Bromfenac Ophthalmic Solution
fa40fc6b-1bd0-4496-b53a-8b3e1c122a50
34391-3
Human Prescription Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Bromfenac ophthalmic solution 0.09% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
Dosage and Administration
Instill one drop into the affected eye once daily beginning 1 day prior to surgery, continued on the day of surgery and through the first 14 days post-surgery (2.1) .
Contraindications
None.
Warnings and Precautions
Sulfite Allergic Reactions (5.1) Slow or Delayed Healing (5.2) Potential for cross-sensitivity (5.3) Increase bleeding of ocular tissues (5.4) Corneal effects including keratitis (5.5) Contact Lens Wear (5.6)
Adverse Reactions
The most commonly reported adverse reactions in 2 to 7% of patients were abnormal sensation in eye, conjunctival hyperemia and eye irritation (including burning/stinging) (6.1) . To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Bromfenac ophthalmic solution 0.09% is a clear, yellow color solution, supplied in an opaque LDPE bottle with opaque LDPE nozzle and HDPE grey color cap as follows: 1.7 mL in 5 mL container NDC 65862-789-17 STORAGE: Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature].
Storage and Handling
Bromfenac ophthalmic solution 0.09% is a clear, yellow color solution, supplied in an opaque LDPE bottle with opaque LDPE nozzle and HDPE grey color cap as follows: 1.7 mL in 5 mL container NDC 65862-789-17 STORAGE: Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature].
Description
Bromfenac ophthalmic solution 0.09% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
Medication Information
Warnings and Precautions
Sulfite Allergic Reactions (5.1) Slow or Delayed Healing (5.2) Potential for cross-sensitivity (5.3) Increase bleeding of ocular tissues (5.4) Corneal effects including keratitis (5.5) Contact Lens Wear (5.6)
Indications and Usage
Bromfenac ophthalmic solution 0.09% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
Dosage and Administration
Instill one drop into the affected eye once daily beginning 1 day prior to surgery, continued on the day of surgery and through the first 14 days post-surgery (2.1) .
Contraindications
None.
Adverse Reactions
The most commonly reported adverse reactions in 2 to 7% of patients were abnormal sensation in eye, conjunctival hyperemia and eye irritation (including burning/stinging) (6.1) . To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Bromfenac ophthalmic solution 0.09% is a clear, yellow color solution, supplied in an opaque LDPE bottle with opaque LDPE nozzle and HDPE grey color cap as follows: 1.7 mL in 5 mL container NDC 65862-789-17 STORAGE: Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature].
How Supplied
Bromfenac ophthalmic solution 0.09% is a clear, yellow color solution, supplied in an opaque LDPE bottle with opaque LDPE nozzle and HDPE grey color cap as follows: 1.7 mL in 5 mL container NDC 65862-789-17 STORAGE: Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature].
Description
Bromfenac ophthalmic solution 0.09% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C.
Reproduction studies performed in rats at oral doses up to 0.9 mg/kg/day (1300 times the recommended human ophthalmic dose [RHOD]) and in rabbits at oral doses up to 7.5 mg/kg/day (11,000 times RHOD) revealed no evidence of teratogenicity due to bromfenac. However, 0.9 mg/kg/day in rats caused embryo-fetal lethality, increased neonatal mortality, and reduced postnatal growth. Pregnant rabbits treated with 7.5 mg/kg/day caused increased post-implantation loss.
There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects:
Because of the known effects of prostaglandin biosynthesis-inhibiting drugs on the fetal cardiovascular system (closure of ductus arteriosus), the use of bromfenac ophthalmic solution during late pregnancy should be avoided.
11 Description
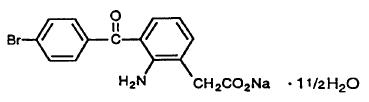
Bromfenac ophthalmic solution 0.09% is a sterile, topical, nonsteroidal anti-inflammatory drug (NSAID) for ophthalmic use. Each mL of bromfenac ophthalmic solution contains 1.035 mg bromfenac sodium (equivalent to 0.9 mg bromfenac free acid). Bromfenac sodium is designated chemically as sodium 2-amino-3-(4-bromobenzoyl) phenylacetate sesquihydrate, with an molecular formula of C15H11BrNNaO3• 1½H2O. The structural structure for bromfenac sodium is:
Bromfenac sodium is a yellow or orange yellow crystalline powder. The molecular weight of bromfenac sodium is 383.17. Bromfenac ophthalmic solution is supplied as a sterile aqueous 0.09% solution, with a pH of 8.3. The osmolality of bromfenac ophthalmic solution is approximately 300 mOsmol/kg.
