These Highlights Do Not Include All The Information Needed To Use Otezla/otezla Xr Safely And Effectively. See Full Prescribing Information For Otezla/otezla Xr.
f6b1f516-4972-4d82-bced-113e47b41cc5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 08/2025 Dosage and Administration ( 2 ) 08/2025
Indications and Usage
OTEZLA/OTEZLA XR, an inhibitor of phosphodiesterase 4 (PDE4), is indicated for the treatment of: Adult patients with: Active psoriatic arthritis ( 1.1 ) Plaque psoriasis who are candidates for phototherapy or systemic therapy ( 1.2 ) Oral ulcers associated with Behçet's Disease ( 1.3 ) Pediatric patients 6 years of age and older with: Active psoriatic arthritis ( 1.1 ) Moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy ( 1.2 ) In the pediatric population, OTEZLA is indicated for patients weighing at least 20 kg, and OTEZLA XR is indicated for patients weighing at least 50 kg.
Dosage and Administration
To reduce the risk of gastrointestinal symptoms, titrate to recommended dosage as follows: Adults with Psoriatic Arthritis, Plaque Psoriasis, or Behçet's Disease See Table 1 for the initial titration schedule. Recommended maintenance dosage is OTEZLA 30 mg twice daily or OTEZLA XR 75 mg once daily ( 2.1 ) Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis See Table 2 for the initial titration schedule ( 2.1 ) For patients weighing 50 kg or more : Recommended maintenance dosage is OTEZLA 30 mg twice daily or OTEZLA XR 75 mg once daily ( 2.1 ) For patients weighing 20 kg to less than 50 kg : Recommended maintenance dosage is OTEZLA 20 mg twice daily ( 2.1 ) Dosage in Patients with Severe Renal Impairment : Adult Patients : For initial dosage titration, titrate using only morning schedule listed in Table 1 and skip afternoon doses. Recommended maintenance dosage is OTEZLA 30 mg once daily ( 2.3 ) Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis : For initial dosage titration, titrate using only morning schedule for appropriate body weight category in Table 2 and skip afternoon doses ( 2.3 ) For patients weighing 50 kg or more: Recommended maintenance dosage is OTEZLA 30 mg once daily ( 2.3 ) For patients weighing 20 kg to less than 50 kg: Recommended maintenance dosage is OTEZLA 20 mg once daily ( 2.3 )
Warnings and Precautions
Hypersensitivity : Cases of angioedema and anaphylaxis have been reported during post marketing surveillance. Avoid the use of OTEZLA/OTEZLA XR in patients with known hypersensitivity to apremilast or to any of the excipients in the formulation. If signs or symptoms of serious hypersensitivity reactions develop during treatment, discontinue OTEZLA/OTEZLA XR and institute appropriate therapy ( 5.1 ) Diarrhea, Nausea, and Vomiting : Consider OTEZLA/OTEZLA XR dosage reduction or suspension if patients develop severe diarrhea, nausea, or vomiting ( 5.2 ) Depression : Advise patients, their caregivers, and families to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes and if such changes occur to contact their healthcare provider. Carefully weigh risks and benefits of treatment with OTEZLA/OTEZLA XR in patients with a history of depression and/or suicidal thoughts or behavior ( 5.3 ) Weight Decrease : Monitor weight regularly. If unexplained or clinically significant weight loss occurs, evaluate weight loss and consider discontinuation of OTEZLA/OTEZLA XR ( 5.4 ) Drug Interactions : Use with strong cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) is not recommended because loss of efficacy may occur ( 5.5 , 7.1 )
Contraindications
OTEZLA/OTEZLA XR is contraindicated in patients with a known hypersensitivity to apremilast or to any of the excipients in the formulation [see Warnings and Precautions (5.1) , see Adverse Reactions (6.1) ] .
Adverse Reactions
The following adverse reactions are described elsewhere in the labeling: Hypersensitivity [see Warnings and Precautions (5.1) ] Diarrhea, Nausea, and Vomiting [see Warnings and Precautions (5.2) ] Depression [see Warnings and Precautions (5.3) ] Weight Decrease [see Warnings and Precautions (5.4) ] Drug Interactions [see Warnings and Precautions (5.5) ]
Drug Interactions
Co-administration of strong cytochrome P450 enzyme inducer, rifampin, resulted in a reduction of systemic exposure of apremilast, which may result in a loss of efficacy of OTEZLA/OTEZLA XR. Therefore, the use of cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) with OTEZLA/OTEZLA XR is not recommended [see Drug Interactions (7.1) and Clinical Pharmacology (12.3) ] .
Storage and Handling
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths: 10 mg pink tablet engraved with "APR" on one side and "10" on the other side; 20 mg brown tablet engraved with "APR" on one side and "20" on the other side; 30 mg beige tablet engraved with "APR" on one side and "30" on the other side. Tablets are supplied in the strengths and package configurations listed in Table 14. Table 14. OTEZLA Package Configurations Package configuration Tablet strength NDC number Configurations for 30 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each), 4 tablets (20 mg each), and 47 tablets (30 mg each) 55513-369-55 60-count bottle 30 mg 55513-137-60 Configurations for 20 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each) and 51 tablets (20 mg each) 55513-508-55 60-count bottle 20 mg 55513-497-60 OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible. Tablets are supplied in the strengths and package configurations listed in Table 15. Table 15. OTEZLA XR Package Configurations Package configuration Tablet strength NDC number 28-day treatment initiation pack 41-tablet blister titration pack including tablets for titration and maintenance dosage: OTEZLA: 4 tablets (10 mg each), 4 tablets (20 mg each), and 19 tablets (30 mg each) and OTEZLA XR: 14 tablets (75 mg each) 55513-516-41 Bottles of 30 OTEZLA XR : 30 tablets (75 mg each) 55513-519-30
How Supplied
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths: 10 mg pink tablet engraved with "APR" on one side and "10" on the other side; 20 mg brown tablet engraved with "APR" on one side and "20" on the other side; 30 mg beige tablet engraved with "APR" on one side and "30" on the other side. Tablets are supplied in the strengths and package configurations listed in Table 14. Table 14. OTEZLA Package Configurations Package configuration Tablet strength NDC number Configurations for 30 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each), 4 tablets (20 mg each), and 47 tablets (30 mg each) 55513-369-55 60-count bottle 30 mg 55513-137-60 Configurations for 20 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each) and 51 tablets (20 mg each) 55513-508-55 60-count bottle 20 mg 55513-497-60 OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible. Tablets are supplied in the strengths and package configurations listed in Table 15. Table 15. OTEZLA XR Package Configurations Package configuration Tablet strength NDC number 28-day treatment initiation pack 41-tablet blister titration pack including tablets for titration and maintenance dosage: OTEZLA: 4 tablets (10 mg each), 4 tablets (20 mg each), and 19 tablets (30 mg each) and OTEZLA XR: 14 tablets (75 mg each) 55513-516-41 Bottles of 30 OTEZLA XR : 30 tablets (75 mg each) 55513-519-30
Medication Information
Warnings and Precautions
Hypersensitivity : Cases of angioedema and anaphylaxis have been reported during post marketing surveillance. Avoid the use of OTEZLA/OTEZLA XR in patients with known hypersensitivity to apremilast or to any of the excipients in the formulation. If signs or symptoms of serious hypersensitivity reactions develop during treatment, discontinue OTEZLA/OTEZLA XR and institute appropriate therapy ( 5.1 ) Diarrhea, Nausea, and Vomiting : Consider OTEZLA/OTEZLA XR dosage reduction or suspension if patients develop severe diarrhea, nausea, or vomiting ( 5.2 ) Depression : Advise patients, their caregivers, and families to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes and if such changes occur to contact their healthcare provider. Carefully weigh risks and benefits of treatment with OTEZLA/OTEZLA XR in patients with a history of depression and/or suicidal thoughts or behavior ( 5.3 ) Weight Decrease : Monitor weight regularly. If unexplained or clinically significant weight loss occurs, evaluate weight loss and consider discontinuation of OTEZLA/OTEZLA XR ( 5.4 ) Drug Interactions : Use with strong cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) is not recommended because loss of efficacy may occur ( 5.5 , 7.1 )
Indications and Usage
OTEZLA/OTEZLA XR, an inhibitor of phosphodiesterase 4 (PDE4), is indicated for the treatment of: Adult patients with: Active psoriatic arthritis ( 1.1 ) Plaque psoriasis who are candidates for phototherapy or systemic therapy ( 1.2 ) Oral ulcers associated with Behçet's Disease ( 1.3 ) Pediatric patients 6 years of age and older with: Active psoriatic arthritis ( 1.1 ) Moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy ( 1.2 ) In the pediatric population, OTEZLA is indicated for patients weighing at least 20 kg, and OTEZLA XR is indicated for patients weighing at least 50 kg.
Dosage and Administration
To reduce the risk of gastrointestinal symptoms, titrate to recommended dosage as follows: Adults with Psoriatic Arthritis, Plaque Psoriasis, or Behçet's Disease See Table 1 for the initial titration schedule. Recommended maintenance dosage is OTEZLA 30 mg twice daily or OTEZLA XR 75 mg once daily ( 2.1 ) Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis See Table 2 for the initial titration schedule ( 2.1 ) For patients weighing 50 kg or more : Recommended maintenance dosage is OTEZLA 30 mg twice daily or OTEZLA XR 75 mg once daily ( 2.1 ) For patients weighing 20 kg to less than 50 kg : Recommended maintenance dosage is OTEZLA 20 mg twice daily ( 2.1 ) Dosage in Patients with Severe Renal Impairment : Adult Patients : For initial dosage titration, titrate using only morning schedule listed in Table 1 and skip afternoon doses. Recommended maintenance dosage is OTEZLA 30 mg once daily ( 2.3 ) Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis : For initial dosage titration, titrate using only morning schedule for appropriate body weight category in Table 2 and skip afternoon doses ( 2.3 ) For patients weighing 50 kg or more: Recommended maintenance dosage is OTEZLA 30 mg once daily ( 2.3 ) For patients weighing 20 kg to less than 50 kg: Recommended maintenance dosage is OTEZLA 20 mg once daily ( 2.3 )
Contraindications
OTEZLA/OTEZLA XR is contraindicated in patients with a known hypersensitivity to apremilast or to any of the excipients in the formulation [see Warnings and Precautions (5.1) , see Adverse Reactions (6.1) ] .
