These Highlights Do Not Include All The Information Needed To Use Ruxience Safely And Effectively. See Full Prescribing Information For Ruxience.
f941fc61-f7a3-4e4a-ab7c-87c1667fa05b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage, Pemphigus Vulgaris (PV) ( 1.5 ) 6/2025 Dosage and Administration ( 2 ) 6/2025 Warnings and Precautions ( 5 ) 6/2025
Indications and Usage
RUXIENCE is a CD20-directed cytolytic antibody indicated for the treatment of: • Adult patients with Non-Hodgkin's Lymphoma (NHL) ( 1.1 ). o Relapsed or refractory, low grade or follicular, CD20-positive B-cell NHL as a single agent. o Previously untreated follicular, CD20-positive, B-cell NHL in combination with first line chemotherapy and, in patients achieving a complete or partial response to a rituximab product in combination with chemotherapy, as single-agent maintenance therapy. o Non-progressing (including stable disease), low-grade, CD20-positive, B-cell NHL as a single agent after first-line cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy. o Previously untreated diffuse large B-cell, CD20-positive NHL in combination with (cyclophosphamide, doxorubicin, vincristine, and prednisone) (CHOP) or other anthracycline-based chemotherapy regimens. • Adult patients with Chronic Lymphocytic Leukemia (CLL) ( 1.2 ). o Previously untreated and previously treated CD20-positive CLL in combination with fludarabine and cyclophosphamide (FC). • Rheumatoid Arthritis (RA) in combination with methotrexate in adult patients with moderately-to-severely-active RA who have inadequate response to one or more TNF antagonist therapies ( 1.3 ). • Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA) in adult patients in combination with glucocorticoids ( 1.4 ). • Moderate to severe Pemphigus Vulgaris (PV) in adult patients ( 1.5 ).
Dosage and Administration
• Administer only as an intravenous infusion ( 2.1 ). • Do not administer as an intravenous push or bolus ( 2.1 ). • RUXIENCE should only be administered by a healthcare professional with appropriate medical support to manage severe infusion-related reactions that can be fatal if they occur ( 2.1 ). • The dose for adult B-cell NHL is 375 mg/m 2 ( 2.2 ). • The dose for CLL is 375 mg/m 2 in the first cycle and 500 mg/m 2 in Cycles 2–6, in combination with FC, administered every 28 days ( 2.3 ). • The dose as a component of Zevalin ® (ibritumomab tiuxetan) Therapeutic Regimen is 250 mg/m 2 ( 2.4 ). • The dose for RA in combination with methotrexate is two-1,000 mg intravenous infusions separated by 2 weeks (one course) every 24 weeks or based on clinical evaluation, but not sooner than every 16 weeks. Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion ( 2.5 ). • The induction dose for adult patients with active GPA and MPA in combination with glucocorticoids is 375 mg/m 2 once weekly for 4 weeks. The follow up dose for adult patients with GPA and MPA who have achieved disease control with induction treatment, in combination with glucocorticoids is two 500 mg intravenous infusions separated by two weeks, followed by a 500 mg intravenous infusion every 6 months thereafter based on clinical evaluation ( 2.6 ). • The dose for PV is two-1,000 mg intravenous infusions separated by 2 weeks in combination with a tapering course of glucocorticoids, then a 500 mg intravenous infusion at Month 12 and every 6 months thereafter or based on clinical evaluation. Dose upon relapse is a 1,000 mg intravenous infusion with considerations to resume or increase the glucocorticoid dose based on clinical evaluation. Subsequent infusions may be no sooner than 16 weeks after the previous infusion ( 2.7 ). Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion ( 2.8 ).
Warnings and Precautions
• Tumor lysis syndrome: Administer aggressive intravenous hydration, anti-hyperuricemic agents, monitor renal function ( 5.5 ). • Infections: Withhold RUXIENCE and institute appropriate anti-infective therapy ( 5.6 ). • Cardiac adverse reactions: Discontinue infusions in case of serious or life-threatening events ( 5.7 ). • Renal toxicity: Discontinue in patients with rising serum creatinine or oliguria ( 5.8 ). • Bowel obstruction and perforation: Consider and evaluate for abdominal pain, vomiting, or related symptoms ( 5.9 ). • Immunizations: Live virus vaccinations prior to or during RUXIENCE treatment are not recommended ( 5.10 ). • Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception ( 5.11 ).
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: • Infusion-related reactions [see Warnings and Precautions (5.1) ] • Severe mucocutaneous reactions [see Warnings and Precautions (5.2) ] • Hepatitis B reactivation with fulminant hepatitis [see Warnings and Precautions (5.3) ] • Progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.4) ] • Tumor lysis syndrome [see Warnings and Precautions (5.5) ] • Infections [see Warnings and Precautions (5.6) ] • Cardiovascular adverse reactions [see Warnings and Precautions (5.7) ] • Renal toxicity [see Warnings and Precautions (5.8) ] • Bowel obstruction and perforation [see Warnings and Precautions (5.9) ]
Drug Interactions
Formal drug interaction studies have not been performed with rituximab products. In patients with CLL, rituximab did not alter systemic exposure to fludarabine or cyclophosphamide. In clinical trials of patients with RA, concomitant administration of methotrexate or cyclophosphamide did not alter the pharmacokinetics of rituximab.
Storage and Handling
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion supplied as follows: Carton contents NDC number One 100 mg/10 mL (10 mg/mL) single-dose vial NDC 0069-0238-01 One 500 mg/50 mL (10 mg/mL) single-dose vial NDC 0069-0249-01
How Supplied
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion supplied as follows: Carton contents NDC number One 100 mg/10 mL (10 mg/mL) single-dose vial NDC 0069-0238-01 One 500 mg/50 mL (10 mg/mL) single-dose vial NDC 0069-0249-01
Medication Information
Warnings and Precautions
• Tumor lysis syndrome: Administer aggressive intravenous hydration, anti-hyperuricemic agents, monitor renal function ( 5.5 ). • Infections: Withhold RUXIENCE and institute appropriate anti-infective therapy ( 5.6 ). • Cardiac adverse reactions: Discontinue infusions in case of serious or life-threatening events ( 5.7 ). • Renal toxicity: Discontinue in patients with rising serum creatinine or oliguria ( 5.8 ). • Bowel obstruction and perforation: Consider and evaluate for abdominal pain, vomiting, or related symptoms ( 5.9 ). • Immunizations: Live virus vaccinations prior to or during RUXIENCE treatment are not recommended ( 5.10 ). • Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception ( 5.11 ).
Indications and Usage
RUXIENCE is a CD20-directed cytolytic antibody indicated for the treatment of: • Adult patients with Non-Hodgkin's Lymphoma (NHL) ( 1.1 ). o Relapsed or refractory, low grade or follicular, CD20-positive B-cell NHL as a single agent. o Previously untreated follicular, CD20-positive, B-cell NHL in combination with first line chemotherapy and, in patients achieving a complete or partial response to a rituximab product in combination with chemotherapy, as single-agent maintenance therapy. o Non-progressing (including stable disease), low-grade, CD20-positive, B-cell NHL as a single agent after first-line cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy. o Previously untreated diffuse large B-cell, CD20-positive NHL in combination with (cyclophosphamide, doxorubicin, vincristine, and prednisone) (CHOP) or other anthracycline-based chemotherapy regimens. • Adult patients with Chronic Lymphocytic Leukemia (CLL) ( 1.2 ). o Previously untreated and previously treated CD20-positive CLL in combination with fludarabine and cyclophosphamide (FC). • Rheumatoid Arthritis (RA) in combination with methotrexate in adult patients with moderately-to-severely-active RA who have inadequate response to one or more TNF antagonist therapies ( 1.3 ). • Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA) in adult patients in combination with glucocorticoids ( 1.4 ). • Moderate to severe Pemphigus Vulgaris (PV) in adult patients ( 1.5 ).
Dosage and Administration
• Administer only as an intravenous infusion ( 2.1 ). • Do not administer as an intravenous push or bolus ( 2.1 ). • RUXIENCE should only be administered by a healthcare professional with appropriate medical support to manage severe infusion-related reactions that can be fatal if they occur ( 2.1 ). • The dose for adult B-cell NHL is 375 mg/m 2 ( 2.2 ). • The dose for CLL is 375 mg/m 2 in the first cycle and 500 mg/m 2 in Cycles 2–6, in combination with FC, administered every 28 days ( 2.3 ). • The dose as a component of Zevalin ® (ibritumomab tiuxetan) Therapeutic Regimen is 250 mg/m 2 ( 2.4 ). • The dose for RA in combination with methotrexate is two-1,000 mg intravenous infusions separated by 2 weeks (one course) every 24 weeks or based on clinical evaluation, but not sooner than every 16 weeks. Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion ( 2.5 ). • The induction dose for adult patients with active GPA and MPA in combination with glucocorticoids is 375 mg/m 2 once weekly for 4 weeks. The follow up dose for adult patients with GPA and MPA who have achieved disease control with induction treatment, in combination with glucocorticoids is two 500 mg intravenous infusions separated by two weeks, followed by a 500 mg intravenous infusion every 6 months thereafter based on clinical evaluation ( 2.6 ). • The dose for PV is two-1,000 mg intravenous infusions separated by 2 weeks in combination with a tapering course of glucocorticoids, then a 500 mg intravenous infusion at Month 12 and every 6 months thereafter or based on clinical evaluation. Dose upon relapse is a 1,000 mg intravenous infusion with considerations to resume or increase the glucocorticoid dose based on clinical evaluation. Subsequent infusions may be no sooner than 16 weeks after the previous infusion ( 2.7 ). Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion ( 2.8 ).
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: • Infusion-related reactions [see Warnings and Precautions (5.1) ] • Severe mucocutaneous reactions [see Warnings and Precautions (5.2) ] • Hepatitis B reactivation with fulminant hepatitis [see Warnings and Precautions (5.3) ] • Progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.4) ] • Tumor lysis syndrome [see Warnings and Precautions (5.5) ] • Infections [see Warnings and Precautions (5.6) ] • Cardiovascular adverse reactions [see Warnings and Precautions (5.7) ] • Renal toxicity [see Warnings and Precautions (5.8) ] • Bowel obstruction and perforation [see Warnings and Precautions (5.9) ]
Drug Interactions
Formal drug interaction studies have not been performed with rituximab products. In patients with CLL, rituximab did not alter systemic exposure to fludarabine or cyclophosphamide. In clinical trials of patients with RA, concomitant administration of methotrexate or cyclophosphamide did not alter the pharmacokinetics of rituximab.
Storage and Handling
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion supplied as follows: Carton contents NDC number One 100 mg/10 mL (10 mg/mL) single-dose vial NDC 0069-0238-01 One 500 mg/50 mL (10 mg/mL) single-dose vial NDC 0069-0249-01
How Supplied
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion supplied as follows: Carton contents NDC number One 100 mg/10 mL (10 mg/mL) single-dose vial NDC 0069-0238-01 One 500 mg/50 mL (10 mg/mL) single-dose vial NDC 0069-0249-01
Description
Indications and Usage, Pemphigus Vulgaris (PV) ( 1.5 ) 6/2025 Dosage and Administration ( 2 ) 6/2025 Warnings and Precautions ( 5 ) 6/2025
Section 42229-5
Prior to First Infusion
Screen all patients for HBV infection by measuring HBsAg and anti-HBc before initiating treatment with RUXIENCE [see Warnings and Precautions (5.3)]. Obtain complete blood counts (CBC) including platelets prior to the first dose.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 6/2025 | ||
|
MEDICATION GUIDE
|
||
|
What is the most important information I should know about RUXIENCE?
|
||
|
|
|
|
||
|
|
|
|
See " What are the possible side effects of RUXIENCE? " for more information about side effects. |
||
|
What is RUXIENCE?
RUXIENCE is not indicated for treatment of children. |
||
|
Before you receive RUXIENCE, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take or have taken:
If you are not sure if your medicine is one listed above, ask your healthcare provider. |
||
|
How will I receive RUXIENCE?
|
||
|
What are the possible side effects of RUXIENCE?
|
||
|
|
|
Your healthcare provider will stop treatment with RUXIENCE if you have severe, serious or life-threatening side effects.
In adults with GPA or MPA the most common side effects of RUXIENCE also include:
Other side effects with RUXIENCE include:
These are not all of the possible side effects with RUXIENCE. |
||
|
General information about the safe and effective use of RUXIENCE.
|
||
|
What are the ingredients in RUXIENCE?
