These Highlights Do Not Include All The Information Needed To Use Methylphenidate Hydrochloride Extended-release Capsules Safely And Effectively. See Full Prescribing Information For Methylphenidate Hydrochloride Extended-release Capsules
f7a7ffd1-bc30-4db8-8391-dddf76acd639
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10) ] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection. Before prescribing methylphenidate hydrochloride extended-release capsules, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout methylphenidate hydrochloride extended-release capsules treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2) ] .
Indications and Usage
Methylphenidate hydrochloride extended-release capsules are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD ) in patients 6 years and older [see Clinical Studies (14) ].
Dosage and Administration
Recommended starting dose for patients 6 years and older: 10 mg once daily with or without food in the morning. Dosage may be increased weekly in increments of 10 mg per day. Daily dosage above 60 mg is not recommended. ( 2.1 ) Capsules may be swallowed whole or opened and the entire contents sprinkled onto applesauce. ( 2.1 )
Warnings and Precautions
Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. ( 5.2 ) Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse. ( 5.3 ) Psychiatric Adverse Reactions: Prior to initiating methylphenidate hydrochloride extended-release capsules, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing methylphenidate hydrochloride extended-release capsules. ( 5.4 ) Priapism: If abnormally sustained or frequent and painful erections occur, patients should seek immediate medical attention. ( 5.5 ) Peripheral Vasculopathy, including Raynaud's Phenomenon: Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release capsules treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. ( 5.6 ) Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. ( 5.7 ) Acute Angle Closure Glaucoma: Methylphenidate hydrochloride extended-release capsules-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist. ( 5.8 ) Increased Intraocular Pressure (IOP) and Glaucoma: Prescribe methylphenidate hydrochloride extended-release capsules to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor patients with a history of increased IOP or open angle glaucoma. ( 5.9 ) Motor and Verbal Tics, and Worsening of Tourette's Syndrome: Before initiating methylphenidate hydrochloride extended-release capsules, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette's syndrome. Discontinue treatment if clinically appropriate. ( 5.10 )
Contraindications
Hypersensitivity to methylphenidate or other components of the product. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.1) ]. Concomitant treatment with monoamine oxidase inhibitors, and also within 14 days following discontinuation of treatment with a monoamine oxidase inhibitor, because of the risk of hypertensive crisis [see Drug Interactions (7.1) ] .
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Abuse, Misuse, and Addiction [see Boxed Warning , Warnings and Precautions (5.1) , and Drug Abuse and Dependence (9.2 , 9.3) ] Hypersensitivity to Methylphenidate [see Contraindications (4) ] Hypertensive Crisis with Concomitant Use of Monoamine Oxidase Inhibitors [see Contraindications (4) and Drug Interactions (7.1) ] Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2) ] Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3) ] Psychiatric Adverse Reactions [see Warnings and Precautions (5.4) ] Priapism [see Warnings and Precautions (5.5) ] Peripheral Vasculopathy, including Raynaud's Phenomenon [see Warnings and Precautions (5.6) ] Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.7) ] Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8) ] Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9) ] Motor and Verbal Tics, and Worsening of Tourette's Syndrome [see Warnings and Precautions (5.10) ]
Drug Interactions
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. ( 7 )
Storage and Handling
Methylphenidate Hydrochloride Extended-Release Capsules are available as follows: 10 mg Extended-Release Capsules - white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting "910". Cap imprinting "RP" Bottles of 90 NDC 42858-075-45 15 mg Extended-Release Capsules - white opaque body and orange cap containing white spherical beads. Body imprinting "915". Cap imprinting "RP" Bottles of 90 NDC 42858-076-45 20 mg Extended-Release Capsules - white opaque body and yellow opaque cap containing white spherical beads. Body imprinting "920". Cap imprinting "RP" Bottles of 90 NDC 42858-077-45 30 mg Extended-Release Capsules - white opaque body and blue violet cap containing white spherical beads. Body imprinting "930". Cap imprinting "RP" Bottles of 90 NDC 42858-078-45 40 mg Extended-Release Capsules - white opaque body and pink opaque cap containing white spherical beads. Body imprinting "940". Cap imprinting "RP" Bottles of 90 NDC 42858-079-45 50 mg Extended-Release Capsules - white opaque body and green opaque cap containing white spherical beads. Body imprinting "950". Cap Imprinting "RP" Bottles of 90 NDC 42858-080-45 60 mg Extended-Release Capsules - white opaque body and gray opaque cap containing white spherical beads. Body imprinting "960". Cap imprinting "RP" Bottles of 90 NDC 42858-081-45
How Supplied
Methylphenidate Hydrochloride Extended-Release Capsules are available as follows: 10 mg Extended-Release Capsules - white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting "910". Cap imprinting "RP" Bottles of 90 NDC 42858-075-45 15 mg Extended-Release Capsules - white opaque body and orange cap containing white spherical beads. Body imprinting "915". Cap imprinting "RP" Bottles of 90 NDC 42858-076-45 20 mg Extended-Release Capsules - white opaque body and yellow opaque cap containing white spherical beads. Body imprinting "920". Cap imprinting "RP" Bottles of 90 NDC 42858-077-45 30 mg Extended-Release Capsules - white opaque body and blue violet cap containing white spherical beads. Body imprinting "930". Cap imprinting "RP" Bottles of 90 NDC 42858-078-45 40 mg Extended-Release Capsules - white opaque body and pink opaque cap containing white spherical beads. Body imprinting "940". Cap imprinting "RP" Bottles of 90 NDC 42858-079-45 50 mg Extended-Release Capsules - white opaque body and green opaque cap containing white spherical beads. Body imprinting "950". Cap Imprinting "RP" Bottles of 90 NDC 42858-080-45 60 mg Extended-Release Capsules - white opaque body and gray opaque cap containing white spherical beads. Body imprinting "960". Cap imprinting "RP" Bottles of 90 NDC 42858-081-45
Medication Information
Warnings and Precautions
Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. ( 5.2 ) Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse. ( 5.3 ) Psychiatric Adverse Reactions: Prior to initiating methylphenidate hydrochloride extended-release capsules, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing methylphenidate hydrochloride extended-release capsules. ( 5.4 ) Priapism: If abnormally sustained or frequent and painful erections occur, patients should seek immediate medical attention. ( 5.5 ) Peripheral Vasculopathy, including Raynaud's Phenomenon: Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release capsules treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. ( 5.6 ) Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. ( 5.7 ) Acute Angle Closure Glaucoma: Methylphenidate hydrochloride extended-release capsules-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist. ( 5.8 ) Increased Intraocular Pressure (IOP) and Glaucoma: Prescribe methylphenidate hydrochloride extended-release capsules to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor patients with a history of increased IOP or open angle glaucoma. ( 5.9 ) Motor and Verbal Tics, and Worsening of Tourette's Syndrome: Before initiating methylphenidate hydrochloride extended-release capsules, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette's syndrome. Discontinue treatment if clinically appropriate. ( 5.10 )
Indications and Usage
Methylphenidate hydrochloride extended-release capsules are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD ) in patients 6 years and older [see Clinical Studies (14) ].