Adverse Reactions
The following adverse reactions are described elsewhere in the labeling: Hypersensitivity [see Warnings and Precautions (5.1) ] Diarrhea, Nausea, and Vomiting [see Warnings and Precautions (5.2) ] Depression [see Warnings and Precautions (5.3) ] Weight Decrease [see Warnings and Precautions (5.4) ] Drug Interactions [see Warnings and Precautions (5.5) ]
Drug Interactions
Co-administration of strong cytochrome P450 enzyme inducer, rifampin, resulted in a reduction of systemic exposure of apremilast, which may result in a loss of efficacy of OTEZLA/OTEZLA XR. Therefore, the use of cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) with OTEZLA/OTEZLA XR is not recommended [see Drug Interactions (7.1) and Clinical Pharmacology (12.3) ] .
Storage and Handling
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths: 10 mg pink tablet engraved with "APR" on one side and "10" on the other side; 20 mg brown tablet engraved with "APR" on one side and "20" on the other side; 30 mg beige tablet engraved with "APR" on one side and "30" on the other side. Tablets are supplied in the strengths and package configurations listed in Table 14. Table 14. OTEZLA Package Configurations Package configuration Tablet strength NDC number Configurations for 30 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each), 4 tablets (20 mg each), and 47 tablets (30 mg each) 55513-369-55 60-count bottle 30 mg 55513-137-60 Configurations for 20 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each) and 51 tablets (20 mg each) 55513-508-55 60-count bottle 20 mg 55513-497-60 OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible. Tablets are supplied in the strengths and package configurations listed in Table 15. Table 15. OTEZLA XR Package Configurations Package configuration Tablet strength NDC number 28-day treatment initiation pack 41-tablet blister titration pack including tablets for titration and maintenance dosage: OTEZLA: 4 tablets (10 mg each), 4 tablets (20 mg each), and 19 tablets (30 mg each) and OTEZLA XR: 14 tablets (75 mg each) 55513-516-41 Bottles of 30 OTEZLA XR : 30 tablets (75 mg each) 55513-519-30
How Supplied
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths: 10 mg pink tablet engraved with "APR" on one side and "10" on the other side; 20 mg brown tablet engraved with "APR" on one side and "20" on the other side; 30 mg beige tablet engraved with "APR" on one side and "30" on the other side. Tablets are supplied in the strengths and package configurations listed in Table 14. Table 14. OTEZLA Package Configurations Package configuration Tablet strength NDC number Configurations for 30 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each), 4 tablets (20 mg each), and 47 tablets (30 mg each) 55513-369-55 60-count bottle 30 mg 55513-137-60 Configurations for 20 mg BID Dosage 28-day treatment initiation pack 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each) and 51 tablets (20 mg each) 55513-508-55 60-count bottle 20 mg 55513-497-60 OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible. Tablets are supplied in the strengths and package configurations listed in Table 15. Table 15. OTEZLA XR Package Configurations Package configuration Tablet strength NDC number 28-day treatment initiation pack 41-tablet blister titration pack including tablets for titration and maintenance dosage: OTEZLA: 4 tablets (10 mg each), 4 tablets (20 mg each), and 19 tablets (30 mg each) and OTEZLA XR: 14 tablets (75 mg each) 55513-516-41 Bottles of 30 OTEZLA XR : 30 tablets (75 mg each) 55513-519-30
Description
Indications and Usage ( 1 ) 08/2025 Dosage and Administration ( 2 ) 08/2025
Section 42229-5
Adult Patients with Psoriatic Arthritis, Plaque Psoriasis, or Behçet's Disease
The recommended initial dosage titration from Day 1 to Day 5 is shown in Table 1. Following the 5-day titration with OTEZLA, the recommended maintenance dosage is OTEZLA 30 mg twice daily or OTEZLA XR 75 mg once daily taken orally starting on Day 6. This titration is intended to reduce the gastrointestinal symptoms associated with initial therapy.
|
OTEZLA Dosage Titration OTEZLA tablets should be used for the initial titration regardless of whether OTEZLA or OTEZLA XR will be used for the maintenance dosage.
|
OTEZLA/OTEZLA XR Maintenance Dosage | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 & thereafter | ||||
| AM | AM | PM | AM | PM | AM | PM | AM | PM | |
| BID = twice daily; QD = once daily | |||||||||
| 10 mg | 10 mg | 10 mg | 10 mg | 20 mg | 20 mg | 20 mg | 20 mg | 30 mg | OTEZLA 30 mg BID OR OTEZLA XR 75 mg QD |
Section 44425-7
Storage and Handling
Store OTEZLA tablets below 30°C (86°F).
Store OTEZLA XR tablets between 20°C and 25°C (68°F and 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
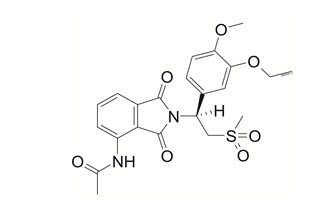
11 Description
The active ingredient in OTEZLA/OTEZLA XR tablets is apremilast. Apremilast drug substance is non-hygroscopic. Apremilast drug substance is practically insoluble in water and slightly soluble in alcohol.
Apremilast is a phosphodiesterase 4 (PDE4) inhibitor. Apremilast is known chemically as
N-[2-[(1S)-1-(3-ethoxy-4-methoxyphenyl)-2-(methylsulfonyl)ethyl]-2,3-dihydro-1,3-dioxo-1H-isoindol-4-yl]acetamide. Its empirical formula is C22H24N2O7S and the molecular weight is 460.5.
The chemical structure is:
OTEZLA (apremilast) tablets are supplied in 10 mg, 20 mg, and 30 mg strengths for oral administration. Each tablet contains apremilast as the active ingredient and the following inactive ingredients: croscarmellose sodium, iron oxide red, iron oxide yellow (20 and 30 mg only), iron oxide black (30 mg only), lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide.
OTEZLA XR (apremilast) extended-release tablets are supplied in a 75 mg strength for oral administration. Each tablet contains apremilast as the active ingredient and the following inactive ingredients: cellulose acetate, colloidal silicon dioxide, ferrosoferric oxide, hydroxypropyl methylcellulose acetate succinate, hypromellose, iron oxide red, magnesium stearate, mannitol, microcrystalline cellulose, polyethylene glycol, polyethylene oxide, sodium chloride, titanium dioxide.
5.3 Depression
Treatment with apremilast is associated with an increased incidence of depression. Before using OTEZLA/OTEZLA XR in patients with a history of depression and/or suicidal thoughts or behavior, carefully weigh the risks and benefits of treatment with OTEZLA/OTEZLA XR. Advise patients, their caregivers, and families of the need to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes, and if such changes occur to contact their healthcare provider. Carefully evaluate the risks and benefits of continuing treatment with OTEZLA/OTEZLA XR if such events occur.
8.5 Geriatric Use
Of the 1493 patients who enrolled in Trials PsA-1, PsA-2, and PsA-3, a total of 146 (9.8%) psoriatic arthritis patients were 65 years of age and older, including 19 (1.3%) patients 75 years and older. No overall differences were observed in the safety profile of geriatric patients ≥ 65 years of age and younger adult patients < 65 years of age in the clinical trials.
Of the 1257 subjects who enrolled in two placebo-controlled plaque psoriasis trials (PSOR-1 and PSOR-2), a total of 108 (8.6%) plaque psoriasis patients were 65 years of age and older, including 9 (0.7%) patients who were 75 years of age and older. No overall differences were observed in the safety or effectiveness in geriatric patients ≥ 65 years of age and younger adult patients < 65 years of age in the clinical trials.
Because patients 65 years of age or older may be at a higher risk of complications such as volume depletion or hypotension from severe diarrhea, nausea, or vomiting, monitor geriatric patients closely for such complications [see Warnings and Precautions (5.2)].
4 Contraindications
OTEZLA/OTEZLA XR is contraindicated in patients with a known hypersensitivity to apremilast or to any of the excipients in the formulation [see Warnings and Precautions (5.1), see Adverse Reactions (6.1)].
5.4 Weight Decrease
Weight loss may occur in adult or pediatric patients treated with OTEZLA/OTEZLA XR.
Regularly monitor the weight of patients treated with OTEZLA/OTEZLA XR. If unexplained or clinically significant weight loss occurs, evaluate weight loss and consider discontinuation of OTEZLA/OTEZLA XR [see Adverse Reactions (6.1)].
6 Adverse Reactions
The following adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Diarrhea, Nausea, and Vomiting [see Warnings and Precautions (5.2)]
- Depression [see Warnings and Precautions (5.3)]
- Weight Decrease [see Warnings and Precautions (5.4)]
- Drug Interactions [see Warnings and Precautions (5.5)]
1.2 Plaque Psoriasis
OTEZLA/OTEZLA XR is indicated for the treatment of adult patients with plaque psoriasis who are candidates for phototherapy or systemic therapy.
OTEZLA is indicated for the treatment of pediatric patients 6 years of age and older and weighing at least 20 kg with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy.