Manufactured by Pfizer Ireland Pharmaceuticals, Cork, Ireland, P43 X336 U.S. License No. 2060
|
Section 43683-2
Section 44425-7
Store RUXIENCE vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton. RUXIENCE vials should be protected from direct sunlight. Do not freeze or shake.
8.2 Lactation
There are limited data on the presence of rituximab in human milk, and the effect on the breastfed child, and there are no data on the effect on milk production. Rituximab is detected in the milk of lactating cynomolgus monkeys, and maternal IgG is present in human breast milk. Rituximab has also been reported to be excreted at low concentrations in human breast milk. Given that the clinical significance of this finding for children is not known, advise women not to breastfeed during treatment with RUXIENCE and for 6 months after the last dose due to the potential of serious adverse reactions in breastfed children.
11 Description
Rituximab-pvvr is a genetically engineered chimeric murine/human monoclonal IgG1 kappa antibody directed against the CD20 antigen. Rituximab-pvvr has an approximate molecular weight of 145 kD.
Rituximab-pvvr is produced by mammalian cell (Chinese Hamster Ovary) suspension culture in a nutrient medium.
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion. RUXIENCE is supplied at a concentration of 10 mg/mL in either 100 mg/10 mL or 500 mg/50 mL single-dose vials. Each mL of solution contains 10 mg rituximab-pvvr, 0.056 mg of edetate disodium dihydrate, 1.2 mg of L-histidine, 2.57 mg of L-histidine hydrochloride monohydrate, 0.2 mg of polysorbate 80, 85 mg of sucrose, and Water for Injection, USP. The pH is 5.8.
5.6 Infections
Serious, including fatal, bacterial, fungal, and new or reactivated viral infections can occur during and following the completion of rituximab product-based therapy. Infections have been reported in some patients with prolonged hypogammaglobulinemia (defined as hypogammaglobulinemia greater than 11 months after rituximab exposure). New or reactivated viral infections included cytomegalovirus, herpes simplex virus, parvovirus B19, varicella zoster virus, West Nile virus, and hepatitis B and C. Discontinue RUXIENCE for serious infections and institute appropriate anti-infective therapy [see Adverse Reactions (6.1 , 6.3)]. RUXIENCE is not recommended for use in patients with severe, active infections.
5.10 Immunization
The safety of immunization with live viral vaccines following rituximab product therapy has not been studied and vaccination with live virus vaccines is not recommended before or during treatment.
For patients treated with RUXIENCE, physicians should review the patient's vaccination status and patients should, if possible, be brought up-to-date with all immunizations in agreement with current immunization guidelines prior to initiating RUXIENCE and administer non live vaccines at least 4 weeks prior to a course of RUXIENCE.
The effect of rituximab on immune responses was assessed in a randomized, controlled study in patients with RA treated with rituximab and methotrexate (MTX) compared to patients treated with MTX alone.
A response to pneumococcal vaccination (a T-cell independent antigen) as measured by an increase in antibody titers to at least 6 of 12 serotypes was lower in patients treated with rituximab plus MTX as compared to patients treated with MTX alone (19% vs. 61%). A lower proportion of patients in the rituximab plus MTX group developed detectable levels of anti-keyhole limpet hemocyanin antibodies (a novel protein antigen) after vaccination compared to patients on MTX alone (47% vs. 93%).
A positive response to tetanus toxoid vaccine (a T-cell dependent antigen with existing immunity) was similar in patients treated with rituximab plus MTX compared to patients on MTX alone (39% vs. 42%). The proportion of patients maintaining a positive Candida skin test (to evaluate delayed type hypersensitivity) was also similar (77% of patients on rituximab plus MTX vs. 70% of patients on MTX alone).
Most patients in the rituximab-treated group had B-cell counts below the lower limit of normal at the time of immunization. The clinical implications of these findings are not known.
8.4 Pediatric Use
The safety and effectiveness of RUXIENCE have not been established in pediatric patients with NHL, CLL, PV or RA.
Rituximab was not studied in pediatric patients with polyarticular juvenile idiopathic arthritis (PJIA) due to concerns regarding the potential for prolonged immunosuppression as a result of B-cell depletion in the developing juvenile immune system.
5.8 Renal Toxicity
Severe, including fatal, renal toxicity can occur after rituximab product administration in patients with NHL. Renal toxicity has occurred in patients who experience tumor lysis syndrome and in patients with NHL administered concomitant cisplatin therapy during clinical trials. The combination of cisplatin and RUXIENCE is not an approved treatment regimen. Monitor closely for signs of renal failure and discontinue RUXIENCE in patients with a rising serum creatinine or oliguria [see Warnings and Precautions (5.5)].
6.2 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of rituximab or of other rituximab products.
Using an ELISA assay, anti-rituximab antibody was detected in 4 of 356 (1.1%) patients with low-grade or follicular NHL receiving single-agent rituximab. Three of the four patients had an objective clinical response.
A total of 273/2578 (11%) patients with RA tested positive for anti-rituximab antibodies at any time after receiving rituximab. Anti-rituximab antibody positivity was not associated with increased rates of infusion-related reactions or other adverse events. Upon further treatment, the proportions of patients with infusion-related reactions were similar between anti-rituximab antibody positive and negative patients, and most reactions were mild to moderate. Four anti-rituximab antibody positive patients had serious infusion-related reactions, and the temporal relationship between anti-rituximab antibody positivity and infusion-related reaction was variable.
A total of 23/99 (23%) rituximab-treated adult patients with GPA and MPA developed anti-rituximab antibodies by 18 months in GPA/MPA Study 1. The clinical relevance of anti-rituximab antibody formation in rituximab-treated adult patients is unclear.
Using a new ELISA assay, a total of 19/34 (56%) patients with PV, who were treated with non-U.S.-licensed rituximab, tested positive for anti-rituximab antibodies by 18 months in PV Study 1. In PV Study 2, a total of 20/63 (32%) rituximab-treated PV patients tested positive for ADA by week 52 (19 patients had treatment-inducted ADA and 1 patient had treatment-enhanced ADA). The clinical relevance of anti-rituximab antibody formation in rituximab-treated PV patients is unclear.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Infusion-related reactions [see Warnings and Precautions (5.1)]
-
•Severe mucocutaneous reactions [see Warnings and Precautions (5.2)]
-
•Hepatitis B reactivation with fulminant hepatitis [see Warnings and Precautions (5.3)]
-
•Progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.4)]
-
•Tumor lysis syndrome [see Warnings and Precautions (5.5)]
-
•Infections [see Warnings and Precautions (5.6)]
-
•Cardiovascular adverse reactions [see Warnings and Precautions (5.7)]
-
•Renal toxicity [see Warnings and Precautions (5.8)]
-
•Bowel obstruction and perforation [see Warnings and Precautions (5.9)]
7 Drug Interactions
Formal drug interaction studies have not been performed with rituximab products. In patients with CLL, rituximab did not alter systemic exposure to fludarabine or cyclophosphamide. In clinical trials of patients with RA, concomitant administration of methotrexate or cyclophosphamide did not alter the pharmacokinetics of rituximab.
1 Indications and Usage
RUXIENCE is a CD20-directed cytolytic antibody indicated for the treatment of:
-
•Adult patients with Non-Hodgkin's Lymphoma (NHL) (1.1).
-
oRelapsed or refractory, low grade or follicular, CD20-positive B-cell NHL as a single agent.
-
oPreviously untreated follicular, CD20-positive, B-cell NHL in combination with first line chemotherapy and, in patients achieving a complete or partial response to a rituximab product in combination with chemotherapy, as single-agent maintenance therapy.
-
oNon-progressing (including stable disease), low-grade, CD20-positive, B-cell NHL as a single agent after first-line cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy.
-
oPreviously untreated diffuse large B-cell, CD20-positive NHL in combination with (cyclophosphamide, doxorubicin, vincristine, and prednisone) (CHOP) or other anthracycline-based chemotherapy regimens.
-
-
•Adult patients with Chronic Lymphocytic Leukemia (CLL) (1.2).
-
oPreviously untreated and previously treated CD20-positive CLL in combination with fludarabine and cyclophosphamide (FC).
-
-
•Rheumatoid Arthritis (RA) in combination with methotrexate in adult patients with moderately-to-severely-active RA who have inadequate response to one or more TNF antagonist therapies (1.3).
-
•Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA) in adult patients in combination with glucocorticoids (1.4).
-
•Moderate to severe Pemphigus Vulgaris (PV) in adult patients (1.5).
12.1 Mechanism of Action
Rituximab-pvvr is a monoclonal antibody. Rituximab products target the CD20 antigen expressed on the surface of pre-B and mature B-lymphocytes. Upon binding to CD20, rituximab products mediate B-cell lysis. Possible mechanisms of cell lysis include complement dependent cytotoxicity (CDC) and antibody dependent cell mediated cytotoxicity (ADCC). B cells are believed to play a role in the pathogenesis of rheumatoid arthritis (RA) and associated chronic synovitis. In this setting, B cells may be acting at multiple sites in the autoimmune/inflammatory process, including through production of rheumatoid factor (RF) and other autoantibodies, antigen presentation, T-cell activation, and/or proinflammatory cytokine production.
5 Warnings and Precautions
-
•Tumor lysis syndrome: Administer aggressive intravenous hydration, anti-hyperuricemic agents, monitor renal function (5.5).
-
•Infections: Withhold RUXIENCE and institute appropriate anti-infective therapy (5.6).
-
•Cardiac adverse reactions: Discontinue infusions in case of serious or life-threatening events (5.7).
-
•Renal toxicity: Discontinue in patients with rising serum creatinine or oliguria (5.8).
-
•Bowel obstruction and perforation: Consider and evaluate for abdominal pain, vomiting, or related symptoms (5.9).
-
•Immunizations: Live virus vaccinations prior to or during RUXIENCE treatment are not recommended (5.10).
-
•Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception (5.11).
5.11 Embryo Fetal Toxicity
Based on human data, rituximab products can cause fetal harm due to B-cell lymphocytopenia in infants exposed in-utero. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception while receiving RUXIENCE and for 12 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
1.5 Pemphigus Vulgaris (pv)
RUXIENCE is indicated for the treatment of adult patients with moderate to severe pemphigus vulgaris.
2 Dosage and Administration
-
•Administer only as an intravenous infusion (2.1).
-
•Do not administer as an intravenous push or bolus (2.1).
-
•RUXIENCE should only be administered by a healthcare professional with appropriate medical support to manage severe infusion-related reactions that can be fatal if they occur (2.1).
-
•The dose for adult B-cell NHL is 375 mg/m2 (2.2).
-
•The dose for CLL is 375 mg/m2 in the first cycle and 500 mg/m2 in Cycles 2–6, in combination with FC, administered every 28 days (2.3).
-
•The dose as a component of Zevalin® (ibritumomab tiuxetan) Therapeutic Regimen is 250 mg/m2 (2.4).
-
•The dose for RA in combination with methotrexate is two-1,000 mg intravenous infusions separated by 2 weeks (one course) every 24 weeks or based on clinical evaluation, but not sooner than every 16 weeks. Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion (2.5).
-
•The induction dose for adult patients with active GPA and MPA in combination with glucocorticoids is 375 mg/m2 once weekly for 4 weeks. The follow up dose for adult patients with GPA and MPA who have achieved disease control with induction treatment, in combination with glucocorticoids is two 500 mg intravenous infusions separated by two weeks, followed by a 500 mg intravenous infusion every 6 months thereafter based on clinical evaluation (2.6).
-
•The dose for PV is two-1,000 mg intravenous infusions separated by 2 weeks in combination with a tapering course of glucocorticoids, then a 500 mg intravenous infusion at Month 12 and every 6 months thereafter or based on clinical evaluation. Dose upon relapse is a 1,000 mg intravenous infusion with considerations to resume or increase the glucocorticoid dose based on clinical evaluation. Subsequent infusions may be no sooner than 16 weeks after the previous infusion (2.7). Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion (2.8).
