Daptomycin For Injection
f69e9306-ffac-41c9-b277-a6d784f2e341
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Daptomycin for Injection is a lipopeptide antibacterial indicated for the treatment of: Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) ( 1.1 ) and, Staphylococcus aureus bloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, ( 1.2 ) Staphylococcus aureus bloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age). ( 1.3 ) Limitations of Use: Daptomycin for Injection is not indicated for the treatment of pneumonia. ( 1.4 ) Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus . ( 1.4 ) Daptomycin for Injection is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. ( 1.4 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5 )
Dosage and Administration
Adult Patients Administer to adult patients intravenously in 0.9% sodium chloride, either by injection over a 2-minute period or by infusion over a 30-minute period. ( 2.1 , 2.7 ) Recommended dosage regimen for adult patients ( 2.2 , 2.4 , 2.6 ): Creatinine Clearance (CL CR ) Dosage Regimen cSSSI For 7 to 14 days S. aureus Bacteremia For 2 to 6 weeks ≥30 mL/min 4 mg/kg once every 24 hours 6 mg/kg once every 24 hours <30 mL/min, including hemodialysis and CAPD 4 mg/kg once every 48 hours* 6 mg/kg once every 48 hours* *Administered following hemodialysis on hemodialysis days. Pediatric Patients Unlike in adults, do NOT administer by injection over a two (2) minute period to pediatric patients. ( 2.1 , 2.7 ) Administer to pediatric patients intravenously in 0.9% sodium chloride, by infusion over a 30-or 60-minute period, based on age. ( 2.1 , 2.7 ) Recommended dosage regimen for pediatric patients (1 to 17 years of age) with cSSSI, based on age ( 2.3 ): Age group Dosage* Duration of therapy 12 to 17 years 5 mg/kg once every 24 hours infused over 30 minutes Up to 14 days 7 to 11 years 7 mg/kg once every 24 hours infused over 30 minutes 2 to 6 years 9 mg/kg once every 24 hours infused over 60 minutes 1 to less than 2 years 10 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. Recommended dosage regimen for pediatric patients (1 to 17 years of age) with S. aureus bacteremia, based on age ( 2.5 ): Age group Dosage* Duration of therapy 12 to 17 years 7 mg/kg once every 24 hours infused over 30 minutes Up to 42 days 7 to 11 years 9 mg/kg once every 24 hours infused over 30 minutes 1 to 6 years 12 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. There are other formulations of daptomycin that have differences concerning storage and reconstitution. Carefully follow the reconstitution and storage procedures in labeling. ( 2.7 ) Do not use in conjunction with ReadyMED ® elastomeric infusion pumps in adult and pediatric patients. ( 2.9 )
Contraindications
Daptomycin for injection is contraindicated in patients with known hypersensitivity to daptomycin. [see Warnings and Precautions ( 5.1 )].
Warnings and Precautions
Anaphylaxis/hypersensitivity reactions (including life-threatening): Discontinue daptomycin and treat signs/symptoms. ( 5.1 ) Myopathy and rhabdomyolysis: Monitor CPK levels and follow muscle pain or weakness; if elevated CPK or myopathy occurs, consider discontinuation of daptomycin. ( 5.2 ) Eosinophilic pneumonia: Discontinue daptomycin and consider treatment with systemic steroids. ( 5.3 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue daptomycin and institute appropriate treatment. ( 5.4 ) Tubulointerstitial Nephritis (TIN): Discontinue daptomycin and institute appropriate treatment. ( 5.5 ) Peripheral neuropathy: Monitor for neuropathy and consider discontinuation. ( 5.6 ) Potential nervous system and/or muscular system effects in pediatric patients younger than 12 months: Avoid use of daptomycin in this age group. ( 5.7 ) Clostridioides difficile –associated diarrhea: Evaluate patients if diarrhea occurs. ( 5.8 ) Persisting or relapsing S. aureus bacteremia/endocarditis: Perform susceptibility testing and rule out sequestered foci of infection. ( 5.9 ) Decreased efficacy was observed in adult patients with moderate baseline renal impairment. ( 5.10 )
Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections: Anaphylaxis/Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Myopathy and Rhabdomyolysis [see Warnings and Precautions ( 5.2 )] Eosinophilic Pneumonia [see Warnings and Precautions ( 5.3 )] Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions ( 5.4 )] Tubulointerstitial Nephritis [see Warnings and Precautions ( 5.5 )] Peripheral Neuropathy [see Warnings and Precautions ( 5.6 )] Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time [see Warnings and Precautions ( 5.11 ) and Drug Interactions ( 7.2 )]
How Supplied
How Supplied Daptomycin for Injection is supplied as a sterile, nonpyrogenic, preservative-free, pale yellow to light brown lyophilized cake in single-dose vials as follows: NDC Daptomycin for Injection Package Factor 16729-434-05 350 mg Single-Dose Vial 1 vial per carton 16729-434-45 350 mg Single-Dose Vial 10 vials per carton The container closure is not made with natural rubber latex.
Storage and Handling
How Supplied Daptomycin for Injection is supplied as a sterile, nonpyrogenic, preservative-free, pale yellow to light brown lyophilized cake in single-dose vials as follows: NDC Daptomycin for Injection Package Factor 16729-434-05 350 mg Single-Dose Vial 1 vial per carton 16729-434-45 350 mg Single-Dose Vial 10 vials per carton The container closure is not made with natural rubber latex.
Description
Daptomycin for Injection is a lipopeptide antibacterial indicated for the treatment of: Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) ( 1.1 ) and, Staphylococcus aureus bloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, ( 1.2 ) Staphylococcus aureus bloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age). ( 1.3 ) Limitations of Use: Daptomycin for Injection is not indicated for the treatment of pneumonia. ( 1.4 ) Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus . ( 1.4 ) Daptomycin for Injection is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. ( 1.4 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5 )
Medication Information
Warnings and Precautions
Anaphylaxis/hypersensitivity reactions (including life-threatening): Discontinue daptomycin and treat signs/symptoms. ( 5.1 ) Myopathy and rhabdomyolysis: Monitor CPK levels and follow muscle pain or weakness; if elevated CPK or myopathy occurs, consider discontinuation of daptomycin. ( 5.2 ) Eosinophilic pneumonia: Discontinue daptomycin and consider treatment with systemic steroids. ( 5.3 ) Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue daptomycin and institute appropriate treatment. ( 5.4 ) Tubulointerstitial Nephritis (TIN): Discontinue daptomycin and institute appropriate treatment. ( 5.5 ) Peripheral neuropathy: Monitor for neuropathy and consider discontinuation. ( 5.6 ) Potential nervous system and/or muscular system effects in pediatric patients younger than 12 months: Avoid use of daptomycin in this age group. ( 5.7 ) Clostridioides difficile –associated diarrhea: Evaluate patients if diarrhea occurs. ( 5.8 ) Persisting or relapsing S. aureus bacteremia/endocarditis: Perform susceptibility testing and rule out sequestered foci of infection. ( 5.9 ) Decreased efficacy was observed in adult patients with moderate baseline renal impairment. ( 5.10 )
Indications and Usage
Daptomycin for Injection is a lipopeptide antibacterial indicated for the treatment of: Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) ( 1.1 ) and, Staphylococcus aureus bloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, ( 1.2 ) Staphylococcus aureus bloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age). ( 1.3 ) Limitations of Use: Daptomycin for Injection is not indicated for the treatment of pneumonia. ( 1.4 ) Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus . ( 1.4 ) Daptomycin for Injection is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. ( 1.4 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5 )
Dosage and Administration
Adult Patients Administer to adult patients intravenously in 0.9% sodium chloride, either by injection over a 2-minute period or by infusion over a 30-minute period. ( 2.1 , 2.7 ) Recommended dosage regimen for adult patients ( 2.2 , 2.4 , 2.6 ): Creatinine Clearance (CL CR ) Dosage Regimen cSSSI For 7 to 14 days S. aureus Bacteremia For 2 to 6 weeks ≥30 mL/min 4 mg/kg once every 24 hours 6 mg/kg once every 24 hours <30 mL/min, including hemodialysis and CAPD 4 mg/kg once every 48 hours* 6 mg/kg once every 48 hours* *Administered following hemodialysis on hemodialysis days. Pediatric Patients Unlike in adults, do NOT administer by injection over a two (2) minute period to pediatric patients. ( 2.1 , 2.7 ) Administer to pediatric patients intravenously in 0.9% sodium chloride, by infusion over a 30-or 60-minute period, based on age. ( 2.1 , 2.7 ) Recommended dosage regimen for pediatric patients (1 to 17 years of age) with cSSSI, based on age ( 2.3 ): Age group Dosage* Duration of therapy 12 to 17 years 5 mg/kg once every 24 hours infused over 30 minutes Up to 14 days 7 to 11 years 7 mg/kg once every 24 hours infused over 30 minutes 2 to 6 years 9 mg/kg once every 24 hours infused over 60 minutes 1 to less than 2 years 10 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. Recommended dosage regimen for pediatric patients (1 to 17 years of age) with S. aureus bacteremia, based on age ( 2.5 ): Age group Dosage* Duration of therapy 12 to 17 years 7 mg/kg once every 24 hours infused over 30 minutes Up to 42 days 7 to 11 years 9 mg/kg once every 24 hours infused over 30 minutes 1 to 6 years 12 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. There are other formulations of daptomycin that have differences concerning storage and reconstitution. Carefully follow the reconstitution and storage procedures in labeling. ( 2.7 ) Do not use in conjunction with ReadyMED ® elastomeric infusion pumps in adult and pediatric patients. ( 2.9 )
Contraindications
Daptomycin for injection is contraindicated in patients with known hypersensitivity to daptomycin. [see Warnings and Precautions ( 5.1 )].
Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections: Anaphylaxis/Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Myopathy and Rhabdomyolysis [see Warnings and Precautions ( 5.2 )] Eosinophilic Pneumonia [see Warnings and Precautions ( 5.3 )] Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions ( 5.4 )] Tubulointerstitial Nephritis [see Warnings and Precautions ( 5.5 )] Peripheral Neuropathy [see Warnings and Precautions ( 5.6 )] Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time [see Warnings and Precautions ( 5.11 ) and Drug Interactions ( 7.2 )]
Storage and Handling
How Supplied Daptomycin for Injection is supplied as a sterile, nonpyrogenic, preservative-free, pale yellow to light brown lyophilized cake in single-dose vials as follows: NDC Daptomycin for Injection Package Factor 16729-434-05 350 mg Single-Dose Vial 1 vial per carton 16729-434-45 350 mg Single-Dose Vial 10 vials per carton The container closure is not made with natural rubber latex.
How Supplied
How Supplied Daptomycin for Injection is supplied as a sterile, nonpyrogenic, preservative-free, pale yellow to light brown lyophilized cake in single-dose vials as follows: NDC Daptomycin for Injection Package Factor 16729-434-05 350 mg Single-Dose Vial 1 vial per carton 16729-434-45 350 mg Single-Dose Vial 10 vials per carton The container closure is not made with natural rubber latex.
Description
Daptomycin for Injection is a lipopeptide antibacterial indicated for the treatment of: Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) ( 1.1 ) and, Staphylococcus aureus bloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, ( 1.2 ) Staphylococcus aureus bloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age). ( 1.3 ) Limitations of Use: Daptomycin for Injection is not indicated for the treatment of pneumonia. ( 1.4 ) Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus . ( 1.4 ) Daptomycin for Injection is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. ( 1.4 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5 )
Section 42229-5
Reconstitution of Daptomycin for Injection Vial
Daptomycin for Injection is supplied in single-dose vials, each containing 350 mg daptomycin as a sterile, lyophilized powder. The contents of a Daptomycin for Injection vial should be reconstituted, using aseptic technique, to 50 mg per mL as follows:
- To minimize foaming, AVOID vigorous agitation or shaking of the vial during or after reconstitution.
- Remove the polypropylene flip-off cap from the Daptomycin for Injection vial to expose the central portion of the rubber stopper.
- Wipe the top of the rubber stopper with an alcohol swab or other antiseptic solution and allow to dry. After cleaning, do not touch the rubber stopper or allow it to touch any other surface.
- Slowly transfer 7 mL of 0.9% sodium chloride injection through the center of the rubber stopper into the Daptomycin for Injection vial, pointing the transfer needle toward the wall of the vial. It is recommended that a beveled sterile transfer needle that is 21 gauge or smaller in diameter, or a needleless device is used, pointing the transfer needle toward the wall of the vial.
- Ensure that all of the Daptomycin for Injection powder is wetted by gently rotating the vial.
- Allow the wetted product to stand undisturbed for 10 minutes.
- Gently rotate or swirl the vial contents for a few minutes, as needed, to obtain a completely reconstituted solution.
Section 44425-7
Storage Conditions
Store refrigerated between 2°C and 8°C (36°F and 46° F); avoid excessive heat. Storage conditions for the reconstituted and diluted solutions are described in another section of the prescribing information [Dosage and Administration (2.7)].
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
1.5 Usage
Appropriate specimens for microbiological examination should be obtained in order to isolate and identify the causative pathogens and to determine their susceptibility to daptomycin.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
When culture and susceptibility information is available, it should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Empiric therapy may be initiated while awaiting test results.
10 Overdosage
In the event of overdosage, supportive care is advised with maintenance of glomerular filtration. Daptomycin is cleared slowly from the body by hemodialysis (approximately 15% of the administered dose is removed over 4 hours) and by peritoneal dialysis (approximately 11% of the administered dose is removed over 48 hours). The use of high-flux dialysis membranes during 4 hours of hemodialysis may increase the percentage of dose removed compared with that removed by low-flux membranes.
15 References
- Liu SL, Howard LC, Van Lier RBL, Markham JK: Teratology studies with daptomycin administered intravenously (iv) to rats and rabbits. Teratology 37(5):475, 1988.
- Stroup JS, Wagner J, Badzinski T: Use of daptomycin in a pregnant patient with Staphylococcus aureus endocarditis. Ann Pharmacother 44(4):746-749, 2010.
- Buitrago MI, Crompton JA, Bertolami S, North DS, Nathan RA. Extremely low excretion of daptomycin into breast milk of a nursing mother with methicillin-resistant Staphylococcus aureuspelvic inflammatory disease. Pharmacotherapy 2009;29(3):347–351.
- Klibanov OM, Vickery S, Nortey C: Successful treatment of infective panniculitis with daptomycin in a pregnant, morbidly obese patient. Ann Pharmacother 48(5):652-655, 2014.
- Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG Jr, Ryan T, Bashore T, Corey GR. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 2000; 30:633–638.
11 Description
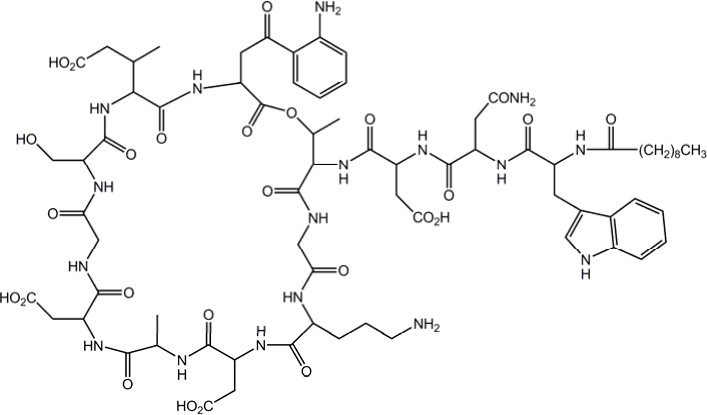
Daptomycin for Injection contains daptomycin, a cyclic lipopeptide antibacterial agent derived from the fermentation of Streptomyces roseosporus.The chemical name is N-decanoyl-L-tryptophyl-D-asparaginyl-L-aspartyl-L-threonylglycyl-L-ornithyl-L-aspartyl-D-alanyl-L-aspartylglycyl-D-seryl- threo-3-methyl-L-glutamyl-3-anthraniloyl-L-alanine ε 1-lactone. The chemical structure is:
The empirical formula is C 72H 101N 17O 26; the molecular weight is 1620.67. Daptomycin for Injection is supplied in a single-dose vial as a sterile, preservative-free, pale yellow to light brown, lyophilized cake containing approximately 350 mg of daptomycin for intravenous (IV) use following reconstitution with 0.9% sodium chloride injection [see Dosage and Administration ( 2.7)] . The only inactive ingredient is sodium hydroxide, which is used for pH adjustment; between 4.0 to 5.0. Freshly reconstituted solutions of Daptomycin for Injection range in color from pale yellow to light brown.