OTEZLA XR is indicated for the treatment of pediatric patients 6 years of age and older and weighing at least 50 kg with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy.
5.1 Hypersensitivity
Hypersensitivity reactions, including cases of angioedema and anaphylaxis, have been reported during post marketing surveillance. Avoid the use of OTEZLA/OTEZLA XR in patients with known hypersensitivity to apremilast or to any of the excipients in the formulation. If signs or symptoms of serious hypersensitivity reactions develop during treatment, discontinue OTEZLA/OTEZLA XR and institute appropriate therapy.
8.6 Renal Impairment
OTEZLA pharmacokinetics were characterized in adult subjects with mild, moderate, and severe renal impairment as defined by a creatinine clearance of 60-89, 30-59, and less than 30 mL per minute, respectively, by the Cockcroft–Gault equation. No dosage adjustment is needed in patients with mild or moderate renal impairment. In adult patients with severe renal impairment, reduce the maintenance dosage to OTEZLA 30 mg once daily. In pediatric patients 6 years of age and older and weighing at least 20 kg with psoriatic arthritis or moderate to severe plaque psoriasis and severe renal impairment, reduce the maintenance dosage to OTEZLA 30 mg once daily for pediatric patients weighing at least 50 kg and to OTEZLA 20 mg once daily for pediatric patients weighing 20 kg to less than 50 kg [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
The pharmacokinetics of OTEZLA XR has not been evaluated in patients with renal impairment. In patients with severe renal impairment, it is recommended that OTEZLA be used instead of OTEZLA XR [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In clinical studies, apremilast decreased IL-17, IL-22 and TNF-alpha levels circulating in blood and reduced expression of these pro-inflammatory cytokine genes in skin. The clinical implications of these pharmacodynamic responses remain unclear.
5.5 Drug Interactions
Co-administration of strong cytochrome P450 enzyme inducer, rifampin, resulted in a reduction of systemic exposure of apremilast, which may result in a loss of efficacy of OTEZLA/OTEZLA XR. Therefore, the use of cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) with OTEZLA/OTEZLA XR is not recommended [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Apremilast pharmacokinetics were characterized in subjects with moderate (Child Pugh B) and severe (Child Pugh C) hepatic impairment. No dosage adjustment is necessary in these patients.
1 Indications and Usage
OTEZLA/OTEZLA XR, an inhibitor of phosphodiesterase 4 (PDE4), is indicated for the treatment of:
Adult patients with:
- Active psoriatic arthritis (1.1)
- Plaque psoriasis who are candidates for phototherapy or systemic therapy (1.2)
- Oral ulcers associated with Behçet's Disease (1.3)
Pediatric patients 6 years of age and older with:
- Active psoriatic arthritis (1.1)
- Moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy (1.2)
In the pediatric population, OTEZLA is indicated for patients weighing at least 20 kg, and OTEZLA XR is indicated for patients weighing at least 50 kg.
1.1 Psoriatic Arthritis
OTEZLA is indicated for the treatment of adult patients and pediatric patients 6 years of age and older and weighing at least 20 kg with active psoriatic arthritis.
OTEZLA XR is indicated for the treatment of adult patients and pediatric patients 6 years of age and older and weighing at least 50 kg with active psoriatic arthritis.
12.1 Mechanism of Action
Apremilast is an oral small molecule inhibitor of phosphodiesterase 4 (PDE4) specific for cyclic adenosine monophosphate (cAMP). PDE4 inhibition results in increased intracellular cAMP levels. The specific mechanism(s) by which apremilast exerts its therapeutic action is not well defined.
5 Warnings and Precautions
- Hypersensitivity: Cases of angioedema and anaphylaxis have been reported during post marketing surveillance. Avoid the use of OTEZLA/OTEZLA XR in patients with known hypersensitivity to apremilast or to any of the excipients in the formulation. If signs or symptoms of serious hypersensitivity reactions develop during treatment, discontinue OTEZLA/OTEZLA XR and institute appropriate therapy (5.1)
- Diarrhea, Nausea, and Vomiting: Consider OTEZLA/OTEZLA XR dosage reduction or suspension if patients develop severe diarrhea, nausea, or vomiting (5.2)
- Depression: Advise patients, their caregivers, and families to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes and if such changes occur to contact their healthcare provider. Carefully weigh risks and benefits of treatment with OTEZLA/OTEZLA XR in patients with a history of depression and/or suicidal thoughts or behavior (5.3)
- Weight Decrease: Monitor weight regularly. If unexplained or clinically significant weight loss occurs, evaluate weight loss and consider discontinuation of OTEZLA/OTEZLA XR (5.4)
- Drug Interactions: Use with strong cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) is not recommended because loss of efficacy may occur (5.5, 7.1)
7.1 Strong Cyp450 Inducers
Co-administration with strong CYP450 inducers (such as rifampin) decreases apremilast exposure and may result in loss of efficacy of OTEZLA/OTEZLA XR [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.3)].
2 Dosage and Administration
To reduce the risk of gastrointestinal symptoms, titrate to recommended dosage as follows:
- Adults with Psoriatic Arthritis, Plaque Psoriasis, or Behçet's Disease
- Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis
-
Dosage in Patients with Severe Renal Impairment:
- Adult Patients: For initial dosage titration, titrate using only morning schedule listed in Table 1 and skip afternoon doses. Recommended maintenance dosage is OTEZLA 30 mg once daily (2.3)
- Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis: For initial dosage titration, titrate using only morning schedule for appropriate body weight category in Table 2 and skip afternoon doses (2.3)
3 Dosage Forms and Strengths
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths:
- 10-mg pink tablet engraved with "APR" on one side and "10" on the other side
- 20-mg brown tablet engraved with "APR" on one side and "20" on the other side
- 30-mg beige tablet engraved with "APR" on one side and "30" on the other side
OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible.
8 Use in Specific Populations
14.1 Adult Psoriatic Arthritis
The safety and efficacy of OTEZLA was evaluated in three multicenter, randomized, double-blind, placebo-controlled trials (PsA-1 [NCT01172938], PsA-2 [NCT01212757], and PsA-3 [NCT01212770]) of similar design. A total of 1493 adult subjects with active PsA (≥ 3 swollen joints and ≥ 3 tender joints) despite prior or current treatment with disease-modifying antirheumatic drug (DMARD) therapy were randomized. Subjects enrolled in these trials had a diagnosis of PsA for at least 6 months. One qualifying psoriatic skin lesion of at least 2 cm in diameter was required in Trial PsA-3. Previous treatment with a biologic, including TNF blockers was allowed (up to 10% could be TNF blocker therapeutic failures). Across the three trials, subjects were randomly assigned to placebo (n = 496), OTEZLA 20 mg (n = 500), or OTEZLA 30 mg (n = 497) given orally twice daily. Titration was used over the first 5 days [see Dosage and Administration (2.1)]. Subjects were allowed to receive stable doses of concomitant methotrexate [MTX (≤ 25 mg/week)], sulfasalazine [SSZ (≤ 2 g/day)], leflunomide [LEF (≤ 20 mg/day)], low dose oral corticosteroids (equivalent to ≤ 10 mg of prednisone a day), and/or nonsteroidal anti-inflammatory drugs (NSAIDs) during the trial. Treatment assignments were stratified based on small molecule DMARD use at baseline in Trials PsA-1, PsA-2 and PsA-3. There was an additional stratification of body surface area (BSA) > 3% with psoriasis in Trial PsA-3. The subjects who were therapeutic failures of > 3 agents for PsA (small molecules or biologics), or > 1 biologic TNF blocker were excluded.
The primary endpoint was the percentage of subjects achieving American College of Rheumatology (ACR) 20 response at Week 16. Placebo-controlled efficacy data were collected and analyzed through Week 24. Subjects whose tender and swollen joint counts had not improved by at least 20% were considered non-responders at Week 16. Placebo non-responders were re-randomized 1:1 in a blinded fashion to either OTEZLA 20 mg twice daily or 30 mg twice daily following the titration schema [see Dosage and Administration (2.1)]. OTEZLA subjects remained on their initial treatment. At Week 24, all remaining placebo subjects were re-randomized to either 20 mg twice daily or 30 mg twice daily.
Subjects with subtypes of PsA were enrolled across the three trials, including symmetric polyarthritis (62.0%), asymmetric oligoarthritis (27.0%), distal interphalangeal (DIP) joint arthritis (6.0%), arthritis mutilans (3.0%), and predominant spondylitis (2.1%). The median duration of PsA disease was 5 years. Subjects received concomitant therapy with at least one DMARD (65.0%), MTX (55.0%), SSZ (9.0%), LEF (7.0%), low dose oral corticosteroids (14.0%), and NSAIDs (71.0%). Prior treatment with small molecule DMARDs only was reported in 76.0% of subjects and prior treatment with biologic DMARDs was reported in 22.0% of subjects, which includes 9.0% who had failed prior biologic DMARD treatment.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
-
Administration Instructions
Instruct patients to take OTEZLA/OTEZLA XR only as prescribed [see Dosage and Administration (2.1 and 2.3)]. Advise patients to take OTEZLA/OTEZLA XR with or without food. Instruct patients to swallow tablets whole and not to crush, split, or chew prior to swallowing [see Dosage and Administration (2.4)]. -
Hypersensitivity
Inform patients that hypersensitivity reactions can occur following administration of OTEZLA/OTEZLA XR. Instruct patients to contact their healthcare provider if they experience symptoms of an allergic reaction [see Warnings and Precautions (5.1)]. -
Diarrhea, Nausea, and Vomiting
Advise patients of the potential complications of severe diarrhea, nausea, or vomiting and instruct them to contact their healthcare provider if they experience these adverse reactions, especially if the patient is 65 years of age or older [see Warnings and Precautions (5.2)]. -
Depression
Inform patients that treatment with OTEZLA/OTEZLA XR is associated with an increased incidence of depression. Advise patients, their caregivers, and families of the need to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes, and if such changes occur to contact their healthcare provider [see Warnings and Precautions (5.3)]. -
Weight Decrease
Inform patients that treatment with OTEZLA/OTEZLA XR is associated with potential weight loss. Instruct patients or caregivers to have their or their child's weight monitored regularly and, if unexplained or clinically significant weight loss occurs, to contact their healthcare provider for evaluation of the weight loss [see Warnings and Precautions (5.4)]. -
Pregnancy
Advise pregnant patients and patients of reproductive potential of the potential risk to a fetus. Advise patients to inform their prescriber of a known or suspected pregnancy [see Use in Specific Populations (8.1)]. -
Residual Tablet Shell
Patients receiving OTEZLA XR may notice an inert tablet shell passing in the stool or via colostomy. Patients should be informed that the active medication has already been absorbed by the time the patient sees the inert tablet shell.