Each mL of bromfenac ophthalmic solution contains:
Active: bromfenac sodium hydrate 0.1035%
Preservative: benzalkonium chloride (0.05 mg/mL)
Inactives: boric acid, edetate disodium (0.2 mg/mL), polysorbate 80 (1.5 mg/mL), povidone (20 mg/mL), sodium borate, sodium sulfite (2 mg/mL), sodium hydroxide to adjust pH, and water for injection.
8.4 Pediatric Use
Safety and efficacy in pediatric patients below the age of 18 have not been established.
8.5 Geriatric Use
There is no evidence that the efficacy or safety profiles for bromfenac ophthalmic solution differ in patients 65 years of age and older compared to younger adult patients.
4 Contraindications
None.
6 Adverse Reactions
The most commonly reported adverse reactions in 2 to 7% of patients were abnormal sensation in eye, conjunctival hyperemia and eye irritation (including burning/stinging) (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
8.3 Nursing Mothers
Caution should be exercised when bromfenac ophthalmic solution is administered to a nursing woman.
12.3 Pharmacokinetics
The plasma concentration of bromfenac following ocular administration of 0.09% bromfenac ophthalmic solution in humans is unknown. Based on the maximum proposed dose of one drop to the eye (0.045 mg) and PK information from other routes of administration, the systemic concentration of bromfenac is estimated to be below the limit of quantification (50 ng/mL) at steady-state in humans.
5.6 Contact Lens Wear
Bromfenac ophthalmic solution should not be administered while wearing contact lenses.
2.1 Recommended Dosing
For the treatment of postoperative inflammation in patients who have undergone cataract extraction, one drop of bromfenac ophthalmic solution should be applied to the affected eye once daily beginning 1 day prior to cataract surgery, continued on the day of surgery, and through the first 14 days of the postoperative period.
1 Indications and Usage
Bromfenac ophthalmic solution 0.09% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
12.1 Mechanism of Action
Bromfenac is a nonsteroidal anti-inflammatory drug (NSAID) that has anti-inflammatory activity. The mechanism of its action is thought to be due to its ability to block prostaglandin synthesis by inhibiting cyclooxygenase 1 and 2.
Prostaglandins have been shown in many animal models to be mediators of certain kinds of intraocular inflammation. In studies performed in animal eyes, prostaglandins have been shown to produce disruption of the blood-aqueous humor barrier, vasodilation, increased vascular permeability, leukocytosis, and increased intraocular pressure.
5 Warnings and Precautions
2 Dosage and Administration
Instill one drop into the affected eye once daily beginning 1 day prior to surgery, continued on the day of surgery and through the first 14 days post-surgery (2.1).
5.2 Slow Or Delayed Healing
All topical nonsteroidal anti-inflammatory drugs (NSAIDs) may slow or delay healing. Topical corticosteroids are also known to slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
5.4 Increased Bleeding Time
With some NSAIDs, there exists the potential for increased bleeding time due to interference with platelet aggregation. There have been reports that ocularly applied NSAIDs may cause increased bleeding of ocular tissues (including hyphemas) in conjunction with ocular surgery.
It is recommended that bromfenac ophthalmic solution be used with caution in patients with known bleeding tendencies or who are receiving other medications which may prolong bleeding time.
3 Dosage Forms and Strengths
Topical ophthalmic solution: bromfenac 0.09%.
17.2 Sterility of Dropper Tip
Patients should be advised to not touch dropper tip to any surface, as this may contaminate the contents.
6.1 Clinical Trial Experience
The most commonly reported adverse experiences reported following use of bromfenac after cataract surgery include: abnormal sensation in eye, conjunctival hyperemia, eye irritation (including burning/stinging), eye pain, eye pruritus, eye redness, headache, and iritis. These events were reported in 2 to 7% of patients.
6.2 Post Marketing Experience
The following events have been identified during post-marketing use of bromfenac ophthalmic solution 0.09% in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to topical bromfenac ophthalmic solution 0.09% or a combination of these factors, include corneal erosion, corneal perforation, corneal thinning, and epithelial breakdown [ see Warnings and Precautions (5) ].
17.1 Slowed Or Delayed Healing
Patients should be advised of the possibility that slow or delayed healing may occur while using NSAIDs.