12.4 Microbiology
Daptomycin belongs to the cyclic lipopeptide class of antibacterials. Daptomycin has clinical utility in the treatment of infections caused by aerobic, Gram-positive bacteria. The in vitrospectrum of activity of daptomycin encompasses most clinically relevant Gram-positive pathogenic bacteria.
Daptomycin exhibits rapid, concentration-dependent bactericidal activity against Gram-positive bacteria in vitro.This has been demonstrated both by time-kill curves and by MBC/MIC (minimum bactericidal concentration/minimum inhibitory concentration) ratios using broth dilution methodology. Daptomycin maintained bactericidal activity in vitroagainst stationary phase S. aureusin simulated endocardial vegetations. The clinical significance of this is not known.
8.4 Pediatric Use
The safety and effectiveness of daptomycin in the treatment of cSSSI and S. aureusbloodstream infections (bacteremia) have been established in the age groups 1 to 17 years of age. Use of daptomycin in these age groups is supported by evidence from adequate and well-controlled studies in adults, with additional data from pharmacokinetic studies in pediatric patients, and from safety, efficacy and PK studies in pediatric patients with cSSSI and S. aureusbloodstream infections [see Adverse Reactions ( 6.1), Clinical Pharmacology ( 12.3), and Clinical Studies ( 14.1, 14.2)].
Safety and effectiveness in pediatric patients below the age of one year have not been established.
Avoid use of daptomycin in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs
[see Warnings and Precautions (
5.7) and Nonclinical Toxicology (
13.2)]
.
Daptomycin is not indicated in pediatric patients with renal impairment because dosage has not been established in these patients.
Daptomycin has not been studied in pediatric patients with other bacterial infections.
8.5 Geriatric Use
Of the 534 adult patients treated with daptomycin in Phase 3 controlled clinical trials of complicated skin and skin structure infections (cSSSI), 27% were 65 years of age or older and 12% were 75 years of age or older. Of the 120 adult patients treated with daptomycin in the Phase 3 controlled clinical trial of S. aureusbacteremia/endocarditis, 25% were 65 years of age or older and 16% were 75 years of age or older. In Phase 3 adult clinical trials of cSSSI and S. aureusbacteremia/endocarditis, clinical success rates were lower in patients ≥65 years of age than in patients <65 years of age. In addition, treatment-emergent adverse events were more common in patients ≥65 years of age than in patients <65 years of age.
The exposure of daptomycin was higher in healthy elderly subjects than in healthy young adult subjects. However, no adjustment of daptomycin dosage is warranted for elderly patients with creatinine clearance (CL CR) ≥30 mL/min [see Dosage and Administration ( 2.6) and Clinical Pharmacology ( 12.3)] .
4 Contraindications
Daptomycin for injection is contraindicated in patients with known hypersensitivity to daptomycin. [see Warnings and Precautions ( 5.1)].
6 Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections:
- Anaphylaxis/Hypersensitivity Reactions [see Warnings and Precautions ( 5.1)]
- Myopathy and Rhabdomyolysis [see Warnings and Precautions ( 5.2)]
- Eosinophilic Pneumonia [see Warnings and Precautions ( 5.3)]
- Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions ( 5.4)]
- Tubulointerstitial Nephritis [see Warnings and Precautions ( 5.5)]
- Peripheral Neuropathy [see Warnings and Precautions ( 5.6)]
- Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time [see Warnings and Precautions ( 5.11) and Drug Interactions ( 7.2)]
12.2 Pharmacodynamics
Based on animal models of infection, the antimicrobial activity of daptomycin appears to correlate with the AUC/MIC (area under the concentration-time curve/minimum inhibitory concentration) ratio for certain pathogens, including S. aureus.The principal pharmacokinetic/pharmacodynamic parameter best associated with clinical and microbiological cure has not been elucidated in clinical trials with daptomycin.
2.9 Incompatibilities
Daptomycin for Injection is not compatible with dextrose-containing diluents.
Daptomycin for Injection should not be used in conjunction with ReadyMED ®elastomeric infusion pumps. Stability studies of daptomycin for injection solutions stored in ReadyMED ®elastomeric infusion pumps identified an impurity (2-mercaptobenzothiazole) leaching from this pump system into the Daptomycin for Injection solution.
Because only limited data are available on the compatibility of daptomycin for injection with other IV substances, additives and other medications should not be added to Daptomycin for Injection single-dose vials or infusion bags, or infused simultaneously with Daptomycin for Injection through the same IV line. If the same IV line is used for sequential infusion of different drugs, the line should be flushed with a compatible intravenous solution before and after infusion with Daptomycin for Injection.
1.4 Limitations of Use
Daptomycin for Injection is not indicated for the treatment of pneumonia.
Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus. The clinical trial of Daptomycin for Injection in adult patients with S. aureusbloodstream infections included limited data from patients with left-sided infective endocarditis; outcomes in these patients were poor [see Clinical Studies ( 14.2)] . Daptomycin for Injection has not been studied in patients with prosthetic valve endocarditis.
Daptomycin for Injection is not recommended in pediatric patients younger than 1 year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs [see Warnings and Precautions ( 5.7) and Nonclinical Toxicology ( 13.2)].
1 Indications and Usage
Daptomycin for Injection is a lipopeptide antibacterial indicated for the treatment of:
- Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) ( 1.1) and,
- Staphylococcus aureusbloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, ( 1.2)
- Staphylococcus aureusbloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age). ( 1.3)
Limitations of Use:
- Daptomycin for Injection is not indicated for the treatment of pneumonia. ( 1.4)
- Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus. ( 1.4)
- Daptomycin for Injection is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. ( 1.4)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5)
12.1 Mechanism of Action
Daptomycin is an antibacterial drug [see Clinical Pharmacology ( 12.4)] .
1.2 Staphylococcus Aureus
Daptomycin for Injection is indicated for the treatment of adult patients with Staphylococcus aureusbloodstream infections (bacteremia), including adult patients with right-sided infective endocarditis, caused by methicillin-susceptible and methicillin-resistant isolates.
1.3 Staphylococcus Aureus
Daptomycin for Injection is indicated for the treatment of pediatric patients (1 to 17 years of age) with Staphylococcus aureusbloodstream infections (bacteremia).
5.6 Peripheral Neuropathy
Cases of peripheral neuropathy have been reported during the daptomycin postmarketing experience [see Adverse Reactions ( 6.2)]. Therefore, physicians should be alert to signs and symptoms of peripheral neuropathy in patients receiving daptomycin. Monitor for neuropathy and consider discontinuation.
5 Warnings and Precautions
- Anaphylaxis/hypersensitivity reactions (including life-threatening): Discontinue daptomycin and treat signs/symptoms. ( 5.1)
- Myopathy and rhabdomyolysis: Monitor CPK levels and follow muscle pain or weakness; if elevated CPK or myopathy occurs, consider discontinuation of daptomycin. ( 5.2)
- Eosinophilic pneumonia: Discontinue daptomycin and consider treatment with systemic steroids. ( 5.3)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue daptomycin and institute appropriate treatment. ( 5.4)
- Tubulointerstitial Nephritis (TIN): Discontinue daptomycin and institute appropriate treatment. ( 5.5)
- Peripheral neuropathy: Monitor for neuropathy and consider discontinuation. ( 5.6)
- Potential nervous system and/or muscular system effects in pediatric patients younger than 12 months: Avoid use of daptomycin in this age group. ( 5.7)
- Clostridioides difficile–associated diarrhea: Evaluate patients if diarrhea occurs. ( 5.8)
- Persisting or relapsing S. aureusbacteremia/endocarditis: Perform susceptibility testing and rule out sequestered foci of infection. ( 5.9)
- Decreased efficacy was observed in adult patients with moderate baseline renal impairment. ( 5.10)
5.3 Eosinophilic Pneumonia
Eosinophilic pneumonia has been reported in patients receiving daptomycin [see Adverse Reactions ( 6.2)] . In reported cases associated with daptomycin, patients developed fever, dyspnea with hypoxic respiratory insufficiency, and diffuse pulmonary infiltrates or organizing pneumonia. In general, patients developed eosinophilic pneumonia 2 to 4 weeks after starting daptomycin and improved when daptomycin was discontinued and steroid therapy was initiated. Recurrence of eosinophilic pneumonia upon re-exposure has been reported. Patients who develop these signs and symptoms while receiving daptomycin should undergo prompt medical evaluation, and daptomycin should be discontinued immediately. Treatment with systemic steroids is recommended.