Dosage and Administration
Recommended starting dose for patients 6 years and older: 10 mg once daily with or without food in the morning. Dosage may be increased weekly in increments of 10 mg per day. Daily dosage above 60 mg is not recommended. ( 2.1 ) Capsules may be swallowed whole or opened and the entire contents sprinkled onto applesauce. ( 2.1 )
Contraindications
Hypersensitivity to methylphenidate or other components of the product. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.1) ]. Concomitant treatment with monoamine oxidase inhibitors, and also within 14 days following discontinuation of treatment with a monoamine oxidase inhibitor, because of the risk of hypertensive crisis [see Drug Interactions (7.1) ] .
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Abuse, Misuse, and Addiction [see Boxed Warning , Warnings and Precautions (5.1) , and Drug Abuse and Dependence (9.2 , 9.3) ] Hypersensitivity to Methylphenidate [see Contraindications (4) ] Hypertensive Crisis with Concomitant Use of Monoamine Oxidase Inhibitors [see Contraindications (4) and Drug Interactions (7.1) ] Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2) ] Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3) ] Psychiatric Adverse Reactions [see Warnings and Precautions (5.4) ] Priapism [see Warnings and Precautions (5.5) ] Peripheral Vasculopathy, including Raynaud's Phenomenon [see Warnings and Precautions (5.6) ] Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.7) ] Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8) ] Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9) ] Motor and Verbal Tics, and Worsening of Tourette's Syndrome [see Warnings and Precautions (5.10) ]
Drug Interactions
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. ( 7 )
Storage and Handling
Methylphenidate Hydrochloride Extended-Release Capsules are available as follows: 10 mg Extended-Release Capsules - white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting "910". Cap imprinting "RP" Bottles of 90 NDC 42858-075-45 15 mg Extended-Release Capsules - white opaque body and orange cap containing white spherical beads. Body imprinting "915". Cap imprinting "RP" Bottles of 90 NDC 42858-076-45 20 mg Extended-Release Capsules - white opaque body and yellow opaque cap containing white spherical beads. Body imprinting "920". Cap imprinting "RP" Bottles of 90 NDC 42858-077-45 30 mg Extended-Release Capsules - white opaque body and blue violet cap containing white spherical beads. Body imprinting "930". Cap imprinting "RP" Bottles of 90 NDC 42858-078-45 40 mg Extended-Release Capsules - white opaque body and pink opaque cap containing white spherical beads. Body imprinting "940". Cap imprinting "RP" Bottles of 90 NDC 42858-079-45 50 mg Extended-Release Capsules - white opaque body and green opaque cap containing white spherical beads. Body imprinting "950". Cap Imprinting "RP" Bottles of 90 NDC 42858-080-45 60 mg Extended-Release Capsules - white opaque body and gray opaque cap containing white spherical beads. Body imprinting "960". Cap imprinting "RP" Bottles of 90 NDC 42858-081-45
How Supplied
Methylphenidate Hydrochloride Extended-Release Capsules are available as follows: 10 mg Extended-Release Capsules - white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting "910". Cap imprinting "RP" Bottles of 90 NDC 42858-075-45 15 mg Extended-Release Capsules - white opaque body and orange cap containing white spherical beads. Body imprinting "915". Cap imprinting "RP" Bottles of 90 NDC 42858-076-45 20 mg Extended-Release Capsules - white opaque body and yellow opaque cap containing white spherical beads. Body imprinting "920". Cap imprinting "RP" Bottles of 90 NDC 42858-077-45 30 mg Extended-Release Capsules - white opaque body and blue violet cap containing white spherical beads. Body imprinting "930". Cap imprinting "RP" Bottles of 90 NDC 42858-078-45 40 mg Extended-Release Capsules - white opaque body and pink opaque cap containing white spherical beads. Body imprinting "940". Cap imprinting "RP" Bottles of 90 NDC 42858-079-45 50 mg Extended-Release Capsules - white opaque body and green opaque cap containing white spherical beads. Body imprinting "950". Cap Imprinting "RP" Bottles of 90 NDC 42858-080-45 60 mg Extended-Release Capsules - white opaque body and gray opaque cap containing white spherical beads. Body imprinting "960". Cap imprinting "RP" Bottles of 90 NDC 42858-081-45
Description
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10) ] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection. Before prescribing methylphenidate hydrochloride extended-release capsules, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout methylphenidate hydrochloride extended-release capsules treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2) ] .
Section 42229-5
Limitations of Use
The use of methylphenidate hydrochloride extended-release capsules is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions(e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7), Use in Specific Populations (8.4)].
