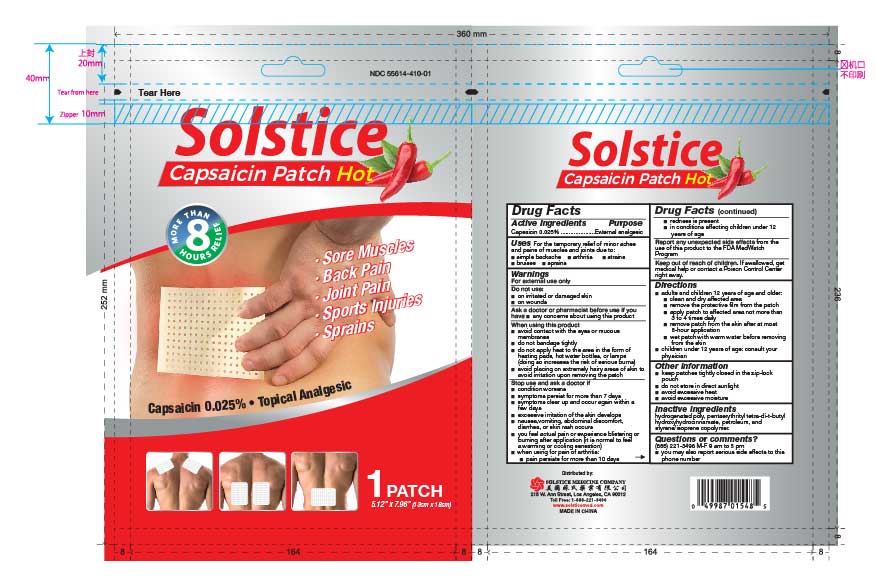
Drug Facts
f74db6e0-853a-4eb3-a4b5-9da2cd3d95cf
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Capsaicin 0.025%
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Uses
For the temporary relief of minor aches and pains of muscles and joints due to:
simple backache
arthritis
strains
bruises
sprains
Dosage and Administration
Directions
adults and children 12 years of age and older:
clean and dry affected area
remove the protective film from the patch
apply patch to affected area not more than 3 to 4 times daily
remove patch from the skin after at most 8-hour application
wet patch with warm water before removing from the skin
children under 12 years of age: consult your physician
Description
Active ingredient Capsaicin 0.025%
Section 34084-4
Report any unexpected side effectsfrom the use of this product to the FDA MedWatch Program
Section 44425-7
Other information
keep patches tightly closed in the zip-lock pouch
avoid excessive heat
Section 50565-1
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
condition worsens
symptoms persist for more than 7 days
symptoms clear up and occur again within a few days
excessive irritation of the skin develops
nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
you feel actual pain or experience blistering or burning after application (it is normal to feel a warming or cooling sensation)
when using for pain of arthritis:
pain persists for more than 10 days
redness is present
in conditions affecting children under 12 years of age
Section 50567-7
When using this product
avoid contact with the eyes or mucous membranes
do not bandage tightly
do not apply heat to the area in the form of heating pads, hot water bottles, or lamps (doing so increases the risk of serious burns)
avoid placing on extremely hairy areas of skin to avoid irritation upon removing the patch
Section 50568-5
Ask a doctor or pharmacist before use if you have
any concerns about using this product
Section 50570-1
Do not use
on irritated or damaged skin on wounds
Section 51727-6
Inactive ingredients hydrogenated poly, pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate, petroleum, and styrene/isoprene copolymer.
Section 51945-4
Solstice Capsaicin Patch Hot
Capsaicin 0.025%
NDC 55614-410-01
1 Patches, Each Patch 5.12 in x 7.09 in (13 cm x 18 cm)
Section 53413-1
Questions or comments?
(888) 221-3496 M-F 9 am to 5 pm
you may also report serious side effects to this phone number
Section 55105-1
Purpose
External analgesic
Section 55106-9
Active ingredient
Capsaicin 0.025%
Structured Label Content
Indications and Usage (34067-9)
Uses
For the temporary relief of minor aches and pains of muscles and joints due to:
simple backache
arthritis
strains
bruises
sprains
Dosage and Administration (34068-7)
Directions
adults and children 12 years of age and older:
clean and dry affected area
remove the protective film from the patch
apply patch to affected area not more than 3 to 4 times daily
remove patch from the skin after at most 8-hour application
wet patch with warm water before removing from the skin
children under 12 years of age: consult your physician
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 34084-4 (34084-4)
Report any unexpected side effectsfrom the use of this product to the FDA MedWatch Program
Section 44425-7 (44425-7)
Other information
keep patches tightly closed in the zip-lock pouch
avoid excessive heat
Section 50565-1 (50565-1)
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
condition worsens
symptoms persist for more than 7 days
symptoms clear up and occur again within a few days
excessive irritation of the skin develops
nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
you feel actual pain or experience blistering or burning after application (it is normal to feel a warming or cooling sensation)
when using for pain of arthritis:
pain persists for more than 10 days
redness is present
in conditions affecting children under 12 years of age
Section 50567-7 (50567-7)
When using this product
avoid contact with the eyes or mucous membranes
do not bandage tightly
do not apply heat to the area in the form of heating pads, hot water bottles, or lamps (doing so increases the risk of serious burns)
avoid placing on extremely hairy areas of skin to avoid irritation upon removing the patch
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you have
any concerns about using this product
Section 50570-1 (50570-1)
Do not use
on irritated or damaged skin on wounds
Section 51727-6 (51727-6)
Inactive ingredients hydrogenated poly, pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate, petroleum, and styrene/isoprene copolymer.
Section 51945-4 (51945-4)
Solstice Capsaicin Patch Hot
Capsaicin 0.025%
NDC 55614-410-01
1 Patches, Each Patch 5.12 in x 7.09 in (13 cm x 18 cm)
Section 53413-1 (53413-1)
Questions or comments?
(888) 221-3496 M-F 9 am to 5 pm
you may also report serious side effects to this phone number
Section 55105-1 (55105-1)
Purpose
External analgesic
Section 55106-9 (55106-9)
Active ingredient
Capsaicin 0.025%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:33.183087 · Updated: 2026-03-14T23:13:34.159935