Zolpidem Tartrate Extended-release Tablets
facfc337-3447-4052-a9aa-8aa55282a3e3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Zolpidem Tartrate Extended-Release Tablets are indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance (as measured by wake time after sleep onset). The clinical trials performed in support of efficacy were up to 3 weeks (using polysomnography measurement up to 2 weeks in both adult and elderly patients) and 24 weeks (using patient-reported assessment in adult patients only) in duration [see Clinical Studies (14) ] .
Dosage and Administration
• Use the lowest dose effective for the patient and must not exceed a total of 12.5 mg daily ( 2.1 ) • Recommended initial dose is a single dose of 6.25 mg for women, and a single dose of 6.25 or 12.5 mg for men, immediately before bedtime with at least 7–8 hours remaining before the planned time of awakening ( 2.1 ) • Geriatric patients and patients with mild to moderate hepatic impairment: Recommended dose is 6.25 mg for men and women ( 2.2 ) • Lower doses of CNS depressants may be necessary when taken concomitantly with Zolpidem Tartrate Extended-Release Tablets ( 2.3 ) • Tablets to be swallowed whole, not to be crushed, divided or chewed ( 2.4 ) • The effect of Zolpidem Tartrate Extended-Release Tablets may be slowed if taken with or immediately after a meal ( 2.4 )
Contraindications
Zolpidem Tartrate Extended-Release Tablets are contraindicated in patients with known hypersensitivity to zolpidem. Observed reactions include anaphylaxis and angioedema [see Warnings and Precautions (5.3) ].
Warnings and Precautions
• CNS depressant effects: Impaired alertness and motor coordination, including risk of morning impairment. Caution patients against driving and other activities requiring complete mental alertness the morning after use. ( 5.1 ) • Need to evaluate for co-morbid diagnoses: Revaluate if insomnia persists after 7 to 10 days of use. ( 5.2 ) • Severe anaphylactic/anaphylactoid reactions: Angioedema and anaphylaxis have been reported. Do not rechallenge if such reactions occur. ( 5.3 ) • "Sleep-driving" and other complex behaviors while not fully awake. Risk increases with dose and use with other CNS depressants and alcohol. Immediately evaluate any new onset behavioral changes. ( 5.4 ) • Depression: Worsening of depression or, suicidal thinking may occur. Prescribe the least amount of tablets feasible to avoid intentional overdose. ( 5. 5 ) • Respiratory Depression: Consider this risk before prescribing in patients with compromised respiratory function. ( 5. 6 ) • Hepatic Impairment: Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment. ( 5.7 ) • Withdrawal effects: Symptoms may occur with rapid dose reduction or discontinuation. ( 5.8 , 9.3 ) • Severe Injuries: Drowsiness may lead to fall including severe injuries. ( 5.9 )
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: • CNS-depressant effects and next-day impairment [see Warnings and Precautions (5.1) ] • Serious anaphylactic and anaphylactoid reactions [see Warnings and Precautions (5.3) ] • Abnormal thinking and behavior changes, and complex behaviors [see Warnings and Precautions (5.4) ] • Withdrawal effects [see Warnings and Precautions (5.8) ]
Drug Interactions
• CNS depressants, including alcohol: Possible adverse additive CNS-depressant effects ( 5.1 , 7.1 ) • Imipramine: Decreased alertness observed ( 7.1 ) • Chlorpromazine: Impaired alertness and psychomotor performance observed ( 7.1 ) • CYP3A4 inducers (rifampin or St. John's wort): Combination use may decrease effect ( 7.2 ) • Ketoconazole: Combination use may increase effect ( 7.2 )
How Supplied
Zolpidem Tartrate Extended-Release 12.5 mg tablets are composed of two layers and are coated, blue, round, bi-convex, debossed with ZCR on one side and supplied as: NDC Number Size 63187-862-15 Bottle of 15 63187-862-30 Bottle of 30 63187-862-60 Bottle of 60 63187-862-90 Bottle of 90
Storage and Handling
Zolpidem Tartrate Extended-Release 12.5 mg tablets are composed of two layers and are coated, blue, round, bi-convex, debossed with ZCR on one side and supplied as: NDC Number Size 63187-862-15 Bottle of 15 63187-862-30 Bottle of 30 63187-862-60 Bottle of 60 63187-862-90 Bottle of 90
Description
Dosage and Administration, Dosage in Adults ( 2.1 ) 08/2016 Dosage and Administration, Special Populations ( 2.2 ) 12/2016 Warnings and Precautions, CNS Depressant Effects and Next-Day Impairment ( 5.1 ) 08/2016 Warnings and Precautions, Precipitation of Hepatic Encephalopathy ( 5.7 ) 12/2016
Medication Information
Warnings and Precautions
• CNS depressant effects: Impaired alertness and motor coordination, including risk of morning impairment. Caution patients against driving and other activities requiring complete mental alertness the morning after use. ( 5.1 ) • Need to evaluate for co-morbid diagnoses: Revaluate if insomnia persists after 7 to 10 days of use. ( 5.2 ) • Severe anaphylactic/anaphylactoid reactions: Angioedema and anaphylaxis have been reported. Do not rechallenge if such reactions occur. ( 5.3 ) • "Sleep-driving" and other complex behaviors while not fully awake. Risk increases with dose and use with other CNS depressants and alcohol. Immediately evaluate any new onset behavioral changes. ( 5.4 ) • Depression: Worsening of depression or, suicidal thinking may occur. Prescribe the least amount of tablets feasible to avoid intentional overdose. ( 5. 5 ) • Respiratory Depression: Consider this risk before prescribing in patients with compromised respiratory function. ( 5. 6 ) • Hepatic Impairment: Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment. ( 5.7 ) • Withdrawal effects: Symptoms may occur with rapid dose reduction or discontinuation. ( 5.8 , 9.3 ) • Severe Injuries: Drowsiness may lead to fall including severe injuries. ( 5.9 )
Indications and Usage
Zolpidem Tartrate Extended-Release Tablets are indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance (as measured by wake time after sleep onset). The clinical trials performed in support of efficacy were up to 3 weeks (using polysomnography measurement up to 2 weeks in both adult and elderly patients) and 24 weeks (using patient-reported assessment in adult patients only) in duration [see Clinical Studies (14) ] .
Dosage and Administration
• Use the lowest dose effective for the patient and must not exceed a total of 12.5 mg daily ( 2.1 ) • Recommended initial dose is a single dose of 6.25 mg for women, and a single dose of 6.25 or 12.5 mg for men, immediately before bedtime with at least 7–8 hours remaining before the planned time of awakening ( 2.1 ) • Geriatric patients and patients with mild to moderate hepatic impairment: Recommended dose is 6.25 mg for men and women ( 2.2 ) • Lower doses of CNS depressants may be necessary when taken concomitantly with Zolpidem Tartrate Extended-Release Tablets ( 2.3 ) • Tablets to be swallowed whole, not to be crushed, divided or chewed ( 2.4 ) • The effect of Zolpidem Tartrate Extended-Release Tablets may be slowed if taken with or immediately after a meal ( 2.4 )
Contraindications
Zolpidem Tartrate Extended-Release Tablets are contraindicated in patients with known hypersensitivity to zolpidem. Observed reactions include anaphylaxis and angioedema [see Warnings and Precautions (5.3) ].
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: • CNS-depressant effects and next-day impairment [see Warnings and Precautions (5.1) ] • Serious anaphylactic and anaphylactoid reactions [see Warnings and Precautions (5.3) ] • Abnormal thinking and behavior changes, and complex behaviors [see Warnings and Precautions (5.4) ] • Withdrawal effects [see Warnings and Precautions (5.8) ]
Drug Interactions
• CNS depressants, including alcohol: Possible adverse additive CNS-depressant effects ( 5.1 , 7.1 ) • Imipramine: Decreased alertness observed ( 7.1 ) • Chlorpromazine: Impaired alertness and psychomotor performance observed ( 7.1 ) • CYP3A4 inducers (rifampin or St. John's wort): Combination use may decrease effect ( 7.2 ) • Ketoconazole: Combination use may increase effect ( 7.2 )
Storage and Handling
Zolpidem Tartrate Extended-Release 12.5 mg tablets are composed of two layers and are coated, blue, round, bi-convex, debossed with ZCR on one side and supplied as: NDC Number Size 63187-862-15 Bottle of 15 63187-862-30 Bottle of 30 63187-862-60 Bottle of 60 63187-862-90 Bottle of 90
How Supplied
Zolpidem Tartrate Extended-Release 12.5 mg tablets are composed of two layers and are coated, blue, round, bi-convex, debossed with ZCR on one side and supplied as: NDC Number Size 63187-862-15 Bottle of 15 63187-862-30 Bottle of 30 63187-862-60 Bottle of 60 63187-862-90 Bottle of 90
Description
Dosage and Administration, Dosage in Adults ( 2.1 ) 08/2016 Dosage and Administration, Special Populations ( 2.2 ) 12/2016 Warnings and Precautions, CNS Depressant Effects and Next-Day Impairment ( 5.1 ) 08/2016 Warnings and Precautions, Precipitation of Hepatic Encephalopathy ( 5.7 ) 12/2016
Section 34077-8
Pregnancy Category C
There are no adequate and well-controlled studies of Zolpidem Tartrate Extended-Release Tablets in pregnant women. Studies in children to assess the effects of prenatal exposure to zolpidem have not been conducted; however, cases of severe neonatal respiratory depression have been reported when zolpidem was used at the end of pregnancy, especially when taken with other CNS depressants. Children born to mothers taking sedative-hypnotic drugs may be at risk for withdrawal symptoms during the postnatal period. Neonatal flaccidity has also been reported in infants born to mothers who received sedative-hypnotic drugs during pregnancy. Zolpidem Tartrate Extended-Release Tablets should be used during pregnancy only if the potential benefit outweighs the potential risk to the fetus.
