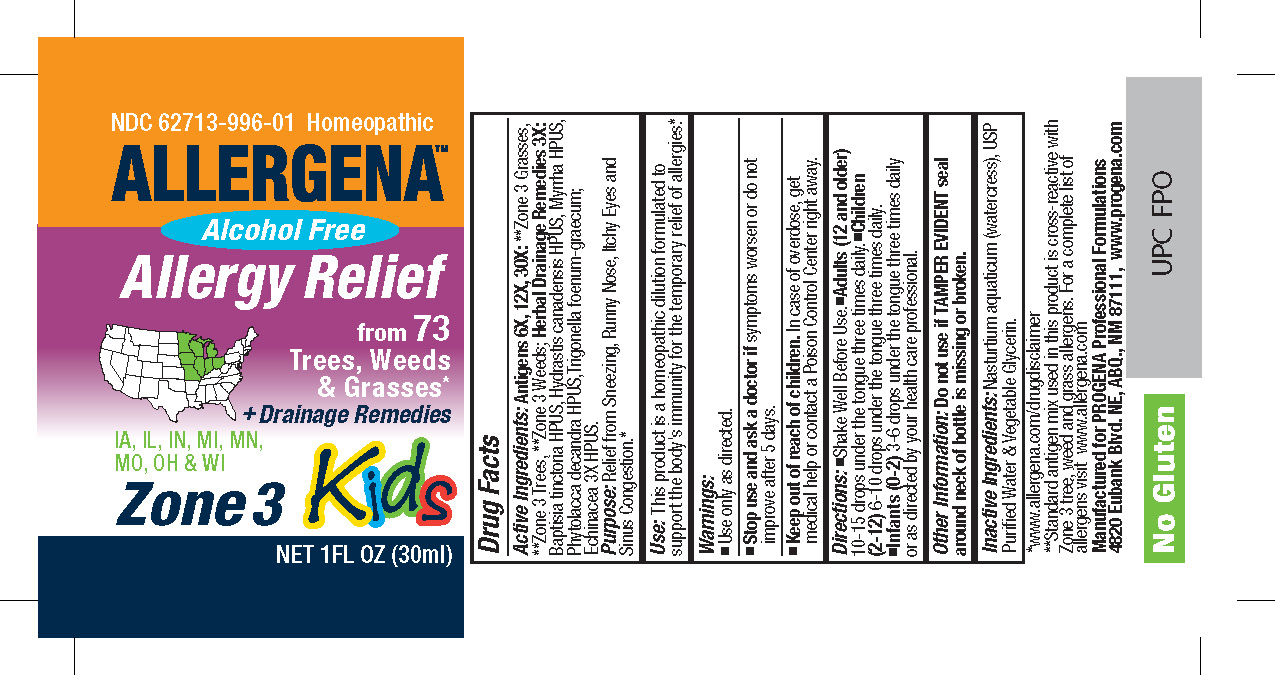
[{"name": "WATER", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "NASTURTIUM OFFICINALE", "unii": "YH89GMV676", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ECHINACEA, UNSPECIFIED", "unii": "4N9P6CC1DX", "classCode": "ACTIB", "strengthNumerator": "3", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "BAPTISIA TINCTORIA ROOT", "unii": "5EF0HWI5WU", "classCode": "ACTIB", "strengthNumerator": "3", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "GOLDENSEAL", "unii": "ZW3Z11D0JV", "classCode": "ACTIB", "strengthNumerator": "3", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "MYRRH", "unii": "JC71GJ1F3L", "classCode": "ACTIB", "strengthNumerator": "3", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "PHYTOLACCA AMERICANA ROOT", "unii": "11E6VI8VEG", "classCode": "ACTIB", "strengthNumerator": "3", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "FENUGREEK LEAF", "unii": "487RI96K8Z", "classCode": "ACTIB", "strengthNumerator": "3", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ALNUS INCANA SUBSP. RUGOSA POLLEN", "unii": "605T96G8Y5", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "FRAXINUS VELUTINA POLLEN", "unii": "LJT6I6Z8FD", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "FRAXINUS PENNSYLVANICA POLLEN", "unii": "2WZG2G15WX", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "FRAXINUS AMERICANA POLLEN", "unii": "G684LX721Q", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "POPULUS TREMULOIDES POLLEN", "unii": "928OC2TJDA", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "FAGUS GRANDIFOLIA POLLEN", "unii": "34X886W1H4", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "BETULA OCCIDENTALIS POLLEN", "unii": "R889N2L976", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "BETULA PAPYRIFERA POLLEN", "unii": "3538FNV8AY", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ACER NEGUNDO POLLEN", "unii": "P6K070AR8V", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "JUNIPERUS ASHEI POLLEN", "unii": "544F8MEY0Y", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "JUNIPERUS VIRGINIANA POLLEN", "unii": "PY0JA16R2G", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "POPULUS DELTOIDES POLLEN", "unii": "476DVV63WP", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ULMUS AMERICANA POLLEN", "unii": "89BAT511BD", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ULMUS PUMILA POLLEN", "unii": "030R993R8E", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "CELTIS OCCIDENTALIS POLLEN", "unii": "68R9X9Y96X", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "CARYA ALBA POLLEN", "unii": "G2A764T54B", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ACER SACCHARUM POLLEN", "unii": "V38QUQ7861", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "MORUS RUBRA POLLEN", "unii": "9LYI4RTZ52", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "MORUS ALBA POLLEN", "unii": "3I9T68187H", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "QUERCUS VELUTINA POLLEN", "unii": "294L626TT0", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "QUERCUS MACROCARPA POLLEN", "unii": "57BTU4547U", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "QUERCUS STELLATA POLLEN", "unii": "W34X0P8636", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "QUERCUS ALBA POLLEN", "unii": "Z4Y9ZSV4KK", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "CARYA ILLINOINENSIS POLLEN", "unii": "PYO4JR720Y", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "PINUS STROBUS POLLEN", "unii": "TX1ER5UV3T", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "POPULUS NIGRA POLLEN", "unii": "0MGE63QPFJ", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "POPULUS ALBA POLLEN", "unii": "VU8C8SB23P", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "LIGUSTRUM VULGARE POLLEN", "unii": "Y3FRX92Z0E", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "LIQUIDAMBAR STYRACIFLUA POLLEN", "unii": "5Q246DS5BS", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "PLATANUS OCCIDENTALIS POLLEN", "unii": "E03U1K03LK", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AILANTHUS ALTISSIMA