These Highlights Do Not Include All The Information Needed To Use Rhofade®
f7dbf12e-a802-4261-bae2-c20977698933
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
RHOFADE ® (oxymetazoline hydrochloride) cream, 1% is indicated for the topical treatment of persistent facial erythema associated with rosacea in adults.
Indications and Usage
RHOFADE ® (oxymetazoline hydrochloride) cream, 1% is indicated for the topical treatment of persistent facial erythema associated with rosacea in adults.
Dosage and Administration
For topical use only. RHOFADE cream is not for oral, ophthalmic, or intravaginal use. Apply a pea-sized amount of RHOFADE cream, once daily in a thin layer to cover the entire face (forehead, nose, each cheek, and chin) avoiding the eyes and lips. Wash hands immediately after applying RHOFADE cream.
Warnings and Precautions
Alpha-adrenergic agonists as a class may impact blood pressure. Advise patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension or hypotension to seek medical care if their condition worsens. ( 5.1 ) Use with caution in patients with cerebral or coronary insufficiency, Raynaud's phenomenon, thromboangiitis obliterans, scleroderma, or Sjögren's syndrome and advise patients to seek medical care if signs and symptoms of potentiation of vascular insufficiency develop. ( 5.2 ) Advise patients to seek immediate medical care if signs and symptoms of acute narrow-angle glaucoma develop. ( 5.3 )
Contraindications
None.
Adverse Reactions
Most common adverse reactions (incidence ≥ 1%) are application site dermatitis, worsening inflammatory lesions of rosacea, application site pruritis, application site erythema, and application site pain. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Mayne Pharma at 1-844-825-8500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
RHOFADE ® (oxymetazoline hydrochloride) cream, 1%, is a white to off-white cream. The product is available in a laminated tube in the following packaging configuration, each with a child-resistant closure: NDC 51862-765-30 30 gram tube
How Supplied
RHOFADE ® (oxymetazoline hydrochloride) cream, 1%, is a white to off-white cream. The product is available in a laminated tube in the following packaging configuration, each with a child-resistant closure: NDC 51862-765-30 30 gram tube
Medication Information
Warnings and Precautions
Alpha-adrenergic agonists as a class may impact blood pressure. Advise patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension or hypotension to seek medical care if their condition worsens. ( 5.1 ) Use with caution in patients with cerebral or coronary insufficiency, Raynaud's phenomenon, thromboangiitis obliterans, scleroderma, or Sjögren's syndrome and advise patients to seek medical care if signs and symptoms of potentiation of vascular insufficiency develop. ( 5.2 ) Advise patients to seek immediate medical care if signs and symptoms of acute narrow-angle glaucoma develop. ( 5.3 )
Indications and Usage
RHOFADE ® (oxymetazoline hydrochloride) cream, 1% is indicated for the topical treatment of persistent facial erythema associated with rosacea in adults.
Dosage and Administration
For topical use only. RHOFADE cream is not for oral, ophthalmic, or intravaginal use. Apply a pea-sized amount of RHOFADE cream, once daily in a thin layer to cover the entire face (forehead, nose, each cheek, and chin) avoiding the eyes and lips. Wash hands immediately after applying RHOFADE cream.
Contraindications
None.
Adverse Reactions
Most common adverse reactions (incidence ≥ 1%) are application site dermatitis, worsening inflammatory lesions of rosacea, application site pruritis, application site erythema, and application site pain. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Mayne Pharma at 1-844-825-8500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
RHOFADE ® (oxymetazoline hydrochloride) cream, 1%, is a white to off-white cream. The product is available in a laminated tube in the following packaging configuration, each with a child-resistant closure: NDC 51862-765-30 30 gram tube
How Supplied
RHOFADE ® (oxymetazoline hydrochloride) cream, 1%, is a white to off-white cream. The product is available in a laminated tube in the following packaging configuration, each with a child-resistant closure: NDC 51862-765-30 30 gram tube
Description
RHOFADE ® (oxymetazoline hydrochloride) cream, 1% is indicated for the topical treatment of persistent facial erythema associated with rosacea in adults.