Administration of zolpidem to pregnant rats and rabbits resulted in adverse effects on offspring development at doses greater than the Zolpidem Tartrate Extended-Release Tablets maximum recommended human dose (MRHD) of 12.5 mg/day (approximately 10 mg/day zolpidem base); however, teratogenicity was not observed.
When zolpidem was administered at oral doses of 4, 20, and 100 mg base/kg/day to pregnant rats during the period of organogenesis, dose-related decreases in fetal skull ossification occurred at all but the lowest dose, which is approximately 4 times the MRHD on a mg/m2 basis. In rabbits treated during organogenesis with zolpidem at oral doses of 1, 4, and 16 mg base/kg/day, increased embryo-fetal death and incomplete fetal skeletal ossification occurred at the highest dose. The no-effect dose for embryo-fetal toxicity in rabbits is approximately 8 times the MRHD on a mg/m2 basis. Administration of zolpidem to rats at oral doses of 4, 20, and 100 mg base/kg/day during the latter part of pregnancy and throughout lactation produced decreased offspring growth and survival at all but the lowest dose, which is approximately 4 times the MRHD on a mg/m2 basis.
Section 42229-5
Associated with discontinuation of treatment: In 3-week clinical trials in adults and elderly patients (> 65 years), 3.5% (7/201) patients receiving Zolpidem Tartrate Extended-Release Tablets 6.25 or 12.5 mg discontinued treatment due to an adverse reaction as compared to 0.9% (2/216) of patients on placebo. The reaction most commonly associated with discontinuation in patients treated with Zolpidem Tartrate Extended-Release Tablets was somnolence (1%).
In a 6-month study in adult patients (18–64 years of age), 8.5% (57/669) of patients receiving Zolpidem Tartrate Extended-Release Tablets 12.5 mg as compared to 4.6% on placebo (16/349) discontinued treatment due to an adverse reaction. Reactions most commonly associated with discontinuation of Zolpidem Tartrate Extended-Release Tablets included anxiety (anxiety, restlessness or agitation) reported in 1.5% (10/669) of patients as compared to 0.3% (1/349) of patients on placebo, and depression (depression, major depression or depressed mood) reported in 1.5% (10/669) of patients as compared to 0.3% (1/349) of patients on placebo.
Data from a clinical study in which selective serotonin reuptake inhibitor- (SSRI-) treated patients were given zolpidem revealed that four of the seven discontinuations during double-blind treatment with zolpidem (n=95) were associated with impaired concentration, continuing or aggravated depression, and manic reaction; one patient treated with placebo (n=97) was discontinued after an attempted suicide.
Section 42231-1
MEDICATION GUIDE
ZOLPIDEM TARTRATE EXTENDED-RELEASE TABLETS C-IV
Read the Medication Guide that comes with ZOLPIDEM TARTRATE Extended-Release Tablets before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is the most important information I should know about ZOLPIDEM TARTRATE Extended-Release Tablets?
-
•Do not take more ZOLPIDEM TARTRATE Extended-Release Tablets than prescribed.
-
•Do not take ZOLPIDEM TARTRATE Extended-Release Tablets unless you are able to stay in bed a full night (7 to 8 hours) before you must be active again.
-
•Take ZOLPIDEM TARTRATE Extended-Release Tablets right before you get in bed, not sooner.
ZOLPIDEM TARTRATE Extended-Release Tablets may cause serious side effects that you may not know are happening to you. These side effects include:
-
•sleepiness during the day
-
•not thinking clearly
-
•act strangely, confused, or upset
-
•"sleep-walking" or doing other activities when you are asleep like:
-
•eating
-
•talking
-
•having sex
-
•driving a car
-
Call your healthcare provider right away if you find out that you have done any of the above activities after taking ZOLPIDEM TARTRATE Extended-Release Tablets.
You should not drive a car or do things that require clear thinking the day after you take ZOLPIDEM TARTRATE Extended-Release Tablets.
Do not take ZOLPIDEM TARTRATE Extended-Release Tablets if you:
-
•drank alcohol that evening or before bed
-
•take other medicines that can make you sleepy. Taking ZOLPIDEM TARTRATE Extended-Release Tablets with other drugs can cause side effects. Talk to your healthcare provider about all of your medicines. Your healthcare provider will tell you if you can take ZOLPIDEM TARTRATE Extended-Release Tablets with your other medicines.
-
•cannot get a full night's sleep
What are ZOLPIDEM TARTRATE Extended-Release Tablets?
ZOLPIDEM TARTRATE Extended-Release Tablets are a sedative-hypnotic (sleep) medicine. ZOLPIDEM TARTRATE Extended-Release Tablets are used in adults for the treatment of a sleep problem called insomnia. Symptoms of insomnia include:
-
•trouble falling asleep
-
•waking up often during the night
It is not known if ZOLPIDEM TARTRATE Extended-Release Tablets are safe and effective in children under the age of 18 years.
|
ZOLPIDEM TARTRATE Extended-Release Tablets are a federally controlled substance (C-IV) because it can be abused or lead to dependence. Keep ZOLPIDEM TARTRATE Extended-Release Tablets in a safe place to prevent misuse and abuse. Selling or giving away ZOLPIDEM TARTRATE Extended-Release Tablets may harm others, and is against the law. Tell your healthcare provider if you have ever abused or have been dependent on alcohol, prescription medicines or street drugs. |
Section 43683-2
Section 44425-7
Store between 15°–25° C (59°–77°F). Limited excursions permissible up to 30° C (86°F)
Section 51945-4
PRINCIPAL DISPLAY PANEL - 12.5 mg Tablet Bottle Label
NDC 63187-862-30
Zolpidem Tartrate
Extended-Release
CIV
12.5 mg
Dispense with Medication Guide
Rx only
30 Tablets
9.2 Abuse
Abuse and addiction are separate and distinct from physical dependence and tolerance. Abuse is characterized by misuse of the drug for non-medical purposes, often in combination with other psychoactive substances. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug effects over time. Tolerance may occur to both desired and undesired effects of drugs and may develop at different rates for different effects.
Addiction is a primary, chronic, neurobiological disease with genetic, psychosocial, and environmental factors influencing its development and manifestations. It is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, using a multidisciplinary approach, but relapse is common.
Studies of abuse potential in former drug abusers found that the effects of single doses of zolpidem tartrate 40 mg were similar, but not identical, to diazepam 20 mg, while zolpidem tartrate 10 mg effects were difficult to distinguish from placebo.
Because persons with a history of addiction to, or abuse of, drugs or alcohol are at increased risk for misuse, abuse and addiction of zolpidem, they should be monitored carefully when receiving zolpidem or any other hypnotic.
11 Description
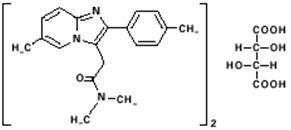
Zolpidem Tartrate Extended-Release Tablets contain zolpidem tartrate, a gamma-aminobutyric acid (GABA) A agonist of the imidazopyridine class. Zolpidem Tartrate Extended-Release Tablets are available in 6.25 mg and 12.5 mg strength tablets for oral administration.
Chemically, zolpidem is N,N,6-trimethyl-2-p-tolylimidazo[1,2-a] pyridine-3-acetamide L-(+)-tartrate (2:1). It has the following structure:
Zolpidem tartrate is a white to off-white crystalline powder that is sparingly soluble in water, alcohol, and propylene glycol. It has a molecular weight of 764.88.
Zolpidem Tartrate Extended-Release Tablet consists of a coated two-layer tablet: one layer that releases its drug content immediately and another layer that allows a slower release of additional drug content. The 6.25 mg Zolpidem Tartrate Extended-Release tablet contains the following inactive ingredients: colloidal silicon dioxide, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, potassium bitartrate, red ferric oxide, sodium starch glycolate, and titanium dioxide. The 12.5 mg Zolpidem Tartrate Extended-Release tablet contains the following inactive ingredients: colloidal silicon dioxide, FD&C Blue #2, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, potassium bitartrate, sodium starch glycolate, titanium dioxide, and yellow ferric oxide.
9.3 Dependence
Physical dependence is a state of adaptation that is manifested by a specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug, and/or administration of an antagonist.
Sedative/hypnotics have produced withdrawal signs and symptoms following abrupt discontinuation. These reported symptoms range from mild dysphoria and insomnia to a withdrawal syndrome that may include abdominal and muscle cramps, vomiting, sweating, tremors, and convulsions. The following adverse events, which are considered to meet the DSM-III-R criteria for uncomplicated sedative/hypnotic withdrawal, were reported during U.S. clinical trials following placebo substitution occurring within 48 hours following last zolpidem treatment: fatigue, nausea, flushing, lightheadedness, uncontrolled crying, emesis, stomach cramps, panic attack, nervousness, and abdominal discomfort. These reported adverse events occurred at an incidence of 1% or less. However, available data cannot provide a reliable estimate of the incidence, if any, of dependence during treatment at recommended doses. Post-marketing reports of abuse, dependence and withdrawal have been received.
8.4 Pediatric Use
Zolpidem Tartrate Extended-Release Tablets are not recommended for use in children. Safety and effectiveness of zolpidem in pediatric patients below the age of 18 years have not been established.