2 Dosage and Administration
Adult Patients
- Administer to adult patientsintravenously in 0.9% sodium chloride, either by injection over a 2-minute period or by infusion over a 30-minute period. ( 2.1, 2.7)
- Recommended dosage regimen for adult patients ( 2.2, 2.4, 2.6):
| Creatinine Clearance (CL CR ) | Dosage Regimen | |
|
cSSSI
For 7 to 14 days |
S. aureusBacteremia
For 2 to 6 weeks |
|
| ≥30 mL/min | 4 mg/kg once every 24 hours | 6 mg/kg once every
24 hours |
| <30 mL/min, including hemodialysis and CAPD | 4 mg/kg once every 48 hours* | 6 mg/kg once every
48 hours* |
|
*Administered following hemodialysis on hemodialysis days. |
Pediatric Patients
- Unlike in adults, do NOT administer by injection over a two (2) minute period to pediatric patients.( 2.1, 2.7)
- Administer to pediatric patients intravenously in 0.9% sodium chloride, by infusion over a 30-or 60-minute period, based on age. ( 2.1, 2.7)
- Recommended dosage regimen for pediatric patients (1 to 17 years of age) with cSSSI, based on age (
2.3):
Age group Dosage* Duration of therapy 12 to 17 years 5 mg/kg once every 24 hours infused over 30 minutes Up to 14 days 7 to 11 years 7 mg/kg once every 24 hours infused over 30 minutes 2 to 6 years 9 mg/kg once every 24 hours infused over 60 minutes 1 to less than 2 years 10 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
- Recommended dosage regimen for pediatric patients (1 to 17 years of age) with S. aureus bacteremia, based on age (
2.5):
Age group Dosage* Duration of therapy 12 to 17 years 7 mg/kg once every 24 hours infused over 30 minutes Up to 42 days 7 to 11 years 9 mg/kg once every 24 hours infused over 30 minutes 1 to 6 years 12 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
3 Dosage Forms and Strengths
For Injection: 350 mg daptomycin as a sterile, pale yellow to light brown lyophilized powder for reconstitution in a single-dose vial.
5.8 Clostridioides Difficile
Clostridioides difficile–associated diarrhea (CDAD) has been reported with the use of nearly all systemic antibacterial agents, including daptomycin, and may range in severity from mild diarrhea to fatal colitis [see Adverse Reactions ( 6.2)] . Treatment with antibacterial agents alters the normal flora of the colon, leading to overgrowth of C. difficile.
C. difficileproduces toxins A and B, which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficilecause increased morbidity and mortality, since these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficilemay need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of daptomycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders:anemia, thrombocytopenia
General and Administration Site Conditions:pyrexia
Immune System Disorders:anaphylaxis; hypersensitivity reactions, including angioedema , pruritus, hives, shortness of breath, difficulty swallowing, truncal erythema, and pulmonary eosinophilia [see Contraindications ( 4) and Warnings and Precautions ( 5.1)]
Infections and Infestations: Clostridioides difficile–associated diarrhea [see Warnings and Precautions ( 5.8)]
Laboratory Investigations:platelet count decreased
Musculoskeletal Disorders:myoglobin increased; rhabdomyolysis (some reports involved patients treated concurrently with daptomycin and HMG-CoA reductase inhibitors) [see Warnings and Precautions ( 5.2), Drug Interactions ( 7.1), and Clinical Pharmacology ( 12.3)]
Respiratory, Thoracic, and Mediastinal Disorders:cough, eosinophilic pneumonia, organizing pneumonia [see Warnings and Precautions ( 5.3)]
Nervous System Disorders:peripheral neuropathy [see Warnings and Precautions ( 5.6)]
Skin and Subcutaneous Tissue Disorders:serious skin reactions, including drug reaction with eosinophilia and systemic symptoms (DRESS), vesiculobullous rash (with or without mucous membrane involvement, including Stevens-Johnson syndrome [SJS] and toxic epidermal necrolysis [TEN]), and acute generalized exanthematous pustulosis [see Warnings and Precautions ( 5.4)]
Gastrointestinal Disorders:nausea, vomiting
Metabolic and Nutritional Disorders:hyperkalemia
Renal and Urinary Disorders:acute kidney injury, renal insufficiency, renal failure, and tubulointerstitial nephritis (TIN) [see Warnings and Precautions ( 5.5)]
Special Senses:visual disturbances
2.2 Dosage in Adults for Csssi
Administer Daptomycin for Injection 4 mg/kg to adult patients intravenously in 0.9% sodium chloride injection once every 24 hours for 7 to 14 days.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.2 Myopathy and Rhabdomyolysis
Myopathy, defined as muscle aching or muscle weakness in conjunction with increases in creatine phosphokinase (CPK) values to greater than 10 times the upper limit of normal (ULN), has been reported with the use of daptomycin. Rhabdomyolysis, with or without acute renal failure, has been reported [see Adverse Reactions ( 6.2)] .
Patients receiving Daptomycin for Injection should be monitored for the development of muscle pain or weakness, particularly of the distal extremities. In patients who receive Daptomycin for Injection, CPK levels should be monitored weekly, and more frequently in patients who received recent prior or concomitant therapy with an HMG-CoA reductase inhibitor or in whom elevations in CPK occur during treatment with Daptomycin for Injection.
In adult patients with renal impairment, both renal function and CPK should be monitored more frequently than once weekly [see Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3)] .
In Phase 1 studies and Phase 2 clinical trials in adults, CPK elevations appeared to be more frequent when daptomycin was dosed more than once daily. Therefore, daptomycin should not be dosed more frequently than once a day.
Daptomycin should be discontinued in patients with unexplained signs and symptoms of myopathy in conjunction with CPK elevations to levels >1,000 U/L (~5× ULN), and in patients without reported symptoms who have marked elevations in CPK, with levels >2,000 U/L (≥10× ULN). In addition, consideration should be given to suspending agents associated with rhabdomyolysis, such as HMG-CoA reductase inhibitors, temporarily in patients receiving daptomycin [see Drug Interactions ( 7.1)] .
7.1 Hmg Coa Reductase Inhibitors
In healthy adult subjects, concomitant administration of daptomycin and simvastatin had no effect on plasma trough concentrations of simvastatin, and there were no reports of skeletal myopathy [see Clinical Pharmacology ( 12.3)] .
However, inhibitors of HMG-CoA reductase may cause myopathy, which is manifested as muscle pain or weakness associated with elevated levels of creatine phosphokinase (CPK). In the adult Phase 3 S. aureusbacteremia/endocarditis trial, some patients who received prior or concomitant treatment with an HMG-CoA reductase inhibitor developed elevated CPK [see Adverse Reactions ( 6.1)] . Experience with the coadministration of HMG-CoA reductase inhibitors and daptomycin in patients is limited; therefore, consideration should be given to suspending use of HMG-CoA reductase inhibitors temporarily in patients receiving daptomycin.
17 Patient Counseling Information
Allergic Reactions
Advise patients that allergic reactions, including serious skin, kidney, lung, or other organ reactions, could occur and that these serious reactions require immediate treatment. Patients should report any previous allergic reactions to daptomycin [see Warnings and Precautions ( 5.1, 5.4, 5.5)].
Muscle Pain or Weakness (Myopathy and Rhabdomyolysis, Peripheral Neuropathy
Advise patients to report muscle pain or weakness, especially in the forearms and lower legs, as well as tingling or numbness. [see Warnings and Precautions ( 5.2, 5.6)].
Cough, Breathlessness or Fever (Eosinophilic Pneumonia)
Advise patients to report any symptoms of cough, breathlessness, or fever [see Warnings and Precautions ( 5.3)].