5.2 Diarrhea, Nausea, and Vomiting
There have been reports of severe diarrhea, nausea, and vomiting associated with the use of OTEZLA. Most events occurred within the first few weeks of treatment. In some cases, patients were hospitalized. Patients 65 years of age or older and patients taking medications that can lead to volume depletion or hypotension may be at a higher risk of complications from severe diarrhea, nausea, or vomiting. Monitor patients who are more susceptible to complications of diarrhea or vomiting. Patients who reduced dosage or discontinued OTEZLA generally improved quickly. Consider OTEZLA/OTEZLA XR dosage reduction or suspension if patients develop severe diarrhea, nausea, or vomiting.
16 How Supplied/storage and Handling
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths: 10 mg pink tablet engraved with "APR" on one side and "10" on the other side; 20 mg brown tablet engraved with "APR" on one side and "20" on the other side; 30 mg beige tablet engraved with "APR" on one side and "30" on the other side.
Tablets are supplied in the strengths and package configurations listed in Table 14.
| Package configuration | Tablet strength | NDC number |
|---|---|---|
| Configurations for 30 mg BID Dosage | ||
| 28-day treatment initiation pack | 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each), 4 tablets (20 mg each), and 47 tablets (30 mg each) | 55513-369-55 |
| 60-count bottle | 30 mg | 55513-137-60 |
| Configurations for 20 mg BID Dosage | ||
| 28-day treatment initiation pack | 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each) and 51 tablets (20 mg each) | 55513-508-55 |
| 60-count bottle | 20 mg | 55513-497-60 |
OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible.
Tablets are supplied in the strengths and package configurations listed in Table 15.
| Package configuration | Tablet strength | NDC number |
|---|---|---|
| 28-day treatment initiation pack | 41-tablet blister titration pack including tablets for titration and maintenance dosage: OTEZLA: 4 tablets (10 mg each), 4 tablets (20 mg each), and 19 tablets (30 mg each) and OTEZLA XR: 14 tablets (75 mg each) |
55513-516-41 |
| Bottles of 30 |
OTEZLA XR: 30 tablets (75 mg each) |
55513-519-30 |
2.4 Important Administration Instructions
- Administer OTEZLA/OTEZLA XR with or without food.
- Swallow tablets whole. Do not crush, split, or chew.
2.2 Switching Between Otezla and Otezla Xr
Patients treated with OTEZLA 30 mg twice daily may be switched to OTEZLA XR 75 mg once daily the day following the last dose of OTEZLA 30 mg.
Patients treated with OTEZLA XR 75 mg once daily may be switched to OTEZLA 30 mg twice daily the day following the last dose of OTEZLA XR 75 mg.
14.4 Adult Mild to Moderate Plaque Psoriasis
A multicenter, randomized, double-blind, placebo-controlled trial (PSOR-4 [NCT03721172]) was conducted in 595 adult subjects with mild to moderate plaque psoriasis (BSA involvement of 2-15%, sPGA score of 2-3 [mild or moderate disease], and PASI score of 2-15). Enrolled subjects had an inadequate response or were intolerant to at least one topical therapy and had not received prior biologic therapy. Subjects were allowed to use unmedicated emollients for lesions on non-scalp areas of the body and non-medicated shampoos for lesions on the scalp.
Subjects were randomized 1:1 to receive either OTEZLA 30 mg twice daily (n = 297) or placebo twice daily (n = 298) for 16 weeks. At Week 16, the placebo group was switched to receive OTEZLA and the OTEZLA group remained on drug through Week 32. The primary endpoint was the proportion of subjects who achieved an sPGA response (defined as an sPGA score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline) at Week 16. Subjects with mild disease (sPGA = 2 at baseline) were required to be clear (sPGA = 0) to achieve an sPGA response. Other evaluated endpoints include the proportion of subjects with a Whole Body Itch NRS response (defined as a ≥ 4-point reduction from baseline) at Week 16 among subjects with a baseline Whole Body Itch NRS ≥ 4 and the proportion of subjects with an ScPGA response (defined as an ScPGA score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline) at Week 16 among subjects with a baseline ScPGA score ≥ 2.
Subjects ranged in age from 18 to 85 years, with an overall median age of 50 years. The mean baseline BSA involvement was 6.4%, the mean baseline PASI score was 6.5, and the proportions of subjects with an sPGA score of 2 (mild) and 3 (moderate) at baseline were 30.6% and 69.4%, respectively.
14.2 Adult Moderate to Severe Plaque Psoriasis
Two multicenter, randomized, double-blind, placebo-controlled trials (PSOR-1 [NCT01194219] and PSOR-2 [NCT01232283]) enrolled a total of 1257 subjects 18 years of age and older with moderate to severe plaque psoriasis [body surface area (BSA) involvement of ≥ 10%, static Physician Global Assessment (sPGA) of ≥ 3 (moderate or severe disease), Psoriasis Area and Severity Index (PASI) score ≥ 12, candidates for phototherapy or systemic therapy]. Subjects were allowed to use low potency topical corticosteroids on the face, axilla and groin. Subjects with plaque psoriasis of the scalp were allowed to use coal tar shampoo and/or salicylic acid scalp preparations on scalp lesions.
Trial PSOR-1 enrolled 844 subjects and Trial PSOR-2 enrolled 413 subjects. In both trials, subjects were randomized 2:1 to OTEZLA 30 mg twice daily (BID) or placebo for 16 weeks. Both trials assessed the proportion of subjects who achieved PASI-75 at Week 16 and the proportion of subjects who achieved an sPGA score of clear (0) or almost clear (1) at Week 16. Across both trials, subjects ranged in age from 18 to 83 years, with an overall median age of 46 years. The mean baseline BSA involvement was 25.2% (median 21.0%), the mean baseline PASI score was 19.1 (median 16.8), and the proportion of subjects with an sPGA score of 3 (moderate) and 4 (severe) at baseline were 70.0% and 29.8%, respectively. Approximately 30% of all subjects had received prior phototherapy and 54% had received prior conventional systemic and/or biologic therapy for the treatment of psoriasis with 37% receiving prior conventional systemic therapy and 30% receiving prior biologic therapy. Approximately one-third of subjects had not received prior phototherapy, conventional systemic nor biologic therapy. A total of 18% of subjects had a history of psoriatic arthritis.
Principal Display Panel 60 Tablet Bottle Label
NDC 55513-137-60
Otezla®
(apremilast) tablets
30 mg
Rx Only
60 Tablets
1.3 Oral Ulcers Associated With Behçet's Disease
OTEZLA/OTEZLA XR is indicated for the treatment of adult patients with oral ulcers associated with Behçet's Disease.
14.3 Pediatric Moderate to Severe Plaque Psoriasis
A multicenter, randomized, double-blind, placebo-controlled trial (PSOR-6 [NCT03701763]) was conducted in 245 pediatric subjects 6 to 17 years of age (inclusive) with moderate to severe plaque psoriasis who were candidates for phototherapy or systemic therapy. Subjects had an sPGA score of ≥ 3 (moderate or severe disease), BSA involvement of ≥ 10%, and PASI score of ≥ 12, with psoriasis that was inadequately controlled by or inappropriate for topical therapy. Subjects were allowed to use low potency or weak topical corticosteroids on the face, axilla, and groin and unmedicated skin moisturizers for body lesions only.
Subjects were randomized 2:1 to receive either OTEZLA (n = 163) or placebo (n = 82) for 16 weeks. Subjects with a baseline weight of 20 kg to < 50 kg received OTEZLA 20 mg twice daily or placebo twice daily, and subjects with a baseline weight ≥ 50 kg received OTEZLA 30 mg twice daily or placebo twice daily. At Week 16, the placebo group was switched to receive OTEZLA (with dosage based on baseline weight) and the OTEZLA group remained on drug (according to their original dosing assignment) through Week 52. The primary endpoint was the proportion of subjects who achieved an sPGA response (defined as a score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline) at Week 16. The key secondary endpoint was the proportion of subjects who achieved a PASI-75 response (at least a 75% reduction in PASI score from baseline) at Week 16.
Enrolled subjects ranged in age from 6 to 17 years, with a median age of 13 years; 41.2% of subjects were 6 to 11 years of age and 58.8% of subjects were 12 to 17 years of age. Of the enrolled subjects, 52.2% were female. For race, 86.9% were White, 3.7% were Asian, 3.3% were Black or African American, 0.8% were American Indian or Alaskan Native, and 5.3% were not reported or unknown. For ethnicity, 81.6% of subjects identified as not Hispanic or Latino, 13.1% identified as Hispanic or Latino, and 5.3% were not reported or unknown. The mean baseline BSA involvement was 31.5% (median 26.0%), the mean baseline PASI score was 19.8 (median 17.2), and the proportions of subjects with an sPGA score of 3 (moderate) and 4 (severe) at baseline were 75.5% and 24.5%, respectively. Of the enrolled subjects, 82.9% did not receive prior conventional systemic therapy and 94.3% were biologic-naïve.