Section 42229-5
Risk Summary
There are no available data on RHOFADE cream use in pregnant women to inform a drug-associated risk for major birth defects and miscarriage. A literature article describing intranasal decongestant use in pregnant women identified a potential association between second-trimester exposure to oxymetazoline (with no decongestant exposure in the first trimester) and renal collecting system anomalies [see Data]. In animal reproduction studies, there were no adverse developmental effects observed after oral administration of oxymetazoline hydrochloride in pregnant rats and rabbits at systemic exposures up to 3 times and 73 times, respectively, the exposure associated with the maximum recommended human dose (MRHD) [see Data]. The estimated background risks of major birth defects and miscarriage for the indicated population are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Section 42230-3
| PATIENT INFORMATION RHOFADE® (roe' fayd) (oxymetazoline hydrochloride) cream |
|---|
| Important: RHOFADE cream is for skin (topical) use on the face only. Do not use RHOFADE cream in your eyes, mouth, or vagina. |
| Keep RHOFADE cream out of the reach of children. |
| Get medical help right away if you, a child, or anyone else swallows RHOFADE cream. |
| What is RHOFADE cream? |
| RHOFADE cream is a prescription medicine used on the skin (topical) to treat facial redness due to rosacea that does not go away (persistent) in adults. |
| It is not known if RHOFADE cream is safe and effective in children under 18 years of age. |
Before you use RHOFADE cream, tell your healthcare provider about all of your medical conditions, including if you:
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, skin products, vitamins, and herbal supplements. Using RHOFADE cream with certain other medicines may affect each other and can cause serious side effects. |
How should I use RHOFADE cream?
|
| What are the possible side effects of RHOFADE cream? |
The most common side effects of RHOFADE cream include application site reactions of:
|
| These are not all the possible side effects of RHOFADE cream. |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store RHOFADE cream?
|
| Keep RHOFADE cream and all medicines out of the reach of children. |
| General information about the safe and effective use of RHOFADE cream |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use RHOFADE cream for a condition for which it was not prescribed. Do not give RHOFADE cream to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about RHOFADE cream that is written for health professionals. |
| What are the ingredients in RHOFADE cream? |
| Active ingredient: oxymetazoline hydrochloride |
| Inactive ingredients: sodium citrate dihydrate, citric acid anhydrous, disodium edetate dihydrate, butylated hydroxytoluene, anhydrous lanolin, medium chain triglycerides, diisopropyl adipate, oleyl alcohol, polyethylene glycol 300, PEG-6 stearate, glycol stearate, PEG-32 stearate, cetostearyl alcohol, ceteareth-6, stearyl alcohol, ceteareth-25, methylparaben, propylparaben, phenoxyethanol, and purified water |
| Distributed by: Mayne Pharma Raleigh, NC 27609 |
| RHOFADE and its design are registered trademarks of Mayne Pharma, LLC. |
| Patented. U.S. Patent Numbers: U.S. 7,812,049; U.S. 8,420,688; U.S. 8,815,929; U.S. 8,883,838; U.S. 9,974,773 and U.S. 10335391. Product of Germany. |
This Patient Information has been approved by the U.S. Food and Drug Administration
Revised 10/2023
Section 44425-7
Storage: Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30ºC (59°F- 86ºF) [see USP Controlled Room Temperature].
Section 59845-8
| INSTRUCTIONS FOR USE RHOFADE® (roe' fayd) (oxymetazoline hydrochloride) cream Tube |
|---|
Important:
|
Read and follow the steps below so that you use your tube of RHOFADE cream correctly:
|
Step 1: Open the tube of RHOFADE cream by gently pressing down on the child-resistant cap and twisting it counterclockwise until the cap is removed. Do not squeeze the tube while opening or closing. Note: When the cap is removed, the tube is not child-resistant. |
|
| Step 2: To apply RHOFADE cream to your face, squeeze a pea- sized amount of RHOFADE cream from the tube onto your fingertip. | |
Step 3: Apply the pea-sized amount of RHOFADE cream to cover your entire face (forehead, nose, each cheek, and chin) 1 time each day. Spread the cream smoothly and evenly in a thin layer over your face.
|
|
| Step 4: To close your RHOFADE cream tube, place the cap back on the tube. Press down on the child-resistant cap and twist clockwise until it stops. The tube is child-resistant again. | |
| Step 5: Wash your hands right away after applying RHOFADE cream. | |
How do I store RHOFADE cream?
|
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by:
Mayne Pharma
Raleigh, NC 27609
RHOFADE and its design are registered trademarks of Mayne Pharma, LLC
Patented. U.S. Patent Numbers: U.S. 7,812,049; U.S. 8,420,688; U.S. 8,815,929; U.S. 8,883,838; U.S. 9,974,773 and U.S. 10335391. Product of Germany.
Revised: 10/2023
10 Overdosage
RHOFADE cream is not for oral use. If oral ingestion occurs, seek medical advice. Monitor patient closely and administer appropriate supportive measures as necessary. Accidental ingestion of topical solutions (nasal sprays) containing imidazoline derivatives (e.g., oxymetazoline) in children has resulted in serious adverse events requiring hospitalization, including nausea, vomiting, lethargy, tachycardia, decreased respiration, bradycardia, hypotension, hypertension, sedation, somnolence, mydriasis, stupor, hypothermia, drooling, and coma. Keep RHOFADE cream out of reach of children.