POLLEN", "unii": "2A64U81OQ3", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "JUGLANS NIGRA POLLEN", "unii": "1BV28146ZR", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "SALIX NIGRA POLLEN", "unii": "6M2JIH93ZN", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMPHIACHYRIS DRACUNCULOIDES POLLEN", "unii": "83X1I1RR5F", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMARANTHUS PALMERI POLLEN", "unii": "1GH3WV23KH", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "XANTHIUM STRUMARIUM POLLEN", "unii": "2QOF601J1M", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "RUMEX CRISPUS POLLEN", "unii": "V825XJG64G", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "SOLIDAGO CANADENSIS POLLEN", "unii": "644CZ16IR5", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "CHENOPODIUM ALBUM POLLEN", "unii": "098LKX5NCN", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "IVA ANNUA POLLEN", "unii": "Y2U5S5PF22", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "CHENOPODIUM AMBROSIOIDES POLLEN", "unii": "WIB701MW2H", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "URTICA DIOICA POLLEN", "unii": "DNB59M1NVU", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMARANTHUS RETROFLEXUS POLLEN", "unii": "73B14PX5FW", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMARANTHUS SPINOSUS POLLEN", "unii": "380W4HYR6N", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "PLANTAGO LANCEOLATA POLLEN", "unii": "DO87T1U2CI", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMBROSIA TRIFIDA POLLEN", "unii": "KU1V1898XX", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMBROSIA ARTEMISIIFOLIA POLLEN", "unii": "K20Y81ACO3", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMBROSIA PSILOSTACHYA POLLEN", "unii": "RX18M46K8L", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ARTEMISIA TRIDENTATA POLLEN", "unii": "YI19RB8YFD", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ARTEMISIA LUDOVICIANA POLLEN", "unii": "57KIJ4772H", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "RUMEX ACETOSELLA POLLEN", "unii": "N52MIQ81ZW", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AMARANTHUS TUBERCULATUS POLLEN", "unii": "92N6W6KO2G", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ARTEMISIA ANNUA POLLEN", "unii": "36R82U4DL6", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ARTEMISIA ABSINTHIUM POLLEN", "unii": "81GS97HVFO", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "CYNODON DACTYLON POLLEN", "unii": "175F461W10", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "POA ANNUA POLLEN", "unii": "7U437HHU5C", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "BROMUS INERMIS POLLEN", "unii": "766QT72BK6", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "PHALARIS ARUNDINACEA POLLEN", "unii": "FAY1Y90VJ9", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "ZEA MAYS POLLEN", "unii": "74PD8J616H", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "SORGHUM HALEPENSE POLLEN", "unii": "577VA5B4HP", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "KOELERIA MACRANTHA POLLEN", "unii": "IIC6H3WF6J", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "POA PRATENSIS POLLEN", "unii": "SCB8J7LS3T", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "FESTUCA PRATENSIS POLLEN", "unii": "A0WFQ8P6N1", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "AVENA SATIVA POLLEN", "unii": "A7IKY24TR7", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "DACTYLIS GLOMERATA POLLEN", "unii": "83N78IDA7P", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "LOLIUM MULTIFLORUM POLLEN", "unii": "VJI0WKK736", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "LOLIUM PERENNE POLLEN", "unii": "4T81LB52R0", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "SORGHUM BICOLOR POLLEN", "unii": "LD795V73G4", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "TRITICUM AESTIVUM POLLEN", "unii": "F1KAH8374D", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}, {"name": "PASCOPYRUM SMITHII POLLEN", "unii": "6AU0ZD8T1O", "classCode": "ACTIB", "strengthNumerator": "30", "strengthDenominator": "1", "strengthNumeratorUnit": "[hp_X]", "strengthDenominatorUnit": "mL"}]