Section 42231-1
| MEDICATION GUIDE Methylphenidate (METH-il-FEN-i-date) Hydrochloride Extended-Release Capsules, CII |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 09/2025 |
|
What is the most important information I should know about methylphenidate hydrochloride extended-release capsules? |
|
| Methylphenidate hydrochloride extended-release capsules may cause serious side effects, including: | |
|
|
| What are methylphenidate hydrochloride extended-release capsules? | |
| Methylphenidate hydrochloride extended-release capsules are a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in people 6 years of age and older. Methylphenidate hydrochloride extended-release capsules may help increase attention and decrease impulsiveness and hyperactivity in people with ADHD. | |
|
|
| Do not take methylphenidate hydrochloride extended-release capsules if you or your child are: | |
|
|
| Before taking methylphenidate hydrochloride extended-release capsules tell your healthcare provider about all medical conditions, including if you or your child: | |
|
|
| Tell your healthcare provider about all the medicines that you or your child take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
| Methylphenidate hydrochloride extended-release capsules and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be changed during treatment with methylphenidate hydrochloride extended-release capsules. Your healthcare provider will decide whether methylphenidate hydrochloride extended-release capsules can be taken with other medicines. | |
| Especially tell your healthcare provider if you or your child take a medicine used to treat depression called monoamine oxidase inhibitor (MAOI). | |
| Know the medicines that you or your child take. Keep a list of the medicines with you to show your healthcare provider and pharmacist. Do not start any new medicine during treatment with methylphenidate hydrochloride extended-release capsules without talking to your healthcare provider first. | |
| How should methylphenidate hydrochloride extended-release capsules be taken? | |
|
|
| What should be avoided during treatment with methylphenidate hydrochloride extended-release capsules? | |
| Avoid drinking alcohol during treatment with methylphenidate hydrochloride extended-release capsules. This may cause a faster release of the methylphenidate hydrochloride extended-release capsules medicine. | |
| What are possible side effects of methylphenidate hydrochloride extended-release capsules? | |
| Methylphenidate hydrochloride extended-release capsules may cause serious side effects, including: | |
| See "What is the most important information I should know about methylphenidate hydrochloride extended-release capsules?" | |
|
|
| The most common side effects of methylphenidate hydrochloride extended-release capsules in children 6 to 17 years of age include stomach pain, decreased appetite, headache, trouble sleeping. | |
| These are not all the possible side effects of methylphenidate hydrochloride extended-release capsules. | |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |
| You may also report side effects to Rhodes Pharmaceuticals at 1-888-827-0616. | |
| How should I store methylphenidate hydrochloride extended-release capsules? | |
|
|
| Keep methylphenidate hydrochloride extended-release capsules and all medicines out of the reach of children. | |
| General information about the safe and effective use of methylphenidate hydrochloride extended-release capsules. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use methylphenidate hydrochloride extended-release capsules for a condition for which it was not prescribed. Do not give methylphenidate hydrochloride extended-release capsules to other people, even if they have the same symptoms. It may harm them and it is against the law. You can ask your healthcare provider or pharmacist for information about methylphenidate hydrochloride extended-release capsules that is written for healthcare professionals. | |
| What are the ingredients in Methylphenidate Hydrochloride Extended-Release Capsules? | |
| Active Ingredient: methylphenidate hydrochloride | |
| Inactive Ingredients: ammonio methacrylate copolymer, type B; colloidal silicon dioxide (added if necessary); gelatin; hypromelloses; methacrylic acid copolymer, type C; polyethylene glycol; sugar spheres; talc; titanium oxide; and triethyl citrate. | |
| Manufactured by: Purdue Pharma L.P., Stamford, CT 06901 | |
| For more information, call Rhodes Pharmaceuticals (the distributor for methylphenidate hydrochloride extended-release capsules) at 1-888-827-0616. |
Section 44425-7
Storage and Handling
Methylphenidate Hydrochloride Extended-Release Capsules should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Dispense in tight container (USP).
9.2 Abuse
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction [see Warnings and Precautions (5.1)]. Methylphenidate hydrochloride extended-release capsules can be diverted for non-medical use into illicit channels or distribution.
Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of methylphenidate may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate use, in both adult and pediatric male patients. Although priapism was not reported with methylphenidate initiation, it developed after some time on methylphenidate, often subsequent to an increase in dosage. Priapism also occured during methylphenidate withdrawal (drug holidays or during discontinuation).
Methylphenidate hydrochloride extended-release capsules patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
11 Description
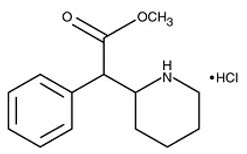
Methylphenidate hydrochloride extended-release capsules contain methylphenidate hydrochloride, a central nervous system (CNS) stimulant. Methylphenidate hydrochloride extended-release capsules contain multi layered beads, which are composed of an immediate-release layer which contains approximately 40% of the methylphenidate dose, and a controlled release layer which contains approximately 60% of the methylphenidate dose. Methylphenidate hydrochloride extended-release capsules are available in seven capsule strengths. Each extended-release capsule for once-a-day oral administration contains 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, 50 mg, or 60 mg of methylphenidate HCl USP, which is equivalent to 8.6 mg, 13.0 mg, 17.3 mg, 25.9 mg, 34.6 mg, 43.2 mg, or 51.9 mg of methylphenidate free base, respectively. Chemically, methylphenidate HCl is d,l (racemic) methyl α-phenyl-2-piperidineacetate hydrochloride. Its molecular formula is C14H19NO2∙HCl. Its structural formula is:
Methylphenidate hydrochloride USP is a white to off-white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone. Its molecular weight is 269.77.
Inactive Ingredients: ammonio methacrylate copolymer, type B; colloidal silicon dioxide (added if necessary); gelatin; hypromelloses; methacrylic acid copolymer, type C; polyethylene glycol; sugar spheres; talc; titanium oxide; and triethyl citrate.
Each strength capsule also contains colorant ingredients in the capsule shell as follows:
10 mg: FD&C Blue No. 1
15 mg: D&C Red No. 28, D&C Yellow No. 10, FD&C Red No. 40
20 mg: D&C Red No. 33, D&C Yellow No. 10
30 mg: FD&C Blue No. 1, FD&C Red No. 3
40 mg: D&C Red No. 28, FD&C Blue No. 1, FD&C Red No. 40
50 mg: D&C Yellow No. 10, FD&C Green No. 3
60 mg: Black Iron Oxide
8.4 Pediatric Use
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules have not been established in pediatric patients below the age of 6 years.
Safety and efficacy of methylphenidate hydrochloride extended-release capsules were evaluated in a multicenter, placebo-controlled, double-blind, parallel group study in 119 children 4 to <6 years of age with ADHD followed by a 12-month open-label extension in 44 of these children. In these studies, patients experienced high rates of adverse reactions, most notably weight loss. Comparing weights prior to initiation of methylphenidate hydrochloride extended-release capsules (in the safety and efficacy study) to weights after 12 months of treatment (in the open-label extension), 20 of 39 patients with data (50%) had lost enough weight to decrease 10 or more percentiles on a Centers for Disease Control growth chart for weight. In addition, systemic drug exposures in patients 4 to <6 years of age were higher than those observed in older children and adolescents at the same dose (2 to 3 fold higher Cmax and AUC). Therefore, the benefits of methylphenidate hydrochloride extended-release capsules do not outweigh the risks in pediatric patients 4 to <6 years of age.