8.2 Lactation
No clinical data are available to assess the effects of oxymetazoline on the quantity or rate of breastmilk production, or to establish the level of oxymetazoline present in human breastmilk post-dose. Oxymetazoline was detected in the milk of lactating rats. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for RHOFADE cream and any potential adverse effects on the breastfed child from RHOFADE cream or from the underlying maternal condition.
11 Description
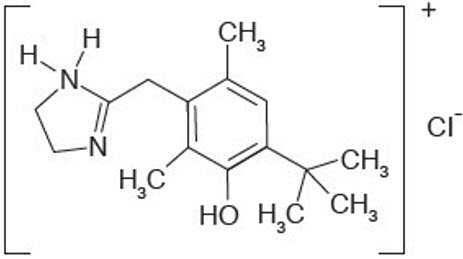
RHOFADE® (oxymetazoline hydrochloride) cream, 1% contains oxymetazoline hydrochloride, an alpha1A adrenoceptor agonist. RHOFADE is a white to off-white cream. It has a chemical name of 3-[(4,5-Dihydro1H-imidazol-2-yl)methyl]-6-(1,1-dimethylethyl)- 2,4-dimethyl-phenol hydrochloride and a molecular weight of 296.8. It is freely soluble in water and ethanol and has a partition coefficient of 0.1 in 1-octanol/water. The molecular formula of oxymetazoline HCl is C16H25ClN2O and its structural formula is:
Each gram of RHOFADE® (oxymetazoline hydrochloride) cream contains 10 mg (1%) oxymetazoline hydrochloride, equivalent to 8.8 mg (0.88%) of oxymetazoline free base. The cream contains the following inactive ingredients: sodium citrate dihydrate, citric acid anhydrous, disodium edetate dihydrate, butylated hydroxytoluene, anhydrous lanolin, medium chain triglycerides, diisopropyl adipate, oleyl alcohol, polyethylene glycol 300, PEG-6 stearate, glycol stearate, PEG-32 stearate, cetostearyl alcohol, ceteareth-6, stearyl alcohol, ceteareth-25, methylparaben, propylparaben, phenoxyethanol, and purified water.
8.4 Pediatric Use
Safety and effectiveness of RHOFADE cream have not been established in pediatric patients below the age of 18 years.
8.5 Geriatric Use
One hundred and ninety-three subjects aged 65 years and older received treatment with RHOFADE cream (n = 135) or vehicle (n = 58) in clinical trials. No overall differences in safety or effectiveness were observed between subjects ≥ 65 years of age and younger subjects, based on available data. Clinical studies of RHOFADE cream did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
14 Clinical Studies
RHOFADE cream was evaluated for the treatment of persistent erythema associated with rosacea in two identical, randomized, double-blind, vehicle-controlled, parallel-group clinical trials. The trials enrolled 885 subjects aged 18 years and older. Overall, 90% of subjects were Caucasian and 79% were female. Subjects applied either RHOFADE cream or vehicle once daily for 29 days.
Disease severity was graded by the clinician using a 5-point clinician erythema assessment (CEA) scale and by the subject on a similar 5-point subject self-assessment (SSA) scale, on which subjects scored either "moderate" or "severe" on both scales.
CEA and SSA were measured over a 12-hour period at equally-spaced timepoints (hours 3, 6, 9, and 12) post dose on Days 1, 15, and 29. The primary efficacy endpoint was defined as the proportion of subjects with at least a 2-grade reduction in erythema (improvement) from baseline (pre-dose on Day 1) on both the CEA and SSA measured at hours 3, 6, 9, and 12 on Day 29. The results from both trials on the composite endpoint for Day 29 are presented in Table 2.
| Time-point (Hour) |
Trial 1 | Trial 2 | ||
|---|---|---|---|---|
| RHOFADE Cream (N-222) |
Vehicle Cream (N-218) |
RHOFADE Cream (N-224) |
Vehicle Cream (N-221) |
|
| 3 | 12% | 6% | 14% | 7% |
| 6 | 16% | 8% | 13% | 5% |
| 9 | 18% | 6% | 16% | 9% |
| 12 | 15% | 6% | 12% | 6% |
4 Contraindications
None.
6 Adverse Reactions
Most common adverse reactions (incidence ≥ 1%) are application site dermatitis, worsening inflammatory lesions of rosacea, application site pruritis, application site erythema, and application site pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Mayne Pharma at 1-844-825-8500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
12.2 Pharmacodynamics
The pharmacodynamics of RHOFADE cream has not been studied.
1 Indications and Usage
RHOFADE® (oxymetazoline hydrochloride) cream, 1% is indicated for the topical treatment of persistent facial erythema associated with rosacea in adults.
