These Highlights Do Not Include All The Information Needed To Use Zoryve Foam, 0.3%, Safely And Effectively. See Full Prescribing Information For Zoryve Foam, 0.3%.
f9e0b4a9-a227-4947-b00c-41fb18d16589
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage, Plaque Psoriasis ( 1.2 ) 5/2025
Indications and Usage
ZORYVE topical foam, 0.3%, is a phosphodiesterase 4 inhibitor indicated for the treatment of seborrheic dermatitis in adult and pediatric patients 9 years of age and older. ( 1.1 ) plaque psoriasis of the scalp and body in adult and pediatric patients 12 years of age and older. ( 1.2 )
Dosage and Administration
Shake can prior to each use. Apply a thin layer of ZORYVE foam, 0.3%, once daily to affected areas of body and/or scalp when they are not wet. Rub in completely. Wash hands after application. Avoid fire, flame, and smoking during and immediately following application [see Warnings and Precautions (5.1) ] . ZORYVE foam, 0.3%, is for topical use only and not for ophthalmic, oral, or intravaginal use.
Warnings and Precautions
Flammability : The propellants in ZORYVE foam, 0.3%, are flammable. Avoid fire, flame, and smoking during and immediately following application. ( 5.1 )
Contraindications
ZORYVE foam, 0.3%, is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C) [see Use in Specific Populations (8.6) , Clinical Pharmacology (12.3) ] .
Adverse Reactions
The most common adverse reactions (reported in ≥ 1% of patients) are: Seborrheic dermatitis : nasopharyngitis, nausea, and headache. ( 6.1 ) Plaque psoriasis of the scalp and body : headache, diarrhea, nausea, and nasopharyngitis. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Arcutis Biotherapeutics, Inc. at 1-844-692-6729 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Co-administration of roflumilast with systemic CYP3A4 inhibitors or dual inhibitors that inhibit both CYP3A4 and CYP1A2 simultaneously may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. ( 7.1 ) Co-administration of roflumilast with oral contraceptives containing gestodene and ethinyl estradiol may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. ( 7.1 )
Medication Information
Warnings and Precautions
Flammability : The propellants in ZORYVE foam, 0.3%, are flammable. Avoid fire, flame, and smoking during and immediately following application. ( 5.1 )
Indications and Usage
ZORYVE topical foam, 0.3%, is a phosphodiesterase 4 inhibitor indicated for the treatment of seborrheic dermatitis in adult and pediatric patients 9 years of age and older. ( 1.1 ) plaque psoriasis of the scalp and body in adult and pediatric patients 12 years of age and older. ( 1.2 )
Dosage and Administration
Shake can prior to each use. Apply a thin layer of ZORYVE foam, 0.3%, once daily to affected areas of body and/or scalp when they are not wet. Rub in completely. Wash hands after application. Avoid fire, flame, and smoking during and immediately following application [see Warnings and Precautions (5.1) ] . ZORYVE foam, 0.3%, is for topical use only and not for ophthalmic, oral, or intravaginal use.
Contraindications
ZORYVE foam, 0.3%, is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C) [see Use in Specific Populations (8.6) , Clinical Pharmacology (12.3) ] .
Adverse Reactions
The most common adverse reactions (reported in ≥ 1% of patients) are: Seborrheic dermatitis : nasopharyngitis, nausea, and headache. ( 6.1 ) Plaque psoriasis of the scalp and body : headache, diarrhea, nausea, and nasopharyngitis. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Arcutis Biotherapeutics, Inc. at 1-844-692-6729 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Co-administration of roflumilast with systemic CYP3A4 inhibitors or dual inhibitors that inhibit both CYP3A4 and CYP1A2 simultaneously may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. ( 7.1 ) Co-administration of roflumilast with oral contraceptives containing gestodene and ethinyl estradiol may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. ( 7.1 )
Description
Indications and Usage, Plaque Psoriasis ( 1.2 ) 5/2025
Section 42229-5
Seborrheic Dermatitis
In two multicenter, randomized, double-blind, vehicle-controlled trials (Trial 203 and STRATUM), 683 adult and pediatric subjects 9 years of age or older with seborrheic dermatitis were treated with ZORYVE foam, 0.3%, or vehicle foam once daily for 8 weeks [see Clinical Studies (14.1)].
Table 1 presents adverse reactions that occurred in at least 1% of subjects treated with ZORYVE foam, 0.3%.
| Adverse Reaction | ZORYVE foam, 0.3% (N=458) n (%) |
Vehicle foam (N=225) n (%) |
|---|---|---|
| Nasopharyngitis | 7 (1.5) | 1 (0.4) |
| Nausea | 6 (1.3) | 0 (0) |
| Headache | 5 (1.1) | 0 (0) |
The following additional adverse reactions were reported in fewer than 1% of subjects treated with ZORYVE foam, 0.3%: diarrhea and insomnia.
The adverse reaction profile in pediatric subjects was consistent with that observed in adults [see Use in Specific Populations (8.4), Clinical Pharmacology (12.3)].
In 408 subjects who continued treatment with ZORYVE foam, 0.3%, for up to 24 to 52 weeks in an open-label, long-term trial, the adverse reaction profile was consistent with that observed in vehicle-controlled trials.