In an 8-week study in pediatric patients (aged 6–17 years) with insomnia associated with attention-deficit/hyperactivity disorder (ADHD) an oral solution of zolpidem tartrate dosed at 0.25 mg/kg at bedtime did not decrease sleep latency compared to placebo. Psychiatric and nervous system disorders comprised the most frequent (> 5%) treatment emergent adverse reactions observed with zolpidem versus placebo and included dizziness (23.5% vs. 1.5%), headache (12.5% vs. 9.2%), and hallucinations were reported in 7% of the pediatric patients who received zolpidem; none of the pediatric patients who received placebo reported hallucinations [see Warnings and Precautions (5.4)]. Ten patients on zolpidem (7.4%) discontinued treatment due to an adverse reaction.
FDA has not required pediatric studies of Zolpidem Tartrate Extended-Release Tablets in the pediatric population based on these efficacy and safety findings.
8.5 Geriatric Use
A total of 99 elderly (≥ 65 years of age) received daily doses of 6.25 mg Zolpidem Tartrate Extended-Release Tablets in a 3-week placebo-controlled study. The adverse reaction profile of Zolpidem Tartrate Extended-Release Tablets 6.25 mg in this population was similar to that of Zolpidem Tartrate Extended-Release Tablets 12.5 mg in younger adults (≤ 64 years of age). Dizziness was reported in 8% of Zolpidem Tartrate Extended-Release Tablets-treated patients compared with 3% of those treated with placebo.
The dose of Zolpidem Tartrate Extended-Release Tablets in elderly patients is 6.25 mg to minimize adverse effects related to impaired motor and/or cognitive performance and unusual sensitivity to sedative/hypnotic drugs [see Warnings and Precautions (5.1)] .
2.4 Administration
Zolpidem Tartrate Extended-Release Tablets should be swallowed whole, and not be divided, crushed, or chewed. The effect of Zolpidem Tartrate Extended-Release Tablets may be slowed by ingestion with or immediately after a meal.
4 Contraindications
Zolpidem Tartrate Extended-Release Tablets are contraindicated in patients with known hypersensitivity to zolpidem. Observed reactions include anaphylaxis and angioedema [see Warnings and Precautions (5.3)].
5.9 Severe Injuries
Zolpidem can cause drowsiness and a decreased level of consciousness, which may lead to falls and consequently to severe injuries. Severe injuries such as hip fractures and intracranial hemorrhage have been reported.
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
-
•CNS-depressant effects and next-day impairment [see Warnings and Precautions (5.1)]
-
•Serious anaphylactic and anaphylactoid reactions [see Warnings and Precautions (5.3)]
-
•Abnormal thinking and behavior changes, and complex behaviors [see Warnings and Precautions (5.4)]
-
•Withdrawal effects [see Warnings and Precautions (5.8)]
7 Drug Interactions
-
•CNS depressants, including alcohol: Possible adverse additive CNS-depressant effects (5.1, 7.1)
-
•Imipramine: Decreased alertness observed (7.1)
-
•Chlorpromazine: Impaired alertness and psychomotor performance observed (7.1)
-
•CYP3A4 inducers (rifampin or St. John's wort): Combination use may decrease effect (7.2)
-
•Ketoconazole: Combination use may increase effect (7.2)
8.3 Nursing Mothers
Zolpidem is excreted in human milk. Caution should be exercised when Zolpidem Tartrate Extended-Release Tablets are administered to a nursing woman.
2.1 Dosage in Adults
Use the lowest effective dose for the patient. The recommended initial dose is 6.25 mg for women and either 6.25 or 12.5 mg for men, taken only once per night immediately before bedtime with at least 7–8 hours remaining before the planned time of awakening. If the 6.25 mg dose is not effective, the dose can be increased to 12.5 mg. In some patients, the higher morning blood levels following use of the 12.5 mg dose increase the risk of next day impairment of driving and other activities that require full alertness [see Warnings and Precautions (5.1) ] . The total dose of Zolpidem Tartrate Extended-Release Tablets should not exceed 12.5 mg once daily immediately before bedtime. Zolpidem Tartrate Extended-Release Tablets should be taken as a single dose and should not be readministered during the same night.
The recommended initial doses for women and men are different because zolpidem clearance is lower in women.
7.1 Cns Active Drugs
Co-administration of zolpidem with other CNS depressants increases the risk of CNS depression. Concomitant use of zolpidem with these drugs may increase drowsiness and psychomotor impairment, including impaired driving ability [see Warnings and Precautions (5.1)]. Zolpidem tartrate was evaluated in healthy volunteers in single-dose interaction studies for several CNS drugs.
12.3 Pharmacokinetics
Zolpidem Tartrate Extended-Release Tablets exhibit biphasic absorption characteristics, which results in rapid initial absorption from the gastrointestinal tract similar to zolpidem tartrate immediate-release, then provides extended plasma concentrations beyond three hours after administration. A study in 24 healthy male subjects was conducted to compare mean zolpidem plasma concentration-time profiles obtained after single oral administration of Zolpidem Tartrate Extended-Release Tablets 12.5 mg and of an immediate-release formulation of zolpidem tartrate (10 mg). The terminal elimination half-life observed with Zolpidem Tartrate Extended-Release Tablets (12.5 mg) was similar to that obtained with immediate-release zolpidem tartrate (10 mg). The mean plasma concentration-time profiles are shown in Figure 1.
Figure 1: Mean plasma concentration-time profiles for Zolpidem Tartrate Extended-Release Tablets (12.5 mg) and immediate-release zolpidem tartrate (10 mg)
In adult and elderly patients treated with Zolpidem Tartrate Extended-Release Tablets, there was no evidence of accumulation after repeated once-daily dosing for up to two weeks.
5.8 Withdrawal Effects
There have been reports of withdrawal signs and symptoms following the rapid dose decrease or abrupt discontinuation of zolpidem. Monitor patients for tolerance, abuse, and dependence [see Drug Abuse and Dependence (9.2) and (9.3)].
8.2 Labor and Delivery
Zolpidem Tartrate Extended-Release Tablets have no established use in labor and delivery [see Pregnancy (8.1)].
8.7 Hepatic Impairment
The recommended dose of Zolpidem Tartrate Extended-Release Tablets in patients with mild to moderate hepatic impairment is 6.25 mg once daily immediately before bedtime. Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment as it may contribute to encephalopathy [see Dosage and Administration (2.2), Warnings and Precautions (5.7), Clinical Pharmacology (12.3)].
1 Indications and Usage
Zolpidem Tartrate Extended-Release Tablets are indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance (as measured by wake time after sleep onset).
The clinical trials performed in support of efficacy were up to 3 weeks (using polysomnography measurement up to 2 weeks in both adult and elderly patients) and 24 weeks (using patient-reported assessment in adult patients only) in duration [see Clinical Studies (14)].
10.1 Signs and Symptoms
In postmarketing experience of overdose with zolpidem tartrate alone, or in combination with CNS-depressant agents, impairment of consciousness ranging from somnolence to coma, cardiovascular and/or respiratory compromise and fatal outcomes have been reported.
2.2 Special Populations
Elderly or debilitated patients may be especially sensitive to the effects of zolpidem tartrate. The recommended dose of Zolpidem Tartrate Extended-Release Tablets in these patients is 6.25 mg once daily immediately before bedtime [see Warnings and Precautions (5.1), Use in Specific Populations (8.5)].
Patients with mild to moderate hepatic impairment do not clear the drug as rapidly as normal subjects. The recommended dose of Zolpidem Tartrate Extended-Release Tablets in these patients is 6.25 mg once daily immediately before bedtime. Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment as it may contribute to encephalopathy [see Warnings and Precautions (5.7) , Use in Specific Populations (8.7) , Clinical Pharmacology (12.3) ] .
12.1 Mechanism of Action
Zolpidem, the active moiety of zolpidem tartrate, is a hypnotic agent with a chemical structure unrelated to benzodiazepines, barbiturates, or other drugs with known hypnotic properties. It interacts with a GABA-BZ receptor complex and shares some of the pharmacological properties of the benzodiazepines. In contrast to the benzodiazepines, which non-selectively bind to and activate all BZ receptor subtypes, zolpidem in vitro binds the BZ1 receptor preferentially with a high affinity ratio of the α1/α5 subunits. This selective binding of zolpidem on the BZ1 receptor is not absolute, but it may explain the relative absence of myorelaxant and anticonvulsant effects in animal studies as well as the preservation of deep sleep (stages 3 and 4) in human studies of zolpidem tartrate at hypnotic doses.
9.1 Controlled Substance
Zolpidem tartrate is classified as a Schedule IV controlled substance by federal regulation.
10.2 Recommended Treatment
General symptomatic and supportive measures should be used along with immediate gastric lavage where appropriate. Intravenous fluids should be administered as needed. Zolpidem's sedative hypnotic effect was shown to be reduced by flumazenil and therefore may be useful; however, flumazenil administration may contribute to the appearance of neurological symptoms (convulsions). As in all cases of drug overdose, respiration, pulse, blood pressure, and other appropriate signs should be monitored and general supportive measures employed. Hypotension and CNS depression should be monitored and treated by appropriate medical intervention. Sedating drugs should be withheld following zolpidem overdosage, even if excitation occurs. The value of dialysis in the treatment of overdosage has not been determined, although hemodialysis studies in patients with renal failure receiving therapeutic doses have demonstrated that zolpidem is not dialyzable.
As with the management of all overdosage, the possibility of multiple drug ingestion should be considered. The physician may wish to consider contacting a poison control center for up-to-date information on the management of hypnotic drug product overdosage.