12.1 Mechanism of Action
Oxymetazoline is an alpha1A adrenoceptor agonist. Oxymetazoline acts as a vasoconstrictor.
5 Warnings and Precautions
- Alpha-adrenergic agonists as a class may impact blood pressure. Advise patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension or hypotension to seek medical care if their condition worsens. (5.1)
- Use with caution in patients with cerebral or coronary insufficiency, Raynaud's phenomenon, thromboangiitis obliterans, scleroderma, or Sjögren's syndrome and advise patients to seek medical care if signs and symptoms of potentiation of vascular insufficiency develop. (5.2)
- Advise patients to seek immediate medical care if signs and symptoms of acute narrow-angle glaucoma develop. (5.3)
2 Dosage and Administration
For topical use only. RHOFADE cream is not for oral, ophthalmic, or intravaginal use.
Apply a pea-sized amount of RHOFADE cream, once daily in a thin layer to cover the entire face (forehead, nose, each cheek, and chin) avoiding the eyes and lips. Wash hands immediately after applying RHOFADE cream.
3 Dosage Forms and Strengths
RHOFADE® (oxymetazoline hydrochloride) cream, 1% is a white to off-white cream. Each gram of cream contains 10 mg (1%) oxymetazoline hydrochloride, equivalent to 8.8 mg (0.88%) of oxymetazoline free base.
6.1 Clinical Studies Experience
Because clinical trials are conducted under varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 489 subjects with persistent facial erythema associated with rosacea were treated with RHOFADE cream once daily for 4 weeks in 3 controlled clinical trials. An additional 440 subjects with persistent facial erythema associated with rosacea were also treated with RHOFADE cream once daily for up to one year in a long-term (open- label) clinical trial. Adverse reactions that occurred in at least 1% of subjects treated with RHOFADE cream through 4 weeks of treatment are presented in Table 1 below.
| Adverse Reaction | Pooled Controlled Clinical Trials | |
|---|---|---|
| RHOFADE Cream (N = 489) |
Vehicle Cream (N = 483) |
|
| Application site dermatitis | 9 (2%) | 0 |
| Worsening inflammatory lesions of rosacea | 7 (1%) | 1 (<1%) |
| Application site pruritus | 5 (1%) | 4 (1%) |
| Application site erythema | 5 (1%) | 2 (<1%) |
| Application site pain | 4 (1%) | 1 (<1%) |
In the long-term (open-label) clinical trial, the rates of adverse reactions over a one-year treatment period were as follows: worsening inflammatory lesions of rosacea (3%), application site dermatitis (3%), application site pruritis (2%), application site pain (2%), and application site erythema (2%). Subjects with persistent erythema along with inflammatory lesions were allowed to use additional therapy for the inflammatory lesions of rosacea.
7.2 Monoamine Oxidase Inhibitors
Caution is advised in patients taking MAO inhibitors which can affect the metabolism and uptake of circulating amines.
17 Patient Counseling Information
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Important Administration Instructions
Advise patients of the following:
- RHOFADE cream is for topical use only.
- Do not apply RHOFADE cream to irritated skin or open wounds.\
- Avoid contact with the eyes and lips.
- Wash hands immediately after application.
- Keep RHOFADE cream out of reach of children.
5.3 Risk of Angle Closure Glaucoma
RHOFADE cream may increase the risk of angle closure glaucoma in patients with narrow-angle glaucoma. Advise patients to seek immediate medical care if signs and symptoms of acute angle closure glaucoma develop.
16 How Supplied/storage and Handling
RHOFADE® (oxymetazoline hydrochloride) cream, 1%, is a white to off-white cream. The product is available in a laminated tube in the following packaging configuration, each with a child-resistant closure:
| NDC 51862-765-30 | 30 gram tube |
Principal Display Panel 3g Tube Carton
NDC 51862-765-00
Rhofade®
(oxymetazoline
hydrochloride) cream, 1%*
*Each gram of RHOFADE® cream contains 10 mg
of oxymetazoline hydrochloride, equivalent to
8.8 mg of oxymetazoline free base
For Topical Use Only
Keep Out of Reach of Children
Rx Only
15 tubes - 3g each
mayne pharma
7.1 Anti Hypertensives/cardiac Glycosides
Alpha-adrenergic agonists, as a class, may impact blood pressure. Caution in using drugs such as beta-blockers, anti-hypertensives and/or cardiac glycosides is advised.
Caution should also be exercised in patients receiving alpha1 adrenergic receptor antagonists such as in the treatment of cardiovascular disease, benign prostatic hypertrophy, or Raynaud's disease.