14.8 Pemphigus Vulgaris (pv)
PV Study 1 (NCT00784589)
Non-U.S.-licensed rituximab in combination with short-term prednisone was compared to prednisone monotherapy as first-line treatment in 90 newly diagnosed adult patients with moderate to severe pemphigus (74 Pemphigus Vulgaris [PV] and 16 Pemphigus Foliaceus [PF]) in this randomized, open-label, controlled, multicenter study (PV Study 1). Patients were between 19 and 79 years of age and had not received prior therapies for pemphigus. In the PV population, 5 (13%) patients in the group treated with non-U.S.-licensed rituximab and 3 (8%) patients in the prednisone group had moderate disease and 33 (87%) patients in the group treated with non-U.S.-licensed rituximab and 33 (92%) patients in the prednisone group had severe disease according to disease severity defined by Harman’s criteria.
Patients were stratified by baseline disease severity (moderate or severe) and randomized 1:1 to receive either the non-U.S.-licensed rituximab and short-term prednisone or long-term prednisone monotherapy. Patients were pre-medicated with antihistamine, acetaminophen and methylprednisolone prior to infusion of the non-U.S.-licensed rituximab. Patients randomized to the group treated with non-U.S.-licensed rituximab received an initial intravenous infusion of 1,000 mg non-U.S.-licensed rituximab on Study Day 1 in combination with a short-term regimen of 0.5 mg/kg/day oral prednisone tapered off over 3 months if they had moderate disease or 1 mg/kg/day oral prednisone tapered off over 6 months if they had severe disease. All patients received a second intravenous infusion of 1,000 mg non-U.S.-licensed rituximab on Study Day 15. Maintenance infusions of 500 mg non-U.S.-licensed rituximab were administered at Months 12 and 18. Patients randomized to the prednisone monotherapy group received an initial 1 mg/kg/day oral prednisone tapered off over 12 months if they had moderate disease or 1.5 mg/kg/day oral prednisone tapered off over 18 months if they had severe disease. Patients in the group treated with non-U.S.-licensed rituximab who relapsed could receive an additional infusion of 1,000 mg non-U.S.-licensed rituximab in combination with reintroduced or escalated prednisone dose. Maintenance and relapse infusions were administered no sooner than 16 weeks following the previous infusion.
The primary endpoint for the study was complete remission (complete epithelialization and absence of new and/or established lesions) at Month 24 without the use of prednisone therapy for 2 months or more (CRoff for greater than or equal to 2 months).
The results of the trial are presented in Table 18.
|
Non-U.S.-Licensed Rituximab + Short-term Prednisone N=46 |
Prednisone N=44 |
|
|
Number of responders (response rate [%]) |
41 (89%) |
15 (34%) |
|
PV patients |
34/38 (90%) |
10/36 (28%) |
|
PF patients |
7/8 (88%) |
5/8 (63%) |
PV Study 2 (NCT02383589)
In a randomized, double-blind, double-dummy, active-comparator, multicenter study, the efficacy and safety of rituximab compared to mycophenolate mofetil (MMF) were evaluated in patients with moderate to severe PV receiving 60-120 mg/day oral prednisone or equivalent (1.0-1.5 mg/kg/day) at study entry and tapered to reach a dose of 60 or 80 mg/day by Day 1. Patients had a confirmed diagnosis of PV within the previous 24 months and evidence of moderate to severe disease defined as a total Pemphigus Disease Area Index (PDAI) activity score of greater than or equal to 15. The study consisted of a screening period of up to 28 days, a 52-week double-blind treatment period, and a 48-week safety follow up period.
One hundred and thirty-five patients were randomized to treatment with rituximab 1,000 mg administered on Day 1, Day 15, Week 24 and Week 26 or oral MMF 2 g/day (starting at 1 g/day on Day 1 and titrated to achieve a goal of 2 g/day by Week 2) for 52 weeks in combination with an initial dose of 60 or 80 mg oral prednisone with the aim of tapering to 0 mg/day by Week 24. Randomization was stratified by duration of PV (within the 1 year prior to screening or greater than 1 year) and geographical region. A dual-assessor approach was used during the study for efficacy and safety evaluations to prevent potential unblinding.
One hundred and twenty-five patients (excluding exploratory data from ten telemedicine patients) were analyzed for efficacy (Modified Intent-to-Treat Population). The primary efficacy endpoint for this study was the proportion of subjects achieving sustained complete remission defined as achieving healing of lesions with no new active lesions (i.e., PDAI activity score of 0) while on 0 mg/day prednisone or equivalent, and maintaining this response for at least 16 consecutive weeks, during the 52-week treatment period.
Secondary endpoints included cumulative oral corticosteroid dose and the total number of disease flares.
The results of the trial are presented in Table 19.
| MMF = Mycophenolate mofetil. CI = Confidence Interval. | |||
|
Rituximab (N=62) |
MMF (N=63) |
Difference (95% CI) |
|
|
Number of responders (response rate [%]) |
25 (40.3%) |
6 (9.5%) |
30.80% (14.70%, 45.15%) |
Glucocorticoid Exposure
The median (min, max) cumulative oral prednisone dose at Week 52 was 2,775 mg (450; 22,180) in the rituximab group compared to 4,005 mg (900; 19,920) in the MMF group. Topical corticosteroid use and pre-infusion IV methylprednisolone were not included in this analysis. Prior to each infusion, the rituximab group received IV methylprednisolone 100 mg and the MMF group received IV saline solution.
Disease Flare
Disease flare was defined as an appearance of 3 or more new lesions a month that do not heal spontaneously within 1 week or by the extension of established lesions in a patient who has achieved disease control. The total number of disease flares was lower in patients treated with rituximab compared to MMF (6 vs. 44).
3 Dosage Forms and Strengths
Injection: RUXIENCE is a clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion:
-
•100 mg/10 mL (10 mg/mL) in a single-dose vial
-
•500 mg/50 mL (10 mg/mL) in a single-dose vial
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of rituximab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
•Hematologic: prolonged pancytopenia, marrow hypoplasia, Grade 3–4 prolonged or late-onset neutropenia, hyperviscosity syndrome in Waldenstrom's macroglobulinemia, prolonged hypogammaglobulinemia [see Warnings and Precautions (5.6)].
-
•Cardiac: fatal cardiac failure.
-
•Immune/Autoimmune Events: uveitis, optic neuritis, systemic vasculitis, pleuritis, lupus-like syndrome, serum sickness, polyarticular arthritis, and vasculitis with rash.
-
•Infection: viral infections, including progressive multifocal leukoencephalopathy (PML), increase in fatal infections in HIV-associated lymphoma, and a reported increased incidence of Grade 3 and 4 infections [see Warnings and Precautions (5.6)].
-
•Neoplasia: disease progression of Kaposi's sarcoma.
-
•Skin: severe mucocutaneous reactions, pyoderma gangrenosum (including genital presentation).
-
•Gastrointestinal: bowel obstruction and perforation.
-
•Pulmonary: fatal bronchiolitis obliterans and fatal interstitial lung disease.
-
•Nervous system: Posterior Reversible Encephalopathy Syndrome (PRES)/Reversible Posterior Leukoencephalopathy Syndrome (RPLS).
1.3 Rheumatoid Arthritis (ra)
RUXIENCE, in combination with methotrexate, is indicated for the treatment of adult patients with moderately-to-severely-active rheumatoid arthritis who have had an inadequate response to one or more Tumor Necrosis Factor (TNF) antagonist therapies.
8 Use in Specific Populations
2.9 Administration and Storage
Use appropriate aseptic technique. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. RUXIENCE should be a clear to slightly opalescent, colorless to pale brownish-yellow liquid. Do not use vial if particulates or discoloration is present.
5.1 Infusion Related Reactions
Rituximab products can cause severe, including fatal, infusion-related reactions. Severe reactions typically occurred during the first infusion with time to onset of 30 to 120 minutes. Rituximab product-induced infusion-related reactions and sequelae include urticaria, hypotension, angioedema, hypoxia, bronchospasm, pulmonary infiltrates, acute respiratory distress syndrome, myocardial infarction, ventricular fibrillation, cardiogenic shock, anaphylactoid events, or death.
Premedicate patients with an antihistamine and acetaminophen prior to dosing. For RA, GPA and MPA, and PV patients, methylprednisolone 100 mg intravenously or its equivalent is recommended 30 minutes prior to each infusion. Institute medical management (e.g., glucocorticoids, epinephrine, bronchodilators, or oxygen) for infusion-related reactions as needed. Depending on the severity of the infusion-related reaction and the required interventions, temporarily or permanently discontinue RUXIENCE. Resume infusion at a minimum 50% reduction in rate after symptoms have resolved. Closely monitor the following patients: those with pre-existing cardiac or pulmonary conditions, those who experienced prior cardiopulmonary adverse reactions, and those with high numbers of circulating malignant cells (greater than or equal to 25,000/mm3) [see Warnings and Precautions (5.7), Adverse Reactions (6.1)].
5.5 Tumor Lysis Syndrome (tls)
Acute renal failure, hyperkalemia, hypocalcemia, hyperuricemia, or hyperphosphatemia from tumor lysis, sometimes fatal, can occur within 12–24 hours after the first infusion of rituximab products in patients with NHL. A high number of circulating malignant cells (greater than or equal to 25,000/mm3) or high tumor burden, confers a greater risk of TLS.
Administer aggressive intravenous hydration and anti-hyperuricemic therapy in patients at high risk for TLS. Correct electrolyte abnormalities, monitor renal function and fluid balance, and administer supportive care, including dialysis as indicated [see Warnings and Precautions (5.8)].
1.1 Non Hodgkin's Lymphoma (nhl)
RUXIENCE is indicated for the treatment of adult patients with:
-
•Relapsed or refractory, low-grade or follicular, CD20-positive, B-cell NHL as a single agent.
-
•Previously untreated follicular, CD20-positive, B-cell NHL in combination with first line chemotherapy and, in patients achieving a complete or partial response to a rituximab product in combination with chemotherapy, as single-agent maintenance therapy.
-
•Non-progressing (including stable disease), low-grade, CD20-positive, B-cell NHL as a single agent after first-line cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy.
-
•Previously untreated diffuse large B-cell, CD20-positive NHL in combination with cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) or other anthracycline-based chemotherapy regimens.
2.1 Important Dosing Information
Administer only as an intravenous infusion [see Dosage and Administration (2.9) ]. Do not administer as an intravenous push or bolus.
RUXIENCE should only be administered by a healthcare professional with appropriate medical support to manage severe infusion-related reactions that can be fatal if they occur [see Warnings and Precautions (5.1)].
Premedicate before each infusion [see Dosage and Administration (2.8) ].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.2 Severe Mucocutaneous Reactions
Mucocutaneous reactions, some with fatal outcome, can occur in patients treated with rituximab products. These reactions include paraneoplastic pemphigus, Stevens-Johnson syndrome, lichenoid dermatitis, vesiculobullous dermatitis, and toxic epidermal necrolysis. The onset of these reactions has been variable and includes reports with onset on the first day of rituximab exposure. Discontinue RUXIENCE in patients who experience a severe mucocutaneous reaction. The safety of re-administration of rituximab products to patients with severe mucocutaneous reactions has not been determined.
16 How Supplied/storage and Handling
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion supplied as follows:
|
Carton contents |
NDC number |
|
One 100 mg/10 mL (10 mg/mL) single-dose vial |
NDC 0069-0238-01 |
|
One 500 mg/50 mL (10 mg/mL) single-dose vial |
NDC 0069-0249-01 |
5.7 Cardiovascular Adverse Reactions
Cardiac adverse reactions, including ventricular fibrillation, myocardial infarction, and cardiogenic shock may occur in patients receiving rituximab products. Discontinue infusions for serious or life-threatening cardiac arrhythmias. Perform cardiac monitoring during and after all infusions of RUXIENCE for patients who develop clinically significant arrhythmias, or who have a history of arrhythmia or angina [see Adverse Reactions (6.1)].
14.3 Diffuse Large B Cell Nhl (dlbcl)
The safety and effectiveness of rituximab were evaluated in three randomized, active-controlled, open-label, multicenter studies with a collective enrollment of 1,854 patients. Patients with previously untreated diffuse large B-cell NHL received rituximab in combination with cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) or other anthracycline-based chemotherapy regimens.
5.9 Bowel Obstruction and Perforation
Abdominal pain, bowel obstruction and perforation, in some cases leading to death, can occur in patients receiving rituximab products in combination with chemotherapy. In postmarketing reports, the mean time to documented gastrointestinal perforation was 6 (range 1–77) days in patients with NHL. Evaluate if symptoms of obstruction such as abdominal pain or repeated vomiting occur.
