight loss. Fourteen pediatric patients experienced ALT and/or AST elevations, nine between 1.2 and 3-fold the upper limit of normal, and five between 3 and 8-fold the upper limit of normal.
8.5 Geriatric Use
Of the total number of subjects in controlled clinical trials (Trials 1, 2, and 3) of leflunomide, 234 subjects were 65 years and over [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dosage adjustment is needed in patients over 65.
5.2 Hepatotoxicity
Severe liver injury, including fatal liver failure, has been reported in some patients treated with leflunomide. Patients with pre-existing acute or chronic liver disease, or those with serum alanine aminotransferase (ALT) of greater than twice the upper limits of normal (>2 × ULN) before initiating treatment, should not be treated with leflunomide. Use caution when leflunomide is given with other potentially hepatotoxic drugs. Monitoring of ALT levels is recommended at least monthly for six months after starting leflunomide, and thereafter every 6 to 8 weeks. If ALT elevation >3-fold ULN occurs, interrupt leflunomide therapy and investigate the cause. If likely leflunomide-induced, perform the accelerated drug elimination procedure and monitor liver tests weekly until normalized [see Warnings and Precautions (5.3)]. If leflunomide-induced liver injury is unlikely because some other cause has been found, resumption of leflunomide therapy may be considered.
If leflunomide and methotrexate are given concomitantly, follow the American College of Rheumatology (ACR) guidelines for monitoring methotrexate liver toxicity with ALT, AST, and serum albumin testing.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of leflunomide in the treatment of rheumatoid arthritis (RA) was demonstrated in three controlled trials showing reduction in signs and symptoms, and inhibition of structural damage. In two placebo-controlled trials, efficacy was demonstrated for improvement in physical function. In these trials, efficacy was evaluated by:
1. Reduction of signs and symptoms
Relief of signs and symptoms was assessed using the American College of Rheumatology (ACR) 20 Responder Index, a composite of clinical, laboratory, and functional measures in rheumatoid arthritis. An "ACR20 Responder" is a patient who had ≥20% improvement in both tender and swollen joint counts and in 3 of the following 5 criteria: physician global assessment, patient global assessment, functional ability measure (Modified Health Assessment Questionnaire [MHAQ]), visual analog pain scale, and erythrocyte sedimentation rate or C-reactive protein. An "ACR20 Responder at Endpoint" is a patient who completed the study and was an ACR20 Responder at the completion of the study.
2. Inhibition of structural damage
Inhibition of structural damage compared to control was assessed using the Sharp score, a composite score of x-ray erosions and joint space narrowing in hands/wrists and forefeet.
3. Improvement in physical function
Improvement in physical function was assessed using the Health Assessment Questionnaire (HAQ) and the Medical Outcomes Survey Short Form (SF-36).
In all leflunomide trials, participants of at least 18 years of age and in ARA functional class of I, II, or III received an initial loading dose of 100 mg leflunomide per day for three days, followed by 20 mg per day thereafter.
Exclusion criteria included patients with a history of hypersensitivity to the study medication; women who were pregnant or breastfeeding and men or women of child bearing age and potential who had not received contraceptives for at least 4 weeks before entering the study and to be maintained throughout the study and for at least 6 months after discontinuing treatment; patients with a history of inflammatory disease, impaired renal function or liver impairment, cardiac failure, congenital or acquired immunodeficiency, impaired coagulation, or a history of recent major traumatic injury; and patients taking intra-articular or systemic concomitant medications which could affect the safety and/or efficacy of the study medication.
4 Contraindications (4 CONTRAINDICATIONS)
Leflunomide is contraindicated in:
- Pregnant women. Leflunomide may cause fetal harm. If a woman becomes pregnant while taking this drug, stop leflunomide, apprise the patient of the potential hazard to the fetus, and begin a drug elimination procedure [see Warnings and Precautions (5.1, 5.3) and Use in Specific Populations (8.1)].
- Patients with severe hepatic impairment [see Warnings and Precautions (5.2)].