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules have been established in pediatric patients ages 6 to 17 years in two adequate and well-controlled clinical trials [see Clinical Studies (14)].
8.5 Geriatric Use
Clinical trials of methylphenidate hydrochloride extended-release capsules did not include any patients aged 65 years and over. In general, dose selection for an elderly patient start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
14 Clinical Studies
The efficacy of methylphenidate hydrochloride extended-release capsules for the treatment of ADHD was established in a randomized, double-blind, single center, placebo-controlled, flexible-dose, cross-over trial in pediatric patients aged 6 to 12 years and a second randomized, double-blind, multicenter, placebo-controlled, fixed–dose trial in pediatric patients 6 to 17 years.
4 Contraindications
- Hypersensitivity to methylphenidate or other components of the product. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.1)].
- Concomitant treatment with monoamine oxidase inhibitors, and also within 14 days following discontinuation of treatment with a monoamine oxidase inhibitor, because of the risk of hypertensive crisis [see Drug Interactions (7.1)].
6 Adverse Reactions
The following are discussed in more detail in other sections of the labeling:
- Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1), and Drug Abuse and Dependence (9.2, 9.3)]
- Hypersensitivity to Methylphenidate [see Contraindications (4)]
- Hypertensive Crisis with Concomitant Use of Monoamine Oxidase Inhibitors [see Contraindications (4) and Drug Interactions (7.1)]
- Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2)]
- Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral Vasculopathy, including Raynaud's Phenomenon [see Warnings and Precautions (5.6)]
- Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.7)]
- Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8)]
- Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9)]
- Motor and Verbal Tics, and Worsening of Tourette's Syndrome [see Warnings and Precautions (5.10)]
7 Drug Interactions
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. (7)
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d- and l-isomers. The d-isomer is more pharmacologically active than the l-isomer. Methylphenidate blocks the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
2.2 Recommended Dosage
The recommended starting dose of methylphenidate hydrochloride extended-release capsules in patients 6 years and older is 10 mg once daily orally in the morning with or without food. Advise patients to establish a routine pattern with regard to meals. The dose should be individualized according to the needs and response of the patient.
The dose may be titrated weekly in increments of 10 mg. Daily doses above 60 mg have not been studied and are not recommended.
1 Indications and Usage
Methylphenidate hydrochloride extended-release capsules are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients 6 years and older [see Clinical Studies (14)].
12.1 Mechanism of Action
Methylphenidate HCl is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
Methylphenidate hydrochloride extended-release capsules contains methylphenidate a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to treating patients with methylphenidate hydrochloride extended-release capsules, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions 5.2].
- the family history and clinically evaluate patients for motor or verbal tics or Tourette's syndrome before initiating methylphenidate hydrochloride extended-release capsules [see Warnings and Precautions (5.10)].
5 Warnings and Precautions
- Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. (5.2)
- Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse. (5.3)
- Psychiatric Adverse Reactions: Prior to initiating methylphenidate hydrochloride extended-release capsules, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing methylphenidate hydrochloride extended-release capsules. (5.4)
- Priapism: If abnormally sustained or frequent and painful erections occur, patients should seek immediate medical attention. (5.5)
- Peripheral Vasculopathy, including Raynaud's Phenomenon: Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release capsules treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. (5.6)
- Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. (5.7)
- Acute Angle Closure Glaucoma: Methylphenidate hydrochloride extended-release capsules-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist. (5.8)
- Increased Intraocular Pressure (IOP) and Glaucoma: Prescribe methylphenidate hydrochloride extended-release capsules to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor patients with a history of increased IOP or open angle glaucoma. (5.9)
- Motor and Verbal Tics, and Worsening of Tourette's Syndrome: Before initiating methylphenidate hydrochloride extended-release capsules, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette's syndrome. Discontinue treatment if clinically appropriate. (5.10)
2 Dosage and Administration
- Recommended starting dose for patients 6 years and older: 10 mg once daily with or without food in the morning. Dosage may be increased weekly in increments of 10 mg per day. Daily dosage above 60 mg is not recommended. (2.1)
- Capsules may be swallowed whole or opened and the entire contents sprinkled onto applesauce. (2.1)
3 Dosage Forms and Strengths
- 10 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting: 910. Cap imprinting: RP
- 15 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body and orange cap containing white spherical beads. Body imprinting: 915. Cap imprinting: RP
- 20 mg Extended-Release Capsules Hard Gelatin Capsules, white opaque body and yellow opaque cap containing white spherical beads. Body imprinting: 920. Cap imprinting: RP
- 30 mg Extended-Release Capsules Hard Gelatin Capsules, white opaque body and blue violet cap containing white spherical beads. Body imprinting: 930. Cap imprinting: RP
- 40 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body and pink opaque cap containing white spherical beads. Body imprinting: 940. Cap imprinting: RP
- 50 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body and green opaque cap containing white spherical beads. Body imprinting: 950. Cap Imprinting: RP
- 60 mg Extended-Release Capsules Hard Gelatin Capsules, white opaque body and gray opaque cap containing white spherical beads. Body imprinting: 960. Cap imprinting: RP
6.2 Post Marketing Experience
The following adverse reactions have been identified during post approval use of methylphenidate products. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These adverse reactions are as follows:
Blood and Lymphatic System Disorders: Pancytopenia, Thrombocytopenia, Thrombocytopenic purpura
Cardiac Disorders: Angina pectoris, Bradycardia, Extrasystole, Supraventricular tachycardia, Ventricular extrasystole
Eye Disorders: Diplopia, Increased intraocular pressure, Mydriasis, Visual impairment
General Disorders: Chest pain, Chest discomfort, Hyperpyrexia
Immune System Disorders: Hypersensitivity reactions such as Angioedema, Anaphylactic reactions, Auricular swelling, Bullous conditions, Exfoliative conditions, Urticarias, Pruritus NEC, Rashes, Eruptions, and Exanthems NEC
Investigations: Alkaline phosphatase increased, Bilirubin increased, Hepatic enzyme increased, Platelet count decreased, White blood cell count abnormal, severe hepatic injury
Musculoskeletal, Connective Tissue and Bone Disorders: Arthralgia, Myalgia, Muscle twitching, Rhabdomyolysis
Nervous System: Convulsion, Grand mal convulsion, Dyskinesia, serotonin syndrome in combination with serotonergic drugs, Motor and Verbal Tics
Psychiatric Disorders: Disorientation, Libido changes
Skin and Subcutaneous Tissue Disorders: Alopecia, Erythema
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
2.3 Administration Instructions
Methylphenidate hydrochloride extended-release capsules may be taken whole or the capsule may be opened and the entire contents sprinkled onto applesauce. If the patient is using the sprinkled administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
5.1 Abuse, Misuse, and Addiction
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse. The use of methylphenidate hydrochloride extended-release capsules exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Methylphenidate hydrochloride extended-release capsules can be diverted for non-medical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2)]. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride extended-release capsules, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store methylphenidate hydrochloride extended-release capsules in a safe place, preferably locked, and instruct patients to not give methylphenidate hydrochloride extended-release capsules to anyone else. Throughout methylphenidate hydrochloride extended-release capsules treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
5.8 Acute Angle Closure Glaucoma
There have been reports of angle closure glaucoma associated with methylphenidate treatment.