5 Warnings and Precautions
-
•CNS depressant effects: Impaired alertness and motor coordination, including risk of morning impairment. Caution patients against driving and other activities requiring complete mental alertness the morning after use. (5.1)
-
•Need to evaluate for co-morbid diagnoses: Revaluate if insomnia persists after 7 to 10 days of use. (5.2)
-
•Severe anaphylactic/anaphylactoid reactions: Angioedema and anaphylaxis have been reported. Do not rechallenge if such reactions occur. (5.3)
-
•"Sleep-driving" and other complex behaviors while not fully awake. Risk increases with dose and use with other CNS depressants and alcohol. Immediately evaluate any new onset behavioral changes. (5.4)
-
•Depression: Worsening of depression or, suicidal thinking may occur. Prescribe the least amount of tablets feasible to avoid intentional overdose. (5. 5)
-
•Respiratory Depression: Consider this risk before prescribing in patients with compromised respiratory function. (5. 6)
-
•Hepatic Impairment: Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment. (5.7)
-
•Withdrawal effects: Symptoms may occur with rapid dose reduction or discontinuation. (5.8, 9.3)
-
•Severe Injuries: Drowsiness may lead to fall including severe injuries. (5.9)
5.6 Respiratory Depression
Although studies with 10 mg zolpidem tartrate did not reveal respiratory depressant effects at hypnotic doses in healthy subjects or in patients with mild-to-moderate chronic obstructive pulmonary disease (COPD), a reduction in the Total Arousal Index, together with a reduction in lowest oxygen saturation and increase in the times of oxygen desaturation below 80% and 90%, was observed in patients with mild-to-moderate sleep apnea when treated with zolpidem compared to placebo. Since sedative-hypnotics have the capacity to depress respiratory drive, precautions should be taken if Zolpidem Tartrate Extended-Release Tablets are prescribed to patients with compromised respiratory function. Post-marketing reports of respiratory insufficiency in patients receiving 10 mg of zolpidem tartrate, most of whom had pre-existing respiratory impairment, have been reported. The risk of respiratory depression should be considered prior to prescribing Zolpidem Tartrate Extended-Release Tablets in patients with respiratory impairment including sleep apnea and myasthenia gravis.
2 Dosage and Administration
-
•Use the lowest dose effective for the patient and must not exceed a total of 12.5 mg daily (2.1)
-
•Recommended initial dose is a single dose of 6.25 mg for women, and a single dose of 6.25 or 12.5 mg for men, immediately before bedtime with at least 7–8 hours remaining before the planned time of awakening (2.1)
-
•Geriatric patients and patients with mild to moderate hepatic impairment: Recommended dose is 6.25 mg for men and women (2.2)
-
•Lower doses of CNS depressants may be necessary when taken concomitantly with Zolpidem Tartrate Extended-Release Tablets (2.3)
-
•Tablets to be swallowed whole, not to be crushed, divided or chewed (2.4)
-
•The effect of Zolpidem Tartrate Extended-Release Tablets may be slowed if taken with or immediately after a meal (2.4)
2.3 Use With Cns Depressants
Dosage adjustment may be necessary when Zolpidem Tartrate Extended-Release Tablets are combined with other CNS depressant drugs because of the potentially additive effects [see Warnings and Precautions (5.1)].
3 Dosage Forms and Strengths
Zolpidem Tartrate Extended-Release Tablets are available as extended-release tablets containing 12.5 mg of zolpidem tartrate for oral administration. Tablets are not scored.
Zolpidem Tartrate Extended-Release Tablets 12.5 mg tablets are blue, round, bi-convex, and debossed with ZCR on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Zolpidem Tartrate Extended-Release Tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Liver and biliary system: acute hepatocellular, cholestatic or mixed liver injury with or without jaundice (i.e., bilirubin >2×ULN, alkaline phosphatase ≥2×ULN, transaminase ≥5×ULN).
8 Use in Specific Populations
-
•Pregnancy: Based on animal data may cause fetal harm (8.1)
-
•Pediatric use: Safety and effectiveness not established. Hallucinations (incidence rate 7%) and other psychiatric and/or nervous system adverse reactions were observed frequently in a study of pediatric patients with Attention-Deficit/Hyperactivity Disorder (5.4, 8.4)
14.1 Controlled Clinical Trials
Zolpidem Tartrate Extended-Release Tablets were evaluated in three placebo-controlled studies for the treatment of patients with chronic primary insomnia (as defined in the APA Diagnostic and Statistical Manual of Mental Disorders, DSM IV).
Adult outpatients (18–64 years) with primary insomnia (N=212) were evaluated in a double-blind, randomized, parallel-group, 3-week trial comparing Zolpidem Tartrate Extended-Release Tablets 12.5 mg and placebo. Zolpidem Tartrate Extended-Release Tablets 12.5 mg decreased wake time after sleep onset (WASO) for the first 7 hours during the first 2 nights and for the first 5 hours after 2 weeks of treatment. Zolpidem Tartrate Extended-Release Tablets 12.5 mg were superior to placebo on objective measures (polysomnography recordings) of sleep induction (by decreasing latency to persistent sleep [LPS]) during the first 2 nights of treatment and after 2 weeks of treatment. Zolpidem Tartrate Extended-Release Tablets 12.5 mg were also superior to placebo on the patient reported global impression regarding the aid to sleep after the first 2 nights and after 3 weeks of treatment.
Elderly outpatients (≥ 65 years) with primary insomnia (N=205) were evaluated in a double-blind, randomized, parallel-group, 3-week trial comparing Zolpidem Tartrate Extended-Release Tablets 6.25 mg and placebo. Zolpidem Tartrate Extended-Release Tablets 6.25 mg decreased wake time after sleep onset (WASO) for the first 6 hours during the first 2 nights and the first 4 hours after 2 weeks of treatment. Zolpidem Tartrate Extended-Release Tablets 6.25 mg were superior to placebo on objective measures (polysomnography recordings) of sleep induction (by decreasing LPS) during the first 2 nights of treatment and after 2 weeks on treatment. Zolpidem Tartrate Extended-Release Tablets 6.25 mg were superior to placebo on the patient reported global impression regarding the aid to sleep after the first 2 nights and after 3 weeks of treatment.
In both studies, in patients treated with Zolpidem Tartrate Extended-Release Tablets, polysomnography showed increased wakefulness at the end of the night compared to placebo-treated patients.
In a 24-week double-blind, placebo controlled, randomized study in adult outpatients (18–64 years) with primary insomnia (N=1025), Zolpidem Tartrate Extended-Release Tablets 12.5 mg administered as needed (3 to 7 nights per week) was superior to placebo over 24 weeks, on patient global impression regarding aid to sleep, and on patient-reported specific sleep parameters for sleep induction and sleep maintenance with no significant increased frequency of drug intake observed over time.
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Medication Guide). Inform patients and their families about the benefits and risks of treatment with Zolpidem Tartrate Extended-Release Tablets. Inform patients of the availability of a Medication Guide and instruct them to read the Medication Guide prior to initiating treatment with Zolpidem Tartrate Extended-Release Tablets and with each prescription refill. Review the Zolpidem Tartrate Extended-Release Tablets Medication Guide with every patient prior to initiation of treatment. Instruct patients or caregivers that Zolpidem Tartrate Extended-Release Tablets should be taken only as prescribed.
5.5 Use in Patients With Depression
In primarily depressed patients treated with sedative-hypnotics, worsening of depression, and suicidal thoughts and actions (including completed suicides), have been reported. Suicidal tendencies may be present in such patients and protective measures may be required. Intentional overdosage is more common in this group of patients; therefore, the lowest number of tablets that is feasible should be prescribed for the patient at any one time.
16 How Supplied/storage and Handling
Zolpidem Tartrate Extended-Release 12.5 mg tablets are composed of two layers and are coated, blue, round, bi-convex, debossed with ZCR on one side and supplied as:
| NDC Number | Size |
|---|---|
|
63187-862-15 |
Bottle of 15 |
|
63187-862-30 |
Bottle of 30 |
|
63187-862-60 |
Bottle of 60 |
|
63187-862-90 |
Bottle of 90 |
8.6 Gender Difference in Pharmacokinetics
Women clear zolpidem tartrate from the body at a lower rate than men. Cmax and AUC parameters of zolpidem from Zolpidem Tartrate Extended-Release Tablets were, respectively, approximately 50% and 75% higher at the same dose in adult female subjects compared to adult male subjects. Between 6 and 12 hours after dosing, zolpidem concentrations were 2- to 3 fold higher in adult female compared to adult male subjects. Given the higher blood levels of zolpidem tartrate in women compared to men at a given dose, the recommended initial dose of Zolpidem Tartrate Extended-Release Tablets for adult women is 6.25 mg, and the recommended dose for adult men is 6.25 or 12.5 mg.
In geriatric patients, clearance of zolpidem is similar in men and women. The recommended dose of Zolpidem Tartrate Extended-Release Tablets in geriatric patients is 6.25 mg regardless of gender.
5.7 Precipitation of Hepatic Encephalopathy
GABA agonists such as zolpidem tartrate have been associated with precipitation of hepatic encephalopathy in patients with hepatic insufficiency. In addition, patients with hepatic insufficiency do not clear zolpidem tartrate as rapidly as patients with normal hepatic function. Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment as it may contribute to encephalopathy [see Dosage and Administration (2.2) , Use in Specific Populations (8.7) , Clinical Pharmacology (12.3) ] .