5.2 Potentiation of Vascular Insufficiency
RHOFADE cream should be used with caution in patients with cerebral or coronary insufficiency, Raynaud's phenomenon, thromboangitis obliterans, scleroderma, or Sjögren's syndrome. Advise patients to seek immediate medical care if signs and symptoms of potentiation of vascular insufficiency develop.
Principal Display Panel 30 G Tube Carton
NDC 51862-765-30
Rhofade®
(oxymetazoline
hydrochloride) cream, 1%*
*Each gram of RHOFADE® cream
contains 10 mg of oxymetazoline
hydrochloride, equivalent to
8.8 mg of oxymetazoline free base
For Topical Use Only
Keep Out of Reach of Children
Rx Only
30 g
mayne pharma
5.1 Potential Impacts On Cardiovascular Disease
Alpha-adrenergic agonists may impact blood pressure. RHOFADE cream should be used with caution in patients with severe or unstable or uncontrolled cardiovascular disease, orthostatic hypotension, and uncontrolled hypertension or hypotension. Advise patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension/hypotension to seek immediate medical care if their condition worsens.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Oxymetazoline hydrochloride was not associated with an increased incidence of neoplastic or proliferative changes in transgenic mice given oral doses of 0.5, 1.0, or 2.5 mg/kg/day oxymetazoline hydrochloride for 6 months.
Oxymetazoline hydrochloride revealed no evidence of mutagenic or clastogenic potential based on the results of two in vitro genotoxicity tests (Ames assay and human lymphocyte chromosomal aberration assay) and one in vivo genotoxicity test (mouse micronucleus assay).
Effects on fertility and early embryonic development were evaluated in rats following oral administration of 0.05, 0.1, or 0.2 mg/kg/day oxymetazoline hydrochloride prior to and during mating and through early pregnancy. Decreased number of corpora lutea and increased post-implantation losses were noted at 0.2 mg/kg/day oxymetazoline hydrochloride (3 times the MRHD on an AUC comparison basis). However, no treatment related effects on fertility or mating parameters were noted at 0.2 mg/kg/day oxymetazoline hydrochloride (3 times the MRHD on an AUC comparison basis).
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are no available data on RHOFADE cream use in pregnant women to inform a drug-associated risk for major birth defects and miscarriage. A literature article describing intranasal decongestant use in pregnant women identified a potential association between second-trimester exposure to oxymetazoline (with no decongestant exposure in the first trimester) and renal collecting system anomalies [see Data]. In animal reproduction studies, there were no adverse developmental effects observed after oral administration of oxymetazoline hydrochloride in pregnant rats and rabbits at systemic exposures up to 3 times and 73 times, respectively, the exposure associated with the maximum recommended human dose (MRHD) [see Data]. The estimated background risks of major birth defects and miscarriage for the indicated population are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Section 42230-3 (42230-3)
| PATIENT INFORMATION RHOFADE® (roe' fayd) (oxymetazoline hydrochloride) cream |
|---|
| Important: RHOFADE cream is for skin (topical) use on the face only. Do not use RHOFADE cream in your eyes, mouth, or vagina. |
| Keep RHOFADE cream out of the reach of children. |
| Get medical help right away if you, a child, or anyone else swallows RHOFADE cream. |
| What is RHOFADE cream? |
| RHOFADE cream is a prescription medicine used on the skin (topical) to treat facial redness due to rosacea that does not go away (persistent) in adults. |
| It is not known if RHOFADE cream is safe and effective in children under 18 years of age. |
Before you use RHOFADE cream, tell your healthcare provider about all of your medical conditions, including if you:
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, skin products, vitamins, and herbal supplements. Using RHOFADE cream with certain other medicines may affect each other and can cause serious side effects. |
How should I use RHOFADE cream?
|
| What are the possible side effects of RHOFADE cream? |
The most common side effects of RHOFADE cream include application site reactions of:
|
| These are not all the possible side effects of RHOFADE cream. |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store RHOFADE cream?
|
| Keep RHOFADE cream and all medicines out of the reach of children. |
| General information about the safe and effective use of RHOFADE cream |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use RHOFADE cream for a condition for which it was not prescribed. Do not give RHOFADE cream to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about RHOFADE cream that is written for health professionals. |
| What are the ingredients in RHOFADE cream? |
| Active ingredient: oxymetazoline hydrochloride |
| Inactive ingredients: sodium citrate dihydrate, citric acid anhydrous, disodium edetate dihydrate, butylated hydroxytoluene, anhydrous lanolin, medium chain triglycerides, diisopropyl adipate, oleyl alcohol, polyethylene glycol 300, PEG-6 stearate, glycol stearate, PEG-32 stearate, cetostearyl alcohol, ceteareth-6, stearyl alcohol, ceteareth-25, methylparaben, propylparaben, phenoxyethanol, and purified water |
| Distributed by: Mayne Pharma Raleigh, NC 27609 |
| RHOFADE and its design are registered trademarks of Mayne Pharma, LLC. |
| Patented. U.S. Patent Numbers: U.S. 7,812,049; U.S. 8,420,688; U.S. 8,815,929; U.S. 8,883,838; U.S. 9,974,773 and U.S. 10335391. Product of Germany. |
This Patient Information has been approved by the U.S. Food and Drug Administration
Revised 10/2023
Section 44425-7 (44425-7)
Storage: Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30ºC (59°F- 86ºF) [see USP Controlled Room Temperature].