Section 42230-3
| Patient Information ZORYVE® (zor-EEV) (roflumilast) topical foam, 0.3% |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 5/2025 |
| Important information: ZORYVE foam is for use on the skin (topical use) only. Do not use ZORYVE foam in or on your eyes, mouth, or vagina. | |
|
What is ZORYVE foam?
ZORYVE foam is a prescription medicine used on the skin (topical) to treat:
|
|
| Do not use ZORYVE foam if you have certain liver problems. | |
Before using ZORYVE foam, tell your healthcare provider about all of your medical conditions, including if you:
|
|
How should I use ZORYVE foam?
|
|
|
What should I avoid while using ZORYVE foam? ZORYVE foam is flammable. Avoid fire, flame, and smoking during and right after you apply ZORYVE foam. |
|
|
What are the possible side effects of ZORYVE foam? The most common side effects of ZORYVE foam in people treated for seborrheic dermatitis include:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Arcutis Biotherapeutics, Inc. by calling 1-844-692-6729. |
|
How should I store ZORYVE foam?
|
|
|
General Information about the safe and effective use of ZORYVE foam.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ZORYVE foam for a condition for which it was not prescribed. Do not give it to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ZORYVE foam that is written for health professionals. |
|
|
What are the ingredients in ZORYVE foam?
Active ingredient: roflumilast Inactive ingredients: ceteareth-10 phosphate, cetearyl phosphate, cetostearyl alcohol, diethylene glycol monoethyl ether, hexylene glycol, isopropyl palmitate, methylparaben, propylparaben, purified water, sodium hydroxide, and white petrolatum. Hydrochloric acid may have been added to adjust pH. Propellants: butane, isobutane, and propane. Marketed by: Arcutis Biotherapeutics, Inc. Westlake Village, CA 91361 For more information on ZORYVE topical foam, call 1-844-692-6729 or visit http://www.zoryve.com. |
v2
Section 43683-2
| Indications and Usage, Plaque Psoriasis (1.2) | 5/2025 |
Section 44425-7
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [See USP Controlled Room Temperature].
Do not freeze.
Store upright.
Flammable. Contents under pressure. Do not puncture or incinerate. Do not expose to heat or store at temperatures above 49°C (120°F) [see Warnings and Precautions (5.1)].
8.3 Fertility
In a human spermatogenesis study, oral roflumilast 500 mcg had no effects on semen parameters or reproductive hormones during the 3-month treatment period and the following 3-month off-treatment period.
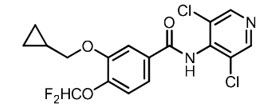
11 Description
ZORYVE (roflumilast) topical foam, 0.3%, is a white to off-white foam for topical use. The active ingredient, roflumilast, is a phosphodiesterase 4 (PDE4) inhibitor.
The chemical name of roflumilast is 3-cyclopropylmethoxy-N-(3,5-dichloropyridin-4-yl)-4-(difluoromethoxy)benzamide with a molecular formula of C17H14Cl2F2N2O3 and the molecular weight of 403.21.
The structural formula of roflumilast is:
Roflumilast is practically insoluble in water and hexane, sparingly soluble in ethanol, and freely soluble in acetone.
Each gram of ZORYVE topical foam, 0.3%, contains 3 mg of roflumilast in a foam base containing ceteareth-10 phosphate, cetearyl phosphate, cetostearyl alcohol, diethylene glycol monoethyl ether, hexylene glycol, isopropyl palmitate, methylparaben, propylparaben, purified water, sodium hydroxide, and white petrolatum. Hydrochloric acid may have been added to adjust pH. ZORYVE topical foam, 0.3%, is dispensed from an aluminum can pressurized with propellant (butane, isobutane, and propane).
5.1 Flammability
The propellants in ZORYVE foam, 0.3%, are flammable. Avoid fire, flame, and smoking during and immediately following application.
4 Contraindications
ZORYVE foam, 0.3%, is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
6 Adverse Reactions
The most common adverse reactions (reported in ≥ 1% of patients) are:
- Seborrheic dermatitis: nasopharyngitis, nausea, and headache. (6.1)
- Plaque psoriasis of the scalp and body: headache, diarrhea, nausea, and nasopharyngitis. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Arcutis Biotherapeutics, Inc. at 1-844-692-6729 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
- Co-administration of roflumilast with systemic CYP3A4 inhibitors or dual inhibitors that inhibit both CYP3A4 and CYP1A2 simultaneously may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. (7.1)
- Co-administration of roflumilast with oral contraceptives containing gestodene and ethinyl estradiol may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. (7.1)
1.2 Plaque Psoriasis
ZORYVE topical foam, 0.3%, is indicated for the treatment of plaque psoriasis of the scalp and body in adult and pediatric patients 12 years of age and older.
Instructions for Use
This Instructions for Use contains information on how to apply ZORYVE foam.
Read this Instructions for Use before you start using ZORYVE foam and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. Use ZORYVE foam exactly as your healthcare provider tells you.
Important information you need to know before applying ZORYVE foam:
- ZORYVE foam is for use on skin only (topical use). ZORYVE foam is not for use in your eyes, mouth, or vagina.
- ZORYVE foam is flammable. Avoid fire, flame, and smoking during and right after you apply ZORYVE foam.