C. difficile-Associated Diarrhea (CDAD)
Advise patients that diarrhea is a common problem caused by antibacterials that usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterials, including daptomycin, patients can develop watery and bloody stools (with or without stomach cramps and fever), even as late as 2 or more months after having received the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible [see Warnings and Precautions ( 5.8)].
Antibacterial Resistance
Patients should be counseled that antibacterial drugs, including Daptomycin for Injection, should be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Daptomycin for Injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be administered exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Daptomycin for Injection or other antibacterial drugs in the future.
Manufactured For:
Accord Healthcare, Inc.,
8041 Arco Corporate Drive,
Suite 200,
Raleigh, NC 27617,
USA.
Manufactured By:
Intas Pharmaceuticals Limited,
Ahmedabad-380 054, India.
10 2865 6 6035362
Issued: June 2025
8.6 Patients With Renal Impairment
Daptomycin is eliminated primarily by the kidneys; therefore, a modification of daptomycin dosage interval is recommended for adult patients with CL CR<30 mL/min, including patients receiving hemodialysis or continuous ambulatory peritoneal dialysis (CAPD). In adult patients with renal impairment, both renal function and creatine phosphokinase (CPK) should be monitored more frequently than once weekly [see Dosage and Administration ( 2.6), Warnings and Precautions ( 5.2, 5.10), and Clinical Pharmacology ( 12.3)] .
The dosage regimen for daptomycin in pediatric patients with renal impairment has not been established.
16 How Supplied/storage and Handling
How Supplied
Daptomycin for Injection is supplied as a sterile, nonpyrogenic, preservative-free, pale yellow to light brown lyophilized cake in single-dose vials as follows:
| NDC | Daptomycin for Injection | Package Factor |
| 16729-434-05 | 350 mg Single-Dose Vial | 1 vial per carton |
| 16729-434-45 | 350 mg Single-Dose Vial | 10 vials per carton |
The container closure is not made with natural rubber latex.
2.8 Compatible Intravenous Solutions
Daptomycin for Injection is compatible with 0.9% sodium chloride injection and Lactated Ringer's injection.
5.9 Persisting Or Relapsing S. Aureus
Patients with persisting or relapsing S. aureusbacteremia/endocarditis or poor clinical response should have repeat blood cultures. If a blood culture is positive for S. aureus, minimum inhibitory concentration (MIC) susceptibility testing of the isolate should be performed using a standardized procedure, and diagnostic evaluation of the patient should be performed to rule out sequestered foci of infection. Appropriate surgical intervention (e.g., debridement, removal of prosthetic devices, valve replacement surgery) and/or consideration of a change in antibacterial regimen may be required.
Failure of treatment due to persisting or relapsing S. aureusbacteremia/endocarditis may be due to reduced daptomycin susceptibility (as evidenced by increasing MIC of the S. aureusisolate) [see Clinical Studies ( 14.2)] .
7.2 Drug Laboratory Test Interactions
Clinically relevant plasma concentrations of daptomycin have been observed to cause a significant concentration-dependent false prolongation of prothrombin time (PT) and elevation of International Normalized Ratio (INR) when certain recombinant thromboplastin reagents are utilized for the assay. The possibility of an erroneously elevated PT/INR result due to interaction with a recombinant thromboplastin reagent may be minimized by drawing specimens for PT or INR testing near the time of trough plasma concentrations of daptomycin. However, sufficient daptomycin concentrations may be present at trough to cause interaction.
If confronted with an abnormally high PT/INR result in a patient being treated with daptomycin, it is recommended that clinicians:
- Repeat the assessment of PT/INR, requesting that the specimen be drawn just prior to the next daptomycin dose (i.e., at trough concentration). If the PT/INR value obtained at trough remains substantially elevated above what would otherwise be expected, consider evaluating PT/INR utilizing an alternative method.
- Evaluate for other causes of abnormally elevated PT/INR results.
5.5 Tubulointerstitial Nephritis (tin)
TIN has been reported in post-marketing experience with daptomycin [see Adverse Reactions ( 6.2)]. Patients who develop new or worsening renal impairment while receiving daptomycin should undergo medical evaluation. If TIN is suspected, discontinue daptomycin promptly and institute appropriate treatment.
5.1 Anaphylaxis/hypersensitivity Reactions
Anaphylaxis/hypersensitivity reactions have been reported with the use of antibacterial agents, including daptomycin for injection, and may be life-threatening. If an allergic reaction to Daptomycin for Injection occurs, discontinue the drug and institute appropriate therapy [see Adverse Reactions ( 6.2)].
5.12 Development of Drug Resistant Bacteria
Prescribing Daptomycin for Injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.6 Dosage in Patients With Renal Impairment
Adult Patients
No dosage adjustment is required in adult patients with creatinine clearance (CL CR) greater than or equal to 30 mL/min. The recommended dosage regimen for Daptomycin for Injection in adult patients with CL CRless than 30 mL/min, including adult patients on hemodialysis or continuous ambulatory peritoneal dialysis (CAPD), is 4 mg/kg (cSSSI) or 6 mg/kg ( S. aureusbloodstream infections) once every 48 hours ( Table 3). When possible, Daptomycin for Injection should be administered following the completion of hemodialysis on hemodialysis days [see Warnings and Precautions ( 5.2, 5.10), Use in Specific Populations ( 8.6), and Clinical Pharmacology ( 12.3)] .
|
* When possible, administer Daptomycin for Injection following the completion of hemodialysis on hemodialysis days. |
||
|
Creatinine Clearance
(CL CR ) |
Dosage Regimen in Adults | |
| cSSSI | S. aureus Bloodstream Infections | |
| Greater than or equal to 30 mL/min | 4 mg/kg once every 24 hours | 6 mg/kg once every 24 hours |
| Less than 30 mL/min, including hemodialysis and CAPD | 4 mg/kg once every 48 hours* | 6 mg/kg once every 48 hours* |
Pediatric Patients
The dosage regimen for Daptomycin for Injection in pediatric patients with renal impairment has not been established.
2.1 Important Administration Duration Instructions
Adults
Administer the appropriate volume of the reconstituted Daptomycin for Injection (concentration of 50 mg per mL) to adult patientsintravenously either by injection over a two (2) minute period or by intravenous infusion over a thirty (30) minute period [see Dosage and Administration ( 2.2, 2.4, 2.7)].
Pediatric Patients (1 to 17 Years of Age)
Unlike in adults, do NOT administer Daptomycin for Injection by injection over a two (2) minute period to pediatric patients.
- Pediatric Patients 7 to 17 years of Age:Administer Daptomycin for Injection intravenously by infusion over a 30-minute period [see Dosage and Administration ( 2.3, 2.5, 2.7)].
- Pediatric Patients 1 to 6 years of Age:Administer Daptomycin for Injection intravenously by infusion over a 60-minute period [see Dosage and Administration ( 2.3, 2.5, 2.7)].
14.1 Complicated Skin and Skin Structure Infections
Adults with cSSSI
Adult patients with clinically documented complicated skin and skin structure infections (cSSSI) ( Table 15) were enrolled in two randomized, multinational, multicenter, investigator-blinded trials comparing daptomycin (4 mg/kg IV every 24h) with either vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g IV per day). Patients could switch to oral therapy after a minimum of 4 days of IV treatment if clinical improvement was demonstrated. Patients known to have bacteremia at baseline were excluded. Patients with creatinine clearance (CL CR) between 30 and 70 mL/min were to receive a lower dose of daptomycin as specified in the protocol; however, the majority of patients in this subpopulation did not have the dose of daptomycin adjusted.
|
* Comparator: vancomycin (1 g IV q12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses). |
|||
|
†The majority of cases were subsequently categorized as complicated cellulitis, major abscesses, or traumatic wound infections. |
|||
| Primary Diagnosis |
Adult Patients
(Daptomycin / Comparator*) |
||
|
Study 9801
N=264 / N=266 |
Study 9901
N=270 / N=292 |
Pooled
N=534 / N=558 |
|
| Wound Infection | 99 (38%) / 116 (44%) | 102 (38%) / 108 (37%) | 201 (38%) / 224 (40%) |
| Major Abscess | 55 (21%) / 43 (16%) | 59 (22%) / 65 (22%) | 114 (21%) / 108 (19%) |
| Ulcer Infection | 71 (27%) / 75 (28%) | 53 (20%) / 68 (23%) | 124 (23%) / 143 (26%) |
| Other Infection † | 39 (15%) / 32 (12%) | 56 (21%) / 51 (18%) | 95 (18%) / 83 (15%) |
One trial was conducted primarily in the United States and South Africa (study 9801), and the second was conducted at non-US sites only (study 9901). The two trials were similar in design but differed in patient characteristics, including history of diabetes and peripheral vascular disease. There were a total of 534 adult patients treated with daptomycin and 558 treated with comparator in the two trials. The majority (89.7%) of patients received IV medication exclusively.