5.1 Sulfite Allergic Reactions
Contains sodium sulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
14.1 Ocular Inflammation and Pain
Clinical efficacy was evaluated in three randomized, double-masked, placebo-controlled trials in which subjects requiring cataract surgery were assigned to bromfenac ophthalmic solution or placebo. Patients were dosed with one drop per eye starting the day before surgery and continuing for 14 days. The primary endpoint was clearing of ocular inflammation by day 15. An additional efficacy endpoint was the number of patients who were pain free on day 1 after cataract surgery.
In 2 of the 3 studies, bromfenac ophthalmic solution had statistically significant higher incidence of completely clearing inflammation (46 to 47% vs. 25 to 29%) and also had a statistically significant higher incidence of subjects that were pain free at day 1 post cataract surgery (83 to 89% vs. 51 to 71%).
5.3 Potential for Cross Sensitivity
There is the potential for cross-sensitivity to acetylsalicylic acid, phenylacetic acid derivatives, and other NSAIDs. Therefore, caution should be used when treating individuals who have previously exhibited sensitivities to these drugs.
5.5 Keratitis and Corneal Reactions
Use of topical NSAIDs may result in keratitis. In some susceptible patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs and should be closely monitored for corneal health.
Post-marketing experience with topical NSAIDs suggests that patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g., dry eye syndrome), rheumatoid arthritis, or repeat ocular surgeries within a short period of time may be at increased risk for corneal adverse events which may become sight threatening. Topical NSAIDs should be used with caution in these patients.
Post-marketing experience with topical NSAIDs also suggests that use more than 24 hours prior to surgery or use beyond 14 days post surgery may increase patient risk for the occurrence and severity of corneal adverse events.
16 How Supplied/storage and Handling
Bromfenac ophthalmic solution 0.09% is a clear, yellow color solution, supplied in an opaque LDPE bottle with opaque LDPE nozzle and HDPE grey color cap as follows:
1.7 mL in 5 mL container NDC 65862-789-17
STORAGE:
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature].
17.3 Concomitant Use of Contact Lenses
Contact lenses should not be worn during the use of this product.
17.4 Concomitant Topical Ocular Therapy
If more than one topical ophthalmic medication is being used, the medicines should be administered at least 5 minutes apart.
Rx Only
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
2.2 Use With Other Topical Ophthalmic Medications
Bromfenac ophthalmic solution may be administered in conjunction with other topical ophthalmic medications such as alpha-agonists, beta-blockers, carbonic anhydrase inhibitors, cycloplegics, and mydriatics. Drops should be administered at least 5 minutes apart.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies in rats and mice given oral doses of bromfenac up to 0.6 mg/kg/day (900 times the recommended human ophthalmic dose [RHOD] of 1.67 mcg/kg in 60 kg person on a mg/kg/basis, assuming 100% absorbed) and 5 mg/kg/day (7500 times RHOD), respectively revealed no significant increases in tumor incidence.
Bromfenac did not show mutagenic potential in various mutagenicity studies, including the reverse mutation, chromosomal aberration, and micronucleus tests.
Bromfenac did not impair fertility when administered orally to male and female rats at doses up to 0.9 mg/kg/day and 0.3 mg/kg/day, respectively (1300 and 450 times RHOD, respectively).
Package Label Principal Display Panel 0.09% (1.7 Ml Carton)
NDC 65862-789-17
Bromfenac
Ophthalmic
Solution
0.09%
For Topical Application in the Eye
Once Daily
Sterile
Rx only 1.7 mL
AUROBINDO
Package Label Principal Display Panel 0.09% (1.7 Ml Container)
NDC 65862-789-17
Bromfenac
Ophthalmic
Solution
0.09%
For Topical Application in the Eye
Once Daily
AUROBINDO
Structured Label Content
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C.
Reproduction studies performed in rats at oral doses up to 0.9 mg/kg/day (1300 times the recommended human ophthalmic dose [RHOD]) and in rabbits at oral doses up to 7.5 mg/kg/day (11,000 times RHOD) revealed no evidence of teratogenicity due to bromfenac. However, 0.9 mg/kg/day in rats caused embryo-fetal lethality, increased neonatal mortality, and reduced postnatal growth. Pregnant rabbits treated with 7.5 mg/kg/day caused increased post-implantation loss.
There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects:
Because of the known effects of prostaglandin biosynthesis-inhibiting drugs on the fetal cardiovascular system (closure of ductus arteriosus), the use of bromfenac ophthalmic solution during late pregnancy should be avoided.