5.2 Need to Evaluate for Co Morbid Diagnoses
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after a careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. Worsening of insomnia or the emergence of new thinking or behavior abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with sedative/hypnotic drugs, including zolpidem.
5.4 Abnormal Thinking and Behavioral Changes
Abnormal thinking and behavior changes have been reported in patients treated with sedative/hypnotics, including Zolpidem Tartrate Extended-Release Tablets. Some of these changes included decreased inhibition (e.g. aggressiveness and extroversion that seemed out of character), bizarre behavior, agitation and depersonalization. Visual and auditory hallucinations have been reported.
In controlled trials, <1% of adults with insomnia reported hallucinations. In a clinical trial, 7% of pediatric patients treated with zolpidem 0.25 mg/kg taken at bedtime reported hallucinations versus 0% treated with placebo [see Use in Specific Populations (8.4)].
Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake after ingestion of a sedative-hypnotic, with amnesia for the event) have been reported in sedative-hypnotic-naive as well as in sedative-hypnotic-experienced persons. Although behaviors such as "sleep-driving" have occurred with Zolpidem Tartrate Extended-Release Tablets alone at therapeutic doses, the co-administration of alcohol and other CNS depressants increases the risk of such behaviors, as does the use of Zolpidem Tartrate Extended-Release Tablets at doses exceeding the maximum recommended dose. Due to the risk to the patient and the community, discontinuation of Zolpidem Tartrate Extended-Release Tablets should be strongly considered for patients who report a "sleep-driving" episode.
Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with "sleep-driving", patients usually do not remember these events. Amnesia, anxiety and other neuro-psychiatric symptoms may also occur.
It can rarely be determined with certainty whether a particular instance of the abnormal behaviors listed above is drug induced, spontaneous in origin, or a result of an underlying psychiatric or physical disorder. Nonetheless, the emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
5.1 Cns Depressant Effects and Next Day Impairment
Zolpidem Tartrate Extended-Release Tablets are a central nervous system (CNS) depressant and can impair daytime function in some patients even when used as prescribed. Prescribers should monitor for excess depressant effects, but impairment can occur in the absence of subjective symptoms, and may not be reliably detected by ordinary clinical exam (i.e. less than formal psychomotor testing). While pharmacodynamic tolerance or adaptation to some adverse depressant effects of Zolpidem Tartrate Extended-Release Tablets may develop, patients using Zolpidem Tartrate Extended-Release Tablets should be cautioned against driving or engaging in other hazardous activities or activities requiring complete mental alertness the day after use.
Additive effects occur with concomitant use of other CNS depressants (e.g. benzodiazepines, opioids, tricyclic antidepressants, alcohol), including daytime use. Downward dose adjustment of Zolpidem Tartrate Extended-Release Tablets and concomitant CNS depressants should be considered [see Dosage and Administration (2.3)].
The use of Zolpidem Tartrate Extended-Release Tablets with other sedative-hypnotics (including other zolpidem products) at bedtime or the middle of the night is not recommended.
The risk of next-day psychomotor impairment is increased if Zolpidem Tartrate Extended-Release Tablets are taken with less than a full night of sleep remaining (7- to 8 hours); if higher than the recommended dose is taken; if co-administered with other CNS depressants or alcohol; or co-administered with other drugs that increase the blood levels of zolpidem. Patients should be warned against driving and other activities requiring complete mental alertness if Zolpidem Tartrate Extended-Release Tablets is taken in these circumstances [see Dosage and Administration (2) and Clinical Studies (14.2) ] .
Vehicle drivers and machine operators should be warned that, as with other hypnotics, there may be a possible risk of adverse reactions including drowsiness, prolonged reaction time, dizziness, sleepiness, blurred/double vision, reduced alertness and impaired driving the morning after therapy. In order to minimize this risk a full night of sleep (7–8 hours) is recommended.
5.3 Severe Anaphylactic and Anaphylactoid Reactions
Cases of angioedema involving the tongue, glottis or larynx have been reported in patients after taking the first or subsequent doses of sedative-hypnotics, including zolpidem. Some patients have had additional symptoms such as dyspnea, throat closing or nausea and vomiting that suggest anaphylaxis. Some patients have required medical therapy in the emergency department. If angioedema involves the throat, glottis or larynx, airway obstruction may occur and be fatal. Patients who develop angioedema after treatment with zolpidem should not be rechallenged with the drug.
7.2 Drugs That Affect Drug Metabolism Via Cytochrome P450
Some compounds known to induce or inhibit CYP3A may affect exposure to zolpidem. The effect of drugs that induce or inhibit other P450 enzymes on the exposure to zolpidem is not known.
Structured Label Content
Section 34077-8 (34077-8)
Pregnancy Category C
There are no adequate and well-controlled studies of Zolpidem Tartrate Extended-Release Tablets in pregnant women. Studies in children to assess the effects of prenatal exposure to zolpidem have not been conducted; however, cases of severe neonatal respiratory depression have been reported when zolpidem was used at the end of pregnancy, especially when taken with other CNS depressants. Children born to mothers taking sedative-hypnotic drugs may be at risk for withdrawal symptoms during the postnatal period. Neonatal flaccidity has also been reported in infants born to mothers who received sedative-hypnotic drugs during pregnancy. Zolpidem Tartrate Extended-Release Tablets should be used during pregnancy only if the potential benefit outweighs the potential risk to the fetus.
Administration of zolpidem to pregnant rats and rabbits resulted in adverse effects on offspring development at doses greater than the Zolpidem Tartrate Extended-Release Tablets maximum recommended human dose (MRHD) of 12.5 mg/day (approximately 10 mg/day zolpidem base); however, teratogenicity was not observed.
When zolpidem was administered at oral doses of 4, 20, and 100 mg base/kg/day to pregnant rats during the period of organogenesis, dose-related decreases in fetal skull ossification occurred at all but the lowest dose, which is approximately 4 times the MRHD on a mg/m2 basis. In rabbits treated during organogenesis with zolpidem at oral doses of 1, 4, and 16 mg base/kg/day, increased embryo-fetal death and incomplete fetal skeletal ossification occurred at the highest dose. The no-effect dose for embryo-fetal toxicity in rabbits is approximately 8 times the MRHD on a mg/m2 basis. Administration of zolpidem to rats at oral doses of 4, 20, and 100 mg base/kg/day during the latter part of pregnancy and throughout lactation produced decreased offspring growth and survival at all but the lowest dose, which is approximately 4 times the MRHD on a mg/m2 basis.
Section 42229-5 (42229-5)
Associated with discontinuation of treatment: In 3-week clinical trials in adults and elderly patients (> 65 years), 3.5% (7/201) patients receiving Zolpidem Tartrate Extended-Release Tablets 6.25 or 12.5 mg discontinued treatment due to an adverse reaction as compared to 0.9% (2/216) of patients on placebo. The reaction most commonly associated with discontinuation in patients treated with Zolpidem Tartrate Extended-Release Tablets was somnolence (1%).
In a 6-month study in adult patients (18–64 years of age), 8.5% (57/669) of patients receiving Zolpidem Tartrate Extended-Release Tablets 12.5 mg as compared to 4.6% on placebo (16/349) discontinued treatment due to an adverse reaction. Reactions most commonly associated with discontinuation of Zolpidem Tartrate Extended-Release Tablets included anxiety (anxiety, restlessness or agitation) reported in 1.5% (10/669) of patients as compared to 0.3% (1/349) of patients on placebo, and depression (depression, major depression or depressed mood) reported in 1.5% (10/669) of patients as compared to 0.3% (1/349) of patients on placebo.
Data from a clinical study in which selective serotonin reuptake inhibitor- (SSRI-) treated patients were given zolpidem revealed that four of the seven discontinuations during double-blind treatment with zolpidem (n=95) were associated with impaired concentration, continuing or aggravated depression, and manic reaction; one patient treated with placebo (n=97) was discontinued after an attempted suicide.
Section 42231-1 (42231-1)
MEDICATION GUIDE
ZOLPIDEM TARTRATE EXTENDED-RELEASE TABLETS C-IV
Read the Medication Guide that comes with ZOLPIDEM TARTRATE Extended-Release Tablets before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is the most important information I should know about ZOLPIDEM TARTRATE Extended-Release Tablets?
-
•Do not take more ZOLPIDEM TARTRATE Extended-Release Tablets than prescribed.
-
•Do not take ZOLPIDEM TARTRATE Extended-Release Tablets unless you are able to stay in bed a full night (7 to 8 hours) before you must be active again.
-
•Take ZOLPIDEM TARTRATE Extended-Release Tablets right before you get in bed, not sooner.
ZOLPIDEM TARTRATE Extended-Release Tablets may cause serious side effects that you may not know are happening to you. These side effects include:
-
•sleepiness during the day
-
•not thinking clearly
-
•act strangely, confused, or upset
-
•"sleep-walking" or doing other activities when you are asleep like:
-
•eating
-
•talking
-
•having sex
-
•driving a car
-
Call your healthcare provider right away if you find out that you have done any of the above activities after taking ZOLPIDEM TARTRATE Extended-Release Tablets.
You should not drive a car or do things that require clear thinking the day after you take ZOLPIDEM TARTRATE Extended-Release Tablets.
Do not take ZOLPIDEM TARTRATE Extended-Release Tablets if you:
-
•drank alcohol that evening or before bed
-
•take other medicines that can make you sleepy. Taking ZOLPIDEM TARTRATE Extended-Release Tablets with other drugs can cause side effects. Talk to your healthcare provider about all of your medicines. Your healthcare provider will tell you if you can take ZOLPIDEM TARTRATE Extended-Release Tablets with your other medicines.