Section 59845-8 (59845-8)
| INSTRUCTIONS FOR USE RHOFADE® (roe' fayd) (oxymetazoline hydrochloride) cream Tube |
|---|
Important:
|
Read and follow the steps below so that you use your tube of RHOFADE cream correctly:
|
Step 1: Open the tube of RHOFADE cream by gently pressing down on the child-resistant cap and twisting it counterclockwise until the cap is removed. Do not squeeze the tube while opening or closing. Note: When the cap is removed, the tube is not child-resistant. |
|
| Step 2: To apply RHOFADE cream to your face, squeeze a pea- sized amount of RHOFADE cream from the tube onto your fingertip. | |
Step 3: Apply the pea-sized amount of RHOFADE cream to cover your entire face (forehead, nose, each cheek, and chin) 1 time each day. Spread the cream smoothly and evenly in a thin layer over your face.
|
|
| Step 4: To close your RHOFADE cream tube, place the cap back on the tube. Press down on the child-resistant cap and twist clockwise until it stops. The tube is child-resistant again. | |
| Step 5: Wash your hands right away after applying RHOFADE cream. | |
How do I store RHOFADE cream?
|
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by:
Mayne Pharma
Raleigh, NC 27609
RHOFADE and its design are registered trademarks of Mayne Pharma, LLC
Patented. U.S. Patent Numbers: U.S. 7,812,049; U.S. 8,420,688; U.S. 8,815,929; U.S. 8,883,838; U.S. 9,974,773 and U.S. 10335391. Product of Germany.
Revised: 10/2023
10 Overdosage (10 OVERDOSAGE)
RHOFADE cream is not for oral use. If oral ingestion occurs, seek medical advice. Monitor patient closely and administer appropriate supportive measures as necessary. Accidental ingestion of topical solutions (nasal sprays) containing imidazoline derivatives (e.g., oxymetazoline) in children has resulted in serious adverse events requiring hospitalization, including nausea, vomiting, lethargy, tachycardia, decreased respiration, bradycardia, hypotension, hypertension, sedation, somnolence, mydriasis, stupor, hypothermia, drooling, and coma. Keep RHOFADE cream out of reach of children.
8.2 Lactation
No clinical data are available to assess the effects of oxymetazoline on the quantity or rate of breastmilk production, or to establish the level of oxymetazoline present in human breastmilk post-dose. Oxymetazoline was detected in the milk of lactating rats. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for RHOFADE cream and any potential adverse effects on the breastfed child from RHOFADE cream or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
RHOFADE® (oxymetazoline hydrochloride) cream, 1% contains oxymetazoline hydrochloride, an alpha1A adrenoceptor agonist. RHOFADE is a white to off-white cream. It has a chemical name of 3-[(4,5-Dihydro1H-imidazol-2-yl)methyl]-6-(1,1-dimethylethyl)- 2,4-dimethyl-phenol hydrochloride and a molecular weight of 296.8. It is freely soluble in water and ethanol and has a partition coefficient of 0.1 in 1-octanol/water. The molecular formula of oxymetazoline HCl is C16H25ClN2O and its structural formula is:
Each gram of RHOFADE® (oxymetazoline hydrochloride) cream contains 10 mg (1%) oxymetazoline hydrochloride, equivalent to 8.8 mg (0.88%) of oxymetazoline free base. The cream contains the following inactive ingredients: sodium citrate dihydrate, citric acid anhydrous, disodium edetate dihydrate, butylated hydroxytoluene, anhydrous lanolin, medium chain triglycerides, diisopropyl adipate, oleyl alcohol, polyethylene glycol 300, PEG-6 stearate, glycol stearate, PEG-32 stearate, cetostearyl alcohol, ceteareth-6, stearyl alcohol, ceteareth-25, methylparaben, propylparaben, phenoxyethanol, and purified water.
8.4 Pediatric Use
Safety and effectiveness of RHOFADE cream have not been established in pediatric patients below the age of 18 years.