Although the mechanism is not clear, methylphenidate hydrochloride extended-release capsules-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
Methylphenidate Hydrochloride Extended-Release Capsules are available as follows:
| 10 mg Extended-Release Capsules - white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting "910". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-075-45 |
| 15 mg Extended-Release Capsules - white opaque body and orange cap containing white spherical beads. Body imprinting "915". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-076-45 |
| 20 mg Extended-Release Capsules - white opaque body and yellow opaque cap containing white spherical beads. Body imprinting "920". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-077-45 |
| 30 mg Extended-Release Capsules - white opaque body and blue violet cap containing white spherical beads. Body imprinting "930". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-078-45 |
| 40 mg Extended-Release Capsules - white opaque body and pink opaque cap containing white spherical beads. Body imprinting "940". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-079-45 |
| 50 mg Extended-Release Capsules - white opaque body and green opaque cap containing white spherical beads. Body imprinting "950". Cap Imprinting "RP" | |
| Bottles of 90 | NDC 42858-080-45 |
| 60 mg Extended-Release Capsules - white opaque body and gray opaque cap containing white spherical beads. Body imprinting "960". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-081-45 |
Warning: Abuse, Misuse, and Addiction
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride extended-release capsules, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout methylphenidate hydrochloride extended-release capsules treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2)].
2.4 Dosage Reduction and Discontinuation
If paradoxical aggravation of symptoms or other adverse reactions occur; the dosage should be reduced, or, if necessary, discontinue methylphenidate hydrochloride extended-release capsules.
If improvement is not observed after appropriate dosage adjustment over a one-month period, discontinue methylphenidate hydrochloride extended-release capsules.
5.3 Increased Blood Pressure and Heart Rate
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm). Some patients may have larger increases.
Monitor all methylphenidate hydrochloride extended-release capsules-treated patients for hypertension and tachycardia.
5.9 Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment [see Adverse Reactions (6.2)].
Prescribe methylphenidate hydrochloride extended-release capsules to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor methylphenidate hydrochloride extended-release capsules-treated patients with a history of abnormally increased IOP or open angle glaucoma.
5.2 Risks to Patients With Serious Cardiac Disease
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosage.
Avoid methylphenidate hydrochloride extended-release capsules use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart arrhythmia, coronary artery disease, or other serious cardiac disease.
Principal Display Panel 10 Mg Capsule Bottle Label
NDC 42858-075-45
CII
Methylphenidate
HCl Extended-Release
Capsules
10 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 15 Mg Capsule Bottle Label
NDC 42858-076-45
CII
Methylphenidate
HCl Extended-Release
Capsules
15 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 20 Mg Capsule Bottle Label
NDC 42858-077-45
CII
Methylphenidate
HCl Extended-Release
Capsules
20 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 30 Mg Capsule Bottle Label
NDC 42858-078-45
CII
Methylphenidate
HCl Extended-Release
Capsules
30 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 40 Mg Capsule Bottle Label
NDC 42858-079-45
CII
Methylphenidate
HCl Extended-Release
Capsules
40 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 50 Mg Capsule Bottle Label
NDC 42858-080-45
CII
Methylphenidate
HCl Extended-Release
Capsules
50 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 60 Mg Capsule Bottle Label
NDC 42858-081-45
CII
Methylphenidate
HCl Extended-Release
Capsules
60 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
5.7 Long Term Suppression of Growth in Pediatric Patients
Methylphenidate hydrochloride extended-release capsules are not approved for use and are not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated pediatric patients over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period.
Closely monitor growth (weight and height) in methylphenidate hydrochloride extended-release capsules-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon
CNS stimulants, including methylphenidate hydrochloride extended-release capsules, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports and at the therapeutic dosages of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release capsules treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for methylphenidate hydrochloride extended-release capsules-treated patients who develop signs or symptoms of peripheral vasculopathy.
5.10 Motor and Verbal Tics, and Worsening of Tourette's Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette's syndrome has also been reported [see Adverse Reactions (6.2)].