Before applying ZORYVE foam for the first time:
| Gently pull back on the nozzle to break the plastic piece at the base. |
Applying ZORYVE foam:
Apply a thin layer of ZORYVE foam 1 time a day to the affected areas of your body and scalp when they are not wet.
Step 1: Shake.
|
|
Step 2: Dispense.
|
|
Step 3: Apply.
|
|
Storing ZORYVE foam
- Store ZORYVE foam at room temperature between 68°F to 77°F (20°C to 25°C).
- ZORYVE foam is flammable. Keep away from heat and flame.
- The contents in ZORYVE foam are under pressure. Do not puncture or burn the can. Do not expose the can to heat or store at temperatures above 120°F (49°C).
- Do not freeze.
- Store the can upright.
Keep ZORYVE foam and all medicines out of the reach of children.
Marketed by:
Arcutis Biotherapeutics, Inc.
Westlake Village, CA 91361
For more information on ZORYVE foam, call 1-844-692-6729 or visit http://www.zoryve.com.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 5/2025
v2
12.2 Pharmacodynamics
Pharmacodynamics of ZORYVE foam, 0.3%, in the treatment of seborrheic dermatitis and plaque psoriasis is unknown.
14.2 Plaque Psoriasis
Two randomized, double-blind, vehicle-controlled trials (ARRECTOR [NCT05028582] and Trial 204 [NCT04128007]) enrolled a total of 736 adult and pediatric subjects 12 years of age and older with mild to severe plaque psoriasis of the scalp and body. In each trial, subjects were randomized 2:1 to receive ZORYVE foam, 0.3%, or vehicle foam applied once daily for 8 weeks. The combined trial population was 55% female, 85% White, 5% Black, 6% Asian, and 4% other races; for ethnicity, 79% identified as non-Hispanic/Latino and 19% identified as Hispanic/Latino. The median age was 47 years (range 12 to 87 years).
In Trial ARRECTOR, the trial population ranged in age from 12 to 87 years, including 2% of subjects who were 12 to 17 years of age and 13% of subjects who were 65 years of age or older. At baseline, 86% of subjects had a Scalp Investigator Global Assessment (S-IGA) score of 3 (moderate) on a 5-point scale of 0 to 4, and 14% had an S-IGA score of 4 (severe); 28% of subjects had a Body Investigator Global Assessment (B-IGA) score of 2 (mild), 67% of subjects had a B-IGA score of 3 (moderate), and 5% had a B-IGA score of 4 (severe). At baseline, 76% of subjects had a Scalp Itch-Numeric Rating Scale (SI-NRS) score of 4 or higher on a scale of 0 to 10 and 73% had a Worst Itch-Numeric Rating Scale (WI-NRS) score of 4 or higher.
In Trial 204, the trial population ranged in age from 12 to 87 years, including 1% of subjects who were 12 to 17 years of age, and 9% who were 65 years of age or older. At baseline, 11% of subjects had an S-IGA score of 2 (mild), 76% of subjects had an S-IGA score of 3 (moderate), and 13% had an S-IGA score of 4 (severe); 36% of subjects had a B-IGA score of 2 (mild), 59% of subjects had a B-IGA score of 3 (moderate), and 5% had a B-IGA score of 4 (severe). At baseline, 89% of subjects had an SI-NRS score of 4 or higher on a scale of 0 to 10.
In both trials, S-IGA treatment success, a primary endpoint in ARRECTOR and Trial 204, and B-IGA treatment success, a primary endpoint in ARRECTOR, were defined as a score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade improvement from baseline.
| Trial ARRECTOR | Trial 204 | |||
|---|---|---|---|---|
| ZORYVE foam, 0.3% | Vehicle foam | ZORYVE foam, 0.3% | Vehicle foam | |
| Number of subjects randomized | N=281 | N=151 | N=200 | N=104 |
| Abbreviations: CI = Confidence Interval | ||||
| S-IGA success S-IGA treatment success and B-IGA treatment success were defined as a score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade score improvement from baseline at Week 8 (Multiple Imputation).
|
66.4% | 27.8% | 56.7% | 11.0% |
| Difference from Vehicle (95% CI) Treatment difference and 95% CI are based on the CMH method stratified by pooled site and baseline IGA strata.
|
37.1% (27.1%, 47.1%) | 47.7% (37.9%, 57.5%) | ||
| B-IGA success | 45.5% | 20.1% | 39.0% | 7.4% |
| Difference from Vehicle (95% CI) | 24.8% (15.0%, 34.6%) | 32.4% (23.3%, 41.6%) |
SI-NRS success and WI-NRS success were defined as a reduction of at least 4 points from baseline with a baseline score of at least 4. In Trial ARRECTOR, among subjects with a baseline SI-NRS score of at least 4 (75% of subjects), a higher percentage of subjects achieved SI-NRS success at Week 8 in the group who received ZORYVE foam, 0.3%, compared to the group who received vehicle foam (65.3% vs. 30.3% for a treatment difference of 35.4% and 95% CI of (23.9, 47.0)). In Trial ARRECTOR, among subjects with a baseline WI-NRS score of at least 4 (72% of subjects), a higher percentage of subjects achieved WI-NRS success at Week 8 in the group who received ZORYVE foam, 0.3%, compared to the group who received vehicle foam (63.1% vs. 30.1% for a treatment difference of 32.8% and 95% CI of (20.3, 45.2)).