- Patients with known hypersensitivity to leflunomide or any of the other components of leflunomide. Known reactions include anaphylaxis [see Adverse Reactions (6.1)].
- Patients being treated with teriflunomide [see Drug Interactions (7)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Immunosuppression [see Warnings and Precautions (5.4)]
- Bone marrow suppression [see Warnings and Precautions (5.4)]
- Serious infections [see Warnings and Precautions (5.4)]
- Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug reactions with eosinophilia and systemic symptoms [see Warnings and Precautions (5.5)]
- Skin ulcers [see Warnings and Precautions (5.6)]
- Peripheral neuropathy [see Warnings and Precautions (5.8)]
- Interstitial lung disease [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Following oral administration, leflunomide is metabolized to an active metabolite, teriflunomide, which is responsible for essentially all of leflunomide's in vivo activity. Drug interaction studies have been conducted with both leflunomide and with its active metabolite, teriflunomide, where the metabolite was directly administered to the test subjects.
8.7 Renal Impairment
Dedicated studies of the effect of renal impairment on leflunomide pharmacokinetics have not been conducted. Given that the kidney plays an important role in drug elimination, caution should be used when leflunomide is administered to these patients.
12.3 Pharmacokinetics
Following oral administration, leflunomide is metabolized to an active metabolite, teriflunomide, which is responsible for essentially all of leflunomide's in vivo activity. Plasma concentrations of the parent drug, leflunomide, have been occasionally seen at very low concentrations. Studies of the pharmacokinetics of leflunomide have primarily examined the plasma concentrations of the active metabolite, teriflunomide.
2.1 Recommended Dosage
The recommended dosage of leflunomide is 20 mg once daily. Treatment may be initiated with or without a loading dose, depending upon the patient's risk of leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression. The loading dose provides steady-state concentrations more rapidly.
- For patients who are at low risk for leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression, the recommended leflunomide loading dose is 100 mg once daily for 3 days. Subsequently administer 20 mg once daily.
- For patients at high risk for leflunomide-associated hepatotoxicity (e.g., those taking concomitant methotrexate) or leflunomide-associated myelosuppression (e.g., patients taking concomitant immunosuppressants), the recommended leflunomide dose is 20 mg once daily without a loading dose [see Warnings and Precautions (5.2, 5.4)].
The maximum recommended daily dose is 20 mg once per day. Consider dosage reduction to 10 mg once daily for patients who are not able to tolerate 20 mg daily (i.e., for patients who experience any adverse events listed in Table 1).
Monitor patients carefully after dosage reduction and after stopping therapy with leflunomide, since the active metabolite of leflunomide, teriflunomide, is slowly eliminated from the plasma [see Clinical Pharmacology (12.3)]. After stopping leflunomide treatment, an accelerated drug elimination procedure is recommended to reduce the plasma concentrations of the active metabolite, teriflunomide [see Warnings and Precautions (5.3)]. Without use of an accelerated drug elimination procedure, it may take up to 2 years to reach undetectable plasma teriflunomide concentrations after stopping leflunomide [see Clinical Pharmacology (12.3)].
8.6 Hepatic Impairment
Dedicated studies of the effect of hepatic impairment on leflunomide pharmacokinetics have not been conducted. Given the need to metabolize leflunomide into the active species, the role of the liver in drug elimination/recycling, and the possible risk of increased hepatic toxicity, the use of leflunomide in patients with hepatic impairment is not recommended.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Leflunomide Tablets, USP are indicated for the treatment of adults with active rheumatoid arthritis (RA).
12.1 Mechanism of Action
Leflunomide is an isoxazole immunomodulatory agent that inhibits dihydroorotate dehydrogenase (a mitochondrial enzyme involved in de novo pyrimidine synthesis) and has antiproliferative activity. Several in vivo and in vitro experimental models have demonstrated an anti-inflammatory effect.