Before initiating methylphenidate hydrochloride extended-release capsules, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor methylphenidate hydrochloride extended-release capsules-treated patients for the emergence or worsening of tics or Tourette's syndrome, and discontinue treatment if clinically appropriate.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
The use of methylphenidate hydrochloride extended-release capsules is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions(e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7), Use in Specific Populations (8.4)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE Methylphenidate (METH-il-FEN-i-date) Hydrochloride Extended-Release Capsules, CII |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 09/2025 |
|
What is the most important information I should know about methylphenidate hydrochloride extended-release capsules? |
|
| Methylphenidate hydrochloride extended-release capsules may cause serious side effects, including: | |
|
|
| What are methylphenidate hydrochloride extended-release capsules? | |
| Methylphenidate hydrochloride extended-release capsules are a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in people 6 years of age and older. Methylphenidate hydrochloride extended-release capsules may help increase attention and decrease impulsiveness and hyperactivity in people with ADHD. | |
|
|
| Do not take methylphenidate hydrochloride extended-release capsules if you or your child are: | |
|
|
| Before taking methylphenidate hydrochloride extended-release capsules tell your healthcare provider about all medical conditions, including if you or your child: | |
|
|
| Tell your healthcare provider about all the medicines that you or your child take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
| Methylphenidate hydrochloride extended-release capsules and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be changed during treatment with methylphenidate hydrochloride extended-release capsules. Your healthcare provider will decide whether methylphenidate hydrochloride extended-release capsules can be taken with other medicines. | |
| Especially tell your healthcare provider if you or your child take a medicine used to treat depression called monoamine oxidase inhibitor (MAOI). | |
| Know the medicines that you or your child take. Keep a list of the medicines with you to show your healthcare provider and pharmacist. Do not start any new medicine during treatment with methylphenidate hydrochloride extended-release capsules without talking to your healthcare provider first. | |
| How should methylphenidate hydrochloride extended-release capsules be taken? | |
|
|
| What should be avoided during treatment with methylphenidate hydrochloride extended-release capsules? | |
| Avoid drinking alcohol during treatment with methylphenidate hydrochloride extended-release capsules. This may cause a faster release of the methylphenidate hydrochloride extended-release capsules medicine. | |
| What are possible side effects of methylphenidate hydrochloride extended-release capsules? | |
| Methylphenidate hydrochloride extended-release capsules may cause serious side effects, including: | |
| See "What is the most important information I should know about methylphenidate hydrochloride extended-release capsules?" | |
|
|
| The most common side effects of methylphenidate hydrochloride extended-release capsules in children 6 to 17 years of age include stomach pain, decreased appetite, headache, trouble sleeping. | |
| These are not all the possible side effects of methylphenidate hydrochloride extended-release capsules. | |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |
| You may also report side effects to Rhodes Pharmaceuticals at 1-888-827-0616. | |
| How should I store methylphenidate hydrochloride extended-release capsules? | |
|
|
| Keep methylphenidate hydrochloride extended-release capsules and all medicines out of the reach of children. | |
| General information about the safe and effective use of methylphenidate hydrochloride extended-release capsules. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use methylphenidate hydrochloride extended-release capsules for a condition for which it was not prescribed. Do not give methylphenidate hydrochloride extended-release capsules to other people, even if they have the same symptoms. It may harm them and it is against the law. You can ask your healthcare provider or pharmacist for information about methylphenidate hydrochloride extended-release capsules that is written for healthcare professionals. | |
| What are the ingredients in Methylphenidate Hydrochloride Extended-Release Capsules? | |
| Active Ingredient: methylphenidate hydrochloride | |
| Inactive Ingredients: ammonio methacrylate copolymer, type B; colloidal silicon dioxide (added if necessary); gelatin; hypromelloses; methacrylic acid copolymer, type C; polyethylene glycol; sugar spheres; talc; titanium oxide; and triethyl citrate. | |
| Manufactured by: Purdue Pharma L.P., Stamford, CT 06901 | |
| For more information, call Rhodes Pharmaceuticals (the distributor for methylphenidate hydrochloride extended-release capsules) at 1-888-827-0616. |
Section 44425-7 (44425-7)
Storage and Handling
Methylphenidate Hydrochloride Extended-Release Capsules should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Dispense in tight container (USP).
9.2 Abuse
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse which can lead to the development of a substance use disorder, including addiction [see Warnings and Precautions (5.1)]. Methylphenidate hydrochloride extended-release capsules can be diverted for non-medical use into illicit channels or distribution.
Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of methylphenidate may cause increased heart rate, respiratory rate, or blood pressure; sweating; dilated pupils; hyperactivity; restlessness; insomnia; decreased appetite; loss of coordination; tremors; flushed skin; vomiting; and/or abdominal pain. Anxiety, psychosis, hostility, aggression, and suicidal or homicidal ideation have also been observed with CNS stimulants abuse and/or misuse. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
5.5 Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate use, in both adult and pediatric male patients. Although priapism was not reported with methylphenidate initiation, it developed after some time on methylphenidate, often subsequent to an increase in dosage. Priapism also occured during methylphenidate withdrawal (drug holidays or during discontinuation).
Methylphenidate hydrochloride extended-release capsules patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
11 Description (11 DESCRIPTION)
Methylphenidate hydrochloride extended-release capsules contain methylphenidate hydrochloride, a central nervous system (CNS) stimulant. Methylphenidate hydrochloride extended-release capsules contain multi layered beads, which are composed of an immediate-release layer which contains approximately 40% of the methylphenidate dose, and a controlled release layer which contains approximately 60% of the methylphenidate dose. Methylphenidate hydrochloride extended-release capsules are available in seven capsule strengths. Each extended-release capsule for once-a-day oral administration contains 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, 50 mg, or 60 mg of methylphenidate HCl USP, which is equivalent to 8.6 mg, 13.0 mg, 17.3 mg, 25.9 mg, 34.6 mg, 43.2 mg, or 51.9 mg of methylphenidate free base, respectively. Chemically, methylphenidate HCl is d,l (racemic) methyl α-phenyl-2-piperidineacetate hydrochloride. Its molecular formula is C14H19NO2∙HCl. Its structural formula is:
Methylphenidate hydrochloride USP is a white to off-white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone. Its molecular weight is 269.77.
Inactive Ingredients: ammonio methacrylate copolymer, type B; colloidal silicon dioxide (added if necessary); gelatin; hypromelloses; methacrylic acid copolymer, type C; polyethylene glycol; sugar spheres; talc; titanium oxide; and triethyl citrate.
Each strength capsule also contains colorant ingredients in the capsule shell as follows:
10 mg: FD&C Blue No. 1
15 mg: D&C Red No. 28, D&C Yellow No. 10, FD&C Red No. 40
20 mg: D&C Red No. 33, D&C Yellow No. 10
30 mg: FD&C Blue No. 1, FD&C Red No. 3
40 mg: D&C Red No. 28, FD&C Blue No. 1, FD&C Red No. 40
50 mg: D&C Yellow No. 10, FD&C Green No. 3
60 mg: Black Iron Oxide
8.4 Pediatric Use
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules have not been established in pediatric patients below the age of 6 years.
Safety and efficacy of methylphenidate hydrochloride extended-release capsules were evaluated in a multicenter, placebo-controlled, double-blind, parallel group study in 119 children 4 to <6 years of age with ADHD followed by a 12-month open-label extension in 44 of these children. In these studies, patients experienced high rates of adverse reactions, most notably weight loss. Comparing weights prior to initiation of methylphenidate hydrochloride extended-release capsules (in the safety and efficacy study) to weights after 12 months of treatment (in the open-label extension), 20 of 39 patients with data (50%) had lost enough weight to decrease 10 or more percentiles on a Centers for Disease Control growth chart for weight. In addition, systemic drug exposures in patients 4 to <6 years of age were higher than those observed in older children and adolescents at the same dose (2 to 3 fold higher Cmax and AUC). Therefore, the benefits of methylphenidate hydrochloride extended-release capsules do not outweigh the risks in pediatric patients 4 to <6 years of age.