Principal Display Panel 75 Mg Tablet Bottle Label
Once-daily
NDC 55513-519-30
Otezla XR™
(apremilast)
extended-release tablets
75 mg
Rx Only
30 Tablets
14.5 Adult Oral Ulcers Associated With Behçet's Disease
A multicenter, randomized, placebo-controlled trial (BCT-002 [NCT02307513]) enrolled a total of 207 adult subjects with BD with active oral ulcers. Subjects were previously treated with at least one nonbiologic BD medication and were candidates for systemic therapy. Subjects met the International Study Group (ISG) Criteria for BD. Subjects had at least 2 oral ulcers at screening and at least 2 oral ulcers at randomization and without currently active major organ involvement. Concomitant treatment for BD was not allowed.
Subjects were randomized 1:1 to receive either OTEZLA 30 mg twice daily (n = 104) or placebo (n = 103) for 12 weeks. After Week 12, all subjects received OTEZLA 30 mg twice daily.
Efficacy was assessed based on the number and pain of oral ulcers.
Subjects ranged in age from 19 to 72 years, with a mean age of 40 years. The mean duration of BD was 6.84 years. All subjects had a history of recurrent oral ulcers that were currently active. Subjects had a history of skin lesions (98.6%), genital ulcers (90.3%), musculoskeletal manifestations (72.5%), ocular manifestations (17.4%), central nervous system (9.7%), gastrointestinal (GI) manifestations (9.2%) and vascular involvement (1.4%). The mean baseline oral ulcer counts were 4.2 and 3.9 in the OTEZLA and placebo groups, respectively.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies were conducted in mice and rats with apremilast to evaluate its carcinogenic potential. No evidence of apremilast-induced tumors was observed in mice at oral doses up to approximately 9-times the MRHD on an AUC basis (1000 mg/kg/day) or in rats at oral doses up to approximately 0.08- and 1.1-times the MRHD, (20 mg/kg/day in males and 3 mg/kg/day in females, respectively).
Apremilast tested negative in the Ames assay, in vitro chromosome aberration assay of human peripheral blood lymphocytes, and the in vivo mouse micronucleus assay.
In a fertility study of male mice, apremilast at oral doses up to approximately 3-times the MRHD based on AUC (up to 50 mg/kg/day) produced no effects on male fertility. In a fertility study of female mice, apremilast was administered at oral doses of 10, 20, 40, or 80 mg/kg/day. At doses ≥ 1.8-times the MRHD (≥ 20 mg/kg/day), estrous cycles were prolonged, due to lengthening of diestrus which resulted in a longer interval until mating. Mice that became pregnant at doses of 20 mg/kg/day and greater also had increased incidences of early post-implantation losses. There was no effect of apremilast approximately 1.0-times the MRHD (10 mg/kg/day).
Principal Display Panel 28 Day Treatment Initiation Pack
NDC 55513-369-55
Otezla®
(apremilast) tablets
10 mg, 20 mg, 30 mg
28 Day
Treatment
Initiation Pack
This pack contains the following
for titration over 5 days up to the
prescribed dose of 30 mg:
Four - 10 mg tablets
Four - 20 mg tablets
Forty-seven - 30 mg tablets
55 TABLETS
Rx Only
AMGEN®
Principal Display Panel 14 Day Treatment Initiation Pack 55513 485 95
NDC 55513-485-95
Otezla®
(apremilast) tablets
10 mg, 20 mg, 30 mg
14 Day
Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
This pack contains the following
for titration over 5 days up to the
prescribed dose of 30 mg:
Four - 10 mg tablets
Four - 20 mg tablets
Nineteen - 30 mg tablets
27 TABLETS
Rx Only
AMGEN®
Principal Display Panel 14 Day Treatment Initiation Pack 55513 485 96
NDC 55513-485-96
14 Day Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
Each pack contains the following for titration
over 5 days up to the prescribed dose of 30 mg:
Four - 10 mg tablets
Four - 20 mg tablets
Nineteen - 30 mg tablets
Five starter packs each containing 27 TABLETS
Rx Only
AMGEN®
Otezla®
(apremilast) tablets
Principal Display Panel 14 Day Treatment Initiation Pack 55513 996 95
NDC 55513-996-95
14 Day Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
Each pack contains the following for titration over
3 days up to the prescribed dose of 20 mg:
Four - 10 mg tablets
Twenty-three - 20 mg tablets
Three starter packs each containing 27 TABLETS
Rx Only
AMGEN®
Otezla®
(apremilast) tablets
Principal Display Panel 14 Day Treatment Initiation Pack 55513 996 96
NDC 55513-996-96
Otezla®
(apremilast) tablets
10 mg, 20 mg
14 Day
Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
This pack contains the following
for titration over 3 days up to the
prescribed dose of 20 mg:
Four - 10 mg tablets
Twenty-three - 20 mg tablets
27 TABLETS
Rx Only
AMGEN®
Principal Display Panel 28 Day Treatment Initiation Pack 55513 508 55
NDC 55513-508-55
Otezla®
(apremilast) tablets
10 mg, 20 mg
28 Day
Treatment
Initiation Pack
This pack contains the following
for titration over 3 days up to the
prescribed dose of 20 mg:
Four - 10 mg tablets
Fifty-one - 20 mg tablets
55 TABLETS
Rx Only
AMGEN®
Principal Display Panel 28 Day Treatment Initiation Pack 55513 516 41
NDC 55513-516-41
Otezla®
(apremilast) tablets
10 mg, 20 mg, 30 mg
Otezla XR™
(apremilast)
extended-release tablets
75 mg
28 Day
Treatment
Initiation Pack
This pack contains the following for titration
up to 30 mg twice daily and transition to the
prescribed dose of 75 mg once daily.
Four - 10 mg tablets
Four - 20 mg tablets
Nineteen - 30 mg tablets
Fourteen - 75 mg extended-release tablets
Contains a total of 41 tablets.
Rx Only
AMGEN
Structured Label Content
Section 42229-5 (42229-5)
Adult Patients with Psoriatic Arthritis, Plaque Psoriasis, or Behçet's Disease
The recommended initial dosage titration from Day 1 to Day 5 is shown in Table 1. Following the 5-day titration with OTEZLA, the recommended maintenance dosage is OTEZLA 30 mg twice daily or OTEZLA XR 75 mg once daily taken orally starting on Day 6. This titration is intended to reduce the gastrointestinal symptoms associated with initial therapy.
|
OTEZLA Dosage Titration OTEZLA tablets should be used for the initial titration regardless of whether OTEZLA or OTEZLA XR will be used for the maintenance dosage.
|
OTEZLA/OTEZLA XR Maintenance Dosage | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 & thereafter | ||||
| AM | AM | PM | AM | PM | AM | PM | AM | PM | |
| BID = twice daily; QD = once daily | |||||||||
| 10 mg | 10 mg | 10 mg | 10 mg | 20 mg | 20 mg | 20 mg | 20 mg | 30 mg | OTEZLA 30 mg BID OR OTEZLA XR 75 mg QD |
Section 44425-7 (44425-7)
Storage and Handling
Store OTEZLA tablets below 30°C (86°F).
Store OTEZLA XR tablets between 20°C and 25°C (68°F and 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
11 Description (11 DESCRIPTION)
The active ingredient in OTEZLA/OTEZLA XR tablets is apremilast. Apremilast drug substance is non-hygroscopic. Apremilast drug substance is practically insoluble in water and slightly soluble in alcohol.
Apremilast is a phosphodiesterase 4 (PDE4) inhibitor. Apremilast is known chemically as
N-[2-[(1S)-1-(3-ethoxy-4-methoxyphenyl)-2-(methylsulfonyl)ethyl]-2,3-dihydro-1,3-dioxo-1H-isoindol-4-yl]acetamide. Its empirical formula is C22H24N2O7S and the molecular weight is 460.5.
The chemical structure is:
OTEZLA (apremilast) tablets are supplied in 10 mg, 20 mg, and 30 mg strengths for oral administration. Each tablet contains apremilast as the active ingredient and the following inactive ingredients: croscarmellose sodium, iron oxide red, iron oxide yellow (20 and 30 mg only), iron oxide black (30 mg only), lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide.
OTEZLA XR (apremilast) extended-release tablets are supplied in a 75 mg strength for oral administration. Each tablet contains apremilast as the active ingredient and the following inactive ingredients: cellulose acetate, colloidal silicon dioxide, ferrosoferric oxide, hydroxypropyl methylcellulose acetate succinate, hypromellose, iron oxide red, magnesium stearate, mannitol, microcrystalline cellulose, polyethylene glycol, polyethylene oxide, sodium chloride, titanium dioxide.
5.3 Depression
Treatment with apremilast is associated with an increased incidence of depression. Before using OTEZLA/OTEZLA XR in patients with a history of depression and/or suicidal thoughts or behavior, carefully weigh the risks and benefits of treatment with OTEZLA/OTEZLA XR. Advise patients, their caregivers, and families of the need to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes, and if such changes occur to contact their healthcare provider. Carefully evaluate the risks and benefits of continuing treatment with OTEZLA/OTEZLA XR if such events occur.
8.5 Geriatric Use
Of the 1493 patients who enrolled in Trials PsA-1, PsA-2, and PsA-3, a total of 146 (9.8%) psoriatic arthritis patients were 65 years of age and older, including 19 (1.3%) patients 75 years and older. No overall differences were observed in the safety profile of geriatric patients ≥ 65 years of age and younger adult patients < 65 years of age in the clinical trials.