5.1 Embryo Fetal Toxicity (5.1 Embryo-Fetal Toxicity)
Leflunomide may cause fetal harm when administered to a pregnant woman. Teratogenicity and embryo-lethality occurred in animal reproduction studies with leflunomide at doses lower than the human exposure level [see Use in Specific Populations (8.1)].
Leflunomide is contraindicated for use in pregnant women [see Contraindications (4)]. Exclude pregnancy before starting treatment with leflunomide in females of reproductive potential [see Dosage and Administration (2.2)]. Advise females of reproductive potential to use effective contraception during leflunomide treatment and during an accelerated drug elimination procedure after leflunomide treatment [see Use in Specific Populations (8.3)]. If a woman becomes pregnant while taking leflunomide, stop treatment with leflunomide, apprise the patient of the potential risk to a fetus, and perform an accelerated drug elimination procedure to achieve nondetectable plasma concentrations of teriflunomide, the active metabolite of leflunomide [see Warnings and Precautions (5.3)].
Upon discontinuing leflunomide, it is recommended that all females of reproductive potential undergo an accelerated drug elimination procedure. Women receiving leflunomide treatment who wish to become pregnant must discontinue leflunomide and undergo an accelerated drug elimination procedure, which includes verification that plasma concentrations of the active metabolite of leflunomide, teriflunomide, are less than 0.02 mg/L (0.02 mcg/mL). Based on animal data, human plasma concentrations of teriflunomide of less than 0.02 mg/L (0.02 mcg/mL) are expected to have minimal embryo-fetal risk [see Contraindications (4), Warnings and Precautions (5.3), and Use in Specific Populations (8.1)].
5.8 Peripheral Neuropathy
Cases of peripheral neuropathy have been reported in patients receiving leflunomide and in clinical studies with teriflunomide, the active metabolite of leflunomide. Most patients recovered after discontinuation of treatment, but some patients had persistent symptoms. Age older than 60 years, concomitant neurotoxic medications, and diabetes may increase the risk for peripheral neuropathy. If a patient taking leflunomide develops a peripheral neuropathy, consider discontinuing leflunomide therapy and performing an accelerated drug elimination procedure [see Warnings and Precautions (5.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- After stopping leflunomide, it is recommended that an accelerated drug elimination procedure be used to reduce the plasma concentrations of the active metabolite, teriflunomide. (5.3)
- Severe infections (including sepsis), pancytopenia, agranulocytosis, and thrombocytopenia: Stop leflunomide and use accelerated elimination procedure. Do not start leflunomide in patients with active infection. Monitor CBCs during treatment with leflunomide. (5.4)
- Stevens-Johnson syndrome and toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS): Stop leflunomide and use accelerated elimination procedure. (5.5)
- Skin ulcers: If skin ulcer is suspected, discontinue leflunomide treatment and consider an accelerated drug elimination procedure. (5.6)
- Peripheral neuropathy: If patient develops symptoms consistent with peripheral neuropathy, evaluate patient and consider discontinuing leflunomide. (5.8)
- Interstitial lung disease: May be fatal. New onset or worsening symptoms may necessitate discontinuation of leflunomide and initiation of accelerated elimination procedure. (5.9)
- Increased blood pressure: Monitor and treat. (5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Loading dosage for patients at low risk for leflunomide-associated hepatotoxicity and leflunomide-associated myelosuppression: 100 mg daily for 3 days. (2.1)
- Maintenance dosage: 20 mg daily. (2.1)
- Screen patients for active and latent tuberculosis, pregnancy test (females), blood pressure, and laboratory tests before starting leflunomide. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Leflunomide Tablets, USP are available in two strengths:
- Tablets: 10 mg, supplied as white to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side.