8.6 Hepatic Impairment
Oral roflumilast 250 mcg once daily for 14 days was studied in subjects with hepatic impairment. The systemic exposure of roflumilast and roflumilast N-oxide were increased in subjects with moderate (Child-Pugh B) hepatic impairment. ZORYVE foam, 0.3%, is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C). No dosage adjustment is needed in patients with mild (Child-Pugh A) hepatic impairment [see Contraindications (4), Clinical Pharmacology (12.3)].
1 Indications and Usage
12.1 Mechanism of Action
Roflumilast and its active metabolite (roflumilast N-oxide) are inhibitors of PDE4. Roflumilast and roflumilast N-oxide inhibition of PDE4 (a major cyclic 3′,5′-adenosine monophosphate (cyclic AMP) metabolizing enzyme) activity leads to accumulation of intracellular cyclic AMP. The specific mechanism(s) by which roflumilast exerts its therapeutic action is not well defined.
1.1 Seborrheic Dermatitis
ZORYVE® topical foam, 0.3%, is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients 9 years of age and older.
14.1 Seborrheic Dermatitis
Two randomized, double-blind, vehicle-controlled trials (STRATUM [NCT04973228] and Trial 203 [NCT04091646]) enrolled a total of 683 adult and pediatric subjects with seborrheic dermatitis involving the scalp, face, and/or body with an Investigator Global Assessment (IGA) of moderate or severe (IGA of 3 or 4 on a 5-point scale from 0 to 4). In each trial, subjects were randomized 2:1 to receive ZORYVE foam, 0.3%, or vehicle foam applied once daily for 8 weeks. The combined trial population was 50% male, 79% White, 11% Black, 5% Asian, and 5% other races; for ethnicity, 79% identified as non-Hispanic/Latino and 21% identified as Hispanic/Latino. In Trial STRATUM, the trial population ranged in age from 9 to 87 years, including 7% of subjects who were 9 to 17 years of age and 12% of subjects who were 65 years of age or older. At baseline, 94% of subjects had an IGA score of 3 (moderate), and 6% had an IGA score of 4 (severe). At baseline, 67% of subjects had a Worst Itch-Numeric Rating Scale (WI-NRS) score of 4 or higher on a scale of 0 to 10. In Trial 203, the trial population ranged in age from 18 to 85 years, including 18% who were 65 years of age or older. At baseline, 93% of subjects had an IGA score of 3 (moderate), and 7% had an IGA score of 4 (severe). At baseline, 81% of subjects had a Worst Itch-Numeric Rating Scale (WI-NRS) score of 4 or higher on a scale of 0 to 10.
The primary endpoint was the proportion of subjects who achieved IGA treatment success at Week 8 (Table 3). Success was defined as a score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade improvement from baseline.
| STRATUM | Trial 203 | |||
|---|---|---|---|---|
| ZORYVE foam, 0.3% | Vehicle foam | ZORYVE foam, 0.3% | Vehicle foam | |
| Number of subjects randomized | N=304 | N=153 | N=154 | N=72 |
| Abbreviations: CI = Confidence Interval | ||||
| IGA success IGA treatment success was defined as an IGA score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade IGA score improvement from baseline at Week 8 (Multiple Imputation).
|
79.5% | 58.0% | 73.1% | 40.8% |
| Difference from Vehicle (95% CI) Treatment difference and 95% CI are based on the CMH method stratified by pooled site and baseline IGA strata.
|
20.6% (11.2%, 30.0%) | 33.8% (20.3%, 47.4%) |
In Trial STRATUM, among subjects with a baseline WI-NRS score of at least 4 (67% of subjects), there was a higher percentage of subjects who achieved a reduction of at least 4 points from baseline at Week 8 in the group who received ZORYVE foam, 0.3%, compared to the group who received vehicle foam (62.8% vs. 40.6% for a treatment difference of 25.7% and 95% CI of (13.4, 38.1)).
5 Warnings and Precautions
Flammability: The propellants in ZORYVE foam, 0.3%, are flammable. Avoid fire, flame, and smoking during and immediately following application. (5.1)
2 Dosage and Administration
Shake can prior to each use. Apply a thin layer of ZORYVE foam, 0.3%, once daily to affected areas of body and/or scalp when they are not wet. Rub in completely.
Wash hands after application.
Avoid fire, flame, and smoking during and immediately following application [see Warnings and Precautions (5.1)].
ZORYVE foam, 0.3%, is for topical use only and not for ophthalmic, oral, or intravaginal use.
3 Dosage Forms and Strengths
Topical foam, 0.3%: 3 mg of roflumilast per gram of white to off-white foam in 60-gram pressurized cans.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient or caregiver to read the FDA-approved patient labeling (Patient Information).
