vcf®
f7a7b388-e55e-4f55-b271-0a60bb7aa694
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Vaginal contraceptive
Description
Drug Facts
Medication Information
Warnings
- For Vaginal Use Only
- Not for rectal (anal) use
Purpose
Vaginal contraceptive
Directions
- before using this product, read the enclosed package insert for complete directions and information
- insert one applicator full of foam in the vagina no more than one hour before each act of intercourse
- if this product is used together with another contraceptive method, there will probably be better protection against pregnancy
- if douching is desired, always wait at least 6 hours after intercourse before douching
- see enclosed instructions for cleaning the applicator after each use
Other Information
- When used correctly every time you have sex, latex condoms greatly reduce, but do not eliminate, the risk of catching or spreading HIV, the virus that causes AIDS.
- Use a latex condom without nonoxynol-9 if you or your sex partner has HIV/AIDS, multiple sex partners, or other HIV risk factors.
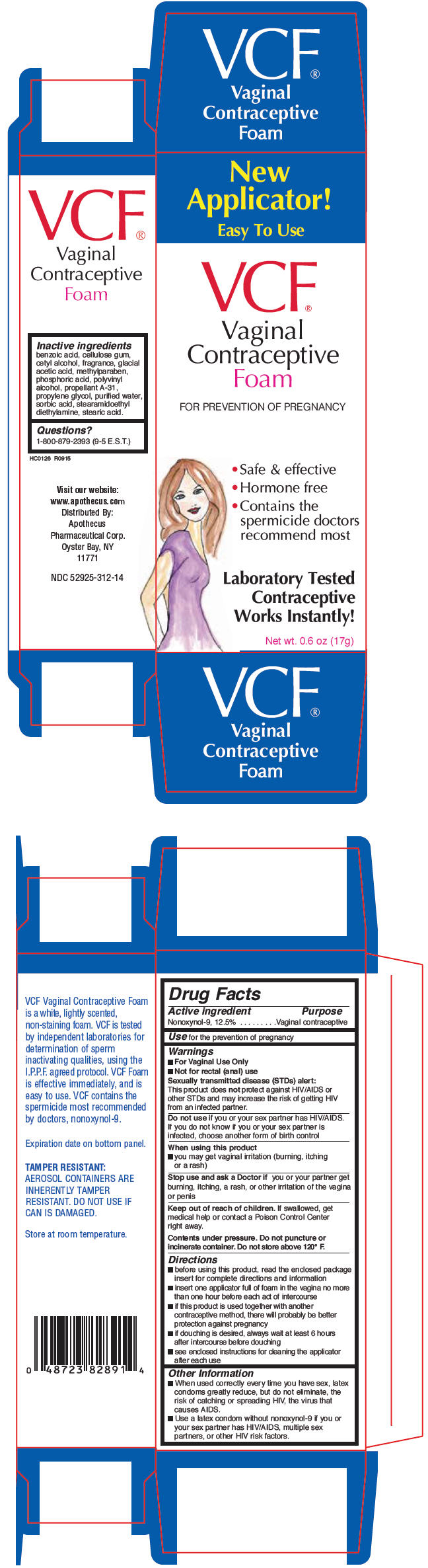
Inactive Ingredients
benzoic acid, cellulose gum, cetyl alcohol, fragrance, glacial acetic acid, methylparaben, phosphoric acid, polyvinyl alcohol, propellant A-31, propylene glycol, purified water, sorbic acid, stearamidoethyl diethylamine, stearic acid.
Description
Drug Facts
Questions?
1-800-879-2393 (9-5 E.S.T.)
Use
for the prevention of pregnancy
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a Doctor if you or your partner get burning, itching, a rash, or other irritation of the vagina or penis
Section 50570-1
Do not use if you or your sex partner has HIV/AIDS. If you do not know if you or your sex partner is infected, choose another form of birth control
Active Ingredient
Nonoxynol-9, 12.5%
When Using This Product
- you may get vaginal irritation (burning, itching or a rash)
Principal Display Panel 17 G Can Box
New
Applicator!
Easy To Use
VCF ®
Vaginal
Contraceptive
Foam
FOR PREVENTION OF PREGNANCY
- Safe & effective
- Hormone free
- Contains the
spermicide doctors
recommend most
Laboratory Tested
Contraceptive
Works Instantly!
Net wt. 0.6 oz (17g)
Sexually Transmitted Disease (stds) Alert
This product does not protect against HIV/AIDS or other STDs and may increase the risk of getting HIV from an infected partner.
Structured Label Content
Warnings
- For Vaginal Use Only
- Not for rectal (anal) use
Purpose
Vaginal contraceptive
Directions
- before using this product, read the enclosed package insert for complete directions and information
- insert one applicator full of foam in the vagina no more than one hour before each act of intercourse
- if this product is used together with another contraceptive method, there will probably be better protection against pregnancy
- if douching is desired, always wait at least 6 hours after intercourse before douching
- see enclosed instructions for cleaning the applicator after each use
Other Information
- When used correctly every time you have sex, latex condoms greatly reduce, but do not eliminate, the risk of catching or spreading HIV, the virus that causes AIDS.
- Use a latex condom without nonoxynol-9 if you or your sex partner has HIV/AIDS, multiple sex partners, or other HIV risk factors.
Inactive Ingredients (Inactive ingredients)
benzoic acid, cellulose gum, cetyl alcohol, fragrance, glacial acetic acid, methylparaben, phosphoric acid, polyvinyl alcohol, propellant A-31, propylene glycol, purified water, sorbic acid, stearamidoethyl diethylamine, stearic acid.
Questions?
1-800-879-2393 (9-5 E.S.T.)
Use
for the prevention of pregnancy
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a Doctor if you or your partner get burning, itching, a rash, or other irritation of the vagina or penis
Section 50570-1 (50570-1)
Do not use if you or your sex partner has HIV/AIDS. If you do not know if you or your sex partner is infected, choose another form of birth control
Active Ingredient (Active ingredient)
Nonoxynol-9, 12.5%
When Using This Product (When using this product)
- you may get vaginal irritation (burning, itching or a rash)
Principal Display Panel 17 G Can Box (PRINCIPAL DISPLAY PANEL - 17 g Can Box)
New
Applicator!
Easy To Use
VCF ®
Vaginal
Contraceptive
Foam
FOR PREVENTION OF PREGNANCY
- Safe & effective
- Hormone free
- Contains the
spermicide doctors
recommend most
Laboratory Tested
Contraceptive
Works Instantly!
Net wt. 0.6 oz (17g)
Sexually Transmitted Disease (stds) Alert (Sexually transmitted disease (STDs) alert)
This product does not protect against HIV/AIDS or other STDs and may increase the risk of getting HIV from an infected partner.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:41.665567 · Updated: 2026-03-14T22:55:20.733713