8.5 Geriatric Use
One hundred and ninety-three subjects aged 65 years and older received treatment with RHOFADE cream (n = 135) or vehicle (n = 58) in clinical trials. No overall differences in safety or effectiveness were observed between subjects ≥ 65 years of age and younger subjects, based on available data. Clinical studies of RHOFADE cream did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
14 Clinical Studies (14 CLINICAL STUDIES)
RHOFADE cream was evaluated for the treatment of persistent erythema associated with rosacea in two identical, randomized, double-blind, vehicle-controlled, parallel-group clinical trials. The trials enrolled 885 subjects aged 18 years and older. Overall, 90% of subjects were Caucasian and 79% were female. Subjects applied either RHOFADE cream or vehicle once daily for 29 days.
Disease severity was graded by the clinician using a 5-point clinician erythema assessment (CEA) scale and by the subject on a similar 5-point subject self-assessment (SSA) scale, on which subjects scored either "moderate" or "severe" on both scales.
CEA and SSA were measured over a 12-hour period at equally-spaced timepoints (hours 3, 6, 9, and 12) post dose on Days 1, 15, and 29. The primary efficacy endpoint was defined as the proportion of subjects with at least a 2-grade reduction in erythema (improvement) from baseline (pre-dose on Day 1) on both the CEA and SSA measured at hours 3, 6, 9, and 12 on Day 29. The results from both trials on the composite endpoint for Day 29 are presented in Table 2.
| Time-point (Hour) |
Trial 1 | Trial 2 | ||
|---|---|---|---|---|
| RHOFADE Cream (N-222) |
Vehicle Cream (N-218) |
RHOFADE Cream (N-224) |
Vehicle Cream (N-221) |
|
| 3 | 12% | 6% | 14% | 7% |
| 6 | 16% | 8% | 13% | 5% |
| 9 | 18% | 6% | 16% | 9% |
| 12 | 15% | 6% | 12% | 6% |
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (incidence ≥ 1%) are application site dermatitis, worsening inflammatory lesions of rosacea, application site pruritis, application site erythema, and application site pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Mayne Pharma at 1-844-825-8500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
12.2 Pharmacodynamics
The pharmacodynamics of RHOFADE cream has not been studied.
1 Indications and Usage (1 INDICATIONS AND USAGE)
RHOFADE® (oxymetazoline hydrochloride) cream, 1% is indicated for the topical treatment of persistent facial erythema associated with rosacea in adults.
12.1 Mechanism of Action
Oxymetazoline is an alpha1A adrenoceptor agonist. Oxymetazoline acts as a vasoconstrictor.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Alpha-adrenergic agonists as a class may impact blood pressure. Advise patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension or hypotension to seek medical care if their condition worsens. (5.1)
- Use with caution in patients with cerebral or coronary insufficiency, Raynaud's phenomenon, thromboangiitis obliterans, scleroderma, or Sjögren's syndrome and advise patients to seek medical care if signs and symptoms of potentiation of vascular insufficiency develop. (5.2)
- Advise patients to seek immediate medical care if signs and symptoms of acute narrow-angle glaucoma develop. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For topical use only. RHOFADE cream is not for oral, ophthalmic, or intravaginal use.
Apply a pea-sized amount of RHOFADE cream, once daily in a thin layer to cover the entire face (forehead, nose, each cheek, and chin) avoiding the eyes and lips. Wash hands immediately after applying RHOFADE cream.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
RHOFADE® (oxymetazoline hydrochloride) cream, 1% is a white to off-white cream. Each gram of cream contains 10 mg (1%) oxymetazoline hydrochloride, equivalent to 8.8 mg (0.88%) of oxymetazoline free base.
6.1 Clinical Studies Experience
Because clinical trials are conducted under varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 489 subjects with persistent facial erythema associated with rosacea were treated with RHOFADE cream once daily for 4 weeks in 3 controlled clinical trials. An additional 440 subjects with persistent facial erythema associated with rosacea were also treated with RHOFADE cream once daily for up to one year in a long-term (open- label) clinical trial. Adverse reactions that occurred in at least 1% of subjects treated with RHOFADE cream through 4 weeks of treatment are presented in Table 1 below.
| Adverse Reaction | Pooled Controlled Clinical Trials | |
|---|---|---|
| RHOFADE Cream (N = 489) |
Vehicle Cream (N = 483) |
|
| Application site dermatitis | 9 (2%) | 0 |
| Worsening inflammatory lesions of rosacea | 7 (1%) | 1 (<1%) |
| Application site pruritus | 5 (1%) | 4 (1%) |
| Application site erythema | 5 (1%) | 2 (<1%) |
| Application site pain | 4 (1%) | 1 (<1%) |
In the long-term (open-label) clinical trial, the rates of adverse reactions over a one-year treatment period were as follows: worsening inflammatory lesions of rosacea (3%), application site dermatitis (3%), application site pruritis (2%), application site pain (2%), and application site erythema (2%). Subjects with persistent erythema along with inflammatory lesions were allowed to use additional therapy for the inflammatory lesions of rosacea.