-
•cannot get a full night's sleep
What are ZOLPIDEM TARTRATE Extended-Release Tablets?
ZOLPIDEM TARTRATE Extended-Release Tablets are a sedative-hypnotic (sleep) medicine. ZOLPIDEM TARTRATE Extended-Release Tablets are used in adults for the treatment of a sleep problem called insomnia. Symptoms of insomnia include:
-
•trouble falling asleep
-
•waking up often during the night
It is not known if ZOLPIDEM TARTRATE Extended-Release Tablets are safe and effective in children under the age of 18 years.
|
ZOLPIDEM TARTRATE Extended-Release Tablets are a federally controlled substance (C-IV) because it can be abused or lead to dependence. Keep ZOLPIDEM TARTRATE Extended-Release Tablets in a safe place to prevent misuse and abuse. Selling or giving away ZOLPIDEM TARTRATE Extended-Release Tablets may harm others, and is against the law. Tell your healthcare provider if you have ever abused or have been dependent on alcohol, prescription medicines or street drugs. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store between 15°–25° C (59°–77°F). Limited excursions permissible up to 30° C (86°F)
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - 12.5 mg Tablet Bottle Label
NDC 63187-862-30
Zolpidem Tartrate
Extended-Release
CIV
12.5 mg
Dispense with Medication Guide
Rx only
30 Tablets
9.2 Abuse
Abuse and addiction are separate and distinct from physical dependence and tolerance. Abuse is characterized by misuse of the drug for non-medical purposes, often in combination with other psychoactive substances. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug effects over time. Tolerance may occur to both desired and undesired effects of drugs and may develop at different rates for different effects.
Addiction is a primary, chronic, neurobiological disease with genetic, psychosocial, and environmental factors influencing its development and manifestations. It is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, using a multidisciplinary approach, but relapse is common.
Studies of abuse potential in former drug abusers found that the effects of single doses of zolpidem tartrate 40 mg were similar, but not identical, to diazepam 20 mg, while zolpidem tartrate 10 mg effects were difficult to distinguish from placebo.
Because persons with a history of addiction to, or abuse of, drugs or alcohol are at increased risk for misuse, abuse and addiction of zolpidem, they should be monitored carefully when receiving zolpidem or any other hypnotic.
11 Description (11 DESCRIPTION)
Zolpidem Tartrate Extended-Release Tablets contain zolpidem tartrate, a gamma-aminobutyric acid (GABA) A agonist of the imidazopyridine class. Zolpidem Tartrate Extended-Release Tablets are available in 6.25 mg and 12.5 mg strength tablets for oral administration.
Chemically, zolpidem is N,N,6-trimethyl-2-p-tolylimidazo[1,2-a] pyridine-3-acetamide L-(+)-tartrate (2:1). It has the following structure:
Zolpidem tartrate is a white to off-white crystalline powder that is sparingly soluble in water, alcohol, and propylene glycol. It has a molecular weight of 764.88.
Zolpidem Tartrate Extended-Release Tablet consists of a coated two-layer tablet: one layer that releases its drug content immediately and another layer that allows a slower release of additional drug content. The 6.25 mg Zolpidem Tartrate Extended-Release tablet contains the following inactive ingredients: colloidal silicon dioxide, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, potassium bitartrate, red ferric oxide, sodium starch glycolate, and titanium dioxide. The 12.5 mg Zolpidem Tartrate Extended-Release tablet contains the following inactive ingredients: colloidal silicon dioxide, FD&C Blue #2, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, potassium bitartrate, sodium starch glycolate, titanium dioxide, and yellow ferric oxide.
9.3 Dependence
Physical dependence is a state of adaptation that is manifested by a specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug, and/or administration of an antagonist.
Sedative/hypnotics have produced withdrawal signs and symptoms following abrupt discontinuation. These reported symptoms range from mild dysphoria and insomnia to a withdrawal syndrome that may include abdominal and muscle cramps, vomiting, sweating, tremors, and convulsions. The following adverse events, which are considered to meet the DSM-III-R criteria for uncomplicated sedative/hypnotic withdrawal, were reported during U.S. clinical trials following placebo substitution occurring within 48 hours following last zolpidem treatment: fatigue, nausea, flushing, lightheadedness, uncontrolled crying, emesis, stomach cramps, panic attack, nervousness, and abdominal discomfort. These reported adverse events occurred at an incidence of 1% or less. However, available data cannot provide a reliable estimate of the incidence, if any, of dependence during treatment at recommended doses. Post-marketing reports of abuse, dependence and withdrawal have been received.
8.4 Pediatric Use
Zolpidem Tartrate Extended-Release Tablets are not recommended for use in children. Safety and effectiveness of zolpidem in pediatric patients below the age of 18 years have not been established.
In an 8-week study in pediatric patients (aged 6–17 years) with insomnia associated with attention-deficit/hyperactivity disorder (ADHD) an oral solution of zolpidem tartrate dosed at 0.25 mg/kg at bedtime did not decrease sleep latency compared to placebo. Psychiatric and nervous system disorders comprised the most frequent (> 5%) treatment emergent adverse reactions observed with zolpidem versus placebo and included dizziness (23.5% vs. 1.5%), headache (12.5% vs. 9.2%), and hallucinations were reported in 7% of the pediatric patients who received zolpidem; none of the pediatric patients who received placebo reported hallucinations [see Warnings and Precautions (5.4)]. Ten patients on zolpidem (7.4%) discontinued treatment due to an adverse reaction.
FDA has not required pediatric studies of Zolpidem Tartrate Extended-Release Tablets in the pediatric population based on these efficacy and safety findings.
8.5 Geriatric Use
A total of 99 elderly (≥ 65 years of age) received daily doses of 6.25 mg Zolpidem Tartrate Extended-Release Tablets in a 3-week placebo-controlled study. The adverse reaction profile of Zolpidem Tartrate Extended-Release Tablets 6.25 mg in this population was similar to that of Zolpidem Tartrate Extended-Release Tablets 12.5 mg in younger adults (≤ 64 years of age). Dizziness was reported in 8% of Zolpidem Tartrate Extended-Release Tablets-treated patients compared with 3% of those treated with placebo.
The dose of Zolpidem Tartrate Extended-Release Tablets in elderly patients is 6.25 mg to minimize adverse effects related to impaired motor and/or cognitive performance and unusual sensitivity to sedative/hypnotic drugs [see Warnings and Precautions (5.1)] .
2.4 Administration
Zolpidem Tartrate Extended-Release Tablets should be swallowed whole, and not be divided, crushed, or chewed. The effect of Zolpidem Tartrate Extended-Release Tablets may be slowed by ingestion with or immediately after a meal.
4 Contraindications (4 CONTRAINDICATIONS)
Zolpidem Tartrate Extended-Release Tablets are contraindicated in patients with known hypersensitivity to zolpidem. Observed reactions include anaphylaxis and angioedema [see Warnings and Precautions (5.3)].
5.9 Severe Injuries
Zolpidem can cause drowsiness and a decreased level of consciousness, which may lead to falls and consequently to severe injuries. Severe injuries such as hip fractures and intracranial hemorrhage have been reported.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
-
•CNS-depressant effects and next-day impairment [see Warnings and Precautions (5.1)]
-
•Serious anaphylactic and anaphylactoid reactions [see Warnings and Precautions (5.3)]
-
•Abnormal thinking and behavior changes, and complex behaviors [see Warnings and Precautions (5.4)]
-
•Withdrawal effects [see Warnings and Precautions (5.8)]
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•CNS depressants, including alcohol: Possible adverse additive CNS-depressant effects (5.1, 7.1)
-
•Imipramine: Decreased alertness observed (7.1)
-
•Chlorpromazine: Impaired alertness and psychomotor performance observed (7.1)
-
•CYP3A4 inducers (rifampin or St. John's wort): Combination use may decrease effect (7.2)
-
•Ketoconazole: Combination use may increase effect (7.2)
8.3 Nursing Mothers
Zolpidem is excreted in human milk. Caution should be exercised when Zolpidem Tartrate Extended-Release Tablets are administered to a nursing woman.
2.1 Dosage in Adults
Use the lowest effective dose for the patient. The recommended initial dose is 6.25 mg for women and either 6.25 or 12.5 mg for men, taken only once per night immediately before bedtime with at least 7–8 hours remaining before the planned time of awakening. If the 6.25 mg dose is not effective, the dose can be increased to 12.5 mg. In some patients, the higher morning blood levels following use of the 12.5 mg dose increase the risk of next day impairment of driving and other activities that require full alertness [see Warnings and Precautions (5.1) ] . The total dose of Zolpidem Tartrate Extended-Release Tablets should not exceed 12.5 mg once daily immediately before bedtime. Zolpidem Tartrate Extended-Release Tablets should be taken as a single dose and should not be readministered during the same night.
The recommended initial doses for women and men are different because zolpidem clearance is lower in women.
7.1 Cns Active Drugs (7.1 CNS-active Drugs)
Co-administration of zolpidem with other CNS depressants increases the risk of CNS depression. Concomitant use of zolpidem with these drugs may increase drowsiness and psychomotor impairment, including impaired driving ability [see Warnings and Precautions (5.1)]. Zolpidem tartrate was evaluated in healthy volunteers in single-dose interaction studies for several CNS drugs.