Principal Display Panel 60 G Can Carton
NDC 80610-430-60
ZORYVE®
(roflumilast)
topical foam, 0.3%
Rx Only
60 grams
For Topical Use Only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies were conducted in hamsters and mice with roflumilast to evaluate its carcinogenic potential. In 2-year oral gavage carcinogenicity studies, roflumilast treatment resulted in dose-related, statistically significant increases in the incidence of undifferentiated carcinomas of nasal epithelium in hamsters at doses greater than or equal to 8 mg/kg/day (11 times the MRHD on an AUC basis). The tumorigenicity of roflumilast appears to be attributed to a reactive metabolite of 4-amino-3,5-dichloropyridine N-oxide (ADCP N-oxide). No evidence of tumorigenicity was observed in mice at roflumilast oral doses up to 12 and 18 mg/kg/day in females and males, respectively (10 and 15 times the MRHD, respectively, on an AUC basis).
In a 2-year dermal mouse carcinogenicity study, no evidence of carcinogenicity was observed at topical doses of roflumilast cream up to 1% applied at 2 mL/kg/day (4 times the MRHD on an AUC basis).
Roflumilast tested positive in an in vivo mouse micronucleus test, but negative in the following assays: the Ames test, an in vitro chromosome aberration assay in human lymphocytes, an in vitro HPRT assay with V79 cells, an in vitro micronucleus test with V79 cells, a DNA adduct formation assay in rat nasal mucosa, liver, and testes, and an in vivo mouse bone marrow chromosome aberration assay. Roflumilast N-oxide was negative in the Ames test and an in vitro micronucleus test with V79 cells.
In a fertility study, oral roflumilast decreased fertility rates in male rats at 1.8 mg/kg/day (20 times the MRHD on a mg/m2 basis). The male rats also showed increases in the incidence of tubular atrophy, degeneration in the testis, and spermiogenic granuloma in the epididymides. No effect on rat fertility rate or male reproductive organ morphology was observed at 0.6 mg/kg/day (7 times the MRHD on a mg/m2 basis). In a female fertility study, no effect on fertility was observed up to the highest roflumilast dose of 1.5 mg/kg/day in rats (17 times the MRHD on a mg/m2 basis).
Structured Label Content
Section 42229-5 (42229-5)
Seborrheic Dermatitis
In two multicenter, randomized, double-blind, vehicle-controlled trials (Trial 203 and STRATUM), 683 adult and pediatric subjects 9 years of age or older with seborrheic dermatitis were treated with ZORYVE foam, 0.3%, or vehicle foam once daily for 8 weeks [see Clinical Studies (14.1)].
Table 1 presents adverse reactions that occurred in at least 1% of subjects treated with ZORYVE foam, 0.3%.
| Adverse Reaction | ZORYVE foam, 0.3% (N=458) n (%) |
Vehicle foam (N=225) n (%) |
|---|---|---|
| Nasopharyngitis | 7 (1.5) | 1 (0.4) |
| Nausea | 6 (1.3) | 0 (0) |
| Headache | 5 (1.1) | 0 (0) |
The following additional adverse reactions were reported in fewer than 1% of subjects treated with ZORYVE foam, 0.3%: diarrhea and insomnia.
The adverse reaction profile in pediatric subjects was consistent with that observed in adults [see Use in Specific Populations (8.4), Clinical Pharmacology (12.3)].
In 408 subjects who continued treatment with ZORYVE foam, 0.3%, for up to 24 to 52 weeks in an open-label, long-term trial, the adverse reaction profile was consistent with that observed in vehicle-controlled trials.
Section 42230-3 (42230-3)
| Patient Information ZORYVE® (zor-EEV) (roflumilast) topical foam, 0.3% |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 5/2025 |
| Important information: ZORYVE foam is for use on the skin (topical use) only. Do not use ZORYVE foam in or on your eyes, mouth, or vagina. | |
|
What is ZORYVE foam?
ZORYVE foam is a prescription medicine used on the skin (topical) to treat:
|
|
| Do not use ZORYVE foam if you have certain liver problems. | |
Before using ZORYVE foam, tell your healthcare provider about all of your medical conditions, including if you:
|
|
How should I use ZORYVE foam?
|
|
|
What should I avoid while using ZORYVE foam? ZORYVE foam is flammable. Avoid fire, flame, and smoking during and right after you apply ZORYVE foam. |
|
|
What are the possible side effects of ZORYVE foam? The most common side effects of ZORYVE foam in people treated for seborrheic dermatitis include:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Arcutis Biotherapeutics, Inc. by calling 1-844-692-6729. |
|
How should I store ZORYVE foam?
|
|
|
General Information about the safe and effective use of ZORYVE foam.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ZORYVE foam for a condition for which it was not prescribed. Do not give it to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ZORYVE foam that is written for health professionals. |
|
|
What are the ingredients in ZORYVE foam?
Active ingredient: roflumilast Inactive ingredients: ceteareth-10 phosphate, cetearyl phosphate, cetostearyl alcohol, diethylene glycol monoethyl ether, hexylene glycol, isopropyl palmitate, methylparaben, propylparaben, purified water, sodium hydroxide, and white petrolatum. Hydrochloric acid may have been added to adjust pH. Propellants: butane, isobutane, and propane. Marketed by: Arcutis Biotherapeutics, Inc. Westlake Village, CA 91361 For more information on ZORYVE topical foam, call 1-844-692-6729 or visit http://www.zoryve.com. |
v2
Section 43683-2 (43683-2)
| Indications and Usage, Plaque Psoriasis (1.2) | 5/2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [See USP Controlled Room Temperature].