The efficacy endpoints in both trials were the clinical success rates in the intent-to-treat (ITT) population and in the clinically evaluable (CE) population. In study 9801, clinical success rates in the ITT population were 62.5% (165/264) in patients treated with daptomycin and 60.9% (162/266) in patients treated with comparator drugs. Clinical success rates in the CE population were 76.0% (158/208) in patients treated with daptomycin and 76.7% (158/206) in patients treated with comparator drugs. In study 9901, clinical success rates in the ITT population were 80.4% (217/270) in patients treated with daptomycin and 80.5% (235/292) in patients treated with comparator drugs. Clinical success rates in the CE population were 89.9% (214/238) in patients treated with daptomycin and 90.4% (226/250) in patients treated with comparator drugs.
The success rates by pathogen for microbiologically evaluable patients are presented in Table 16.
|
* Comparator: vancomycin (1 g IV q12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses). |
||
|
† As determined by the central laboratory. |
||
| Pathogen |
Success Rate
n/N (%) |
|
| Daptomycin | Comparator* | |
| Methicillin-susceptible Staphylococcus aureus(MSSA) † | 170/198 (86%) | 180/207 (87%) |
| Methicillin-resistant Staphylococcus aureus(MRSA) † | 21/28 (75%) | 25/36 (69%) |
| Streptococcus pyogenes | 79/84 (94%) | 80/88 (91%) |
| Streptococcus agalactiae | 23/27 (85%) | 22/29 (76%) |
| Streptococcus dysgalactiaesubsp. equisimilis | 8/8 (100%) | 9/11 (82%) |
| Enterococcus faecalis(vancomycin-susceptible only) | 27/37 (73%) | 40/53 (76%) |
Pediatric Patients (1 to 17 Years of Age) with cSSSI
The cSSSI pediatric trial was a single prospective multi-center, randomized, comparative trial. A total of 396 pediatric patients aged 1 to 17 years with cSSSI caused by Gram positive pathogens were enrolled into the study. Patients known to have bacteremia, osteomyelitis, endocarditis, and pneumonia at baseline were excluded. Patients were enrolled in a stepwise approach into four age groups and given age-dependent doses of daptomycin once daily for up to 14 days. The different age groups and doses evaluated were as follows: Adolescents (12 to 17 years) treated with 5 mg/kg of daptomycin (n=113), Children (7 to 11 years) treated with 7 mg/kg of daptomycin (n=113), Children (2 to 6 years) treated with 9 mg/kg of daptomycin (n=125) and Infants (1 to <2 years) treated with 10 mg/kg (n= 45).
Patients were randomized 2:1 to receive daptomycin or a standard of care (SOC) comparator, which included intravenous therapy with either vancomycin, clindamycin, or an anti-staphylococcal semi-synthetic penicillin (nafcillin, oxacillin, or cloxacillin). Patients could switch to oral therapy after clinical improvement was demonstrated (no minimum IV dosing was required).
The primary objective of this study was to evaluate the safety of daptomycin. The clinical outcome was determined by resolution or improvement of symptoms at the End-of-Treatment (EOT), 3 days after the last dose, and Test-of-Cure (TOC), 7 to 14 days after the last dose. Investigator observed outcomes were verified in a blinded fashion. Of the 396 subjects randomized in the study, 389 subjects were treated with daptomycin or comparator and included in the ITT population. Of these, 257 subjects were randomized to the daptomycin group and 132 subjects were randomized to the comparator group. Approximately 95% of subjects switched to oral therapy. The mean day of switch was day 4, and ranged from day 1 to day 14. The clinical success rates determined at 7 to 14 days after last dose of therapy (IV and oral) (TOC visit) were 88% (227/257) for daptomycin and 86% (114/132) for comparator.
2.4 Dosage in Adult Patients With Staphylococcus Aureus
Administer Daptomycin for Injection 6 mg/kg to adult patients intravenously in 0.9% sodium chloride injection once every 24 hours for 2 to 6 weeks. There are limited safety data for the use of Daptomycin for Injection for more than 28 days of therapy. In the Phase 3 trial, there were a total of 14 adult patients who were treated with daptomycin for more than 28 days.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies in animals have not been conducted to evaluate the carcinogenic potential of daptomycin. However, neither mutagenic nor clastogenic potential was found in a battery of genotoxicity tests, including the Ames assay, a mammalian cell gene mutation assay, a test for chromosomal aberrations in Chinese hamster ovary cells, an in vivomicronucleus assay, an in vitroDNA repair assay, and an in vivosister chromatid exchange assay in Chinese hamsters.
Daptomycin did not affect the fertility or reproductive performance of male and female rats when administered intravenously at doses of 25, 75, or 150 mg/kg/day, which is approximately up to 9 times the estimated human exposure level based upon AUCs (or approximately up to 4 times the recommended human dose of 6 mg/kg based on body surface area comparison).
1.1 Complicated Skin and Skin Structure Infections (csssi)
Daptomycin for Injection is indicated for the treatment of adult and pediatric patients (1 to 17 years of age) with complicated skin and skin structure infections (cSSSI) caused by susceptible isolates of the following Gram-positive bacteria: Staphylococcus aureus(including methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiaesubsp. equisimilis,and Enterococcus faecalis(vancomycin-susceptible isolates only).
2.7 Preparation and Administration of Daptomycin for Injection
There are other formulations of daptomycin that have differences concerning reconstitution and storage. Carefully follow the reconstitution and storage procedures described in this labeling.
2.3 Dosage in Pediatric Patients (1 to 17 Years of Age) for Csssi
The recommended dosage regimens based on age for pediatric patients with cSSSI are shown in Table 1. Administer Daptomycin for Injection intravenously in 0.9% sodium chloride injection once every 24 hours for up to 14 days.
| Age Range | Dosage Regimen* | Duration of therapy |
| 12 to 17 years | 5 mg/kg once every 24 hours infused over 30 minutes | Up to 14 days |
| 7 to 11 years | 7 mg/kg once every 24 hours infused over 30 minutes | |
| 2 to 6 years | 9 mg/kg once every 24 hours infused over 60 minutes | |
| 1 to less than 2 years | 10 mg/kg once every 24 hours infused over 60 minutes | |
|
*Recommended dosage regimen is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. |
5.4 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)
DRESS has been reported in post-marketing experience with daptomycin [see Adverse Reactions ( 6.2)]. Patients who develop skin rash, fever, peripheral eosinophilia, and systemic organ (for example, hepatic, renal, pulmonary) impairment while receiving daptomycin should undergo medical evaluation. If DRESS is suspected, discontinue daptomycin promptly and institute appropriate treatment.
5.10 Decreased Efficacy in Patients With Moderate Baseline Renal Impairment
Limited data are available from the two Phase 3 complicated skin and skin structure infection (cSSSI) trials regarding clinical efficacy of daptomycin treatment in adult patients with creatinine clearance (CL CR) <50 mL/min; only 31/534 (6%) patients treated with daptomycin in the intent-to-treat (ITT) population had a baseline CL CR<50 mL/min. Table 4shows the number of adult patients by renal function and treatment group who were clinical successes in the Phase 3 cSSSI trials.
| CL CR |
Success Rate
n/N (%) |
|
|
Daptomycin
4 mg/kg every 24h |
Comparator | |
| 50 to 70 mL/min | 25/38 (66%) | 30/48 (63%) |
| 30 to <50 mL/min | 7/15 (47%) | 20/35 (57%) |
In a subgroup analysis of the ITT population in the Phase 3 S. aureusbacteremia/endocarditis trial, clinical success rates, as determined by a treatment-blinded Adjudication Committee [see Clinical Studies ( 14.2)] , in the daptomycin-treated adult patients were lower in patients with baseline CL CR<50 mL/min (see Table 5). A decrease of the magnitude shown in Table 5was not observed in comparator-treated patients.
| Baseline CL CR |
Success Rate
n/N (%) |
|||
|
Daptomycin
6 mg/kg every 24h |
Comparator | |||
| Bacteremia | Right-Sided Infective Endocarditis | Bacteremia | Right-Sided Infective Endocarditis | |
| >80 mL/min | 30/50 (60%) | 7/14 (50%) | 19/42 (45%) | 5/11 (46%) |
| 50 to 80 mL/min | 12/26 (46%) | 1/4 (25%) | 13/31 (42%) | 1/2 (50%) |
| 30 to <50 mL/min | 2/14 (14%) | 0/1 (0%) | 7/17 (41%) | 1/1 (100%) |
Consider these data when selecting antibacterial therapy for use in adult patients with baseline moderate to severe renal impairment.