The safety and effectiveness of methylphenidate hydrochloride extended-release capsules have been established in pediatric patients ages 6 to 17 years in two adequate and well-controlled clinical trials [see Clinical Studies (14)].
8.5 Geriatric Use
Clinical trials of methylphenidate hydrochloride extended-release capsules did not include any patients aged 65 years and over. In general, dose selection for an elderly patient start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of methylphenidate hydrochloride extended-release capsules for the treatment of ADHD was established in a randomized, double-blind, single center, placebo-controlled, flexible-dose, cross-over trial in pediatric patients aged 6 to 12 years and a second randomized, double-blind, multicenter, placebo-controlled, fixed–dose trial in pediatric patients 6 to 17 years.
4 Contraindications (4 CONTRAINDICATIONS)
- Hypersensitivity to methylphenidate or other components of the product. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.1)].
- Concomitant treatment with monoamine oxidase inhibitors, and also within 14 days following discontinuation of treatment with a monoamine oxidase inhibitor, because of the risk of hypertensive crisis [see Drug Interactions (7.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more detail in other sections of the labeling:
- Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1), and Drug Abuse and Dependence (9.2, 9.3)]
- Hypersensitivity to Methylphenidate [see Contraindications (4)]
- Hypertensive Crisis with Concomitant Use of Monoamine Oxidase Inhibitors [see Contraindications (4) and Drug Interactions (7.1)]
- Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2)]
- Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Priapism [see Warnings and Precautions (5.5)]
- Peripheral Vasculopathy, including Raynaud's Phenomenon [see Warnings and Precautions (5.6)]
- Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.7)]
- Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8)]
- Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9)]
- Motor and Verbal Tics, and Worsening of Tourette's Syndrome [see Warnings and Precautions (5.10)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. (7)
12.2 Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d- and l-isomers. The d-isomer is more pharmacologically active than the l-isomer. Methylphenidate blocks the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
2.2 Recommended Dosage
The recommended starting dose of methylphenidate hydrochloride extended-release capsules in patients 6 years and older is 10 mg once daily orally in the morning with or without food. Advise patients to establish a routine pattern with regard to meals. The dose should be individualized according to the needs and response of the patient.
The dose may be titrated weekly in increments of 10 mg. Daily doses above 60 mg have not been studied and are not recommended.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Methylphenidate hydrochloride extended-release capsules are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients 6 years and older [see Clinical Studies (14)].
12.1 Mechanism of Action
Methylphenidate HCl is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
9.1 Controlled Substance
Methylphenidate hydrochloride extended-release capsules contains methylphenidate a Schedule II controlled substance.
2.1 Pretreatment Screening
Prior to treating patients with methylphenidate hydrochloride extended-release capsules, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions 5.2].
- the family history and clinically evaluate patients for motor or verbal tics or Tourette's syndrome before initiating methylphenidate hydrochloride extended-release capsules [see Warnings and Precautions (5.10)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. (5.2)
- Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse. (5.3)
- Psychiatric Adverse Reactions: Prior to initiating methylphenidate hydrochloride extended-release capsules, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing methylphenidate hydrochloride extended-release capsules. (5.4)
- Priapism: If abnormally sustained or frequent and painful erections occur, patients should seek immediate medical attention. (5.5)
- Peripheral Vasculopathy, including Raynaud's Phenomenon: Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release capsules treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. (5.6)
- Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. (5.7)
- Acute Angle Closure Glaucoma: Methylphenidate hydrochloride extended-release capsules-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist. (5.8)
- Increased Intraocular Pressure (IOP) and Glaucoma: Prescribe methylphenidate hydrochloride extended-release capsules to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor patients with a history of increased IOP or open angle glaucoma. (5.9)
- Motor and Verbal Tics, and Worsening of Tourette's Syndrome: Before initiating methylphenidate hydrochloride extended-release capsules, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette's syndrome. Discontinue treatment if clinically appropriate. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended starting dose for patients 6 years and older: 10 mg once daily with or without food in the morning. Dosage may be increased weekly in increments of 10 mg per day. Daily dosage above 60 mg is not recommended. (2.1)
- Capsules may be swallowed whole or opened and the entire contents sprinkled onto applesauce. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 10 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting: 910. Cap imprinting: RP
- 15 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body and orange cap containing white spherical beads. Body imprinting: 915. Cap imprinting: RP
- 20 mg Extended-Release Capsules Hard Gelatin Capsules, white opaque body and yellow opaque cap containing white spherical beads. Body imprinting: 920. Cap imprinting: RP
- 30 mg Extended-Release Capsules Hard Gelatin Capsules, white opaque body and blue violet cap containing white spherical beads. Body imprinting: 930. Cap imprinting: RP
- 40 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body and pink opaque cap containing white spherical beads. Body imprinting: 940. Cap imprinting: RP
- 50 mg Extended-Release Capsules - Hard Gelatin Capsules, white opaque body and green opaque cap containing white spherical beads. Body imprinting: 950. Cap Imprinting: RP
- 60 mg Extended-Release Capsules Hard Gelatin Capsules, white opaque body and gray opaque cap containing white spherical beads. Body imprinting: 960. Cap imprinting: RP
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post approval use of methylphenidate products. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These adverse reactions are as follows:
Blood and Lymphatic System Disorders: Pancytopenia, Thrombocytopenia, Thrombocytopenic purpura
Cardiac Disorders: Angina pectoris, Bradycardia, Extrasystole, Supraventricular tachycardia, Ventricular extrasystole
Eye Disorders: Diplopia, Increased intraocular pressure, Mydriasis, Visual impairment
General Disorders: Chest pain, Chest discomfort, Hyperpyrexia
Immune System Disorders: Hypersensitivity reactions such as Angioedema, Anaphylactic reactions, Auricular swelling, Bullous conditions, Exfoliative conditions, Urticarias, Pruritus NEC, Rashes, Eruptions, and Exanthems NEC
Investigations: Alkaline phosphatase increased, Bilirubin increased, Hepatic enzyme increased, Platelet count decreased, White blood cell count abnormal, severe hepatic injury
Musculoskeletal, Connective Tissue and Bone Disorders: Arthralgia, Myalgia, Muscle twitching, Rhabdomyolysis
Nervous System: Convulsion, Grand mal convulsion, Dyskinesia, serotonin syndrome in combination with serotonergic drugs, Motor and Verbal Tics
Psychiatric Disorders: Disorientation, Libido changes
Skin and Subcutaneous Tissue Disorders: Alopecia, Erythema
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
2.3 Administration Instructions
Methylphenidate hydrochloride extended-release capsules may be taken whole or the capsule may be opened and the entire contents sprinkled onto applesauce. If the patient is using the sprinkled administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
5.1 Abuse, Misuse, and Addiction
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse. The use of methylphenidate hydrochloride extended-release capsules exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Methylphenidate hydrochloride extended-release capsules can be diverted for non-medical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2)]. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride extended-release capsules, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store methylphenidate hydrochloride extended-release capsules in a safe place, preferably locked, and instruct patients to not give methylphenidate hydrochloride extended-release capsules to anyone else. Throughout methylphenidate hydrochloride extended-release capsules treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
5.8 Acute Angle Closure Glaucoma
There have been reports of angle closure glaucoma associated with methylphenidate treatment.
