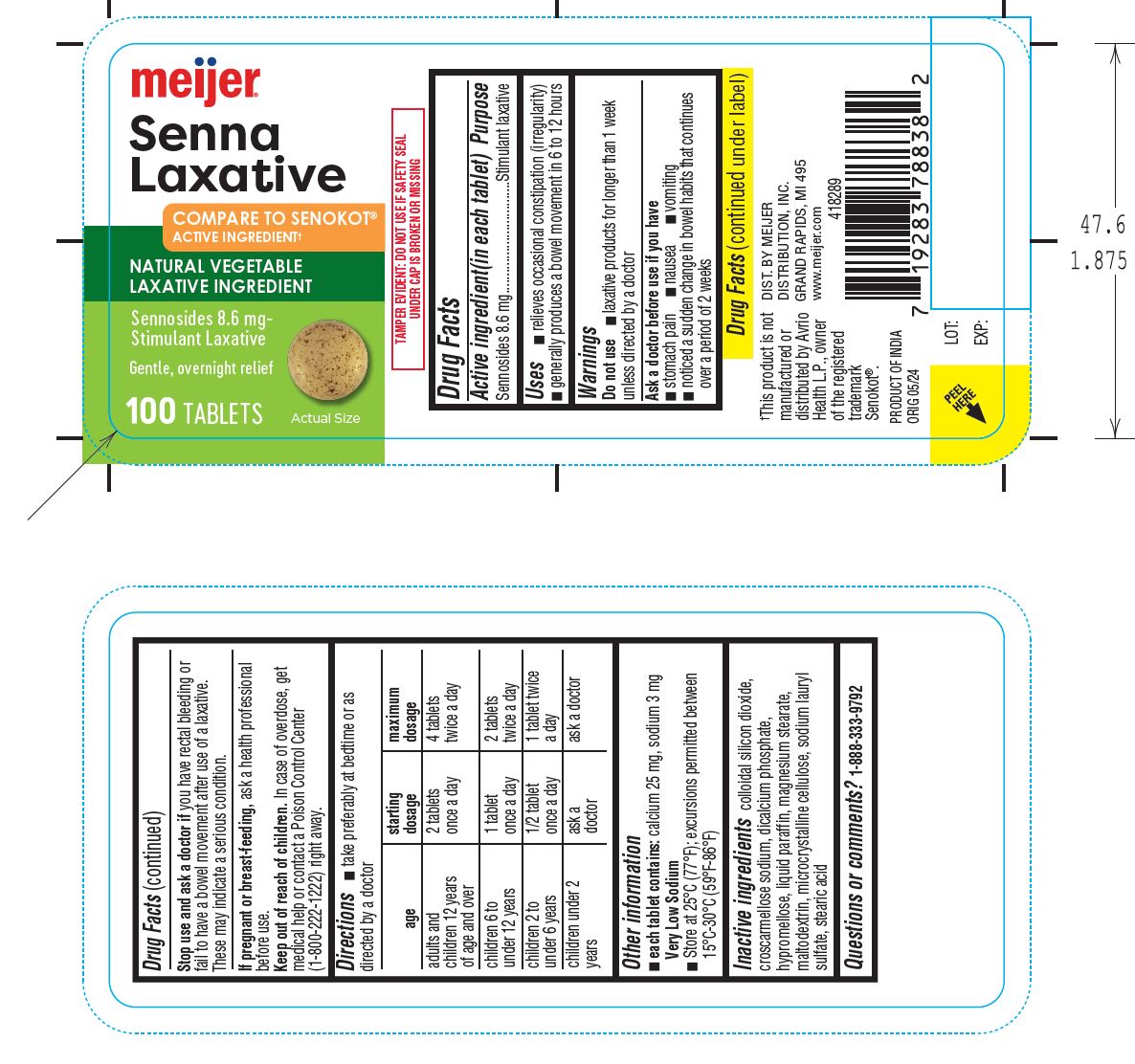
Senna Laxative Tablets
fb72816f-2dec-4dd9-a60c-9868b28297c8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Medication Information
Uses
Warnings
Purpose
Directions
Do Not Use
Drug Facts
Other Information
Inactive Ingredients
Principal Display Panel
If Pregnant Or Breast Feeding,
Keep Out of Reach of Children.
Stop Use and Ask A Doctor If
Active Ingredient(in Each Tablet)
Ask A Doctor Before Use If You Have
Questions Or Comments? 1 888 333 9792
Structured Label Content
Uses
Warnings
Purpose
Directions
Do Not Use (Do not use)
Drug Facts
Other Information (Other information)
Inactive Ingredients (Inactive ingredients)
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
Keep Out of Reach of Children. (Keep out of reach of children.)
Stop Use and Ask A Doctor If (Stop use and ask a doctor if )
Active Ingredient(in Each Tablet) (Active ingredient(in each tablet))
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
Questions Or Comments? 1 888 333 9792 (Questions or comments? 1-888-333-9792)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:00.140532 · Updated: 2026-03-14T23:06:16.981214