- Tablets: 20 mg, supplied as white to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post approval use of leflunomide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System: agranulocytosis, leukopenia, neutropenia, pancytopenia
Infection: opportunistic infections, severe infections including sepsis
Gastrointestinal: acute hepatic necrosis, colitis, including microscopic colitis, hepatitis, jaundice/cholestasis, pancreatitis, severe liver injury such as hepatic failure
Immune System: angioedema
Nervous system: peripheral neuropathy
Respiratory: interstitial lung disease, including interstitial pneumonitis and pulmonary fibrosis, which may be fatal, pulmonary hypertension
Skin and Appendages: erythema multiforme, vasculitis including cutaneous necrotizing vasculitis, cutaneous lupus erythematosus, pustular psoriasis or worsening psoriasis
5.9 Interstitial Lung Disease
Interstitial lung disease and worsening of pre-existing interstitial lung disease have been reported during treatment with leflunomide and has been associated with fatal outcomes [see Adverse Reactions (6.2)]. The risk of leflunomide-associated interstitial lung disease is increased in patients with a history of interstitial lung disease. Interstitial lung disease is a potentially fatal disorder that may occur acutely at any time during therapy and has a variable clinical presentation. New onset or worsening pulmonary symptoms, such as cough and dyspnea, with or without associated fever, may be a reason for discontinuation of leflunomide therapy and for further investigation as appropriate. If discontinuation of leflunomide is necessary, consider performing an accelerated drug elimination procedure [see Warnings and Precautions (5.3)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.11 Blood Pressure Monitoring
In placebo-controlled studies with the active metabolite of leflunomide, teriflunomide, elevations in blood pressure were observed in some subjects. Blood pressure should be checked before starting treatment with leflunomide and monitored periodically thereafter [see Adverse Reactions (6.1)].
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
In clinical studies (Trials 1, 2, and 3), 1,865 patients were treated with leflunomide administered as either monotherapy or in combination with methotrexate or sulfasalazine. Patients ranged in age from 19 to 85 years, with an overall median age of 58 years. The mean duration of RA was 6 years ranging from 0 to 45 years.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
Leflunomide Tablets, USP
| Strength | Quantity | NDC Number | Description |
| 10 mg | 30 count bottle | 35573-447-30 | White to off-white, round, biconvex film-coated tablets debossed with "10" on one side and "LFL" on the other side. |
| 20 mg | 30 count bottle | 35573-448-30 | White to off-white, round, biconvex film-coated tablets debossed with "20" on one side and "LFL" on the other side. |
Principal Display Panel 10 Mg Carton (PRINCIPAL DISPLAY PANEL - 10 mg Carton)
NDC 35573-447-30
burelpharma
Leflunomide
Tablets, USP
10mg
30 Tablets
Rx only
Principal Display Panel 20 Mg Carton (PRINCIPAL DISPLAY PANEL - 20 mg Carton)
NDC 35573-448-30
burelpharma
Leflunomide
Tablets, USP
20mg
30 Tablets
Rx only
8.3 Females and Males of Reproductive Potential
Leflunomide may cause fetal harm if administered during pregnancy [see Use in Specific Populations (8.1)].
5.7 Malignancy and Lymphoproliferative Disorders
The risk of malignancy, particularly lymphoproliferative disorders, is increased with the use of some immunosuppression medications. There is a potential for immunosuppression with leflunomide. No apparent increase in the incidence of malignancies and lymphoproliferative disorders was reported in the clinical trials of leflunomide, but larger dosages and longer-term studies would be needed to determine whether there is an increased risk of malignancy or lymphoproliferative disorders with leflunomide.
Warning: Embryo Fetal Toxicity and Hepatotoxicity (WARNING: EMBRYO-FETAL TOXICITY and HEPATOTOXICITY)
WARNING: EMBRYO-FETAL TOXICITY and HEPATOTOXICITY
See full prescribing information for complete boxed warning.