1.2 Chronic Lymphocytic Leukemia (cll)
RUXIENCE, in combination with fludarabine and cyclophosphamide (FC), is indicated for the treatment of adult patients with previously untreated and previously treated CD20-positive CLL.
14.5 Chronic Lymphocytic Leukemia (cll)
The safety and effectiveness of rituximab were evaluated in two randomized (1:1) multicenter open-label studies comparing FC alone or in combination with rituximab for up to 6 cycles in patients with previously untreated CLL [CLL Study 1 (n=817)] or previously treated CLL [CLL Study 2 (n=552)]. Patients received fludarabine 25 mg/m2/day and cyclophosphamide 250 mg/m2/day on days 1, 2, and 3 of each cycle, with or without rituximab. In both studies, seventy-one percent of CLL patients received 6 cycles and 90% received at least 3 cycles of rituximab-based therapy.
In CLL Study 1, 30% of patients were 65 years or older, 31% were Binet stage C, 45% had B symptoms, more than 99% had ECOG performance status (PS) 0–1, 74% were male, and 100% were White. In CLL Study 2, 44% of patients were 65 years or older, 28% had B symptoms, 82% received a prior alkylating drug, 18% received prior fludarabine, 100% had ECOG PS 0–1, 67% were male, and 98% were White.
The main outcome measure in both studies was progression-free survival (PFS), defined as the time from randomization to progression, relapse, or death, as determined by investigators (CLL Study 1) or an independent review committee (CLL Study 2). The investigator assessed results in CLL Study 2 were supportive of those obtained by the independent review committee. Efficacy results are presented in Table 11.
|
CLL Study 1 As defined in 1996 National Cancer Institute Working Group guidelines.
(Previously untreated) |
CLL Study 2 (Previously treated) |
|||
|
R-FC N=408 |
FC N=409 |
R-FC N=276 |
FC N=276 |
|
|
Median PFS (months) |
39.8 |
31.5 |
26.7 |
21.7 |
|
Hazard ratio (95% CI) |
0.56 (0.43, 0.71) |
0.76 (0.6, 0.96) |
||
|
P value (Log-Rank test) |
<0.01 |
0.02 |
||
|
Response rate |
86% |
73% |
54% |
45% |
|
(95% CI) |
(82, 89) |
(68, 77) |
(48, 60) |
(37, 51) |
Across both studies, 243 of 676 rituximab-treated patients (36%) were 65 years of age or older and 100 rituximab-treated patients (15%) were 70 years of age or older. The results of exploratory subset analyses in elderly patients are presented in Table 12.
|
Age subgroup |
CLL Study 1 |
CLL Study 2 |
||
|
Number of Patients |
Hazard Ratio for PFS (95% CI) |
Number of Patients |
Hazard Ratio for PFS (95% CI) |
|
|
Age less than 65 yrs |
572 |
0.52 (0.39, 0.70) |
313 |
0.61 (0.45, 0.84) |
|
Age greater than or |
245 |
0.62 (0.39, 0.99) |
233 |
0.99 (0.70, 1.40) |
|
Age less than 70 yrs |
736 |
0.51 (0.39, 0.67) |
438 |
0.67 (0.51, 0.87) |
|
Age greater than or |
81 |
1.17 (0.51, 2.66) |
108 |
1.22 (0.73, 2.04) |
5.3 Hepatitis B Virus (hbv) Reactivation
Hepatitis B virus (HBV) reactivation, in some cases resulting in fulminant hepatitis, hepatic failure, and death, can occur in patients treated with drugs classified as CD20-directed cytolytic antibodies, including rituximab products. Cases have been reported in patients who are hepatitis B surface antigen (HBsAg) positive and also in patients who are HBsAg negative but are hepatitis B core antibody (anti-HBc) positive. Reactivation also has occurred in patients who appear to have resolved hepatitis B infection (i.e., HBsAg negative, anti-HBc positive and hepatitis B surface antibody [anti-HBs] positive).
HBV reactivation is defined as an abrupt increase in HBV replication manifesting as a rapid increase in serum HBV DNA levels or detection of HBsAg in a person who was previously HBsAg negative and anti-HBc positive. Reactivation of HBV replication is often followed by hepatitis, i.e., increase in transaminase levels. In severe cases increase in bilirubin levels, liver failure, and death can occur.
Screen all patients for HBV infection by measuring HBsAg and anti-HBc before initiating treatment with RUXIENCE. For patients who show evidence of prior hepatitis B infection (HBsAg positive [regardless of antibody status] or HBsAg negative but anti-HBc positive), consult with physicians with expertise in managing hepatitis B regarding monitoring and consideration for HBV antiviral therapy before and/or during RUXIENCE treatment.
Monitor patients with evidence of current or prior HBV infection for clinical and laboratory signs of hepatitis or HBV reactivation during and for several months following RUXIENCE therapy. HBV reactivation has been reported up to 24 months following completion of rituximab therapy.
In patients who develop reactivation of HBV while on RUXIENCE, immediately discontinue RUXIENCE and any concomitant chemotherapy, and institute appropriate treatment. Insufficient data exist regarding the safety of resuming RUXIENCE treatment in patients who develop HBV reactivation. Resumption of RUXIENCE treatment in patients whose HBV reactivation resolves should be discussed with physicians with expertise in managing HBV.
Principal Display Panel 10 Ml Vial Label
ALWAYS DISPENSE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0238-01
Rx only
Ruxience®
(rituximab-pvvr)
Injection
100 mg/10 mL
(10 mg/mL)
For Intravenous Use after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Principal Display Panel 50 Ml Vial Label
ALWAYS DISPENSE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0249-01
Ruxience®
(rituximab-pvvr)
Injection
500 mg/50 mL
(10 mg/mL)
For Intravenous Use after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Rx only
Principal Display Panel 10 Ml Vial Carton
ALWAYS DISPENSE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0238-01
Ruxience®
(rituximab-pvvr)
Injection
100 mg/10 mL
(10 mg/mL)
For Intravenous Use
after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Rx only
Pfizer Oncology
Principal Display Panel 50 Ml Vial Carton
ALWAYS DISPENSE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0249-01
Ruxience®
(rituximab-pvvr)
Injection
500 mg/50 mL
(10 mg/mL)
For Intravenous Use
after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Rx only
Pfizer Oncology
8.3 Females and Males of Reproductive Potential
Rituximab products can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
2.4 Recommended Dose As A Component of Zevalin®
When used as part of the Zevalin therapeutic regimen, infuse 250 mg/m2 in accordance with the Zevalin package insert. Refer to the Zevalin package insert for full prescribing information regarding the Zevalin therapeutic regimen.
2.7 Recommended Dose for Pemphigus Vulgaris (pv)
-
•Administer RUXIENCE as two-1,000 mg intravenous infusions separated by 2 weeks in combination with a tapering course of glucocorticoids.
-
•Maintenance treatment
Administer RUXIENCE as a 500 mg intravenous infusion at Month 12 and every 6 months thereafter or based on clinical evaluation. -
•Treatment of relapse
Administer RUXIENCE as a 1,000 mg intravenous infusion on relapse, and consider resuming or increasing the glucocorticoid dose based on clinical evaluation.
Subsequent infusions of RUXIENCE may be administered no sooner than 16 weeks following the previous infusion.
2.5 Recommended Dose for Rheumatoid Arthritis (ra)
-
•Administer RUXIENCE as two-1,000 mg intravenous infusions separated by 2 weeks.
-
•Glucocorticoids administered as methylprednisolone 100 mg intravenous or its equivalent 30 minutes prior to each infusion are recommended to reduce the incidence and severity of infusion-related reactions.
-
•Subsequent courses should be administered every 24 weeks or based on clinical evaluation, but not sooner than every 16 weeks.
-
•RUXIENCE is given in combination with methotrexate.
5.4 Progressive Multifocal Leukoencephalopathy (pml)
JC virus infection resulting in PML and death can occur in rituximab product-treated patients with hematologic malignancies or with autoimmune diseases. The majority of patients with hematologic malignancies diagnosed with PML received rituximab in combination with chemotherapy or as part of a hematopoietic stem cell transplant. The patients with autoimmune diseases had prior or concurrent immunosuppressive therapy. Most cases of PML were diagnosed within 12 months of their last infusion of rituximab.
Consider the diagnosis of PML in any patient presenting with new-onset neurologic manifestations. Evaluation of PML includes, but is not limited to, consultation with a neurologist, brain MRI, and lumbar puncture.
Discontinue RUXIENCE and consider discontinuation or reduction of any concomitant chemotherapy or immunosuppressive therapy in patients who develop PML.
2.2 Recommended Dose for Non Hodgkin's Lymphoma (nhl)
The recommended dose is 375 mg/m2 as an intravenous infusion according to the following schedules:
-
•Relapsed or Refractory, Low-Grade or Follicular, CD20-Positive, B-Cell NHL
Administer once weekly for 4 or 8 doses. -
•Retreatment for Relapsed or Refractory, Low-Grade or Follicular, CD20-Positive, B-Cell NHL
Administer once weekly for 4 doses. -
•Previously Untreated, Follicular, CD20-Positive, B-Cell NHL
Administer on Day 1 of each cycle of chemotherapy for up to 8 doses. In patients with complete or partial response, initiate RUXIENCE maintenance eight weeks following completion of a rituximab product in combination with chemotherapy. Administer RUXIENCE as a single agent every 8 weeks for 12 doses. -
•Non-progressing, Low-Grade, CD20-Positive, B-Cell NHL, after first-line CVP chemotherapy
Following completion of 6–8 cycles of CVP chemotherapy, administer once weekly for 4 doses at 6-month intervals to a maximum of 16 doses. -
•Diffuse Large B-Cell NHL
Administer on Day 1 of each cycle of chemotherapy for up to 8 infusions.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to establish the carcinogenic or mutagenic potential of rituximab products or to determine potential effects on fertility in males or females.
2.3 Recommended Dose for Chronic Lymphocytic Leukemia (cll)
The recommended dose is 375 mg/m2 the day prior to the initiation of FC chemotherapy, then 500 mg/m2 on Day 1 of Cycles 2–6 (every 28 days).
2.8 Recommended Dose for Premedication and Prophylactic Medications
Premedicate with acetaminophen and an antihistamine before each infusion of RUXIENCE. For adult patients administered RUXIENCE according to the 90-minute infusion rate, the glucocorticoid component of their chemotherapy regimen should be administered prior to infusion [see Clinical Studies (14.4)].
For RA, GPA and MPA, and PV patients, methylprednisolone 100 mg intravenously or its equivalent is recommended 30 minutes prior to each infusion.
Provide prophylaxis treatment for Pneumocystis jirovecii pneumonia (PCP) and herpes virus infections for patients with CLL during treatment and for up to 12 months following treatment as appropriate [see Warnings and Precautions (5.6)].
PCP prophylaxis is also recommended for patients with GPA and MPA during treatment and for at least 6 months following the last RUXIENCE infusion.
PCP prophylaxis should be considered for patients with PV during and following RUXIENCE treatment.
14.2 Previously Untreated, Low Grade Or Follicular, Cd20 Positive, B Cell Nhl
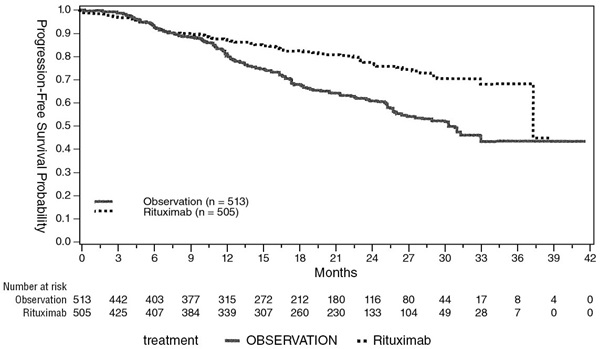
The safety and effectiveness of rituximab in previously untreated, low-grade or follicular, CD20+ NHL were demonstrated in 3 randomized, controlled trials enrolling 1,662 patients.