Do not freeze.
Store upright.
Flammable. Contents under pressure. Do not puncture or incinerate. Do not expose to heat or store at temperatures above 49°C (120°F) [see Warnings and Precautions (5.1)].
8.3 Fertility
In a human spermatogenesis study, oral roflumilast 500 mcg had no effects on semen parameters or reproductive hormones during the 3-month treatment period and the following 3-month off-treatment period.
11 Description (11 DESCRIPTION)
ZORYVE (roflumilast) topical foam, 0.3%, is a white to off-white foam for topical use. The active ingredient, roflumilast, is a phosphodiesterase 4 (PDE4) inhibitor.
The chemical name of roflumilast is 3-cyclopropylmethoxy-N-(3,5-dichloropyridin-4-yl)-4-(difluoromethoxy)benzamide with a molecular formula of C17H14Cl2F2N2O3 and the molecular weight of 403.21.
The structural formula of roflumilast is:
Roflumilast is practically insoluble in water and hexane, sparingly soluble in ethanol, and freely soluble in acetone.
Each gram of ZORYVE topical foam, 0.3%, contains 3 mg of roflumilast in a foam base containing ceteareth-10 phosphate, cetearyl phosphate, cetostearyl alcohol, diethylene glycol monoethyl ether, hexylene glycol, isopropyl palmitate, methylparaben, propylparaben, purified water, sodium hydroxide, and white petrolatum. Hydrochloric acid may have been added to adjust pH. ZORYVE topical foam, 0.3%, is dispensed from an aluminum can pressurized with propellant (butane, isobutane, and propane).
5.1 Flammability
The propellants in ZORYVE foam, 0.3%, are flammable. Avoid fire, flame, and smoking during and immediately following application.
4 Contraindications (4 CONTRAINDICATIONS)
ZORYVE foam, 0.3%, is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C) [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (reported in ≥ 1% of patients) are:
- Seborrheic dermatitis: nasopharyngitis, nausea, and headache. (6.1)
- Plaque psoriasis of the scalp and body: headache, diarrhea, nausea, and nasopharyngitis. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Arcutis Biotherapeutics, Inc. at 1-844-692-6729 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Co-administration of roflumilast with systemic CYP3A4 inhibitors or dual inhibitors that inhibit both CYP3A4 and CYP1A2 simultaneously may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. (7.1)
- Co-administration of roflumilast with oral contraceptives containing gestodene and ethinyl estradiol may increase roflumilast systemic exposure and may result in increased adverse reactions. If these products are co-administered with ZORYVE foam, 0.3%, weigh the potential for increased adverse reactions against benefit. (7.1)
1.2 Plaque Psoriasis
ZORYVE topical foam, 0.3%, is indicated for the treatment of plaque psoriasis of the scalp and body in adult and pediatric patients 12 years of age and older.
Instructions for Use (INSTRUCTIONS FOR USE)
This Instructions for Use contains information on how to apply ZORYVE foam.
Read this Instructions for Use before you start using ZORYVE foam and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. Use ZORYVE foam exactly as your healthcare provider tells you.
Important information you need to know before applying ZORYVE foam:
- ZORYVE foam is for use on skin only (topical use). ZORYVE foam is not for use in your eyes, mouth, or vagina.
- ZORYVE foam is flammable. Avoid fire, flame, and smoking during and right after you apply ZORYVE foam.
Before applying ZORYVE foam for the first time:
| Gently pull back on the nozzle to break the plastic piece at the base. |
Applying ZORYVE foam:
Apply a thin layer of ZORYVE foam 1 time a day to the affected areas of your body and scalp when they are not wet.
Step 1: Shake.
|
|
Step 2: Dispense.
|
|
Step 3: Apply.
|
|
Storing ZORYVE foam
- Store ZORYVE foam at room temperature between 68°F to 77°F (20°C to 25°C).
- ZORYVE foam is flammable. Keep away from heat and flame.
- The contents in ZORYVE foam are under pressure. Do not puncture or burn the can. Do not expose the can to heat or store at temperatures above 120°F (49°C).
- Do not freeze.
- Store the can upright.
Keep ZORYVE foam and all medicines out of the reach of children.
Marketed by:
Arcutis Biotherapeutics, Inc.
Westlake Village, CA 91361
For more information on ZORYVE foam, call 1-844-692-6729 or visit http://www.zoryve.com.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 5/2025
v2
12.2 Pharmacodynamics
Pharmacodynamics of ZORYVE foam, 0.3%, in the treatment of seborrheic dermatitis and plaque psoriasis is unknown.
14.2 Plaque Psoriasis
Two randomized, double-blind, vehicle-controlled trials (ARRECTOR [NCT05028582] and Trial 204 [NCT04128007]) enrolled a total of 736 adult and pediatric subjects 12 years of age and older with mild to severe plaque psoriasis of the scalp and body. In each trial, subjects were randomized 2:1 to receive ZORYVE foam, 0.3%, or vehicle foam applied once daily for 8 weeks. The combined trial population was 55% female, 85% White, 5% Black, 6% Asian, and 4% other races; for ethnicity, 79% identified as non-Hispanic/Latino and 19% identified as Hispanic/Latino. The median age was 47 years (range 12 to 87 years).