Embryo-Fetal Toxicity
- Teratogenicity and embryo-lethality occurred in animals administered leflunomide (5.1, 8.1)
- Exclude pregnancy prior to initiating leflunomide therapy. (5.1, 8.3)
- Advise use of effective contraception in females of reproductive potential during treatment and during a drug elimination procedure. (5.1, 5.3, 8.3)
- Stop leflunomide and use accelerated drug elimination procedure if the patient becomes pregnant. (5.1, 5.3, 8.1)
Hepatotoxicity
- Severe liver injury and fatal liver failure have been reported. (5.2)
- Avoid leflunomide use in patients with pre-existing liver disease, or those with serum alanine aminotransferase (ALT) >2 × ULN. (5.2, 8.6)
- Use caution when leflunomide is given with other potentially hepatotoxic drugs. (5.2)
- Monitor ALT levels. Interrupt leflunomide treatment if ALT elevation >3-fold ULN. If likely leflunomide-induced, start accelerated drug elimination procedure and monitor liver tests weekly until normalized. (5.2, 5.3)
2.2 Evaluation and Testing Prior to Starting Leflunomide
Prior to starting leflunomide treatment, the following evaluations and tests are recommended:
- Evaluate patients for active tuberculosis and screen patients for latent tuberculosis infection [see Warnings and Precautions (5.4)]
- Laboratory tests including serum alanine aminotransferase (ALT); and white blood cell, hemoglobin or hematocrit, and platelet counts [see Warnings and Precautions (5.2, 5.4)]
- For females of reproductive potential, pregnancy testing [see Warnings and Precautions (5.1)]
- Check blood pressure [see Warnings and Precautions (5.11)]
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenicity was observed in a 2-year bioassay in rats at oral doses of leflunomide up to the maximally tolerated dose of 6 mg/kg (approximately 1/40 the maximum human teriflunomide systemic exposure based on AUC). However, male mice in a 2-year bioassay exhibited an increased incidence in lymphoma at an oral dose of 15 mg/kg, the highest dose studied (1.7 times the human teriflunomide exposure based on AUC). Female mice, in the same study, exhibited a dose-related increased incidence of bronchoalveolar adenomas and carcinomas combined beginning at 1.5 mg/kg (approximately 1/10 the human teriflunomide exposure based on AUC). The significance of the findings in mice relative to the clinical use of leflunomide is not known.
Leflunomide was not mutagenic in the Ames assay, the unscheduled DNA synthesis assay, or in the HGPRT gene mutation assay. In addition, leflunomide was not clastogenic in the in vivo mouse micronucleus assay or in the in vivo Chinese hamster bone marrow cell cytogenic test. However, 4-trifluoromethylaniline (TFMA), a minor metabolite of leflunomide, was mutagenic in the Ames assay and in the HGPRT gene mutation assay, and was clastogenic in the in vitro Chinese hamster cell chromosomal aberration assay. TFMA was not clastogenic in the in vivo mouse micronucleus assay or in the in vivo Chinese hamster bone marrow cell cytogenic test.
Leflunomide had no effect on fertility or reproductive performance in either male or female rats at oral doses up to 4.0 mg/kg (approximately 1/30 the human teriflunomide exposure based on AUC) [see Use in Specific Populations (8.1, 8.6)].
5.4 Immunosuppression, Bone Marrow Suppression, and Risk of Serious Infections
Leflunomide is not recommended for patients with severe immunodeficiency, bone marrow dysplasia, or severe, uncontrolled infections. If a serious infection occurs, consider interrupting leflunomide therapy and initiating the accelerated drug elimination procedure [see Warnings and Precautions (5.3)]. Medications like leflunomide that have immunosuppression potential may cause patients to be more susceptible to infections, including opportunistic infections, especially Pneumocystis jirovecii pneumonia, tuberculosis (including extra-pulmonary tuberculosis), and aspergillosis. Severe infections including sepsis, which may be fatal, have been reported in patients receiving leflunomide, especially Pneumocystis jirovecii pneumonia and aspergillosis. Most of the reports were confounded by concomitant immunosuppressant therapy and/or comorbid illness which, in addition to rheumatoid arthritis, may predispose patients to infection.