14.4 Ninety Minute Infusions in Previously Untreated Follicular Nhl and Dlbcl
In NHL Study 10, a total of 363 patients with previously untreated follicular NHL (n=113) or DLBCL (n=250) were evaluated in a prospective, open-label, multicenter, single-arm trial for the safety of 90-minute rituximab infusions. Patients with follicular NHL received rituximab 375 mg/m2 plus CVP chemotherapy. Patients with DLBCL received rituximab 375 mg/m2 plus CHOP chemotherapy. Patients with clinically significant cardiovascular disease were excluded from the study. Patients were eligible for a 90-minute infusion at Cycle 2 if they did not experience a Grade 3–4 infusion-related adverse event with Cycle 1 and had a circulating lymphocyte count less than or equal to 5,000/mm3 before Cycle 2. All patients were pre-medicated with acetaminophen and an antihistamine and received the glucocorticoid component of their chemotherapy prior to rituximab infusion. The main outcome measure was the development of Grade 3–4 infusion-related reactions on the day of, or day after, the 90-minute infusion at Cycle 2 [see Adverse Reactions (6.1)].
Eligible patients received their Cycle 2 rituximab infusion over 90 minutes as follows: 20% of the total dose given in the first 30 minutes and the remaining 80% of the total dose given over the next 60 minutes [see Dosage and Administration (2.1)]. Patients who tolerated the 90-minute rituximab infusion at Cycle 2 continued to receive subsequent rituximab infusions at the 90-minute infusion rate for the remainder of the treatment regimen (through Cycle 6 or Cycle 8).
The incidence of Grade 3–4 infusion-related reactions at Cycle 2 was 1.1% (95% CI [0.3%, 2.8%]) among all patients, 3.5% (95% CI [1.0%, 8.8%]) for those patients treated with R-CVP, and 0.0% (95% CI [0.0%, 1.5%]) for those patients treated with R-CHOP. For Cycles 2–8, the incidence of Grade 3–4 infusion-related reactions was 2.8% (95% CI [1.3%, 5.0%]). No acute fatal infusion-related reactions were observed.
14.1 Relapsed Or Refractory, Low Grade Or Follicular, Cd20 Positive, B Cell Nhl
The safety and effectiveness of rituximab in relapsed, refractory CD20+ NHL were demonstrated in 3 single-arm studies enrolling 296 patients.
1.4 Granulomatosis With Polyangiitis (gpa) (wegener's Granulomatosis) and Microscopic Polyangiitis (mpa)
RUXIENCE, in combination with glucocorticoids, is indicated for the treatment of adult patients with Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA).
5.12 Concomitant Use With Other Biologic Agents and Dmards Other Than Methotrexate in Ra, Gpa and Mpa, Pv
Limited data are available on the safety of the use of biologic agents or disease modifying anti-rheumatic drugs (DMARDs) other than methotrexate in RA patients exhibiting peripheral B-cell depletion following treatment with rituximab. Observe patients closely for signs of infection if biologic agents and/or DMARDs are used concomitantly. Use of concomitant immunosuppressants other than corticosteroids has not been studied in GPA or MPA or PV patients exhibiting peripheral B-cell depletion following treatment with rituximab products.
5.13 Use in Ra Patients Who Have Not Had Prior Inadequate Response to Tumor Necrosis Factor (tnf) Antagonists
While the efficacy of rituximab was supported in four controlled trials in patients with RA with prior inadequate responses to non-biologic DMARDs, and in a controlled trial in MTX-naïve patients, a favorable risk-benefit relationship has not been established in these populations. The use of RUXIENCE in patients with RA who have not had prior inadequate response to one or more TNF antagonists is not recommended [see Clinical Studies (14.6)].
2.6 Recommended Dose for Granulomatosis With Polyangiitis (gpa) (wegener's Granulomatosis) and Microscopic Polyangiitis (mpa)
Warning: Fatal Infusion Related Reactions, Severe Mucocutaneous Reactions, Hepatitis B Virus Reactivation and Progressive Multifocal Leukoencephalopathy
Infusion-Related Reactions
Administration of rituximab products can result in serious, including fatal, infusion-related reactions. Deaths within 24 hours of rituximab infusion have occurred. Approximately 80% of fatal infusion-related reactions occurred in association with the first infusion. Monitor patients closely. Discontinue RUXIENCE infusion for severe reactions and provide medical treatment for Grade 3 or 4 infusion-related reactions [see Warnings and Precautions (5.1), Adverse Reactions (6.1)].
Severe Mucocutaneous Reactions
Severe, including fatal, mucocutaneous reactions can occur in patients receiving rituximab products [see Warnings and Precautions (5.2)].
Hepatitis B Virus (HBV) Reactivation
HBV reactivation can occur in patients treated with rituximab products, in some cases resulting in fulminant hepatitis, hepatic failure, and death. Screen all patients for HBV infection before treatment initiation, and monitor patients during and after treatment with RUXIENCE. Discontinue RUXIENCE and concomitant medications in the event of HBV reactivation [see Warnings and Precautions (5.3)].
Progressive Multifocal Leukoencephalopathy (PML)
Progressive Multifocal Leukoencephalopathy (PML), including fatal PML, can occur in patients receiving rituximab products [see Warnings and Precautions (5.4), Adverse Reactions (6.3)].
Structured Label Content
Section 42229-5 (42229-5)
Prior to First Infusion
Screen all patients for HBV infection by measuring HBsAg and anti-HBc before initiating treatment with RUXIENCE [see Warnings and Precautions (5.3)]. Obtain complete blood counts (CBC) including platelets prior to the first dose.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 6/2025 | ||
|
MEDICATION GUIDE
|
||
|
What is the most important information I should know about RUXIENCE?
|
||
|
|
|
|
||
|
|
|
|
See " What are the possible side effects of RUXIENCE? " for more information about side effects. |
||
|
What is RUXIENCE?
RUXIENCE is not indicated for treatment of children. |
||
|
Before you receive RUXIENCE, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take or have taken:
If you are not sure if your medicine is one listed above, ask your healthcare provider. |
||
|
How will I receive RUXIENCE?
|
||
|
What are the possible side effects of RUXIENCE?
|
||
|
|
|
Your healthcare provider will stop treatment with RUXIENCE if you have severe, serious or life-threatening side effects.
In adults with GPA or MPA the most common side effects of RUXIENCE also include:
Other side effects with RUXIENCE include:
These are not all of the possible side effects with RUXIENCE. |
||
|
General information about the safe and effective use of RUXIENCE.
|
||
|
What are the ingredients in RUXIENCE?
Manufactured by Pfizer Ireland Pharmaceuticals, Cork, Ireland, P43 X336 U.S. License No. 2060
|
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store RUXIENCE vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton. RUXIENCE vials should be protected from direct sunlight. Do not freeze or shake.
8.2 Lactation
There are limited data on the presence of rituximab in human milk, and the effect on the breastfed child, and there are no data on the effect on milk production. Rituximab is detected in the milk of lactating cynomolgus monkeys, and maternal IgG is present in human breast milk. Rituximab has also been reported to be excreted at low concentrations in human breast milk. Given that the clinical significance of this finding for children is not known, advise women not to breastfeed during treatment with RUXIENCE and for 6 months after the last dose due to the potential of serious adverse reactions in breastfed children.
11 Description (11 DESCRIPTION)
Rituximab-pvvr is a genetically engineered chimeric murine/human monoclonal IgG1 kappa antibody directed against the CD20 antigen. Rituximab-pvvr has an approximate molecular weight of 145 kD.
Rituximab-pvvr is produced by mammalian cell (Chinese Hamster Ovary) suspension culture in a nutrient medium.
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion. RUXIENCE is supplied at a concentration of 10 mg/mL in either 100 mg/10 mL or 500 mg/50 mL single-dose vials. Each mL of solution contains 10 mg rituximab-pvvr, 0.056 mg of edetate disodium dihydrate, 1.2 mg of L-histidine, 2.57 mg of L-histidine hydrochloride monohydrate, 0.2 mg of polysorbate 80, 85 mg of sucrose, and Water for Injection, USP. The pH is 5.8.
5.6 Infections
Serious, including fatal, bacterial, fungal, and new or reactivated viral infections can occur during and following the completion of rituximab product-based therapy. Infections have been reported in some patients with prolonged hypogammaglobulinemia (defined as hypogammaglobulinemia greater than 11 months after rituximab exposure). New or reactivated viral infections included cytomegalovirus, herpes simplex virus, parvovirus B19, varicella zoster virus, West Nile virus, and hepatitis B and C. Discontinue RUXIENCE for serious infections and institute appropriate anti-infective therapy [see Adverse Reactions (6.1 , 6.3)]. RUXIENCE is not recommended for use in patients with severe, active infections.
5.10 Immunization
The safety of immunization with live viral vaccines following rituximab product therapy has not been studied and vaccination with live virus vaccines is not recommended before or during treatment.
For patients treated with RUXIENCE, physicians should review the patient's vaccination status and patients should, if possible, be brought up-to-date with all immunizations in agreement with current immunization guidelines prior to initiating RUXIENCE and administer non live vaccines at least 4 weeks prior to a course of RUXIENCE.
The effect of rituximab on immune responses was assessed in a randomized, controlled study in patients with RA treated with rituximab and methotrexate (MTX) compared to patients treated with MTX alone.
A response to pneumococcal vaccination (a T-cell independent antigen) as measured by an increase in antibody titers to at least 6 of 12 serotypes was lower in patients treated with rituximab plus MTX as compared to patients treated with MTX alone (19% vs. 61%). A lower proportion of patients in the rituximab plus MTX group developed detectable levels of anti-keyhole limpet hemocyanin antibodies (a novel protein antigen) after vaccination compared to patients on MTX alone (47% vs. 93%).
A positive response to tetanus toxoid vaccine (a T-cell dependent antigen with existing immunity) was similar in patients treated with rituximab plus MTX compared to patients on MTX alone (39% vs. 42%). The proportion of patients maintaining a positive Candida skin test (to evaluate delayed type hypersensitivity) was also similar (77% of patients on rituximab plus MTX vs. 70% of patients on MTX alone).
Most patients in the rituximab-treated group had B-cell counts below the lower limit of normal at the time of immunization. The clinical implications of these findings are not known.
8.4 Pediatric Use
The safety and effectiveness of RUXIENCE have not been established in pediatric patients with NHL, CLL, PV or RA.
Rituximab was not studied in pediatric patients with polyarticular juvenile idiopathic arthritis (PJIA) due to concerns regarding the potential for prolonged immunosuppression as a result of B-cell depletion in the developing juvenile immune system.
5.8 Renal Toxicity
Severe, including fatal, renal toxicity can occur after rituximab product administration in patients with NHL. Renal toxicity has occurred in patients who experience tumor lysis syndrome and in patients with NHL administered concomitant cisplatin therapy during clinical trials. The combination of cisplatin and RUXIENCE is not an approved treatment regimen. Monitor closely for signs of renal failure and discontinue RUXIENCE in patients with a rising serum creatinine or oliguria [see Warnings and Precautions (5.5)].
6.2 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of rituximab or of other rituximab products.
Using an ELISA assay, anti-rituximab antibody was detected in 4 of 356 (1.1%) patients with low-grade or follicular NHL receiving single-agent rituximab. Three of the four patients had an objective clinical response.
A total of 273/2578 (11%) patients with RA tested positive for anti-rituximab antibodies at any time after receiving rituximab. Anti-rituximab antibody positivity was not associated with increased rates of infusion-related reactions or other adverse events. Upon further treatment, the proportions of patients with infusion-related reactions were similar between anti-rituximab antibody positive and negative patients, and most reactions were mild to moderate. Four anti-rituximab antibody positive patients had serious infusion-related reactions, and the temporal relationship between anti-rituximab antibody positivity and infusion-related reaction was variable.
A total of 23/99 (23%) rituximab-treated adult patients with GPA and MPA developed anti-rituximab antibodies by 18 months in GPA/MPA Study 1. The clinical relevance of anti-rituximab antibody formation in rituximab-treated adult patients is unclear.
Using a new ELISA assay, a total of 19/34 (56%) patients with PV, who were treated with non-U.S.-licensed rituximab, tested positive for anti-rituximab antibodies by 18 months in PV Study 1. In PV Study 2, a total of 20/63 (32%) rituximab-treated PV patients tested positive for ADA by week 52 (19 patients had treatment-inducted ADA and 1 patient had treatment-enhanced ADA). The clinical relevance of anti-rituximab antibody formation in rituximab-treated PV patients is unclear.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Infusion-related reactions [see Warnings and Precautions (5.1)]
-
•Severe mucocutaneous reactions [see Warnings and Precautions (5.2)]
-
•Hepatitis B reactivation with fulminant hepatitis [see Warnings and Precautions (5.3)]
-
•Progressive multifocal leukoencephalopathy [see Warnings and Precautions (5.4)]
-
•Tumor lysis syndrome [see Warnings and Precautions (5.5)]
-
•Infections [see Warnings and Precautions (5.6)]
-
•Cardiovascular adverse reactions [see Warnings and Precautions (5.7)]
-
•Renal toxicity [see Warnings and Precautions (5.8)]
-
•Bowel obstruction and perforation [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Formal drug interaction studies have not been performed with rituximab products. In patients with CLL, rituximab did not alter systemic exposure to fludarabine or cyclophosphamide. In clinical trials of patients with RA, concomitant administration of methotrexate or cyclophosphamide did not alter the pharmacokinetics of rituximab.