11 Description (11 DESCRIPTION)
Bromfenac ophthalmic solution 0.09% is a sterile, topical, nonsteroidal anti-inflammatory drug (NSAID) for ophthalmic use. Each mL of bromfenac ophthalmic solution contains 1.035 mg bromfenac sodium (equivalent to 0.9 mg bromfenac free acid). Bromfenac sodium is designated chemically as sodium 2-amino-3-(4-bromobenzoyl) phenylacetate sesquihydrate, with an molecular formula of C15H11BrNNaO3• 1½H2O. The structural structure for bromfenac sodium is:
Bromfenac sodium is a yellow or orange yellow crystalline powder. The molecular weight of bromfenac sodium is 383.17. Bromfenac ophthalmic solution is supplied as a sterile aqueous 0.09% solution, with a pH of 8.3. The osmolality of bromfenac ophthalmic solution is approximately 300 mOsmol/kg.
Each mL of bromfenac ophthalmic solution contains:
Active: bromfenac sodium hydrate 0.1035%
Preservative: benzalkonium chloride (0.05 mg/mL)
Inactives: boric acid, edetate disodium (0.2 mg/mL), polysorbate 80 (1.5 mg/mL), povidone (20 mg/mL), sodium borate, sodium sulfite (2 mg/mL), sodium hydroxide to adjust pH, and water for injection.
8.4 Pediatric Use
Safety and efficacy in pediatric patients below the age of 18 have not been established.
8.5 Geriatric Use
There is no evidence that the efficacy or safety profiles for bromfenac ophthalmic solution differ in patients 65 years of age and older compared to younger adult patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most commonly reported adverse reactions in 2 to 7% of patients were abnormal sensation in eye, conjunctival hyperemia and eye irritation (including burning/stinging) (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
8.3 Nursing Mothers
Caution should be exercised when bromfenac ophthalmic solution is administered to a nursing woman.
12.3 Pharmacokinetics
The plasma concentration of bromfenac following ocular administration of 0.09% bromfenac ophthalmic solution in humans is unknown. Based on the maximum proposed dose of one drop to the eye (0.045 mg) and PK information from other routes of administration, the systemic concentration of bromfenac is estimated to be below the limit of quantification (50 ng/mL) at steady-state in humans.
5.6 Contact Lens Wear
Bromfenac ophthalmic solution should not be administered while wearing contact lenses.
2.1 Recommended Dosing
For the treatment of postoperative inflammation in patients who have undergone cataract extraction, one drop of bromfenac ophthalmic solution should be applied to the affected eye once daily beginning 1 day prior to cataract surgery, continued on the day of surgery, and through the first 14 days of the postoperative period.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Bromfenac ophthalmic solution 0.09% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
12.1 Mechanism of Action
Bromfenac is a nonsteroidal anti-inflammatory drug (NSAID) that has anti-inflammatory activity. The mechanism of its action is thought to be due to its ability to block prostaglandin synthesis by inhibiting cyclooxygenase 1 and 2.
Prostaglandins have been shown in many animal models to be mediators of certain kinds of intraocular inflammation. In studies performed in animal eyes, prostaglandins have been shown to produce disruption of the blood-aqueous humor barrier, vasodilation, increased vascular permeability, leukocytosis, and increased intraocular pressure.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Instill one drop into the affected eye once daily beginning 1 day prior to surgery, continued on the day of surgery and through the first 14 days post-surgery (2.1).
5.2 Slow Or Delayed Healing (5.2 Slow or Delayed Healing)
All topical nonsteroidal anti-inflammatory drugs (NSAIDs) may slow or delay healing. Topical corticosteroids are also known to slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
5.4 Increased Bleeding Time
With some NSAIDs, there exists the potential for increased bleeding time due to interference with platelet aggregation. There have been reports that ocularly applied NSAIDs may cause increased bleeding of ocular tissues (including hyphemas) in conjunction with ocular surgery.
It is recommended that bromfenac ophthalmic solution be used with caution in patients with known bleeding tendencies or who are receiving other medications which may prolong bleeding time.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical ophthalmic solution: bromfenac 0.09%.
17.2 Sterility of Dropper Tip
Patients should be advised to not touch dropper tip to any surface, as this may contaminate the contents.