5.11 Increased International Normalized Ratio (inr)/prolonged Prothrombin Time
Clinically relevant plasma concentrations of daptomycin have been observed to cause a significant concentration-dependent false prolongation of prothrombin time (PT) and elevation of International Normalized Ratio (INR) when certain recombinant thromboplastin reagents are utilized for the assay [see Drug Interactions ( 7.2)].
2.5 Dosage in Pediatric Patients (1 to 17 Years of Age) With Staphylococcus Aureus
The recommended dosage regimens based on age for pediatric patients with S. aureusbloodstream infections (bacteremia) are shown in Table 2. Administer Daptomycin for Injection intravenously in 0.9% sodium chloride injection once every 24 hours for up to 42 days.
| Age group | Dosage* | Duration of therapy |
| 12 to 17 years | 7 mg/kg once every 24 hours infused over 30 minutes | Up to 42 days |
| 7 to 11 years | 9 mg/kg once every 24 hours infused over 30 minutes | |
| 1 to 6 years | 12 mg/kg once every 24 hours infused over 60 minutes | |
|
*Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. |
5.7 Potential Nervous System And/or Muscular System Effects in Pediatric Patients Younger Than 12 Months
Avoid use of daptomycin in pediatric patients younger than 12 months due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs with intravenous daptomycin [see Nonclinical Toxicology ( 13.2)] .
Structured Label Content
Section 42229-5 (42229-5)
Reconstitution of Daptomycin for Injection Vial
Daptomycin for Injection is supplied in single-dose vials, each containing 350 mg daptomycin as a sterile, lyophilized powder. The contents of a Daptomycin for Injection vial should be reconstituted, using aseptic technique, to 50 mg per mL as follows:
- To minimize foaming, AVOID vigorous agitation or shaking of the vial during or after reconstitution.
- Remove the polypropylene flip-off cap from the Daptomycin for Injection vial to expose the central portion of the rubber stopper.
- Wipe the top of the rubber stopper with an alcohol swab or other antiseptic solution and allow to dry. After cleaning, do not touch the rubber stopper or allow it to touch any other surface.
- Slowly transfer 7 mL of 0.9% sodium chloride injection through the center of the rubber stopper into the Daptomycin for Injection vial, pointing the transfer needle toward the wall of the vial. It is recommended that a beveled sterile transfer needle that is 21 gauge or smaller in diameter, or a needleless device is used, pointing the transfer needle toward the wall of the vial.
- Ensure that all of the Daptomycin for Injection powder is wetted by gently rotating the vial.
- Allow the wetted product to stand undisturbed for 10 minutes.
- Gently rotate or swirl the vial contents for a few minutes, as needed, to obtain a completely reconstituted solution.
Section 44425-7 (44425-7)
Storage Conditions
Store refrigerated between 2°C and 8°C (36°F and 46° F); avoid excessive heat. Storage conditions for the reconstituted and diluted solutions are described in another section of the prescribing information [Dosage and Administration (2.7)].
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
1.5 Usage
Appropriate specimens for microbiological examination should be obtained in order to isolate and identify the causative pathogens and to determine their susceptibility to daptomycin.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
When culture and susceptibility information is available, it should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Empiric therapy may be initiated while awaiting test results.
10 Overdosage (10 OVERDOSAGE)
In the event of overdosage, supportive care is advised with maintenance of glomerular filtration. Daptomycin is cleared slowly from the body by hemodialysis (approximately 15% of the administered dose is removed over 4 hours) and by peritoneal dialysis (approximately 11% of the administered dose is removed over 48 hours). The use of high-flux dialysis membranes during 4 hours of hemodialysis may increase the percentage of dose removed compared with that removed by low-flux membranes.
15 References (15 REFERENCES)
- Liu SL, Howard LC, Van Lier RBL, Markham JK: Teratology studies with daptomycin administered intravenously (iv) to rats and rabbits. Teratology 37(5):475, 1988.
- Stroup JS, Wagner J, Badzinski T: Use of daptomycin in a pregnant patient with Staphylococcus aureus endocarditis. Ann Pharmacother 44(4):746-749, 2010.
- Buitrago MI, Crompton JA, Bertolami S, North DS, Nathan RA. Extremely low excretion of daptomycin into breast milk of a nursing mother with methicillin-resistant Staphylococcus aureuspelvic inflammatory disease. Pharmacotherapy 2009;29(3):347–351.
- Klibanov OM, Vickery S, Nortey C: Successful treatment of infective panniculitis with daptomycin in a pregnant, morbidly obese patient. Ann Pharmacother 48(5):652-655, 2014.
- Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG Jr, Ryan T, Bashore T, Corey GR. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 2000; 30:633–638.
11 Description (11 DESCRIPTION)
Daptomycin for Injection contains daptomycin, a cyclic lipopeptide antibacterial agent derived from the fermentation of Streptomyces roseosporus.The chemical name is N-decanoyl-L-tryptophyl-D-asparaginyl-L-aspartyl-L-threonylglycyl-L-ornithyl-L-aspartyl-D-alanyl-L-aspartylglycyl-D-seryl- threo-3-methyl-L-glutamyl-3-anthraniloyl-L-alanine ε 1-lactone. The chemical structure is:
The empirical formula is C 72H 101N 17O 26; the molecular weight is 1620.67. Daptomycin for Injection is supplied in a single-dose vial as a sterile, preservative-free, pale yellow to light brown, lyophilized cake containing approximately 350 mg of daptomycin for intravenous (IV) use following reconstitution with 0.9% sodium chloride injection [see Dosage and Administration ( 2.7)] . The only inactive ingredient is sodium hydroxide, which is used for pH adjustment; between 4.0 to 5.0. Freshly reconstituted solutions of Daptomycin for Injection range in color from pale yellow to light brown.
12.4 Microbiology
Daptomycin belongs to the cyclic lipopeptide class of antibacterials. Daptomycin has clinical utility in the treatment of infections caused by aerobic, Gram-positive bacteria. The in vitrospectrum of activity of daptomycin encompasses most clinically relevant Gram-positive pathogenic bacteria.
Daptomycin exhibits rapid, concentration-dependent bactericidal activity against Gram-positive bacteria in vitro.This has been demonstrated both by time-kill curves and by MBC/MIC (minimum bactericidal concentration/minimum inhibitory concentration) ratios using broth dilution methodology. Daptomycin maintained bactericidal activity in vitroagainst stationary phase S. aureusin simulated endocardial vegetations. The clinical significance of this is not known.
8.4 Pediatric Use
The safety and effectiveness of daptomycin in the treatment of cSSSI and S. aureusbloodstream infections (bacteremia) have been established in the age groups 1 to 17 years of age. Use of daptomycin in these age groups is supported by evidence from adequate and well-controlled studies in adults, with additional data from pharmacokinetic studies in pediatric patients, and from safety, efficacy and PK studies in pediatric patients with cSSSI and S. aureusbloodstream infections [see Adverse Reactions ( 6.1), Clinical Pharmacology ( 12.3), and Clinical Studies ( 14.1, 14.2)].
Safety and effectiveness in pediatric patients below the age of one year have not been established.
Avoid use of daptomycin in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs
[see Warnings and Precautions (
5.7) and Nonclinical Toxicology (
13.2)]
.
Daptomycin is not indicated in pediatric patients with renal impairment because dosage has not been established in these patients.