Of the 1257 subjects who enrolled in two placebo-controlled plaque psoriasis trials (PSOR-1 and PSOR-2), a total of 108 (8.6%) plaque psoriasis patients were 65 years of age and older, including 9 (0.7%) patients who were 75 years of age and older. No overall differences were observed in the safety or effectiveness in geriatric patients ≥ 65 years of age and younger adult patients < 65 years of age in the clinical trials.
Because patients 65 years of age or older may be at a higher risk of complications such as volume depletion or hypotension from severe diarrhea, nausea, or vomiting, monitor geriatric patients closely for such complications [see Warnings and Precautions (5.2)].
4 Contraindications (4 CONTRAINDICATIONS)
OTEZLA/OTEZLA XR is contraindicated in patients with a known hypersensitivity to apremilast or to any of the excipients in the formulation [see Warnings and Precautions (5.1), see Adverse Reactions (6.1)].
5.4 Weight Decrease
Weight loss may occur in adult or pediatric patients treated with OTEZLA/OTEZLA XR.
Regularly monitor the weight of patients treated with OTEZLA/OTEZLA XR. If unexplained or clinically significant weight loss occurs, evaluate weight loss and consider discontinuation of OTEZLA/OTEZLA XR [see Adverse Reactions (6.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Diarrhea, Nausea, and Vomiting [see Warnings and Precautions (5.2)]
- Depression [see Warnings and Precautions (5.3)]
- Weight Decrease [see Warnings and Precautions (5.4)]
- Drug Interactions [see Warnings and Precautions (5.5)]
1.2 Plaque Psoriasis
OTEZLA/OTEZLA XR is indicated for the treatment of adult patients with plaque psoriasis who are candidates for phototherapy or systemic therapy.
OTEZLA is indicated for the treatment of pediatric patients 6 years of age and older and weighing at least 20 kg with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy.
OTEZLA XR is indicated for the treatment of pediatric patients 6 years of age and older and weighing at least 50 kg with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy.
5.1 Hypersensitivity
Hypersensitivity reactions, including cases of angioedema and anaphylaxis, have been reported during post marketing surveillance. Avoid the use of OTEZLA/OTEZLA XR in patients with known hypersensitivity to apremilast or to any of the excipients in the formulation. If signs or symptoms of serious hypersensitivity reactions develop during treatment, discontinue OTEZLA/OTEZLA XR and institute appropriate therapy.
8.6 Renal Impairment
OTEZLA pharmacokinetics were characterized in adult subjects with mild, moderate, and severe renal impairment as defined by a creatinine clearance of 60-89, 30-59, and less than 30 mL per minute, respectively, by the Cockcroft–Gault equation. No dosage adjustment is needed in patients with mild or moderate renal impairment. In adult patients with severe renal impairment, reduce the maintenance dosage to OTEZLA 30 mg once daily. In pediatric patients 6 years of age and older and weighing at least 20 kg with psoriatic arthritis or moderate to severe plaque psoriasis and severe renal impairment, reduce the maintenance dosage to OTEZLA 30 mg once daily for pediatric patients weighing at least 50 kg and to OTEZLA 20 mg once daily for pediatric patients weighing 20 kg to less than 50 kg [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
The pharmacokinetics of OTEZLA XR has not been evaluated in patients with renal impairment. In patients with severe renal impairment, it is recommended that OTEZLA be used instead of OTEZLA XR [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In clinical studies, apremilast decreased IL-17, IL-22 and TNF-alpha levels circulating in blood and reduced expression of these pro-inflammatory cytokine genes in skin. The clinical implications of these pharmacodynamic responses remain unclear.
5.5 Drug Interactions
Co-administration of strong cytochrome P450 enzyme inducer, rifampin, resulted in a reduction of systemic exposure of apremilast, which may result in a loss of efficacy of OTEZLA/OTEZLA XR. Therefore, the use of cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) with OTEZLA/OTEZLA XR is not recommended [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Apremilast pharmacokinetics were characterized in subjects with moderate (Child Pugh B) and severe (Child Pugh C) hepatic impairment. No dosage adjustment is necessary in these patients.
1 Indications and Usage (1 INDICATIONS AND USAGE)
OTEZLA/OTEZLA XR, an inhibitor of phosphodiesterase 4 (PDE4), is indicated for the treatment of:
Adult patients with:
- Active psoriatic arthritis (1.1)
- Plaque psoriasis who are candidates for phototherapy or systemic therapy (1.2)
- Oral ulcers associated with Behçet's Disease (1.3)
Pediatric patients 6 years of age and older with:
- Active psoriatic arthritis (1.1)
- Moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy (1.2)
In the pediatric population, OTEZLA is indicated for patients weighing at least 20 kg, and OTEZLA XR is indicated for patients weighing at least 50 kg.
1.1 Psoriatic Arthritis
OTEZLA is indicated for the treatment of adult patients and pediatric patients 6 years of age and older and weighing at least 20 kg with active psoriatic arthritis.
OTEZLA XR is indicated for the treatment of adult patients and pediatric patients 6 years of age and older and weighing at least 50 kg with active psoriatic arthritis.
12.1 Mechanism of Action
Apremilast is an oral small molecule inhibitor of phosphodiesterase 4 (PDE4) specific for cyclic adenosine monophosphate (cAMP). PDE4 inhibition results in increased intracellular cAMP levels. The specific mechanism(s) by which apremilast exerts its therapeutic action is not well defined.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity: Cases of angioedema and anaphylaxis have been reported during post marketing surveillance. Avoid the use of OTEZLA/OTEZLA XR in patients with known hypersensitivity to apremilast or to any of the excipients in the formulation. If signs or symptoms of serious hypersensitivity reactions develop during treatment, discontinue OTEZLA/OTEZLA XR and institute appropriate therapy (5.1)
- Diarrhea, Nausea, and Vomiting: Consider OTEZLA/OTEZLA XR dosage reduction or suspension if patients develop severe diarrhea, nausea, or vomiting (5.2)
- Depression: Advise patients, their caregivers, and families to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes and if such changes occur to contact their healthcare provider. Carefully weigh risks and benefits of treatment with OTEZLA/OTEZLA XR in patients with a history of depression and/or suicidal thoughts or behavior (5.3)
- Weight Decrease: Monitor weight regularly. If unexplained or clinically significant weight loss occurs, evaluate weight loss and consider discontinuation of OTEZLA/OTEZLA XR (5.4)
- Drug Interactions: Use with strong cytochrome P450 enzyme inducers (e.g., rifampin, phenobarbital, carbamazepine, phenytoin) is not recommended because loss of efficacy may occur (5.5, 7.1)
7.1 Strong Cyp450 Inducers (7.1 Strong CYP450 Inducers)
Co-administration with strong CYP450 inducers (such as rifampin) decreases apremilast exposure and may result in loss of efficacy of OTEZLA/OTEZLA XR [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.3)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
To reduce the risk of gastrointestinal symptoms, titrate to recommended dosage as follows:
- Adults with Psoriatic Arthritis, Plaque Psoriasis, or Behçet's Disease
- Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis
-
Dosage in Patients with Severe Renal Impairment:
- Adult Patients: For initial dosage titration, titrate using only morning schedule listed in Table 1 and skip afternoon doses. Recommended maintenance dosage is OTEZLA 30 mg once daily (2.3)
- Pediatric Patients 6 Years of Age and Older and Weighing at Least 20 kg with Psoriatic Arthritis or Moderate to Severe Plaque Psoriasis: For initial dosage titration, titrate using only morning schedule for appropriate body weight category in Table 2 and skip afternoon doses (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths:
- 10-mg pink tablet engraved with "APR" on one side and "10" on the other side
- 20-mg brown tablet engraved with "APR" on one side and "20" on the other side
- 30-mg beige tablet engraved with "APR" on one side and "30" on the other side
OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
14.1 Adult Psoriatic Arthritis
The safety and efficacy of OTEZLA was evaluated in three multicenter, randomized, double-blind, placebo-controlled trials (PsA-1 [NCT01172938], PsA-2 [NCT01212757], and PsA-3 [NCT01212770]) of similar design. A total of 1493 adult subjects with active PsA (≥ 3 swollen joints and ≥ 3 tender joints) despite prior or current treatment with disease-modifying antirheumatic drug (DMARD) therapy were randomized. Subjects enrolled in these trials had a diagnosis of PsA for at least 6 months. One qualifying psoriatic skin lesion of at least 2 cm in diameter was required in Trial PsA-3. Previous treatment with a biologic, including TNF blockers was allowed (up to 10% could be TNF blocker therapeutic failures). Across the three trials, subjects were randomly assigned to placebo (n = 496), OTEZLA 20 mg (n = 500), or OTEZLA 30 mg (n = 497) given orally twice daily. Titration was used over the first 5 days [see Dosage and Administration (2.1)]. Subjects were allowed to receive stable doses of concomitant methotrexate [MTX (≤ 25 mg/week)], sulfasalazine [SSZ (≤ 2 g/day)], leflunomide [LEF (≤ 20 mg/day)], low dose oral corticosteroids (equivalent to ≤ 10 mg of prednisone a day), and/or nonsteroidal anti-inflammatory drugs (NSAIDs) during the trial. Treatment assignments were stratified based on small molecule DMARD use at baseline in Trials PsA-1, PsA-2 and PsA-3. There was an additional stratification of body surface area (BSA) > 3% with psoriasis in Trial PsA-3. The subjects who were therapeutic failures of > 3 agents for PsA (small molecules or biologics), or > 1 biologic TNF blocker were excluded.