1 Indications and Usage (1 INDICATIONS AND USAGE)
RUXIENCE is a CD20-directed cytolytic antibody indicated for the treatment of:
-
•Adult patients with Non-Hodgkin's Lymphoma (NHL) (1.1).
-
oRelapsed or refractory, low grade or follicular, CD20-positive B-cell NHL as a single agent.
-
oPreviously untreated follicular, CD20-positive, B-cell NHL in combination with first line chemotherapy and, in patients achieving a complete or partial response to a rituximab product in combination with chemotherapy, as single-agent maintenance therapy.
-
oNon-progressing (including stable disease), low-grade, CD20-positive, B-cell NHL as a single agent after first-line cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy.
-
oPreviously untreated diffuse large B-cell, CD20-positive NHL in combination with (cyclophosphamide, doxorubicin, vincristine, and prednisone) (CHOP) or other anthracycline-based chemotherapy regimens.
-
-
•Adult patients with Chronic Lymphocytic Leukemia (CLL) (1.2).
-
oPreviously untreated and previously treated CD20-positive CLL in combination with fludarabine and cyclophosphamide (FC).
-
-
•Rheumatoid Arthritis (RA) in combination with methotrexate in adult patients with moderately-to-severely-active RA who have inadequate response to one or more TNF antagonist therapies (1.3).
-
•Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA) in adult patients in combination with glucocorticoids (1.4).
-
•Moderate to severe Pemphigus Vulgaris (PV) in adult patients (1.5).
12.1 Mechanism of Action
Rituximab-pvvr is a monoclonal antibody. Rituximab products target the CD20 antigen expressed on the surface of pre-B and mature B-lymphocytes. Upon binding to CD20, rituximab products mediate B-cell lysis. Possible mechanisms of cell lysis include complement dependent cytotoxicity (CDC) and antibody dependent cell mediated cytotoxicity (ADCC). B cells are believed to play a role in the pathogenesis of rheumatoid arthritis (RA) and associated chronic synovitis. In this setting, B cells may be acting at multiple sites in the autoimmune/inflammatory process, including through production of rheumatoid factor (RF) and other autoantibodies, antigen presentation, T-cell activation, and/or proinflammatory cytokine production.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Tumor lysis syndrome: Administer aggressive intravenous hydration, anti-hyperuricemic agents, monitor renal function (5.5).
-
•Infections: Withhold RUXIENCE and institute appropriate anti-infective therapy (5.6).
-
•Cardiac adverse reactions: Discontinue infusions in case of serious or life-threatening events (5.7).
-
•Renal toxicity: Discontinue in patients with rising serum creatinine or oliguria (5.8).
-
•Bowel obstruction and perforation: Consider and evaluate for abdominal pain, vomiting, or related symptoms (5.9).
-
•Immunizations: Live virus vaccinations prior to or during RUXIENCE treatment are not recommended (5.10).
-
•Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception (5.11).
5.11 Embryo Fetal Toxicity (5.11 Embryo-Fetal Toxicity)
Based on human data, rituximab products can cause fetal harm due to B-cell lymphocytopenia in infants exposed in-utero. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception while receiving RUXIENCE and for 12 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
1.5 Pemphigus Vulgaris (pv) (1.5 Pemphigus Vulgaris (PV))
RUXIENCE is indicated for the treatment of adult patients with moderate to severe pemphigus vulgaris.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Administer only as an intravenous infusion (2.1).
-
•Do not administer as an intravenous push or bolus (2.1).
-
•RUXIENCE should only be administered by a healthcare professional with appropriate medical support to manage severe infusion-related reactions that can be fatal if they occur (2.1).
-
•The dose for adult B-cell NHL is 375 mg/m2 (2.2).
-
•The dose for CLL is 375 mg/m2 in the first cycle and 500 mg/m2 in Cycles 2–6, in combination with FC, administered every 28 days (2.3).
-
•The dose as a component of Zevalin® (ibritumomab tiuxetan) Therapeutic Regimen is 250 mg/m2 (2.4).
-
•The dose for RA in combination with methotrexate is two-1,000 mg intravenous infusions separated by 2 weeks (one course) every 24 weeks or based on clinical evaluation, but not sooner than every 16 weeks. Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion (2.5).
-
•The induction dose for adult patients with active GPA and MPA in combination with glucocorticoids is 375 mg/m2 once weekly for 4 weeks. The follow up dose for adult patients with GPA and MPA who have achieved disease control with induction treatment, in combination with glucocorticoids is two 500 mg intravenous infusions separated by two weeks, followed by a 500 mg intravenous infusion every 6 months thereafter based on clinical evaluation (2.6).
-
•The dose for PV is two-1,000 mg intravenous infusions separated by 2 weeks in combination with a tapering course of glucocorticoids, then a 500 mg intravenous infusion at Month 12 and every 6 months thereafter or based on clinical evaluation. Dose upon relapse is a 1,000 mg intravenous infusion with considerations to resume or increase the glucocorticoid dose based on clinical evaluation. Subsequent infusions may be no sooner than 16 weeks after the previous infusion (2.7). Methylprednisolone 100 mg intravenous or equivalent glucocorticoid is recommended 30 minutes prior to each infusion (2.8).
14.8 Pemphigus Vulgaris (pv) (14.8 Pemphigus Vulgaris (PV))
PV Study 1 (NCT00784589)
Non-U.S.-licensed rituximab in combination with short-term prednisone was compared to prednisone monotherapy as first-line treatment in 90 newly diagnosed adult patients with moderate to severe pemphigus (74 Pemphigus Vulgaris [PV] and 16 Pemphigus Foliaceus [PF]) in this randomized, open-label, controlled, multicenter study (PV Study 1). Patients were between 19 and 79 years of age and had not received prior therapies for pemphigus. In the PV population, 5 (13%) patients in the group treated with non-U.S.-licensed rituximab and 3 (8%) patients in the prednisone group had moderate disease and 33 (87%) patients in the group treated with non-U.S.-licensed rituximab and 33 (92%) patients in the prednisone group had severe disease according to disease severity defined by Harman’s criteria.
Patients were stratified by baseline disease severity (moderate or severe) and randomized 1:1 to receive either the non-U.S.-licensed rituximab and short-term prednisone or long-term prednisone monotherapy. Patients were pre-medicated with antihistamine, acetaminophen and methylprednisolone prior to infusion of the non-U.S.-licensed rituximab. Patients randomized to the group treated with non-U.S.-licensed rituximab received an initial intravenous infusion of 1,000 mg non-U.S.-licensed rituximab on Study Day 1 in combination with a short-term regimen of 0.5 mg/kg/day oral prednisone tapered off over 3 months if they had moderate disease or 1 mg/kg/day oral prednisone tapered off over 6 months if they had severe disease. All patients received a second intravenous infusion of 1,000 mg non-U.S.-licensed rituximab on Study Day 15. Maintenance infusions of 500 mg non-U.S.-licensed rituximab were administered at Months 12 and 18. Patients randomized to the prednisone monotherapy group received an initial 1 mg/kg/day oral prednisone tapered off over 12 months if they had moderate disease or 1.5 mg/kg/day oral prednisone tapered off over 18 months if they had severe disease. Patients in the group treated with non-U.S.-licensed rituximab who relapsed could receive an additional infusion of 1,000 mg non-U.S.-licensed rituximab in combination with reintroduced or escalated prednisone dose. Maintenance and relapse infusions were administered no sooner than 16 weeks following the previous infusion.
The primary endpoint for the study was complete remission (complete epithelialization and absence of new and/or established lesions) at Month 24 without the use of prednisone therapy for 2 months or more (CRoff for greater than or equal to 2 months).
The results of the trial are presented in Table 18.
|
Non-U.S.-Licensed Rituximab + Short-term Prednisone N=46 |
Prednisone N=44 |
|
|
Number of responders (response rate [%]) |
41 (89%) |
15 (34%) |
|
PV patients |
34/38 (90%) |
10/36 (28%) |
|
PF patients |
7/8 (88%) |
5/8 (63%) |
PV Study 2 (NCT02383589)
In a randomized, double-blind, double-dummy, active-comparator, multicenter study, the efficacy and safety of rituximab compared to mycophenolate mofetil (MMF) were evaluated in patients with moderate to severe PV receiving 60-120 mg/day oral prednisone or equivalent (1.0-1.5 mg/kg/day) at study entry and tapered to reach a dose of 60 or 80 mg/day by Day 1. Patients had a confirmed diagnosis of PV within the previous 24 months and evidence of moderate to severe disease defined as a total Pemphigus Disease Area Index (PDAI) activity score of greater than or equal to 15. The study consisted of a screening period of up to 28 days, a 52-week double-blind treatment period, and a 48-week safety follow up period.
One hundred and thirty-five patients were randomized to treatment with rituximab 1,000 mg administered on Day 1, Day 15, Week 24 and Week 26 or oral MMF 2 g/day (starting at 1 g/day on Day 1 and titrated to achieve a goal of 2 g/day by Week 2) for 52 weeks in combination with an initial dose of 60 or 80 mg oral prednisone with the aim of tapering to 0 mg/day by Week 24. Randomization was stratified by duration of PV (within the 1 year prior to screening or greater than 1 year) and geographical region. A dual-assessor approach was used during the study for efficacy and safety evaluations to prevent potential unblinding.
One hundred and twenty-five patients (excluding exploratory data from ten telemedicine patients) were analyzed for efficacy (Modified Intent-to-Treat Population). The primary efficacy endpoint for this study was the proportion of subjects achieving sustained complete remission defined as achieving healing of lesions with no new active lesions (i.e., PDAI activity score of 0) while on 0 mg/day prednisone or equivalent, and maintaining this response for at least 16 consecutive weeks, during the 52-week treatment period.
Secondary endpoints included cumulative oral corticosteroid dose and the total number of disease flares.
The results of the trial are presented in Table 19.
| MMF = Mycophenolate mofetil. CI = Confidence Interval. | |||
|
Rituximab (N=62) |
MMF (N=63) |
Difference (95% CI) |
|
|
Number of responders (response rate [%]) |
25 (40.3%) |
6 (9.5%) |
30.80% (14.70%, 45.15%) |
Glucocorticoid Exposure
The median (min, max) cumulative oral prednisone dose at Week 52 was 2,775 mg (450; 22,180) in the rituximab group compared to 4,005 mg (900; 19,920) in the MMF group. Topical corticosteroid use and pre-infusion IV methylprednisolone were not included in this analysis. Prior to each infusion, the rituximab group received IV methylprednisolone 100 mg and the MMF group received IV saline solution.
Disease Flare
Disease flare was defined as an appearance of 3 or more new lesions a month that do not heal spontaneously within 1 week or by the extension of established lesions in a patient who has achieved disease control. The total number of disease flares was lower in patients treated with rituximab compared to MMF (6 vs. 44).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: RUXIENCE is a clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion:
-
•100 mg/10 mL (10 mg/mL) in a single-dose vial
-
•500 mg/50 mL (10 mg/mL) in a single-dose vial
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of rituximab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
•Hematologic: prolonged pancytopenia, marrow hypoplasia, Grade 3–4 prolonged or late-onset neutropenia, hyperviscosity syndrome in Waldenstrom's macroglobulinemia, prolonged hypogammaglobulinemia [see Warnings and Precautions (5.6)].
-
•Cardiac: fatal cardiac failure.
-
•Immune/Autoimmune Events: uveitis, optic neuritis, systemic vasculitis, pleuritis, lupus-like syndrome, serum sickness, polyarticular arthritis, and vasculitis with rash.
-
•Infection: viral infections, including progressive multifocal leukoencephalopathy (PML), increase in fatal infections in HIV-associated lymphoma, and a reported increased incidence of Grade 3 and 4 infections [see Warnings and Precautions (5.6)].
