f6e923e8-46e8-4287-bd62-5b954829cd73
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
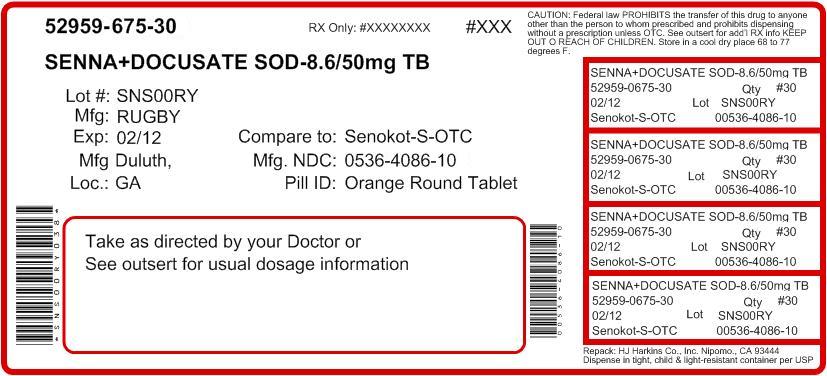
Active ingredients (in each tablet) Docusate sodium 50 mg Sennosides 8.6 mg
Medication Information
Warnings and Precautions
Do not use if your are taking mineral oil; for longer than one week; when abdominal pain, nausea or vomiting are present
Indications and Usage
RELIEVES OCCASIONAL CONSTIPATION
GENERALLY PRODUCES BOWEL MOVEMENT IN 6-12 HOURS
Dosage and Administration
Dosage and Administration
Adults and children 12 years and over - 2 tablets once a day - maximum dosage - 4 tablets twice a day
children 6 to under 12 years - 1 tablet once a day maximum dosage - 2 tablets twice a day
children 2 to under 6 years - 1/2 tablet once a day - maximum dosage- 1 tablet twice a day
children under 2 years - ask a doctor
Description
Active ingredients (in each tablet) Docusate sodium 50 mg Sennosides 8.6 mg
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if: you have rectal bleeding; you fail to have a bowel movement after use of this product. These may indicate a serious condition.
Section 50567-7
Directions take preferably at bedtime or as directed by a doctor
If you do not have a comfortable bowel movement by the second day, increase dose by one tablet (do not exceed maxiumum dosage)
Section 50568-5
Ask a doctor or pharmacist berfore use if you are taking any other drug. Laxatives may affect how other drugs work.
Section 50569-3
ASK A DOCTOR BEFORE USE IF YOU HAVE A SUDDEN CHANGE IN BOWEL HABITS THAT LASTS OVER TWO WEEKS
Section 51727-6
Inactive ingredients
carnauba wax, colloidal silicon dioxide, croscarmellose sodium
D-C yellow #10 aluminum lake, dibasic calcium phosphate
dihydrate, FD-C yellow #6 aluminum lake, hypromellose,
magnesium stearate, microcrystalline cellulose, polyethylene
glycol, sodium benzoate, stearic acide, Titanium dioxide
Section 51945-4
Enter section text here
Section 53413-1
call 1 800 645-2158 9 am - 5 pm Monday - Friday
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Uses: relieves occasional constipation; generally produces bowel movement in 6-12 hours.
Section 55106-9
Active ingredients (in each tablet)
Docusate sodium 50 mg
Sennosides 8.6 mg
Structured Label Content
Indications and Usage (34067-9)
RELIEVES OCCASIONAL CONSTIPATION
GENERALLY PRODUCES BOWEL MOVEMENT IN 6-12 HOURS
Dosage and Administration (34068-7)
Dosage and Administration
Adults and children 12 years and over - 2 tablets once a day - maximum dosage - 4 tablets twice a day
children 6 to under 12 years - 1 tablet once a day maximum dosage - 2 tablets twice a day
children 2 to under 6 years - 1/2 tablet once a day - maximum dosage- 1 tablet twice a day
children under 2 years - ask a doctor
Warnings and Precautions (34071-1)
Do not use if your are taking mineral oil; for longer than one week; when abdominal pain, nausea or vomiting are present
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if: you have rectal bleeding; you fail to have a bowel movement after use of this product. These may indicate a serious condition.
Section 50567-7 (50567-7)
Directions take preferably at bedtime or as directed by a doctor
If you do not have a comfortable bowel movement by the second day, increase dose by one tablet (do not exceed maxiumum dosage)
Section 50568-5 (50568-5)
Ask a doctor or pharmacist berfore use if you are taking any other drug. Laxatives may affect how other drugs work.
Section 50569-3 (50569-3)
ASK A DOCTOR BEFORE USE IF YOU HAVE A SUDDEN CHANGE IN BOWEL HABITS THAT LASTS OVER TWO WEEKS
Section 51727-6 (51727-6)
Inactive ingredients
carnauba wax, colloidal silicon dioxide, croscarmellose sodium
D-C yellow #10 aluminum lake, dibasic calcium phosphate
dihydrate, FD-C yellow #6 aluminum lake, hypromellose,
magnesium stearate, microcrystalline cellulose, polyethylene
glycol, sodium benzoate, stearic acide, Titanium dioxide
Section 51945-4 (51945-4)
Enter section text here
Section 53413-1 (53413-1)
call 1 800 645-2158 9 am - 5 pm Monday - Friday
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Uses: relieves occasional constipation; generally produces bowel movement in 6-12 hours.
Section 55106-9 (55106-9)
Active ingredients (in each tablet)
Docusate sodium 50 mg
Sennosides 8.6 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:23.743854 · Updated: 2026-03-14T22:52:57.217022