Hydrocortisone
fbb876c0-a026-4eec-ae3e-f1d61c4661de
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Hydrocortisone 1.0%
Purpose
Anti-itch
Medication Information
Purpose
Anti-itch
Description
Hydrocortisone 1.0%
Uses
for the temporary relief of itching associated with minor skin irritations, inflammation and rashes due to:
- cosmetics
- soaps
- jewelry
- insect bites
- psoriasis
- eczema
- detergents
- seborrheic dermatitis
- external genital and anal itching
- poison ivy, poison oak, or poison sumac
Other uses of this product should be only under the advice and supervision of a doctor.
Section 42229-5
Made in the USA
for Qualitest Pharmaceuticals
Huntsville, AL 35811
Rev. 5/15 R6
8265556 0534
Directions
Adults and children 2 years of age and older
- apply to affected area not more than 3 to 4 times daily
Children under 2 years of age
- do not use. Consult a doctor.
For external anal itching
- adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly or by patting or blotting with toilet tissue or a soft cloth before application of this product
- children under 12 years of age with external anal itching: consult a doctor
Do Not Use
- for the treatment of diaper rash. Consult a doctor.
- for external genital itching if you have vaginal discharge. Consult a doctor.
Active Ingredient
Hydrocortisone 1.0%
Other Information
- store at 15° to 30°C (59° to 86°F)
- lot number and expiration date: See crimp of tube or box
You may report serious side effects to: 130 Vintage Drive, Huntsville, AL 35811.
Inactive Ingredients
cetyl alcohol, diazolidinyl urea, isopropyl palmitate, mineral oil/lanolin alcohol, polysorbate 40, propylene glycol, propylene glycol monostearate, purified water, sorbic acid, sorbitan monopalmitate, stearyl alcohol, xanthan gum
When Using This Product
- avoid contact with eyes
- do not use more than directed unless told to do so by a doctor
- do not put directly into the rectum by using fingers or any mechanical device or applicator
Stop Use and Ask A Doctor If
- condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days, and do not begin use of any other hydrocortisone product unless you have asked a doctor
- rectal bleeding occurs
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center immediately.
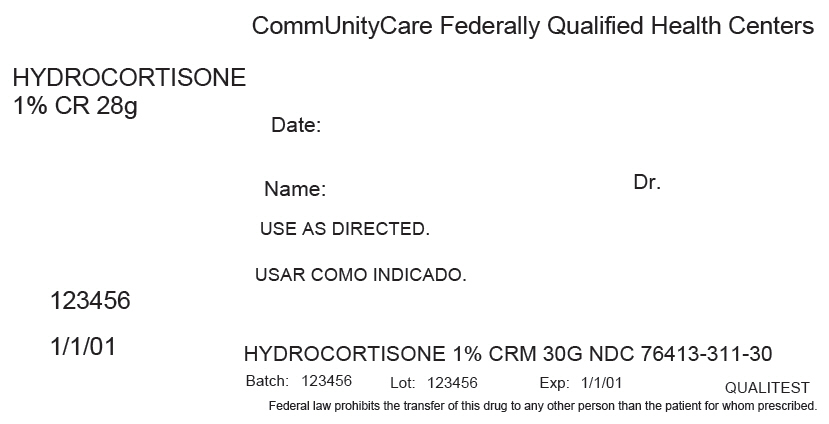
Principal Display Panel 28 G Tube Label
CommUnityCare Federally Qualified Health Centers
HYDROCORTISONE
1% CR 28g
Date:
Name:
Dr.
USE AS DIRECTED.
123456
1/1/01
HYDROCORTISONE 1% CRM 30G NDC 76413-311-30
Batch: 123456
Lot: 123456
Exp: 1/1/01
QUALITEST
Federal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
Structured Label Content
Uses
for the temporary relief of itching associated with minor skin irritations, inflammation and rashes due to:
- cosmetics
- soaps
- jewelry
- insect bites
- psoriasis
- eczema
- detergents
- seborrheic dermatitis
- external genital and anal itching
- poison ivy, poison oak, or poison sumac
Other uses of this product should be only under the advice and supervision of a doctor.
Section 42229-5 (42229-5)
Made in the USA
for Qualitest Pharmaceuticals
Huntsville, AL 35811
Rev. 5/15 R6
8265556 0534
Purpose
Anti-itch
Directions
Adults and children 2 years of age and older
- apply to affected area not more than 3 to 4 times daily
Children under 2 years of age
- do not use. Consult a doctor.
For external anal itching
- adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly or by patting or blotting with toilet tissue or a soft cloth before application of this product
- children under 12 years of age with external anal itching: consult a doctor
Do Not Use (Do not use)
- for the treatment of diaper rash. Consult a doctor.
- for external genital itching if you have vaginal discharge. Consult a doctor.
Active Ingredient (Active ingredient)
Hydrocortisone 1.0%
Other Information (Other information)
- store at 15° to 30°C (59° to 86°F)
- lot number and expiration date: See crimp of tube or box
You may report serious side effects to: 130 Vintage Drive, Huntsville, AL 35811.
Inactive Ingredients (Inactive ingredients)
cetyl alcohol, diazolidinyl urea, isopropyl palmitate, mineral oil/lanolin alcohol, polysorbate 40, propylene glycol, propylene glycol monostearate, purified water, sorbic acid, sorbitan monopalmitate, stearyl alcohol, xanthan gum
When Using This Product (When using this product)
- avoid contact with eyes
- do not use more than directed unless told to do so by a doctor
- do not put directly into the rectum by using fingers or any mechanical device or applicator
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days, and do not begin use of any other hydrocortisone product unless you have asked a doctor
- rectal bleeding occurs
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center immediately.
Principal Display Panel 28 G Tube Label (PRINCIPAL DISPLAY PANEL - 28 g Tube Label)
CommUnityCare Federally Qualified Health Centers
HYDROCORTISONE
1% CR 28g
Date:
Name:
Dr.
USE AS DIRECTED.
123456
1/1/01
HYDROCORTISONE 1% CRM 30G NDC 76413-311-30
Batch: 123456
Lot: 123456
Exp: 1/1/01
QUALITEST
Federal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:24.778455 · Updated: 2026-03-14T22:54:50.171645