-
•Neoplasia: disease progression of Kaposi's sarcoma.
-
•Skin: severe mucocutaneous reactions, pyoderma gangrenosum (including genital presentation).
-
•Gastrointestinal: bowel obstruction and perforation.
-
•Pulmonary: fatal bronchiolitis obliterans and fatal interstitial lung disease.
-
•Nervous system: Posterior Reversible Encephalopathy Syndrome (PRES)/Reversible Posterior Leukoencephalopathy Syndrome (RPLS).
1.3 Rheumatoid Arthritis (ra) (1.3 Rheumatoid Arthritis (RA))
RUXIENCE, in combination with methotrexate, is indicated for the treatment of adult patients with moderately-to-severely-active rheumatoid arthritis who have had an inadequate response to one or more Tumor Necrosis Factor (TNF) antagonist therapies.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
2.9 Administration and Storage
Use appropriate aseptic technique. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. RUXIENCE should be a clear to slightly opalescent, colorless to pale brownish-yellow liquid. Do not use vial if particulates or discoloration is present.
5.1 Infusion Related Reactions (5.1 Infusion-Related Reactions)
Rituximab products can cause severe, including fatal, infusion-related reactions. Severe reactions typically occurred during the first infusion with time to onset of 30 to 120 minutes. Rituximab product-induced infusion-related reactions and sequelae include urticaria, hypotension, angioedema, hypoxia, bronchospasm, pulmonary infiltrates, acute respiratory distress syndrome, myocardial infarction, ventricular fibrillation, cardiogenic shock, anaphylactoid events, or death.
Premedicate patients with an antihistamine and acetaminophen prior to dosing. For RA, GPA and MPA, and PV patients, methylprednisolone 100 mg intravenously or its equivalent is recommended 30 minutes prior to each infusion. Institute medical management (e.g., glucocorticoids, epinephrine, bronchodilators, or oxygen) for infusion-related reactions as needed. Depending on the severity of the infusion-related reaction and the required interventions, temporarily or permanently discontinue RUXIENCE. Resume infusion at a minimum 50% reduction in rate after symptoms have resolved. Closely monitor the following patients: those with pre-existing cardiac or pulmonary conditions, those who experienced prior cardiopulmonary adverse reactions, and those with high numbers of circulating malignant cells (greater than or equal to 25,000/mm3) [see Warnings and Precautions (5.7), Adverse Reactions (6.1)].
5.5 Tumor Lysis Syndrome (tls) (5.5 Tumor Lysis Syndrome (TLS))
Acute renal failure, hyperkalemia, hypocalcemia, hyperuricemia, or hyperphosphatemia from tumor lysis, sometimes fatal, can occur within 12–24 hours after the first infusion of rituximab products in patients with NHL. A high number of circulating malignant cells (greater than or equal to 25,000/mm3) or high tumor burden, confers a greater risk of TLS.
Administer aggressive intravenous hydration and anti-hyperuricemic therapy in patients at high risk for TLS. Correct electrolyte abnormalities, monitor renal function and fluid balance, and administer supportive care, including dialysis as indicated [see Warnings and Precautions (5.8)].
1.1 Non Hodgkin's Lymphoma (nhl) (1.1 Non-Hodgkin's Lymphoma (NHL))
RUXIENCE is indicated for the treatment of adult patients with:
-
•Relapsed or refractory, low-grade or follicular, CD20-positive, B-cell NHL as a single agent.
-
•Previously untreated follicular, CD20-positive, B-cell NHL in combination with first line chemotherapy and, in patients achieving a complete or partial response to a rituximab product in combination with chemotherapy, as single-agent maintenance therapy.
-
•Non-progressing (including stable disease), low-grade, CD20-positive, B-cell NHL as a single agent after first-line cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy.
-
•Previously untreated diffuse large B-cell, CD20-positive NHL in combination with cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) or other anthracycline-based chemotherapy regimens.
2.1 Important Dosing Information
Administer only as an intravenous infusion [see Dosage and Administration (2.9) ]. Do not administer as an intravenous push or bolus.
RUXIENCE should only be administered by a healthcare professional with appropriate medical support to manage severe infusion-related reactions that can be fatal if they occur [see Warnings and Precautions (5.1)].
Premedicate before each infusion [see Dosage and Administration (2.8) ].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.2 Severe Mucocutaneous Reactions
Mucocutaneous reactions, some with fatal outcome, can occur in patients treated with rituximab products. These reactions include paraneoplastic pemphigus, Stevens-Johnson syndrome, lichenoid dermatitis, vesiculobullous dermatitis, and toxic epidermal necrolysis. The onset of these reactions has been variable and includes reports with onset on the first day of rituximab exposure. Discontinue RUXIENCE in patients who experience a severe mucocutaneous reaction. The safety of re-administration of rituximab products to patients with severe mucocutaneous reactions has not been determined.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
RUXIENCE (rituximab-pvvr) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to pale brownish-yellow solution for intravenous infusion supplied as follows:
|
Carton contents |
NDC number |
|
One 100 mg/10 mL (10 mg/mL) single-dose vial |
NDC 0069-0238-01 |
|
One 500 mg/50 mL (10 mg/mL) single-dose vial |
NDC 0069-0249-01 |
5.7 Cardiovascular Adverse Reactions
Cardiac adverse reactions, including ventricular fibrillation, myocardial infarction, and cardiogenic shock may occur in patients receiving rituximab products. Discontinue infusions for serious or life-threatening cardiac arrhythmias. Perform cardiac monitoring during and after all infusions of RUXIENCE for patients who develop clinically significant arrhythmias, or who have a history of arrhythmia or angina [see Adverse Reactions (6.1)].
14.3 Diffuse Large B Cell Nhl (dlbcl) (14.3 Diffuse Large B-Cell NHL (DLBCL))
The safety and effectiveness of rituximab were evaluated in three randomized, active-controlled, open-label, multicenter studies with a collective enrollment of 1,854 patients. Patients with previously untreated diffuse large B-cell NHL received rituximab in combination with cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) or other anthracycline-based chemotherapy regimens.
5.9 Bowel Obstruction and Perforation
Abdominal pain, bowel obstruction and perforation, in some cases leading to death, can occur in patients receiving rituximab products in combination with chemotherapy. In postmarketing reports, the mean time to documented gastrointestinal perforation was 6 (range 1–77) days in patients with NHL. Evaluate if symptoms of obstruction such as abdominal pain or repeated vomiting occur.
1.2 Chronic Lymphocytic Leukemia (cll) (1.2 Chronic Lymphocytic Leukemia (CLL))
RUXIENCE, in combination with fludarabine and cyclophosphamide (FC), is indicated for the treatment of adult patients with previously untreated and previously treated CD20-positive CLL.
14.5 Chronic Lymphocytic Leukemia (cll) (14.5 Chronic Lymphocytic Leukemia (CLL))
The safety and effectiveness of rituximab were evaluated in two randomized (1:1) multicenter open-label studies comparing FC alone or in combination with rituximab for up to 6 cycles in patients with previously untreated CLL [CLL Study 1 (n=817)] or previously treated CLL [CLL Study 2 (n=552)]. Patients received fludarabine 25 mg/m2/day and cyclophosphamide 250 mg/m2/day on days 1, 2, and 3 of each cycle, with or without rituximab. In both studies, seventy-one percent of CLL patients received 6 cycles and 90% received at least 3 cycles of rituximab-based therapy.
In CLL Study 1, 30% of patients were 65 years or older, 31% were Binet stage C, 45% had B symptoms, more than 99% had ECOG performance status (PS) 0–1, 74% were male, and 100% were White. In CLL Study 2, 44% of patients were 65 years or older, 28% had B symptoms, 82% received a prior alkylating drug, 18% received prior fludarabine, 100% had ECOG PS 0–1, 67% were male, and 98% were White.
The main outcome measure in both studies was progression-free survival (PFS), defined as the time from randomization to progression, relapse, or death, as determined by investigators (CLL Study 1) or an independent review committee (CLL Study 2). The investigator assessed results in CLL Study 2 were supportive of those obtained by the independent review committee. Efficacy results are presented in Table 11.
|
CLL Study 1 As defined in 1996 National Cancer Institute Working Group guidelines.
(Previously untreated) |
CLL Study 2 (Previously treated) |
|||
|
R-FC N=408 |
FC N=409 |
R-FC N=276 |
FC N=276 |
|
|
Median PFS (months) |
39.8 |
31.5 |
26.7 |
21.7 |
|
Hazard ratio (95% CI) |
0.56 (0.43, 0.71) |
0.76 (0.6, 0.96) |
||
|
P value (Log-Rank test) |
<0.01 |
0.02 |
||
|
Response rate |
86% |
73% |
54% |
45% |
|
(95% CI) |
(82, 89) |
(68, 77) |
(48, 60) |
(37, 51) |
Across both studies, 243 of 676 rituximab-treated patients (36%) were 65 years of age or older and 100 rituximab-treated patients (15%) were 70 years of age or older. The results of exploratory subset analyses in elderly patients are presented in Table 12.
|
Age subgroup |
CLL Study 1 |
CLL Study 2 |
||
|
Number of Patients |
Hazard Ratio for PFS (95% CI) |
Number of Patients |
Hazard Ratio for PFS (95% CI) |
|
|
Age less than 65 yrs |
572 |
0.52 (0.39, 0.70) |
313 |
0.61 (0.45, 0.84) |
|
Age greater than or |
245 |
0.62 (0.39, 0.99) |
233 |
0.99 (0.70, 1.40) |
|
Age less than 70 yrs |
736 |
0.51 (0.39, 0.67) |
438 |
0.67 (0.51, 0.87) |
|
Age greater than or |
81 |
1.17 (0.51, 2.66) |
108 |
1.22 (0.73, 2.04) |
5.3 Hepatitis B Virus (hbv) Reactivation (5.3 Hepatitis B Virus (HBV) Reactivation)
Hepatitis B virus (HBV) reactivation, in some cases resulting in fulminant hepatitis, hepatic failure, and death, can occur in patients treated with drugs classified as CD20-directed cytolytic antibodies, including rituximab products. Cases have been reported in patients who are hepatitis B surface antigen (HBsAg) positive and also in patients who are HBsAg negative but are hepatitis B core antibody (anti-HBc) positive. Reactivation also has occurred in patients who appear to have resolved hepatitis B infection (i.e., HBsAg negative, anti-HBc positive and hepatitis B surface antibody [anti-HBs] positive).
HBV reactivation is defined as an abrupt increase in HBV replication manifesting as a rapid increase in serum HBV DNA levels or detection of HBsAg in a person who was previously HBsAg negative and anti-HBc positive. Reactivation of HBV replication is often followed by hepatitis, i.e., increase in transaminase levels. In severe cases increase in bilirubin levels, liver failure, and death can occur.
Screen all patients for HBV infection by measuring HBsAg and anti-HBc before initiating treatment with RUXIENCE. For patients who show evidence of prior hepatitis B infection (HBsAg positive [regardless of antibody status] or HBsAg negative but anti-HBc positive), consult with physicians with expertise in managing hepatitis B regarding monitoring and consideration for HBV antiviral therapy before and/or during RUXIENCE treatment.
Monitor patients with evidence of current or prior HBV infection for clinical and laboratory signs of hepatitis or HBV reactivation during and for several months following RUXIENCE therapy. HBV reactivation has been reported up to 24 months following completion of rituximab therapy.
In patients who develop reactivation of HBV while on RUXIENCE, immediately discontinue RUXIENCE and any concomitant chemotherapy, and institute appropriate treatment. Insufficient data exist regarding the safety of resuming RUXIENCE treatment in patients who develop HBV reactivation. Resumption of RUXIENCE treatment in patients whose HBV reactivation resolves should be discussed with physicians with expertise in managing HBV.