Cases of tuberculosis were observed in clinical studies with teriflunomide, the metabolite of leflunomide. Prior to initiating leflunomide, all patients should be screened for active and inactive ("latent") tuberculosis infection as per commonly used diagnostic tests. Leflunomide has not been studied in patients with a positive tuberculosis screen, and the safety of leflunomide in individuals with latent tuberculosis infection is unknown. Patients testing positive in tuberculosis screening should be treated by standard medical practice prior to therapy with leflunomide and monitored carefully during leflunomide treatment for possible reactivation of the infection.
Pancytopenia, agranulocytosis and thrombocytopenia have been reported in patients receiving leflunomide alone. These events have been reported most frequently in patients who received concomitant treatment with methotrexate or other immunosuppressive agents, or who had recently discontinued these therapies; in some cases, patients had a prior history of a significant hematologic abnormality.
Patients taking leflunomide should have platelet, white blood cell count and hemoglobin or hematocrit monitored at baseline and monthly for six months following initiation of therapy and every 6 to 8 weeks thereafter. If used with concomitant methotrexate and/or other potential immunosuppressive agents, chronic monitoring should be monthly. If evidence of bone marrow suppression occurs in a patient taking leflunomide, stop treatment with leflunomide and perform an accelerated drug elimination procedure to reduce the plasma concentration of the leflunomide active metabolite, teriflunomide [see Warnings and Precautions (5.3)].
In any situation in which the decision is made to switch from leflunomide to another antirheumatic agent with a known potential for hematologic suppression, it would be prudent to monitor for hematologic toxicity, because there will be overlap of systemic exposure to both compounds.
5.3 Procedure for Accelerated Elimination of Leflunomide and Its Active Metabolite (5.3 Procedure for Accelerated Elimination of Leflunomide and its Active Metabolite)
The active metabolite of leflunomide, teriflunomide, is eliminated slowly from the plasma [see Clinical Pharmacology (12.3)].
Use of an accelerated drug elimination procedure will rapidly reduce plasma concentrations of leflunomide and its active metabolite, teriflunomide. Therefore, an accelerated elimination procedure should be considered at any time after discontinuation of leflunomide, and in particular, when a patient has experienced a severe adverse reaction (e.g., hepatotoxicity, serious infection, bone marrow suppression, Stevens-Johnson Syndrome, toxic epidermal necrolysis, peripheral neuropathy, interstitial lung disease), suspected hypersensitivity, or has become pregnant. It is recommended that all women of childbearing potential undergo an accelerated elimination procedure after stopping leflunomide treatment.
Without use of an accelerated drug elimination procedure, it may take up to 2 years to reach plasma teriflunomide concentrations of less than 0.02 mg/L, the plasma concentration not associated with embryo-fetal toxicity in animals.
Elimination can be accelerated by the following procedures:
- Administer cholestyramine 8 grams orally 3 times daily for 11 days.
- Alternatively, administer 50 grams of activated charcoal powder (made into a suspension) orally every 12 hours for 11 days.
Verify plasma teriflunomide concentrations of less than 0.02 mg/L (0.02 µg/mL) by two separate tests at least 14 days apart. If plasma teriflunomide concentrations are higher than 0.02 mg/L, repeat cholestyramine and/or activated charcoal treatment.
The duration of accelerated drug elimination treatment may be modified based on the clinical status and tolerability of the elimination procedure. The procedure may be repeated as needed, based on teriflunomide concentrations and clinical status.
Use of the accelerated drug elimination procedure may potentially result in return of disease activity if the patient had been responding to leflunomide treatment.
5.5 Stevens Johnson Syndrome, Toxic Epidermal Necrolysis, and Drug Reactions With Eosinophilia and Systemic Symptoms (5.5 Stevens-Johnson Syndrome, Toxic Epidermal Necrolysis, and Drug Reactions with Eosinophilia and Systemic Symptoms)
Rare cases of Stevens-Johnson syndrome and toxic epidermal necrolysis, and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported in patients receiving leflunomide. If a patient taking leflunomide develops any of these conditions, stop leflunomide treatment and perform an accelerated drug elimination procedure [see Warnings and Precautions (5.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:49.457913 · Updated: 2026-03-14T22:25:41.517877