65517-0040
f6f5dc9a-b578-70c1-e053-6394a90acdb0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Povidone-Iodine USP 10%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Povidone-Iodine USP 10%
Use
Patient preoperative skin preparation. Helps to reduc bacteria that potentially can cause skin infection.
Section 50565-1
- Keep out of reach of children
- If swallowed or gets in eyes, get medical help or contact Poison Control Center right away.
Section 50566-9
Stop use and ask doctor if infection occurs, redness, irritation or swelling develops.
Section 50569-3
- Ask a doctor before use if injuries are deep wounds, puncture wounds, serious burns.
Section 51945-4
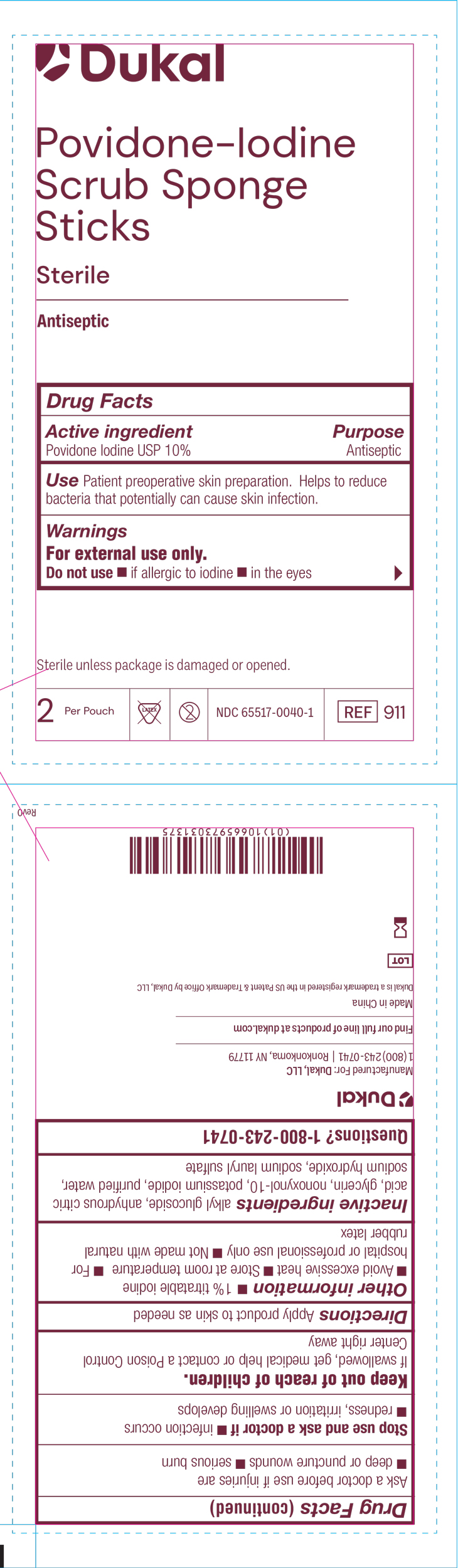
Dukal™
Povidone-Iodine
Scrub Sponge
Sticks
Sterile
Antispetic
Drug Facts
Active ingredient Purpose
Povidone Iodine USP 10% Antiseptic
Use Patient preoperative skin preparation. Helps to reduce bacteria
that potentially can cause skin infection.
Warnings
For external use only.
Do no use if allergic to iodine, in the eyes
Sterile unless package is damagedor opened
2 Per Pouch Not Made with Natural Rubber Latex Do not re-use NDC-66517-0040-1 REF 911
Warnings:
For external use only.
- Do not apply to persons allergic to iodine. Do not use in the eyes.
Directions
Apply product to skin as needed.
Active Ingredient
Povidone-Iodine USP 10%
Other Information:
1% titratable iodine, Avoid excessive heat, Store at room temperature, For hospital or professional use only, Not made with natural rubber latex
Inactive Ingredients:
Alkyl Glucoside, Anhydrous Citric Acid, Glycerin, Nonoxynol-10, Potassium Iodide, Purified Water, Sodium Hydroxide, Sodium Lauryl Sulfate
Structured Label Content
Use
Patient preoperative skin preparation. Helps to reduc bacteria that potentially can cause skin infection.
Section 50565-1 (50565-1)
- Keep out of reach of children
- If swallowed or gets in eyes, get medical help or contact Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask doctor if infection occurs, redness, irritation or swelling develops.
Section 50569-3 (50569-3)
- Ask a doctor before use if injuries are deep wounds, puncture wounds, serious burns.
Section 51945-4 (51945-4)
Dukal™
Povidone-Iodine
Scrub Sponge
Sticks
Sterile
Antispetic
Drug Facts
Active ingredient Purpose
Povidone Iodine USP 10% Antiseptic
Use Patient preoperative skin preparation. Helps to reduce bacteria
that potentially can cause skin infection.
Warnings
For external use only.
Do no use if allergic to iodine, in the eyes
Sterile unless package is damagedor opened
2 Per Pouch Not Made with Natural Rubber Latex Do not re-use NDC-66517-0040-1 REF 911
Purpose
Antiseptic
Warnings:
For external use only.
- Do not apply to persons allergic to iodine. Do not use in the eyes.
Directions
Apply product to skin as needed.
Active Ingredient
Povidone-Iodine USP 10%
Other Information:
1% titratable iodine, Avoid excessive heat, Store at room temperature, For hospital or professional use only, Not made with natural rubber latex
Inactive Ingredients:
Alkyl Glucoside, Anhydrous Citric Acid, Glycerin, Nonoxynol-10, Potassium Iodide, Purified Water, Sodium Hydroxide, Sodium Lauryl Sulfate
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:18.841790 · Updated: 2026-03-14T23:10:35.893664