Principal Display Panel 10 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 10 mL Vial Label)
ALWAYS DISPENSE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0238-01
Rx only
Ruxience®
(rituximab-pvvr)
Injection
100 mg/10 mL
(10 mg/mL)
For Intravenous Use after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Principal Display Panel 50 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 50 mL Vial Label)
ALWAYS DISPENSE ENCLOSED MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0249-01
Ruxience®
(rituximab-pvvr)
Injection
500 mg/50 mL
(10 mg/mL)
For Intravenous Use after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Rx only
Principal Display Panel 10 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton)
ALWAYS DISPENSE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0238-01
Ruxience®
(rituximab-pvvr)
Injection
100 mg/10 mL
(10 mg/mL)
For Intravenous Use
after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Rx only
Pfizer Oncology
Principal Display Panel 50 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 50 mL Vial Carton)
ALWAYS DISPENSE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT
NDC 0069-0249-01
Ruxience®
(rituximab-pvvr)
Injection
500 mg/50 mL
(10 mg/mL)
For Intravenous Use
after dilution
ONE SINGLE-DOSE VIAL
DISCARD UNUSED PORTION.
Rx only
Pfizer Oncology
8.3 Females and Males of Reproductive Potential
Rituximab products can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
2.4 Recommended Dose As A Component of Zevalin® (2.4 Recommended Dose as a Component of Zevalin®)
When used as part of the Zevalin therapeutic regimen, infuse 250 mg/m2 in accordance with the Zevalin package insert. Refer to the Zevalin package insert for full prescribing information regarding the Zevalin therapeutic regimen.
2.7 Recommended Dose for Pemphigus Vulgaris (pv) (2.7 Recommended Dose for Pemphigus Vulgaris (PV))
-
•Administer RUXIENCE as two-1,000 mg intravenous infusions separated by 2 weeks in combination with a tapering course of glucocorticoids.
-
•Maintenance treatment
Administer RUXIENCE as a 500 mg intravenous infusion at Month 12 and every 6 months thereafter or based on clinical evaluation. -
•Treatment of relapse
Administer RUXIENCE as a 1,000 mg intravenous infusion on relapse, and consider resuming or increasing the glucocorticoid dose based on clinical evaluation.
Subsequent infusions of RUXIENCE may be administered no sooner than 16 weeks following the previous infusion.
2.5 Recommended Dose for Rheumatoid Arthritis (ra) (2.5 Recommended Dose for Rheumatoid Arthritis (RA))
-
•Administer RUXIENCE as two-1,000 mg intravenous infusions separated by 2 weeks.
-
•Glucocorticoids administered as methylprednisolone 100 mg intravenous or its equivalent 30 minutes prior to each infusion are recommended to reduce the incidence and severity of infusion-related reactions.
-
•Subsequent courses should be administered every 24 weeks or based on clinical evaluation, but not sooner than every 16 weeks.
-
•RUXIENCE is given in combination with methotrexate.
5.4 Progressive Multifocal Leukoencephalopathy (pml) (5.4 Progressive Multifocal Leukoencephalopathy (PML))
JC virus infection resulting in PML and death can occur in rituximab product-treated patients with hematologic malignancies or with autoimmune diseases. The majority of patients with hematologic malignancies diagnosed with PML received rituximab in combination with chemotherapy or as part of a hematopoietic stem cell transplant. The patients with autoimmune diseases had prior or concurrent immunosuppressive therapy. Most cases of PML were diagnosed within 12 months of their last infusion of rituximab.
Consider the diagnosis of PML in any patient presenting with new-onset neurologic manifestations. Evaluation of PML includes, but is not limited to, consultation with a neurologist, brain MRI, and lumbar puncture.
Discontinue RUXIENCE and consider discontinuation or reduction of any concomitant chemotherapy or immunosuppressive therapy in patients who develop PML.
2.2 Recommended Dose for Non Hodgkin's Lymphoma (nhl) (2.2 Recommended Dose for Non-Hodgkin's Lymphoma (NHL))
The recommended dose is 375 mg/m2 as an intravenous infusion according to the following schedules:
-
•Relapsed or Refractory, Low-Grade or Follicular, CD20-Positive, B-Cell NHL
Administer once weekly for 4 or 8 doses. -
•Retreatment for Relapsed or Refractory, Low-Grade or Follicular, CD20-Positive, B-Cell NHL
Administer once weekly for 4 doses. -
•Previously Untreated, Follicular, CD20-Positive, B-Cell NHL
Administer on Day 1 of each cycle of chemotherapy for up to 8 doses. In patients with complete or partial response, initiate RUXIENCE maintenance eight weeks following completion of a rituximab product in combination with chemotherapy. Administer RUXIENCE as a single agent every 8 weeks for 12 doses. -
•Non-progressing, Low-Grade, CD20-Positive, B-Cell NHL, after first-line CVP chemotherapy
Following completion of 6–8 cycles of CVP chemotherapy, administer once weekly for 4 doses at 6-month intervals to a maximum of 16 doses. -
•Diffuse Large B-Cell NHL
Administer on Day 1 of each cycle of chemotherapy for up to 8 infusions.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to establish the carcinogenic or mutagenic potential of rituximab products or to determine potential effects on fertility in males or females.
2.3 Recommended Dose for Chronic Lymphocytic Leukemia (cll) (2.3 Recommended Dose for Chronic Lymphocytic Leukemia (CLL))
The recommended dose is 375 mg/m2 the day prior to the initiation of FC chemotherapy, then 500 mg/m2 on Day 1 of Cycles 2–6 (every 28 days).
2.8 Recommended Dose for Premedication and Prophylactic Medications
Premedicate with acetaminophen and an antihistamine before each infusion of RUXIENCE. For adult patients administered RUXIENCE according to the 90-minute infusion rate, the glucocorticoid component of their chemotherapy regimen should be administered prior to infusion [see Clinical Studies (14.4)].
For RA, GPA and MPA, and PV patients, methylprednisolone 100 mg intravenously or its equivalent is recommended 30 minutes prior to each infusion.
Provide prophylaxis treatment for Pneumocystis jirovecii pneumonia (PCP) and herpes virus infections for patients with CLL during treatment and for up to 12 months following treatment as appropriate [see Warnings and Precautions (5.6)].
PCP prophylaxis is also recommended for patients with GPA and MPA during treatment and for at least 6 months following the last RUXIENCE infusion.
PCP prophylaxis should be considered for patients with PV during and following RUXIENCE treatment.
14.2 Previously Untreated, Low Grade Or Follicular, Cd20 Positive, B Cell Nhl (14.2 Previously Untreated, Low-Grade or Follicular, CD20-Positive, B-Cell NHL)
The safety and effectiveness of rituximab in previously untreated, low-grade or follicular, CD20+ NHL were demonstrated in 3 randomized, controlled trials enrolling 1,662 patients.
14.4 Ninety Minute Infusions in Previously Untreated Follicular Nhl and Dlbcl (14.4 Ninety-Minute Infusions in Previously Untreated Follicular NHL and DLBCL)
In NHL Study 10, a total of 363 patients with previously untreated follicular NHL (n=113) or DLBCL (n=250) were evaluated in a prospective, open-label, multicenter, single-arm trial for the safety of 90-minute rituximab infusions. Patients with follicular NHL received rituximab 375 mg/m2 plus CVP chemotherapy. Patients with DLBCL received rituximab 375 mg/m2 plus CHOP chemotherapy. Patients with clinically significant cardiovascular disease were excluded from the study. Patients were eligible for a 90-minute infusion at Cycle 2 if they did not experience a Grade 3–4 infusion-related adverse event with Cycle 1 and had a circulating lymphocyte count less than or equal to 5,000/mm3 before Cycle 2. All patients were pre-medicated with acetaminophen and an antihistamine and received the glucocorticoid component of their chemotherapy prior to rituximab infusion. The main outcome measure was the development of Grade 3–4 infusion-related reactions on the day of, or day after, the 90-minute infusion at Cycle 2 [see Adverse Reactions (6.1)].
Eligible patients received their Cycle 2 rituximab infusion over 90 minutes as follows: 20% of the total dose given in the first 30 minutes and the remaining 80% of the total dose given over the next 60 minutes [see Dosage and Administration (2.1)]. Patients who tolerated the 90-minute rituximab infusion at Cycle 2 continued to receive subsequent rituximab infusions at the 90-minute infusion rate for the remainder of the treatment regimen (through Cycle 6 or Cycle 8).
The incidence of Grade 3–4 infusion-related reactions at Cycle 2 was 1.1% (95% CI [0.3%, 2.8%]) among all patients, 3.5% (95% CI [1.0%, 8.8%]) for those patients treated with R-CVP, and 0.0% (95% CI [0.0%, 1.5%]) for those patients treated with R-CHOP. For Cycles 2–8, the incidence of Grade 3–4 infusion-related reactions was 2.8% (95% CI [1.3%, 5.0%]). No acute fatal infusion-related reactions were observed.
14.1 Relapsed Or Refractory, Low Grade Or Follicular, Cd20 Positive, B Cell Nhl (14.1 Relapsed or Refractory, Low-Grade or Follicular, CD20-Positive, B-Cell NHL)
The safety and effectiveness of rituximab in relapsed, refractory CD20+ NHL were demonstrated in 3 single-arm studies enrolling 296 patients.
1.4 Granulomatosis With Polyangiitis (gpa) (wegener's Granulomatosis) and Microscopic Polyangiitis (mpa) (1.4 Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA))
RUXIENCE, in combination with glucocorticoids, is indicated for the treatment of adult patients with Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA).
5.12 Concomitant Use With Other Biologic Agents and Dmards Other Than Methotrexate in Ra, Gpa and Mpa, Pv (5.12 Concomitant Use with Other Biologic Agents and DMARDS other than Methotrexate in RA, GPA and MPA, PV)
Limited data are available on the safety of the use of biologic agents or disease modifying anti-rheumatic drugs (DMARDs) other than methotrexate in RA patients exhibiting peripheral B-cell depletion following treatment with rituximab. Observe patients closely for signs of infection if biologic agents and/or DMARDs are used concomitantly. Use of concomitant immunosuppressants other than corticosteroids has not been studied in GPA or MPA or PV patients exhibiting peripheral B-cell depletion following treatment with rituximab products.
5.13 Use in Ra Patients Who Have Not Had Prior Inadequate Response to Tumor Necrosis Factor (tnf) Antagonists (5.13 Use in RA Patients Who Have Not Had Prior Inadequate Response to Tumor Necrosis Factor (TNF) Antagonists)
While the efficacy of rituximab was supported in four controlled trials in patients with RA with prior inadequate responses to non-biologic DMARDs, and in a controlled trial in MTX-naïve patients, a favorable risk-benefit relationship has not been established in these populations. The use of RUXIENCE in patients with RA who have not had prior inadequate response to one or more TNF antagonists is not recommended [see Clinical Studies (14.6)].
2.6 Recommended Dose for Granulomatosis With Polyangiitis (gpa) (wegener's Granulomatosis) and Microscopic Polyangiitis (mpa) (2.6 Recommended Dose for Granulomatosis with Polyangiitis (GPA) (Wegener's Granulomatosis) and Microscopic Polyangiitis (MPA))
Warning: Fatal Infusion Related Reactions, Severe Mucocutaneous Reactions, Hepatitis B Virus Reactivation and Progressive Multifocal Leukoencephalopathy (WARNING: FATAL INFUSION-RELATED REACTIONS, SEVERE MUCOCUTANEOUS REACTIONS, HEPATITIS B VIRUS REACTIVATION and PROGRESSIVE MULTIFOCAL LEUKOENCEPHALOPATHY)
Infusion-Related Reactions
Administration of rituximab products can result in serious, including fatal, infusion-related reactions. Deaths within 24 hours of rituximab infusion have occurred. Approximately 80% of fatal infusion-related reactions occurred in association with the first infusion. Monitor patients closely. Discontinue RUXIENCE infusion for severe reactions and provide medical treatment for Grade 3 or 4 infusion-related reactions [see Warnings and Precautions (5.1), Adverse Reactions (6.1)].
Severe Mucocutaneous Reactions
Severe, including fatal, mucocutaneous reactions can occur in patients receiving rituximab products [see Warnings and Precautions (5.2)].
Hepatitis B Virus (HBV) Reactivation
HBV reactivation can occur in patients treated with rituximab products, in some cases resulting in fulminant hepatitis, hepatic failure, and death. Screen all patients for HBV infection before treatment initiation, and monitor patients during and after treatment with RUXIENCE. Discontinue RUXIENCE and concomitant medications in the event of HBV reactivation [see Warnings and Precautions (5.3)].
Progressive Multifocal Leukoencephalopathy (PML)
Progressive Multifocal Leukoencephalopathy (PML), including fatal PML, can occur in patients receiving rituximab products [see Warnings and Precautions (5.4), Adverse Reactions (6.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:48.408418 · Updated: 2026-03-14T22:28:06.461439