In Trial ARRECTOR, the trial population ranged in age from 12 to 87 years, including 2% of subjects who were 12 to 17 years of age and 13% of subjects who were 65 years of age or older. At baseline, 86% of subjects had a Scalp Investigator Global Assessment (S-IGA) score of 3 (moderate) on a 5-point scale of 0 to 4, and 14% had an S-IGA score of 4 (severe); 28% of subjects had a Body Investigator Global Assessment (B-IGA) score of 2 (mild), 67% of subjects had a B-IGA score of 3 (moderate), and 5% had a B-IGA score of 4 (severe). At baseline, 76% of subjects had a Scalp Itch-Numeric Rating Scale (SI-NRS) score of 4 or higher on a scale of 0 to 10 and 73% had a Worst Itch-Numeric Rating Scale (WI-NRS) score of 4 or higher.
In Trial 204, the trial population ranged in age from 12 to 87 years, including 1% of subjects who were 12 to 17 years of age, and 9% who were 65 years of age or older. At baseline, 11% of subjects had an S-IGA score of 2 (mild), 76% of subjects had an S-IGA score of 3 (moderate), and 13% had an S-IGA score of 4 (severe); 36% of subjects had a B-IGA score of 2 (mild), 59% of subjects had a B-IGA score of 3 (moderate), and 5% had a B-IGA score of 4 (severe). At baseline, 89% of subjects had an SI-NRS score of 4 or higher on a scale of 0 to 10.
In both trials, S-IGA treatment success, a primary endpoint in ARRECTOR and Trial 204, and B-IGA treatment success, a primary endpoint in ARRECTOR, were defined as a score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade improvement from baseline.
| Trial ARRECTOR | Trial 204 | |||
|---|---|---|---|---|
| ZORYVE foam, 0.3% | Vehicle foam | ZORYVE foam, 0.3% | Vehicle foam | |
| Number of subjects randomized | N=281 | N=151 | N=200 | N=104 |
| Abbreviations: CI = Confidence Interval | ||||
| S-IGA success S-IGA treatment success and B-IGA treatment success were defined as a score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade score improvement from baseline at Week 8 (Multiple Imputation).
|
66.4% | 27.8% | 56.7% | 11.0% |
| Difference from Vehicle (95% CI) Treatment difference and 95% CI are based on the CMH method stratified by pooled site and baseline IGA strata.
|
37.1% (27.1%, 47.1%) | 47.7% (37.9%, 57.5%) | ||
| B-IGA success | 45.5% | 20.1% | 39.0% | 7.4% |
| Difference from Vehicle (95% CI) | 24.8% (15.0%, 34.6%) | 32.4% (23.3%, 41.6%) |
SI-NRS success and WI-NRS success were defined as a reduction of at least 4 points from baseline with a baseline score of at least 4. In Trial ARRECTOR, among subjects with a baseline SI-NRS score of at least 4 (75% of subjects), a higher percentage of subjects achieved SI-NRS success at Week 8 in the group who received ZORYVE foam, 0.3%, compared to the group who received vehicle foam (65.3% vs. 30.3% for a treatment difference of 35.4% and 95% CI of (23.9, 47.0)). In Trial ARRECTOR, among subjects with a baseline WI-NRS score of at least 4 (72% of subjects), a higher percentage of subjects achieved WI-NRS success at Week 8 in the group who received ZORYVE foam, 0.3%, compared to the group who received vehicle foam (63.1% vs. 30.1% for a treatment difference of 32.8% and 95% CI of (20.3, 45.2)).
8.6 Hepatic Impairment
Oral roflumilast 250 mcg once daily for 14 days was studied in subjects with hepatic impairment. The systemic exposure of roflumilast and roflumilast N-oxide were increased in subjects with moderate (Child-Pugh B) hepatic impairment. ZORYVE foam, 0.3%, is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C). No dosage adjustment is needed in patients with mild (Child-Pugh A) hepatic impairment [see Contraindications (4), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
Roflumilast and its active metabolite (roflumilast N-oxide) are inhibitors of PDE4. Roflumilast and roflumilast N-oxide inhibition of PDE4 (a major cyclic 3′,5′-adenosine monophosphate (cyclic AMP) metabolizing enzyme) activity leads to accumulation of intracellular cyclic AMP. The specific mechanism(s) by which roflumilast exerts its therapeutic action is not well defined.
1.1 Seborrheic Dermatitis
ZORYVE® topical foam, 0.3%, is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients 9 years of age and older.