6.1 Clinical Trial Experience
The most commonly reported adverse experiences reported following use of bromfenac after cataract surgery include: abnormal sensation in eye, conjunctival hyperemia, eye irritation (including burning/stinging), eye pain, eye pruritus, eye redness, headache, and iritis. These events were reported in 2 to 7% of patients.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following events have been identified during post-marketing use of bromfenac ophthalmic solution 0.09% in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to topical bromfenac ophthalmic solution 0.09% or a combination of these factors, include corneal erosion, corneal perforation, corneal thinning, and epithelial breakdown [ see Warnings and Precautions (5) ].
17.1 Slowed Or Delayed Healing (17.1 Slowed or Delayed Healing)
Patients should be advised of the possibility that slow or delayed healing may occur while using NSAIDs.
5.1 Sulfite Allergic Reactions
Contains sodium sulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
14.1 Ocular Inflammation and Pain
Clinical efficacy was evaluated in three randomized, double-masked, placebo-controlled trials in which subjects requiring cataract surgery were assigned to bromfenac ophthalmic solution or placebo. Patients were dosed with one drop per eye starting the day before surgery and continuing for 14 days. The primary endpoint was clearing of ocular inflammation by day 15. An additional efficacy endpoint was the number of patients who were pain free on day 1 after cataract surgery.
In 2 of the 3 studies, bromfenac ophthalmic solution had statistically significant higher incidence of completely clearing inflammation (46 to 47% vs. 25 to 29%) and also had a statistically significant higher incidence of subjects that were pain free at day 1 post cataract surgery (83 to 89% vs. 51 to 71%).
5.3 Potential for Cross Sensitivity (5.3 Potential for Cross-Sensitivity)
There is the potential for cross-sensitivity to acetylsalicylic acid, phenylacetic acid derivatives, and other NSAIDs. Therefore, caution should be used when treating individuals who have previously exhibited sensitivities to these drugs.
5.5 Keratitis and Corneal Reactions
Use of topical NSAIDs may result in keratitis. In some susceptible patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs and should be closely monitored for corneal health.
Post-marketing experience with topical NSAIDs suggests that patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g., dry eye syndrome), rheumatoid arthritis, or repeat ocular surgeries within a short period of time may be at increased risk for corneal adverse events which may become sight threatening. Topical NSAIDs should be used with caution in these patients.
Post-marketing experience with topical NSAIDs also suggests that use more than 24 hours prior to surgery or use beyond 14 days post surgery may increase patient risk for the occurrence and severity of corneal adverse events.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Bromfenac ophthalmic solution 0.09% is a clear, yellow color solution, supplied in an opaque LDPE bottle with opaque LDPE nozzle and HDPE grey color cap as follows:
1.7 mL in 5 mL container NDC 65862-789-17
STORAGE:
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature].
17.3 Concomitant Use of Contact Lenses
Contact lenses should not be worn during the use of this product.
17.4 Concomitant Topical Ocular Therapy
If more than one topical ophthalmic medication is being used, the medicines should be administered at least 5 minutes apart.
Rx Only
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
2.2 Use With Other Topical Ophthalmic Medications (2.2 Use with Other Topical Ophthalmic Medications)
Bromfenac ophthalmic solution may be administered in conjunction with other topical ophthalmic medications such as alpha-agonists, beta-blockers, carbonic anhydrase inhibitors, cycloplegics, and mydriatics. Drops should be administered at least 5 minutes apart.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies in rats and mice given oral doses of bromfenac up to 0.6 mg/kg/day (900 times the recommended human ophthalmic dose [RHOD] of 1.67 mcg/kg in 60 kg person on a mg/kg/basis, assuming 100% absorbed) and 5 mg/kg/day (7500 times RHOD), respectively revealed no significant increases in tumor incidence.
Bromfenac did not show mutagenic potential in various mutagenicity studies, including the reverse mutation, chromosomal aberration, and micronucleus tests.
Bromfenac did not impair fertility when administered orally to male and female rats at doses up to 0.9 mg/kg/day and 0.3 mg/kg/day, respectively (1300 and 450 times RHOD, respectively).
Package Label Principal Display Panel 0.09% (1.7 Ml Carton) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL 0.09% (1.7 mL Carton))
NDC 65862-789-17
Bromfenac
Ophthalmic
Solution
0.09%
For Topical Application in the Eye
Once Daily
Sterile
Rx only 1.7 mL
AUROBINDO
Package Label Principal Display Panel 0.09% (1.7 Ml Container) (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL 0.09% (1.7 mL Container))
NDC 65862-789-17
Bromfenac
Ophthalmic
Solution
0.09%
For Topical Application in the Eye
Once Daily
AUROBINDO
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:47.633118 · Updated: 2026-03-14T22:06:44.115888