7.2 Monoamine Oxidase Inhibitors
Caution is advised in patients taking MAO inhibitors which can affect the metabolism and uptake of circulating amines.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Important Administration Instructions
Advise patients of the following:
- RHOFADE cream is for topical use only.
- Do not apply RHOFADE cream to irritated skin or open wounds.\
- Avoid contact with the eyes and lips.
- Wash hands immediately after application.
- Keep RHOFADE cream out of reach of children.
5.3 Risk of Angle Closure Glaucoma
RHOFADE cream may increase the risk of angle closure glaucoma in patients with narrow-angle glaucoma. Advise patients to seek immediate medical care if signs and symptoms of acute angle closure glaucoma develop.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
RHOFADE® (oxymetazoline hydrochloride) cream, 1%, is a white to off-white cream. The product is available in a laminated tube in the following packaging configuration, each with a child-resistant closure:
| NDC 51862-765-30 | 30 gram tube |
Principal Display Panel 3g Tube Carton (PRINCIPAL DISPLAY PANEL - 3g Tube Carton)
NDC 51862-765-00
Rhofade®
(oxymetazoline
hydrochloride) cream, 1%*
*Each gram of RHOFADE® cream contains 10 mg
of oxymetazoline hydrochloride, equivalent to
8.8 mg of oxymetazoline free base
For Topical Use Only
Keep Out of Reach of Children
Rx Only
15 tubes - 3g each
mayne pharma
7.1 Anti Hypertensives/cardiac Glycosides (7.1 Anti-hypertensives/Cardiac Glycosides)
Alpha-adrenergic agonists, as a class, may impact blood pressure. Caution in using drugs such as beta-blockers, anti-hypertensives and/or cardiac glycosides is advised.
Caution should also be exercised in patients receiving alpha1 adrenergic receptor antagonists such as in the treatment of cardiovascular disease, benign prostatic hypertrophy, or Raynaud's disease.
5.2 Potentiation of Vascular Insufficiency
RHOFADE cream should be used with caution in patients with cerebral or coronary insufficiency, Raynaud's phenomenon, thromboangitis obliterans, scleroderma, or Sjögren's syndrome. Advise patients to seek immediate medical care if signs and symptoms of potentiation of vascular insufficiency develop.
Principal Display Panel 30 G Tube Carton (PRINCIPAL DISPLAY PANEL - 30 g Tube Carton)
NDC 51862-765-30
Rhofade®
(oxymetazoline
hydrochloride) cream, 1%*
*Each gram of RHOFADE® cream
contains 10 mg of oxymetazoline
hydrochloride, equivalent to
8.8 mg of oxymetazoline free base
For Topical Use Only
Keep Out of Reach of Children
Rx Only
30 g
mayne pharma
5.1 Potential Impacts On Cardiovascular Disease (5.1 Potential Impacts on Cardiovascular Disease)
Alpha-adrenergic agonists may impact blood pressure. RHOFADE cream should be used with caution in patients with severe or unstable or uncontrolled cardiovascular disease, orthostatic hypotension, and uncontrolled hypertension or hypotension. Advise patients with cardiovascular disease, orthostatic hypotension, and/or uncontrolled hypertension/hypotension to seek immediate medical care if their condition worsens.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Oxymetazoline hydrochloride was not associated with an increased incidence of neoplastic or proliferative changes in transgenic mice given oral doses of 0.5, 1.0, or 2.5 mg/kg/day oxymetazoline hydrochloride for 6 months.
Oxymetazoline hydrochloride revealed no evidence of mutagenic or clastogenic potential based on the results of two in vitro genotoxicity tests (Ames assay and human lymphocyte chromosomal aberration assay) and one in vivo genotoxicity test (mouse micronucleus assay).
Effects on fertility and early embryonic development were evaluated in rats following oral administration of 0.05, 0.1, or 0.2 mg/kg/day oxymetazoline hydrochloride prior to and during mating and through early pregnancy. Decreased number of corpora lutea and increased post-implantation losses were noted at 0.2 mg/kg/day oxymetazoline hydrochloride (3 times the MRHD on an AUC comparison basis). However, no treatment related effects on fertility or mating parameters were noted at 0.2 mg/kg/day oxymetazoline hydrochloride (3 times the MRHD on an AUC comparison basis).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:06.161146 · Updated: 2026-03-14T22:29:01.799450