Although the mechanism is not clear, methylphenidate hydrochloride extended-release capsules-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Methylphenidate Hydrochloride Extended-Release Capsules are available as follows:
| 10 mg Extended-Release Capsules - white opaque body & light turquoise blue opaque cap containing white spherical beads. Body Imprinting "910". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-075-45 |
| 15 mg Extended-Release Capsules - white opaque body and orange cap containing white spherical beads. Body imprinting "915". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-076-45 |
| 20 mg Extended-Release Capsules - white opaque body and yellow opaque cap containing white spherical beads. Body imprinting "920". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-077-45 |
| 30 mg Extended-Release Capsules - white opaque body and blue violet cap containing white spherical beads. Body imprinting "930". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-078-45 |
| 40 mg Extended-Release Capsules - white opaque body and pink opaque cap containing white spherical beads. Body imprinting "940". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-079-45 |
| 50 mg Extended-Release Capsules - white opaque body and green opaque cap containing white spherical beads. Body imprinting "950". Cap Imprinting "RP" | |
| Bottles of 90 | NDC 42858-080-45 |
| 60 mg Extended-Release Capsules - white opaque body and gray opaque cap containing white spherical beads. Body imprinting "960". Cap imprinting "RP" | |
| Bottles of 90 | NDC 42858-081-45 |
Warning: Abuse, Misuse, and Addiction (WARNING: ABUSE, MISUSE, AND ADDICTION)
Methylphenidate hydrochloride extended-release capsules have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release capsules, can result in overdose and death [see Overdosage (10)], and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride extended-release capsules, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout methylphenidate hydrochloride extended-release capsules treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2)].
2.4 Dosage Reduction and Discontinuation
If paradoxical aggravation of symptoms or other adverse reactions occur; the dosage should be reduced, or, if necessary, discontinue methylphenidate hydrochloride extended-release capsules.
If improvement is not observed after appropriate dosage adjustment over a one-month period, discontinue methylphenidate hydrochloride extended-release capsules.
5.3 Increased Blood Pressure and Heart Rate
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm). Some patients may have larger increases.
Monitor all methylphenidate hydrochloride extended-release capsules-treated patients for hypertension and tachycardia.
5.9 Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment [see Adverse Reactions (6.2)].
Prescribe methylphenidate hydrochloride extended-release capsules to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor methylphenidate hydrochloride extended-release capsules-treated patients with a history of abnormally increased IOP or open angle glaucoma.
5.2 Risks to Patients With Serious Cardiac Disease (5.2 Risks to Patients with Serious Cardiac Disease)
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosage.
Avoid methylphenidate hydrochloride extended-release capsules use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart arrhythmia, coronary artery disease, or other serious cardiac disease.
Principal Display Panel 10 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Label)
NDC 42858-075-45
CII
Methylphenidate
HCl Extended-Release
Capsules
10 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 15 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg Capsule Bottle Label)
NDC 42858-076-45
CII
Methylphenidate
HCl Extended-Release
Capsules
15 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 20 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label)
NDC 42858-077-45
CII
Methylphenidate
HCl Extended-Release
Capsules
20 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 30 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Label)
NDC 42858-078-45
CII
Methylphenidate
HCl Extended-Release
Capsules
30 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 40 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 40 mg Capsule Bottle Label)
NDC 42858-079-45
CII
Methylphenidate
HCl Extended-Release
Capsules
40 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 50 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 50 mg Capsule Bottle Label)
NDC 42858-080-45
CII
Methylphenidate
HCl Extended-Release
Capsules
50 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
Principal Display Panel 60 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 60 mg Capsule Bottle Label)
NDC 42858-081-45
CII
Methylphenidate
HCl Extended-Release
Capsules
60 mg
Dispense the accompanying
Medication Guide to each patient.
Rhodes
Rx only
Once Daily
90 Capsules
5.7 Long Term Suppression of Growth in Pediatric Patients (5.7 Long-Term Suppression of Growth in Pediatric Patients)
Methylphenidate hydrochloride extended-release capsules are not approved for use and are not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated pediatric patients over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period.
Closely monitor growth (weight and height) in methylphenidate hydrochloride extended-release capsules-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
5.6 Peripheral Vasculopathy, Including Raynaud's Phenomenon (5.6 Peripheral Vasculopathy, including Raynaud's Phenomenon)
CNS stimulants, including methylphenidate hydrochloride extended-release capsules, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports and at the therapeutic dosages of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release capsules treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for methylphenidate hydrochloride extended-release capsules-treated patients who develop signs or symptoms of peripheral vasculopathy.
5.10 Motor and Verbal Tics, and Worsening of Tourette's Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette's syndrome has also been reported [see Adverse Reactions (6.2)].
Before initiating methylphenidate hydrochloride extended-release capsules, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor methylphenidate hydrochloride extended-release capsules-treated patients for the emergence or worsening of tics or Tourette's syndrome, and discontinue treatment if clinically appropriate.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:06.059874 · Updated: 2026-03-14T22:35:47.693311