12.3 Pharmacokinetics
Zolpidem Tartrate Extended-Release Tablets exhibit biphasic absorption characteristics, which results in rapid initial absorption from the gastrointestinal tract similar to zolpidem tartrate immediate-release, then provides extended plasma concentrations beyond three hours after administration. A study in 24 healthy male subjects was conducted to compare mean zolpidem plasma concentration-time profiles obtained after single oral administration of Zolpidem Tartrate Extended-Release Tablets 12.5 mg and of an immediate-release formulation of zolpidem tartrate (10 mg). The terminal elimination half-life observed with Zolpidem Tartrate Extended-Release Tablets (12.5 mg) was similar to that obtained with immediate-release zolpidem tartrate (10 mg). The mean plasma concentration-time profiles are shown in Figure 1.
Figure 1: Mean plasma concentration-time profiles for Zolpidem Tartrate Extended-Release Tablets (12.5 mg) and immediate-release zolpidem tartrate (10 mg)
In adult and elderly patients treated with Zolpidem Tartrate Extended-Release Tablets, there was no evidence of accumulation after repeated once-daily dosing for up to two weeks.
5.8 Withdrawal Effects
There have been reports of withdrawal signs and symptoms following the rapid dose decrease or abrupt discontinuation of zolpidem. Monitor patients for tolerance, abuse, and dependence [see Drug Abuse and Dependence (9.2) and (9.3)].
8.2 Labor and Delivery
Zolpidem Tartrate Extended-Release Tablets have no established use in labor and delivery [see Pregnancy (8.1)].
8.7 Hepatic Impairment
The recommended dose of Zolpidem Tartrate Extended-Release Tablets in patients with mild to moderate hepatic impairment is 6.25 mg once daily immediately before bedtime. Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment as it may contribute to encephalopathy [see Dosage and Administration (2.2), Warnings and Precautions (5.7), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Zolpidem Tartrate Extended-Release Tablets are indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance (as measured by wake time after sleep onset).
The clinical trials performed in support of efficacy were up to 3 weeks (using polysomnography measurement up to 2 weeks in both adult and elderly patients) and 24 weeks (using patient-reported assessment in adult patients only) in duration [see Clinical Studies (14)].
10.1 Signs and Symptoms
In postmarketing experience of overdose with zolpidem tartrate alone, or in combination with CNS-depressant agents, impairment of consciousness ranging from somnolence to coma, cardiovascular and/or respiratory compromise and fatal outcomes have been reported.
2.2 Special Populations
Elderly or debilitated patients may be especially sensitive to the effects of zolpidem tartrate. The recommended dose of Zolpidem Tartrate Extended-Release Tablets in these patients is 6.25 mg once daily immediately before bedtime [see Warnings and Precautions (5.1), Use in Specific Populations (8.5)].
Patients with mild to moderate hepatic impairment do not clear the drug as rapidly as normal subjects. The recommended dose of Zolpidem Tartrate Extended-Release Tablets in these patients is 6.25 mg once daily immediately before bedtime. Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment as it may contribute to encephalopathy [see Warnings and Precautions (5.7) , Use in Specific Populations (8.7) , Clinical Pharmacology (12.3) ] .
12.1 Mechanism of Action
Zolpidem, the active moiety of zolpidem tartrate, is a hypnotic agent with a chemical structure unrelated to benzodiazepines, barbiturates, or other drugs with known hypnotic properties. It interacts with a GABA-BZ receptor complex and shares some of the pharmacological properties of the benzodiazepines. In contrast to the benzodiazepines, which non-selectively bind to and activate all BZ receptor subtypes, zolpidem in vitro binds the BZ1 receptor preferentially with a high affinity ratio of the α1/α5 subunits. This selective binding of zolpidem on the BZ1 receptor is not absolute, but it may explain the relative absence of myorelaxant and anticonvulsant effects in animal studies as well as the preservation of deep sleep (stages 3 and 4) in human studies of zolpidem tartrate at hypnotic doses.
9.1 Controlled Substance
Zolpidem tartrate is classified as a Schedule IV controlled substance by federal regulation.
10.2 Recommended Treatment
General symptomatic and supportive measures should be used along with immediate gastric lavage where appropriate. Intravenous fluids should be administered as needed. Zolpidem's sedative hypnotic effect was shown to be reduced by flumazenil and therefore may be useful; however, flumazenil administration may contribute to the appearance of neurological symptoms (convulsions). As in all cases of drug overdose, respiration, pulse, blood pressure, and other appropriate signs should be monitored and general supportive measures employed. Hypotension and CNS depression should be monitored and treated by appropriate medical intervention. Sedating drugs should be withheld following zolpidem overdosage, even if excitation occurs. The value of dialysis in the treatment of overdosage has not been determined, although hemodialysis studies in patients with renal failure receiving therapeutic doses have demonstrated that zolpidem is not dialyzable.
As with the management of all overdosage, the possibility of multiple drug ingestion should be considered. The physician may wish to consider contacting a poison control center for up-to-date information on the management of hypnotic drug product overdosage.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•CNS depressant effects: Impaired alertness and motor coordination, including risk of morning impairment. Caution patients against driving and other activities requiring complete mental alertness the morning after use. (5.1)
-
•Need to evaluate for co-morbid diagnoses: Revaluate if insomnia persists after 7 to 10 days of use. (5.2)
-
•Severe anaphylactic/anaphylactoid reactions: Angioedema and anaphylaxis have been reported. Do not rechallenge if such reactions occur. (5.3)
-
•"Sleep-driving" and other complex behaviors while not fully awake. Risk increases with dose and use with other CNS depressants and alcohol. Immediately evaluate any new onset behavioral changes. (5.4)
-
•Depression: Worsening of depression or, suicidal thinking may occur. Prescribe the least amount of tablets feasible to avoid intentional overdose. (5. 5)
-
•Respiratory Depression: Consider this risk before prescribing in patients with compromised respiratory function. (5. 6)
-
•Hepatic Impairment: Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment. (5.7)
-
•Withdrawal effects: Symptoms may occur with rapid dose reduction or discontinuation. (5.8, 9.3)
-
•Severe Injuries: Drowsiness may lead to fall including severe injuries. (5.9)
5.6 Respiratory Depression
Although studies with 10 mg zolpidem tartrate did not reveal respiratory depressant effects at hypnotic doses in healthy subjects or in patients with mild-to-moderate chronic obstructive pulmonary disease (COPD), a reduction in the Total Arousal Index, together with a reduction in lowest oxygen saturation and increase in the times of oxygen desaturation below 80% and 90%, was observed in patients with mild-to-moderate sleep apnea when treated with zolpidem compared to placebo. Since sedative-hypnotics have the capacity to depress respiratory drive, precautions should be taken if Zolpidem Tartrate Extended-Release Tablets are prescribed to patients with compromised respiratory function. Post-marketing reports of respiratory insufficiency in patients receiving 10 mg of zolpidem tartrate, most of whom had pre-existing respiratory impairment, have been reported. The risk of respiratory depression should be considered prior to prescribing Zolpidem Tartrate Extended-Release Tablets in patients with respiratory impairment including sleep apnea and myasthenia gravis.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Use the lowest dose effective for the patient and must not exceed a total of 12.5 mg daily (2.1)
-
•Recommended initial dose is a single dose of 6.25 mg for women, and a single dose of 6.25 or 12.5 mg for men, immediately before bedtime with at least 7–8 hours remaining before the planned time of awakening (2.1)
-
•Geriatric patients and patients with mild to moderate hepatic impairment: Recommended dose is 6.25 mg for men and women (2.2)
-
•Lower doses of CNS depressants may be necessary when taken concomitantly with Zolpidem Tartrate Extended-Release Tablets (2.3)
-
•Tablets to be swallowed whole, not to be crushed, divided or chewed (2.4)
-
•The effect of Zolpidem Tartrate Extended-Release Tablets may be slowed if taken with or immediately after a meal (2.4)
2.3 Use With Cns Depressants (2.3 Use with CNS Depressants)
Dosage adjustment may be necessary when Zolpidem Tartrate Extended-Release Tablets are combined with other CNS depressant drugs because of the potentially additive effects [see Warnings and Precautions (5.1)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Zolpidem Tartrate Extended-Release Tablets are available as extended-release tablets containing 12.5 mg of zolpidem tartrate for oral administration. Tablets are not scored.
Zolpidem Tartrate Extended-Release Tablets 12.5 mg tablets are blue, round, bi-convex, and debossed with ZCR on one side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Zolpidem Tartrate Extended-Release Tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Liver and biliary system: acute hepatocellular, cholestatic or mixed liver injury with or without jaundice (i.e., bilirubin >2×ULN, alkaline phosphatase ≥2×ULN, transaminase ≥5×ULN).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Pregnancy: Based on animal data may cause fetal harm (8.1)
-
•Pediatric use: Safety and effectiveness not established. Hallucinations (incidence rate 7%) and other psychiatric and/or nervous system adverse reactions were observed frequently in a study of pediatric patients with Attention-Deficit/Hyperactivity Disorder (5.4, 8.4)
14.1 Controlled Clinical Trials
Zolpidem Tartrate Extended-Release Tablets were evaluated in three placebo-controlled studies for the treatment of patients with chronic primary insomnia (as defined in the APA Diagnostic and Statistical Manual of Mental Disorders, DSM IV).