14.1 Seborrheic Dermatitis
Two randomized, double-blind, vehicle-controlled trials (STRATUM [NCT04973228] and Trial 203 [NCT04091646]) enrolled a total of 683 adult and pediatric subjects with seborrheic dermatitis involving the scalp, face, and/or body with an Investigator Global Assessment (IGA) of moderate or severe (IGA of 3 or 4 on a 5-point scale from 0 to 4). In each trial, subjects were randomized 2:1 to receive ZORYVE foam, 0.3%, or vehicle foam applied once daily for 8 weeks. The combined trial population was 50% male, 79% White, 11% Black, 5% Asian, and 5% other races; for ethnicity, 79% identified as non-Hispanic/Latino and 21% identified as Hispanic/Latino. In Trial STRATUM, the trial population ranged in age from 9 to 87 years, including 7% of subjects who were 9 to 17 years of age and 12% of subjects who were 65 years of age or older. At baseline, 94% of subjects had an IGA score of 3 (moderate), and 6% had an IGA score of 4 (severe). At baseline, 67% of subjects had a Worst Itch-Numeric Rating Scale (WI-NRS) score of 4 or higher on a scale of 0 to 10. In Trial 203, the trial population ranged in age from 18 to 85 years, including 18% who were 65 years of age or older. At baseline, 93% of subjects had an IGA score of 3 (moderate), and 7% had an IGA score of 4 (severe). At baseline, 81% of subjects had a Worst Itch-Numeric Rating Scale (WI-NRS) score of 4 or higher on a scale of 0 to 10.
The primary endpoint was the proportion of subjects who achieved IGA treatment success at Week 8 (Table 3). Success was defined as a score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade improvement from baseline.
| STRATUM | Trial 203 | |||
|---|---|---|---|---|
| ZORYVE foam, 0.3% | Vehicle foam | ZORYVE foam, 0.3% | Vehicle foam | |
| Number of subjects randomized | N=304 | N=153 | N=154 | N=72 |
| Abbreviations: CI = Confidence Interval | ||||
| IGA success IGA treatment success was defined as an IGA score of "Clear" (0) or "Almost Clear" (1), plus a 2-grade IGA score improvement from baseline at Week 8 (Multiple Imputation).
|
79.5% | 58.0% | 73.1% | 40.8% |
| Difference from Vehicle (95% CI) Treatment difference and 95% CI are based on the CMH method stratified by pooled site and baseline IGA strata.
|
20.6% (11.2%, 30.0%) | 33.8% (20.3%, 47.4%) |
In Trial STRATUM, among subjects with a baseline WI-NRS score of at least 4 (67% of subjects), there was a higher percentage of subjects who achieved a reduction of at least 4 points from baseline at Week 8 in the group who received ZORYVE foam, 0.3%, compared to the group who received vehicle foam (62.8% vs. 40.6% for a treatment difference of 25.7% and 95% CI of (13.4, 38.1)).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Flammability: The propellants in ZORYVE foam, 0.3%, are flammable. Avoid fire, flame, and smoking during and immediately following application. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Shake can prior to each use. Apply a thin layer of ZORYVE foam, 0.3%, once daily to affected areas of body and/or scalp when they are not wet. Rub in completely.
Wash hands after application.
Avoid fire, flame, and smoking during and immediately following application [see Warnings and Precautions (5.1)].
ZORYVE foam, 0.3%, is for topical use only and not for ophthalmic, oral, or intravaginal use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical foam, 0.3%: 3 mg of roflumilast per gram of white to off-white foam in 60-gram pressurized cans.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient or caregiver to read the FDA-approved patient labeling (Patient Information).
Principal Display Panel 60 G Can Carton (PRINCIPAL DISPLAY PANEL - 60 g Can Carton)
NDC 80610-430-60
ZORYVE®
(roflumilast)
topical foam, 0.3%
Rx Only
60 grams
For Topical Use Only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies were conducted in hamsters and mice with roflumilast to evaluate its carcinogenic potential. In 2-year oral gavage carcinogenicity studies, roflumilast treatment resulted in dose-related, statistically significant increases in the incidence of undifferentiated carcinomas of nasal epithelium in hamsters at doses greater than or equal to 8 mg/kg/day (11 times the MRHD on an AUC basis). The tumorigenicity of roflumilast appears to be attributed to a reactive metabolite of 4-amino-3,5-dichloropyridine N-oxide (ADCP N-oxide). No evidence of tumorigenicity was observed in mice at roflumilast oral doses up to 12 and 18 mg/kg/day in females and males, respectively (10 and 15 times the MRHD, respectively, on an AUC basis).
In a 2-year dermal mouse carcinogenicity study, no evidence of carcinogenicity was observed at topical doses of roflumilast cream up to 1% applied at 2 mL/kg/day (4 times the MRHD on an AUC basis).
Roflumilast tested positive in an in vivo mouse micronucleus test, but negative in the following assays: the Ames test, an in vitro chromosome aberration assay in human lymphocytes, an in vitro HPRT assay with V79 cells, an in vitro micronucleus test with V79 cells, a DNA adduct formation assay in rat nasal mucosa, liver, and testes, and an in vivo mouse bone marrow chromosome aberration assay. Roflumilast N-oxide was negative in the Ames test and an in vitro micronucleus test with V79 cells.
In a fertility study, oral roflumilast decreased fertility rates in male rats at 1.8 mg/kg/day (20 times the MRHD on a mg/m2 basis). The male rats also showed increases in the incidence of tubular atrophy, degeneration in the testis, and spermiogenic granuloma in the epididymides. No effect on rat fertility rate or male reproductive organ morphology was observed at 0.6 mg/kg/day (7 times the MRHD on a mg/m2 basis). In a female fertility study, no effect on fertility was observed up to the highest roflumilast dose of 1.5 mg/kg/day in rats (17 times the MRHD on a mg/m2 basis).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:02.872410 · Updated: 2026-03-14T22:45:20.597363