The primary endpoint was the percentage of subjects achieving American College of Rheumatology (ACR) 20 response at Week 16. Placebo-controlled efficacy data were collected and analyzed through Week 24. Subjects whose tender and swollen joint counts had not improved by at least 20% were considered non-responders at Week 16. Placebo non-responders were re-randomized 1:1 in a blinded fashion to either OTEZLA 20 mg twice daily or 30 mg twice daily following the titration schema [see Dosage and Administration (2.1)]. OTEZLA subjects remained on their initial treatment. At Week 24, all remaining placebo subjects were re-randomized to either 20 mg twice daily or 30 mg twice daily.
Subjects with subtypes of PsA were enrolled across the three trials, including symmetric polyarthritis (62.0%), asymmetric oligoarthritis (27.0%), distal interphalangeal (DIP) joint arthritis (6.0%), arthritis mutilans (3.0%), and predominant spondylitis (2.1%). The median duration of PsA disease was 5 years. Subjects received concomitant therapy with at least one DMARD (65.0%), MTX (55.0%), SSZ (9.0%), LEF (7.0%), low dose oral corticosteroids (14.0%), and NSAIDs (71.0%). Prior treatment with small molecule DMARDs only was reported in 76.0% of subjects and prior treatment with biologic DMARDs was reported in 22.0% of subjects, which includes 9.0% who had failed prior biologic DMARD treatment.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
-
Administration Instructions
Instruct patients to take OTEZLA/OTEZLA XR only as prescribed [see Dosage and Administration (2.1 and 2.3)]. Advise patients to take OTEZLA/OTEZLA XR with or without food. Instruct patients to swallow tablets whole and not to crush, split, or chew prior to swallowing [see Dosage and Administration (2.4)]. -
Hypersensitivity
Inform patients that hypersensitivity reactions can occur following administration of OTEZLA/OTEZLA XR. Instruct patients to contact their healthcare provider if they experience symptoms of an allergic reaction [see Warnings and Precautions (5.1)]. -
Diarrhea, Nausea, and Vomiting
Advise patients of the potential complications of severe diarrhea, nausea, or vomiting and instruct them to contact their healthcare provider if they experience these adverse reactions, especially if the patient is 65 years of age or older [see Warnings and Precautions (5.2)]. -
Depression
Inform patients that treatment with OTEZLA/OTEZLA XR is associated with an increased incidence of depression. Advise patients, their caregivers, and families of the need to be alert for the emergence or worsening of depression, suicidal thoughts or other mood changes, and if such changes occur to contact their healthcare provider [see Warnings and Precautions (5.3)]. -
Weight Decrease
Inform patients that treatment with OTEZLA/OTEZLA XR is associated with potential weight loss. Instruct patients or caregivers to have their or their child's weight monitored regularly and, if unexplained or clinically significant weight loss occurs, to contact their healthcare provider for evaluation of the weight loss [see Warnings and Precautions (5.4)]. -
Pregnancy
Advise pregnant patients and patients of reproductive potential of the potential risk to a fetus. Advise patients to inform their prescriber of a known or suspected pregnancy [see Use in Specific Populations (8.1)]. -
Residual Tablet Shell
Patients receiving OTEZLA XR may notice an inert tablet shell passing in the stool or via colostomy. Patients should be informed that the active medication has already been absorbed by the time the patient sees the inert tablet shell.
5.2 Diarrhea, Nausea, and Vomiting
There have been reports of severe diarrhea, nausea, and vomiting associated with the use of OTEZLA. Most events occurred within the first few weeks of treatment. In some cases, patients were hospitalized. Patients 65 years of age or older and patients taking medications that can lead to volume depletion or hypotension may be at a higher risk of complications from severe diarrhea, nausea, or vomiting. Monitor patients who are more susceptible to complications of diarrhea or vomiting. Patients who reduced dosage or discontinued OTEZLA generally improved quickly. Consider OTEZLA/OTEZLA XR dosage reduction or suspension if patients develop severe diarrhea, nausea, or vomiting.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
OTEZLA is available as diamond-shaped, film-coated tablets in the following dosage strengths: 10 mg pink tablet engraved with "APR" on one side and "10" on the other side; 20 mg brown tablet engraved with "APR" on one side and "20" on the other side; 30 mg beige tablet engraved with "APR" on one side and "30" on the other side.
Tablets are supplied in the strengths and package configurations listed in Table 14.
| Package configuration | Tablet strength | NDC number |
|---|---|---|
| Configurations for 30 mg BID Dosage | ||
| 28-day treatment initiation pack | 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each), 4 tablets (20 mg each), and 47 tablets (30 mg each) | 55513-369-55 |
| 60-count bottle | 30 mg | 55513-137-60 |
| Configurations for 20 mg BID Dosage | ||
| 28-day treatment initiation pack | 55-tablet blister pack including tablets for titration and maintenance dosage: 4 tablets (10 mg each) and 51 tablets (20 mg each) | 55513-508-55 |
| 60-count bottle | 20 mg | 55513-497-60 |
OTEZLA XR is available as 75 mg round, biconvex, pink, film-coated extended-release tablets with "APR 75" printed in black on one side and a hole or indentation on either side of the tablet, which may or may not be visible.
Tablets are supplied in the strengths and package configurations listed in Table 15.
| Package configuration | Tablet strength | NDC number |
|---|---|---|
| 28-day treatment initiation pack | 41-tablet blister titration pack including tablets for titration and maintenance dosage: OTEZLA: 4 tablets (10 mg each), 4 tablets (20 mg each), and 19 tablets (30 mg each) and OTEZLA XR: 14 tablets (75 mg each) |
55513-516-41 |
| Bottles of 30 |
OTEZLA XR: 30 tablets (75 mg each) |
55513-519-30 |
2.4 Important Administration Instructions
- Administer OTEZLA/OTEZLA XR with or without food.
- Swallow tablets whole. Do not crush, split, or chew.
2.2 Switching Between Otezla and Otezla Xr (2.2 Switching Between OTEZLA and OTEZLA XR)
Patients treated with OTEZLA 30 mg twice daily may be switched to OTEZLA XR 75 mg once daily the day following the last dose of OTEZLA 30 mg.
Patients treated with OTEZLA XR 75 mg once daily may be switched to OTEZLA 30 mg twice daily the day following the last dose of OTEZLA XR 75 mg.
14.4 Adult Mild to Moderate Plaque Psoriasis
A multicenter, randomized, double-blind, placebo-controlled trial (PSOR-4 [NCT03721172]) was conducted in 595 adult subjects with mild to moderate plaque psoriasis (BSA involvement of 2-15%, sPGA score of 2-3 [mild or moderate disease], and PASI score of 2-15). Enrolled subjects had an inadequate response or were intolerant to at least one topical therapy and had not received prior biologic therapy. Subjects were allowed to use unmedicated emollients for lesions on non-scalp areas of the body and non-medicated shampoos for lesions on the scalp.
Subjects were randomized 1:1 to receive either OTEZLA 30 mg twice daily (n = 297) or placebo twice daily (n = 298) for 16 weeks. At Week 16, the placebo group was switched to receive OTEZLA and the OTEZLA group remained on drug through Week 32. The primary endpoint was the proportion of subjects who achieved an sPGA response (defined as an sPGA score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline) at Week 16. Subjects with mild disease (sPGA = 2 at baseline) were required to be clear (sPGA = 0) to achieve an sPGA response. Other evaluated endpoints include the proportion of subjects with a Whole Body Itch NRS response (defined as a ≥ 4-point reduction from baseline) at Week 16 among subjects with a baseline Whole Body Itch NRS ≥ 4 and the proportion of subjects with an ScPGA response (defined as an ScPGA score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline) at Week 16 among subjects with a baseline ScPGA score ≥ 2.
Subjects ranged in age from 18 to 85 years, with an overall median age of 50 years. The mean baseline BSA involvement was 6.4%, the mean baseline PASI score was 6.5, and the proportions of subjects with an sPGA score of 2 (mild) and 3 (moderate) at baseline were 30.6% and 69.4%, respectively.
14.2 Adult Moderate to Severe Plaque Psoriasis
Two multicenter, randomized, double-blind, placebo-controlled trials (PSOR-1 [NCT01194219] and PSOR-2 [NCT01232283]) enrolled a total of 1257 subjects 18 years of age and older with moderate to severe plaque psoriasis [body surface area (BSA) involvement of ≥ 10%, static Physician Global Assessment (sPGA) of ≥ 3 (moderate or severe disease), Psoriasis Area and Severity Index (PASI) score ≥ 12, candidates for phototherapy or systemic therapy]. Subjects were allowed to use low potency topical corticosteroids on the face, axilla and groin. Subjects with plaque psoriasis of the scalp were allowed to use coal tar shampoo and/or salicylic acid scalp preparations on scalp lesions.
Trial PSOR-1 enrolled 844 subjects and Trial PSOR-2 enrolled 413 subjects. In both trials, subjects were randomized 2:1 to OTEZLA 30 mg twice daily (BID) or placebo for 16 weeks. Both trials assessed the proportion of subjects who achieved PASI-75 at Week 16 and the proportion of subjects who achieved an sPGA score of clear (0) or almost clear (1) at Week 16. Across both trials, subjects ranged in age from 18 to 83 years, with an overall median age of 46 years. The mean baseline BSA involvement was 25.2% (median 21.0%), the mean baseline PASI score was 19.1 (median 16.8), and the proportion of subjects with an sPGA score of 3 (moderate) and 4 (severe) at baseline were 70.0% and 29.8%, respectively. Approximately 30% of all subjects had received prior phototherapy and 54% had received prior conventional systemic and/or biologic therapy for the treatment of psoriasis with 37% receiving prior conventional systemic therapy and 30% receiving prior biologic therapy. Approximately one-third of subjects had not received prior phototherapy, conventional systemic nor biologic therapy. A total of 18% of subjects had a history of psoriatic arthritis.