Adult outpatients (18–64 years) with primary insomnia (N=212) were evaluated in a double-blind, randomized, parallel-group, 3-week trial comparing Zolpidem Tartrate Extended-Release Tablets 12.5 mg and placebo. Zolpidem Tartrate Extended-Release Tablets 12.5 mg decreased wake time after sleep onset (WASO) for the first 7 hours during the first 2 nights and for the first 5 hours after 2 weeks of treatment. Zolpidem Tartrate Extended-Release Tablets 12.5 mg were superior to placebo on objective measures (polysomnography recordings) of sleep induction (by decreasing latency to persistent sleep [LPS]) during the first 2 nights of treatment and after 2 weeks of treatment. Zolpidem Tartrate Extended-Release Tablets 12.5 mg were also superior to placebo on the patient reported global impression regarding the aid to sleep after the first 2 nights and after 3 weeks of treatment.
Elderly outpatients (≥ 65 years) with primary insomnia (N=205) were evaluated in a double-blind, randomized, parallel-group, 3-week trial comparing Zolpidem Tartrate Extended-Release Tablets 6.25 mg and placebo. Zolpidem Tartrate Extended-Release Tablets 6.25 mg decreased wake time after sleep onset (WASO) for the first 6 hours during the first 2 nights and the first 4 hours after 2 weeks of treatment. Zolpidem Tartrate Extended-Release Tablets 6.25 mg were superior to placebo on objective measures (polysomnography recordings) of sleep induction (by decreasing LPS) during the first 2 nights of treatment and after 2 weeks on treatment. Zolpidem Tartrate Extended-Release Tablets 6.25 mg were superior to placebo on the patient reported global impression regarding the aid to sleep after the first 2 nights and after 3 weeks of treatment.
In both studies, in patients treated with Zolpidem Tartrate Extended-Release Tablets, polysomnography showed increased wakefulness at the end of the night compared to placebo-treated patients.
In a 24-week double-blind, placebo controlled, randomized study in adult outpatients (18–64 years) with primary insomnia (N=1025), Zolpidem Tartrate Extended-Release Tablets 12.5 mg administered as needed (3 to 7 nights per week) was superior to placebo over 24 weeks, on patient global impression regarding aid to sleep, and on patient-reported specific sleep parameters for sleep induction and sleep maintenance with no significant increased frequency of drug intake observed over time.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Medication Guide). Inform patients and their families about the benefits and risks of treatment with Zolpidem Tartrate Extended-Release Tablets. Inform patients of the availability of a Medication Guide and instruct them to read the Medication Guide prior to initiating treatment with Zolpidem Tartrate Extended-Release Tablets and with each prescription refill. Review the Zolpidem Tartrate Extended-Release Tablets Medication Guide with every patient prior to initiation of treatment. Instruct patients or caregivers that Zolpidem Tartrate Extended-Release Tablets should be taken only as prescribed.
5.5 Use in Patients With Depression (5.5 Use in Patients with Depression)
In primarily depressed patients treated with sedative-hypnotics, worsening of depression, and suicidal thoughts and actions (including completed suicides), have been reported. Suicidal tendencies may be present in such patients and protective measures may be required. Intentional overdosage is more common in this group of patients; therefore, the lowest number of tablets that is feasible should be prescribed for the patient at any one time.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Zolpidem Tartrate Extended-Release 12.5 mg tablets are composed of two layers and are coated, blue, round, bi-convex, debossed with ZCR on one side and supplied as:
| NDC Number | Size |
|---|---|
|
63187-862-15 |
Bottle of 15 |
|
63187-862-30 |
Bottle of 30 |
|
63187-862-60 |
Bottle of 60 |
|
63187-862-90 |
Bottle of 90 |
8.6 Gender Difference in Pharmacokinetics
Women clear zolpidem tartrate from the body at a lower rate than men. Cmax and AUC parameters of zolpidem from Zolpidem Tartrate Extended-Release Tablets were, respectively, approximately 50% and 75% higher at the same dose in adult female subjects compared to adult male subjects. Between 6 and 12 hours after dosing, zolpidem concentrations were 2- to 3 fold higher in adult female compared to adult male subjects. Given the higher blood levels of zolpidem tartrate in women compared to men at a given dose, the recommended initial dose of Zolpidem Tartrate Extended-Release Tablets for adult women is 6.25 mg, and the recommended dose for adult men is 6.25 or 12.5 mg.
In geriatric patients, clearance of zolpidem is similar in men and women. The recommended dose of Zolpidem Tartrate Extended-Release Tablets in geriatric patients is 6.25 mg regardless of gender.
5.7 Precipitation of Hepatic Encephalopathy
GABA agonists such as zolpidem tartrate have been associated with precipitation of hepatic encephalopathy in patients with hepatic insufficiency. In addition, patients with hepatic insufficiency do not clear zolpidem tartrate as rapidly as patients with normal hepatic function. Avoid Zolpidem Tartrate Extended-Release Tablets use in patients with severe hepatic impairment as it may contribute to encephalopathy [see Dosage and Administration (2.2) , Use in Specific Populations (8.7) , Clinical Pharmacology (12.3) ] .
5.2 Need to Evaluate for Co Morbid Diagnoses (5.2 Need to Evaluate for Co-morbid Diagnoses)
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after a careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated. Worsening of insomnia or the emergence of new thinking or behavior abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with sedative/hypnotic drugs, including zolpidem.
5.4 Abnormal Thinking and Behavioral Changes
Abnormal thinking and behavior changes have been reported in patients treated with sedative/hypnotics, including Zolpidem Tartrate Extended-Release Tablets. Some of these changes included decreased inhibition (e.g. aggressiveness and extroversion that seemed out of character), bizarre behavior, agitation and depersonalization. Visual and auditory hallucinations have been reported.
In controlled trials, <1% of adults with insomnia reported hallucinations. In a clinical trial, 7% of pediatric patients treated with zolpidem 0.25 mg/kg taken at bedtime reported hallucinations versus 0% treated with placebo [see Use in Specific Populations (8.4)].
Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake after ingestion of a sedative-hypnotic, with amnesia for the event) have been reported in sedative-hypnotic-naive as well as in sedative-hypnotic-experienced persons. Although behaviors such as "sleep-driving" have occurred with Zolpidem Tartrate Extended-Release Tablets alone at therapeutic doses, the co-administration of alcohol and other CNS depressants increases the risk of such behaviors, as does the use of Zolpidem Tartrate Extended-Release Tablets at doses exceeding the maximum recommended dose. Due to the risk to the patient and the community, discontinuation of Zolpidem Tartrate Extended-Release Tablets should be strongly considered for patients who report a "sleep-driving" episode.
Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with "sleep-driving", patients usually do not remember these events. Amnesia, anxiety and other neuro-psychiatric symptoms may also occur.
It can rarely be determined with certainty whether a particular instance of the abnormal behaviors listed above is drug induced, spontaneous in origin, or a result of an underlying psychiatric or physical disorder. Nonetheless, the emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
5.1 Cns Depressant Effects and Next Day Impairment (5.1 CNS Depressant Effects and Next-Day Impairment)
Zolpidem Tartrate Extended-Release Tablets are a central nervous system (CNS) depressant and can impair daytime function in some patients even when used as prescribed. Prescribers should monitor for excess depressant effects, but impairment can occur in the absence of subjective symptoms, and may not be reliably detected by ordinary clinical exam (i.e. less than formal psychomotor testing). While pharmacodynamic tolerance or adaptation to some adverse depressant effects of Zolpidem Tartrate Extended-Release Tablets may develop, patients using Zolpidem Tartrate Extended-Release Tablets should be cautioned against driving or engaging in other hazardous activities or activities requiring complete mental alertness the day after use.
Additive effects occur with concomitant use of other CNS depressants (e.g. benzodiazepines, opioids, tricyclic antidepressants, alcohol), including daytime use. Downward dose adjustment of Zolpidem Tartrate Extended-Release Tablets and concomitant CNS depressants should be considered [see Dosage and Administration (2.3)].
The use of Zolpidem Tartrate Extended-Release Tablets with other sedative-hypnotics (including other zolpidem products) at bedtime or the middle of the night is not recommended.
The risk of next-day psychomotor impairment is increased if Zolpidem Tartrate Extended-Release Tablets are taken with less than a full night of sleep remaining (7- to 8 hours); if higher than the recommended dose is taken; if co-administered with other CNS depressants or alcohol; or co-administered with other drugs that increase the blood levels of zolpidem. Patients should be warned against driving and other activities requiring complete mental alertness if Zolpidem Tartrate Extended-Release Tablets is taken in these circumstances [see Dosage and Administration (2) and Clinical Studies (14.2) ] .
Vehicle drivers and machine operators should be warned that, as with other hypnotics, there may be a possible risk of adverse reactions including drowsiness, prolonged reaction time, dizziness, sleepiness, blurred/double vision, reduced alertness and impaired driving the morning after therapy. In order to minimize this risk a full night of sleep (7–8 hours) is recommended.
5.3 Severe Anaphylactic and Anaphylactoid Reactions
Cases of angioedema involving the tongue, glottis or larynx have been reported in patients after taking the first or subsequent doses of sedative-hypnotics, including zolpidem. Some patients have had additional symptoms such as dyspnea, throat closing or nausea and vomiting that suggest anaphylaxis. Some patients have required medical therapy in the emergency department. If angioedema involves the throat, glottis or larynx, airway obstruction may occur and be fatal. Patients who develop angioedema after treatment with zolpidem should not be rechallenged with the drug.
7.2 Drugs That Affect Drug Metabolism Via Cytochrome P450 (7.2 Drugs that Affect Drug Metabolism via Cytochrome P450)
Some compounds known to induce or inhibit CYP3A may affect exposure to zolpidem. The effect of drugs that induce or inhibit other P450 enzymes on the exposure to zolpidem is not known.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:57.327877 · Updated: 2026-03-14T21:41:56.665367