Drug Facts
f7f9b406-6108-480e-a6b6-03a356d40c43
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
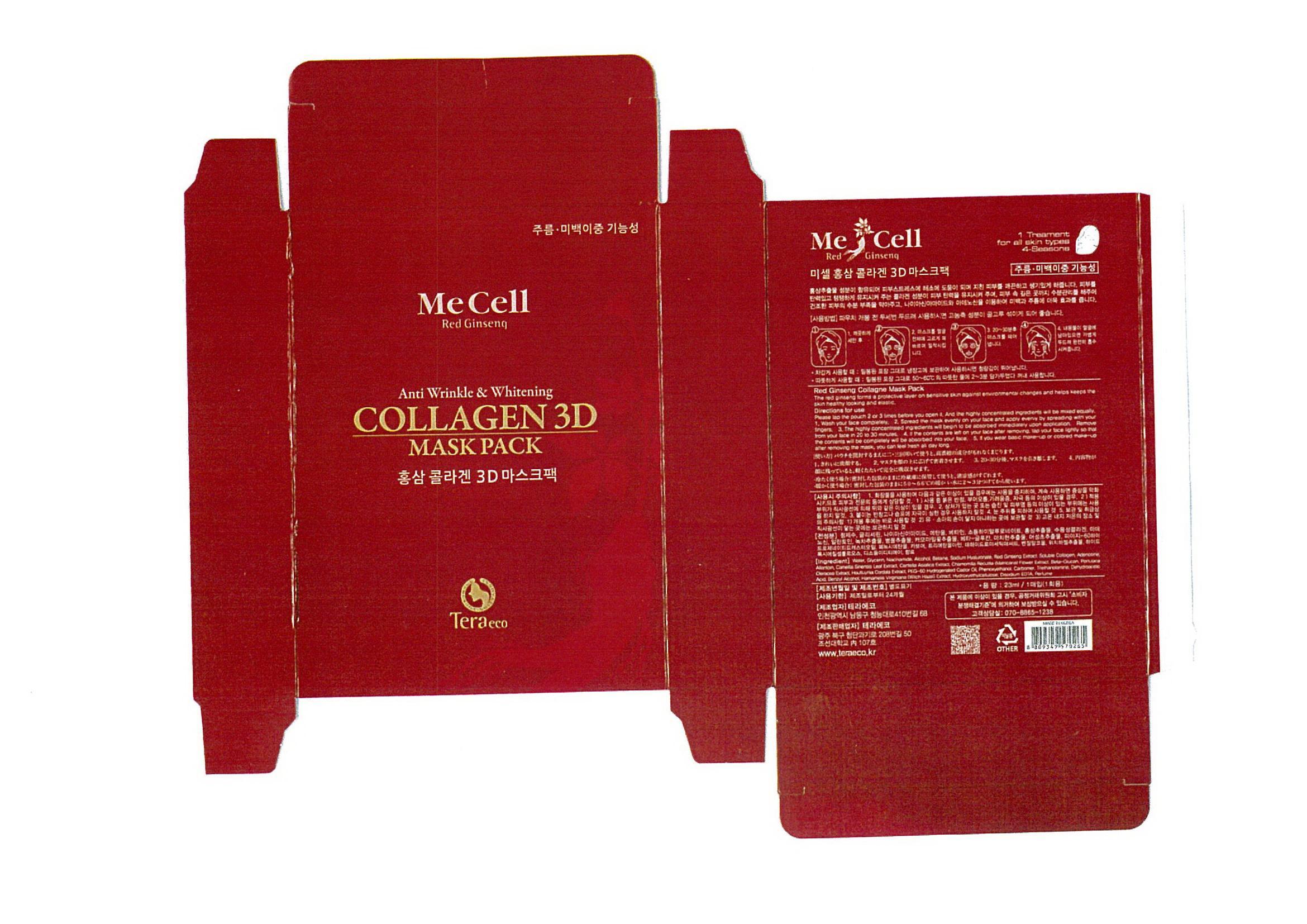
Description
glycerin
Medication Information
Warnings and Precautions
keep in a cool placedo not store in high temperature condition
Indications and Usage
apply to the skin and leave on for 20 to 30 minutes
Dosage and Administration
for external use only
Description
glycerin
Section 50565-1
keep out of reach of the children
Section 51727-6
water, niacinamide, adenosine, ethanol, betaine, sodium hyaluronate, etc
Section 51945-4
Section 55105-1
anti wrinkle
whitening
Section 55106-9
glycerin
Structured Label Content
Indications and Usage (34067-9)
apply to the skin and leave on for 20 to 30 minutes
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
keep in a cool placedo not store in high temperature condition
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
water, niacinamide, adenosine, ethanol, betaine, sodium hyaluronate, etc
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
anti wrinkle
whitening
Section 55106-9 (55106-9)
glycerin
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:52.977035 · Updated: 2026-03-14T22:53:54.250290