Principal Display Panel 60 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 60 Tablet Bottle Label)
NDC 55513-137-60
Otezla®
(apremilast) tablets
30 mg
Rx Only
60 Tablets
1.3 Oral Ulcers Associated With Behçet's Disease (1.3 Oral Ulcers Associated with Behçet's Disease)
OTEZLA/OTEZLA XR is indicated for the treatment of adult patients with oral ulcers associated with Behçet's Disease.
14.3 Pediatric Moderate to Severe Plaque Psoriasis
A multicenter, randomized, double-blind, placebo-controlled trial (PSOR-6 [NCT03701763]) was conducted in 245 pediatric subjects 6 to 17 years of age (inclusive) with moderate to severe plaque psoriasis who were candidates for phototherapy or systemic therapy. Subjects had an sPGA score of ≥ 3 (moderate or severe disease), BSA involvement of ≥ 10%, and PASI score of ≥ 12, with psoriasis that was inadequately controlled by or inappropriate for topical therapy. Subjects were allowed to use low potency or weak topical corticosteroids on the face, axilla, and groin and unmedicated skin moisturizers for body lesions only.
Subjects were randomized 2:1 to receive either OTEZLA (n = 163) or placebo (n = 82) for 16 weeks. Subjects with a baseline weight of 20 kg to < 50 kg received OTEZLA 20 mg twice daily or placebo twice daily, and subjects with a baseline weight ≥ 50 kg received OTEZLA 30 mg twice daily or placebo twice daily. At Week 16, the placebo group was switched to receive OTEZLA (with dosage based on baseline weight) and the OTEZLA group remained on drug (according to their original dosing assignment) through Week 52. The primary endpoint was the proportion of subjects who achieved an sPGA response (defined as a score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline) at Week 16. The key secondary endpoint was the proportion of subjects who achieved a PASI-75 response (at least a 75% reduction in PASI score from baseline) at Week 16.
Enrolled subjects ranged in age from 6 to 17 years, with a median age of 13 years; 41.2% of subjects were 6 to 11 years of age and 58.8% of subjects were 12 to 17 years of age. Of the enrolled subjects, 52.2% were female. For race, 86.9% were White, 3.7% were Asian, 3.3% were Black or African American, 0.8% were American Indian or Alaskan Native, and 5.3% were not reported or unknown. For ethnicity, 81.6% of subjects identified as not Hispanic or Latino, 13.1% identified as Hispanic or Latino, and 5.3% were not reported or unknown. The mean baseline BSA involvement was 31.5% (median 26.0%), the mean baseline PASI score was 19.8 (median 17.2), and the proportions of subjects with an sPGA score of 3 (moderate) and 4 (severe) at baseline were 75.5% and 24.5%, respectively. Of the enrolled subjects, 82.9% did not receive prior conventional systemic therapy and 94.3% were biologic-naïve.
Principal Display Panel 75 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 75 mg Tablet Bottle Label)
Once-daily
NDC 55513-519-30
Otezla XR™
(apremilast)
extended-release tablets
75 mg
Rx Only
30 Tablets
14.5 Adult Oral Ulcers Associated With Behçet's Disease (14.5 Adult Oral Ulcers Associated with Behçet's Disease)
A multicenter, randomized, placebo-controlled trial (BCT-002 [NCT02307513]) enrolled a total of 207 adult subjects with BD with active oral ulcers. Subjects were previously treated with at least one nonbiologic BD medication and were candidates for systemic therapy. Subjects met the International Study Group (ISG) Criteria for BD. Subjects had at least 2 oral ulcers at screening and at least 2 oral ulcers at randomization and without currently active major organ involvement. Concomitant treatment for BD was not allowed.
Subjects were randomized 1:1 to receive either OTEZLA 30 mg twice daily (n = 104) or placebo (n = 103) for 12 weeks. After Week 12, all subjects received OTEZLA 30 mg twice daily.
Efficacy was assessed based on the number and pain of oral ulcers.
Subjects ranged in age from 19 to 72 years, with a mean age of 40 years. The mean duration of BD was 6.84 years. All subjects had a history of recurrent oral ulcers that were currently active. Subjects had a history of skin lesions (98.6%), genital ulcers (90.3%), musculoskeletal manifestations (72.5%), ocular manifestations (17.4%), central nervous system (9.7%), gastrointestinal (GI) manifestations (9.2%) and vascular involvement (1.4%). The mean baseline oral ulcer counts were 4.2 and 3.9 in the OTEZLA and placebo groups, respectively.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies were conducted in mice and rats with apremilast to evaluate its carcinogenic potential. No evidence of apremilast-induced tumors was observed in mice at oral doses up to approximately 9-times the MRHD on an AUC basis (1000 mg/kg/day) or in rats at oral doses up to approximately 0.08- and 1.1-times the MRHD, (20 mg/kg/day in males and 3 mg/kg/day in females, respectively).
Apremilast tested negative in the Ames assay, in vitro chromosome aberration assay of human peripheral blood lymphocytes, and the in vivo mouse micronucleus assay.
In a fertility study of male mice, apremilast at oral doses up to approximately 3-times the MRHD based on AUC (up to 50 mg/kg/day) produced no effects on male fertility. In a fertility study of female mice, apremilast was administered at oral doses of 10, 20, 40, or 80 mg/kg/day. At doses ≥ 1.8-times the MRHD (≥ 20 mg/kg/day), estrous cycles were prolonged, due to lengthening of diestrus which resulted in a longer interval until mating. Mice that became pregnant at doses of 20 mg/kg/day and greater also had increased incidences of early post-implantation losses. There was no effect of apremilast approximately 1.0-times the MRHD (10 mg/kg/day).
Principal Display Panel 28 Day Treatment Initiation Pack (PRINCIPAL DISPLAY PANEL - 28 Day Treatment Initiation Pack)
NDC 55513-369-55
Otezla®
(apremilast) tablets
10 mg, 20 mg, 30 mg
28 Day
Treatment
Initiation Pack
This pack contains the following
for titration over 5 days up to the
prescribed dose of 30 mg:
Four - 10 mg tablets
Four - 20 mg tablets
Forty-seven - 30 mg tablets
55 TABLETS
Rx Only
AMGEN®
Principal Display Panel 14 Day Treatment Initiation Pack 55513 485 95 (PRINCIPAL DISPLAY PANEL - 14 Day Treatment Initiation Pack - 55513-485-95)
NDC 55513-485-95
Otezla®
(apremilast) tablets
10 mg, 20 mg, 30 mg
14 Day
Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
This pack contains the following
for titration over 5 days up to the
prescribed dose of 30 mg:
Four - 10 mg tablets
Four - 20 mg tablets
Nineteen - 30 mg tablets
27 TABLETS
Rx Only
AMGEN®
Principal Display Panel 14 Day Treatment Initiation Pack 55513 485 96 (PRINCIPAL DISPLAY PANEL - 14 Day Treatment Initiation Pack - 55513-485-96)
NDC 55513-485-96
14 Day Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
Each pack contains the following for titration
over 5 days up to the prescribed dose of 30 mg:
Four - 10 mg tablets
Four - 20 mg tablets
Nineteen - 30 mg tablets
Five starter packs each containing 27 TABLETS
Rx Only
AMGEN®
Otezla®
(apremilast) tablets
Principal Display Panel 14 Day Treatment Initiation Pack 55513 996 95 (PRINCIPAL DISPLAY PANEL - 14 Day Treatment Initiation Pack - 55513-996-95)
NDC 55513-996-95
14 Day Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
Each pack contains the following for titration over
3 days up to the prescribed dose of 20 mg:
Four - 10 mg tablets
Twenty-three - 20 mg tablets
Three starter packs each containing 27 TABLETS
Rx Only
AMGEN®
Otezla®
(apremilast) tablets
Principal Display Panel 14 Day Treatment Initiation Pack 55513 996 96 (PRINCIPAL DISPLAY PANEL - 14 Day Treatment Initiation Pack - 55513-996-96)
NDC 55513-996-96
Otezla®
(apremilast) tablets
10 mg, 20 mg
14 Day
Treatment
Initiation Pack
SAMPLE - NOT FOR SALE
This pack contains the following
for titration over 3 days up to the
prescribed dose of 20 mg:
Four - 10 mg tablets
Twenty-three - 20 mg tablets
27 TABLETS
Rx Only
AMGEN®
Principal Display Panel 28 Day Treatment Initiation Pack 55513 508 55 (PRINCIPAL DISPLAY PANEL - 28 Day Treatment Initiation Pack - 55513-508-55)
NDC 55513-508-55
Otezla®
(apremilast) tablets
10 mg, 20 mg
28 Day
Treatment
Initiation Pack
This pack contains the following
for titration over 3 days up to the
prescribed dose of 20 mg:
Four - 10 mg tablets
Fifty-one - 20 mg tablets
55 TABLETS
Rx Only
AMGEN®
Principal Display Panel 28 Day Treatment Initiation Pack 55513 516 41 (PRINCIPAL DISPLAY PANEL - 28 Day Treatment Initiation Pack - 55513-516-41)
NDC 55513-516-41
Otezla®
(apremilast) tablets
10 mg, 20 mg, 30 mg
Otezla XR™
(apremilast)
extended-release tablets
75 mg
28 Day
Treatment
Initiation Pack
This pack contains the following for titration
up to 30 mg twice daily and transition to the
prescribed dose of 75 mg once daily.
Four - 10 mg tablets
Four - 20 mg tablets
Nineteen - 30 mg tablets
Fourteen - 75 mg extended-release tablets
Contains a total of 41 tablets.
Rx Only
AMGEN
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:37.075866 · Updated: 2026-03-14T22:44:06.575433