Daptomycin has not been studied in pediatric patients with other bacterial infections.
8.5 Geriatric Use
Of the 534 adult patients treated with daptomycin in Phase 3 controlled clinical trials of complicated skin and skin structure infections (cSSSI), 27% were 65 years of age or older and 12% were 75 years of age or older. Of the 120 adult patients treated with daptomycin in the Phase 3 controlled clinical trial of S. aureusbacteremia/endocarditis, 25% were 65 years of age or older and 16% were 75 years of age or older. In Phase 3 adult clinical trials of cSSSI and S. aureusbacteremia/endocarditis, clinical success rates were lower in patients ≥65 years of age than in patients <65 years of age. In addition, treatment-emergent adverse events were more common in patients ≥65 years of age than in patients <65 years of age.
The exposure of daptomycin was higher in healthy elderly subjects than in healthy young adult subjects. However, no adjustment of daptomycin dosage is warranted for elderly patients with creatinine clearance (CL CR) ≥30 mL/min [see Dosage and Administration ( 2.6) and Clinical Pharmacology ( 12.3)] .
4 Contraindications (4 CONTRAINDICATIONS)
Daptomycin for injection is contraindicated in patients with known hypersensitivity to daptomycin. [see Warnings and Precautions ( 5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described, or described in greater detail, in other sections:
- Anaphylaxis/Hypersensitivity Reactions [see Warnings and Precautions ( 5.1)]
- Myopathy and Rhabdomyolysis [see Warnings and Precautions ( 5.2)]
- Eosinophilic Pneumonia [see Warnings and Precautions ( 5.3)]
- Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions ( 5.4)]
- Tubulointerstitial Nephritis [see Warnings and Precautions ( 5.5)]
- Peripheral Neuropathy [see Warnings and Precautions ( 5.6)]
- Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time [see Warnings and Precautions ( 5.11) and Drug Interactions ( 7.2)]
12.2 Pharmacodynamics
Based on animal models of infection, the antimicrobial activity of daptomycin appears to correlate with the AUC/MIC (area under the concentration-time curve/minimum inhibitory concentration) ratio for certain pathogens, including S. aureus.The principal pharmacokinetic/pharmacodynamic parameter best associated with clinical and microbiological cure has not been elucidated in clinical trials with daptomycin.
2.9 Incompatibilities
Daptomycin for Injection is not compatible with dextrose-containing diluents.
Daptomycin for Injection should not be used in conjunction with ReadyMED ®elastomeric infusion pumps. Stability studies of daptomycin for injection solutions stored in ReadyMED ®elastomeric infusion pumps identified an impurity (2-mercaptobenzothiazole) leaching from this pump system into the Daptomycin for Injection solution.
Because only limited data are available on the compatibility of daptomycin for injection with other IV substances, additives and other medications should not be added to Daptomycin for Injection single-dose vials or infusion bags, or infused simultaneously with Daptomycin for Injection through the same IV line. If the same IV line is used for sequential infusion of different drugs, the line should be flushed with a compatible intravenous solution before and after infusion with Daptomycin for Injection.
1.4 Limitations of Use
Daptomycin for Injection is not indicated for the treatment of pneumonia.
Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus. The clinical trial of Daptomycin for Injection in adult patients with S. aureusbloodstream infections included limited data from patients with left-sided infective endocarditis; outcomes in these patients were poor [see Clinical Studies ( 14.2)] . Daptomycin for Injection has not been studied in patients with prosthetic valve endocarditis.
Daptomycin for Injection is not recommended in pediatric patients younger than 1 year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs [see Warnings and Precautions ( 5.7) and Nonclinical Toxicology ( 13.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Daptomycin for Injection is a lipopeptide antibacterial indicated for the treatment of:
- Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) ( 1.1) and,
- Staphylococcus aureusbloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, ( 1.2)
- Staphylococcus aureusbloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age). ( 1.3)
Limitations of Use:
- Daptomycin for Injection is not indicated for the treatment of pneumonia. ( 1.4)
- Daptomycin for Injection is not indicated for the treatment of left-sided infective endocarditis due to S. aureus. ( 1.4)
- Daptomycin for Injection is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. ( 1.4)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Daptomycin for Injection and other antibacterial drugs, Daptomycin for Injection should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5)
12.1 Mechanism of Action
Daptomycin is an antibacterial drug [see Clinical Pharmacology ( 12.4)] .
1.2 Staphylococcus Aureus (1.2 Staphylococcus aureus)
Daptomycin for Injection is indicated for the treatment of adult patients with Staphylococcus aureusbloodstream infections (bacteremia), including adult patients with right-sided infective endocarditis, caused by methicillin-susceptible and methicillin-resistant isolates.
1.3 Staphylococcus Aureus (1.3 Staphylococcus aureus)
Daptomycin for Injection is indicated for the treatment of pediatric patients (1 to 17 years of age) with Staphylococcus aureusbloodstream infections (bacteremia).
5.6 Peripheral Neuropathy
Cases of peripheral neuropathy have been reported during the daptomycin postmarketing experience [see Adverse Reactions ( 6.2)]. Therefore, physicians should be alert to signs and symptoms of peripheral neuropathy in patients receiving daptomycin. Monitor for neuropathy and consider discontinuation.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Anaphylaxis/hypersensitivity reactions (including life-threatening): Discontinue daptomycin and treat signs/symptoms. ( 5.1)
- Myopathy and rhabdomyolysis: Monitor CPK levels and follow muscle pain or weakness; if elevated CPK or myopathy occurs, consider discontinuation of daptomycin. ( 5.2)
- Eosinophilic pneumonia: Discontinue daptomycin and consider treatment with systemic steroids. ( 5.3)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue daptomycin and institute appropriate treatment. ( 5.4)
- Tubulointerstitial Nephritis (TIN): Discontinue daptomycin and institute appropriate treatment. ( 5.5)
- Peripheral neuropathy: Monitor for neuropathy and consider discontinuation. ( 5.6)
- Potential nervous system and/or muscular system effects in pediatric patients younger than 12 months: Avoid use of daptomycin in this age group. ( 5.7)
- Clostridioides difficile–associated diarrhea: Evaluate patients if diarrhea occurs. ( 5.8)
- Persisting or relapsing S. aureusbacteremia/endocarditis: Perform susceptibility testing and rule out sequestered foci of infection. ( 5.9)
- Decreased efficacy was observed in adult patients with moderate baseline renal impairment. ( 5.10)
5.3 Eosinophilic Pneumonia
Eosinophilic pneumonia has been reported in patients receiving daptomycin [see Adverse Reactions ( 6.2)] . In reported cases associated with daptomycin, patients developed fever, dyspnea with hypoxic respiratory insufficiency, and diffuse pulmonary infiltrates or organizing pneumonia. In general, patients developed eosinophilic pneumonia 2 to 4 weeks after starting daptomycin and improved when daptomycin was discontinued and steroid therapy was initiated. Recurrence of eosinophilic pneumonia upon re-exposure has been reported. Patients who develop these signs and symptoms while receiving daptomycin should undergo prompt medical evaluation, and daptomycin should be discontinued immediately. Treatment with systemic steroids is recommended.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Adult Patients
- Administer to adult patientsintravenously in 0.9% sodium chloride, either by injection over a 2-minute period or by infusion over a 30-minute period. ( 2.1, 2.7)
- Recommended dosage regimen for adult patients ( 2.2, 2.4, 2.6):
| Creatinine Clearance (CL CR ) | Dosage Regimen | |
|
cSSSI
For 7 to 14 days |
S. aureusBacteremia
For 2 to 6 weeks |
|
| ≥30 mL/min | 4 mg/kg once every 24 hours | 6 mg/kg once every
24 hours |
| <30 mL/min, including hemodialysis and CAPD | 4 mg/kg once every 48 hours* | 6 mg/kg once every
48 hours* |
|
*Administered following hemodialysis on hemodialysis days. |
Pediatric Patients
- Unlike in adults, do NOT administer by injection over a two (2) minute period to pediatric patients.( 2.1, 2.7)
- Administer to pediatric patients intravenously in 0.9% sodium chloride, by infusion over a 30-or 60-minute period, based on age. ( 2.1, 2.7)
- Recommended dosage regimen for pediatric patients (1 to 17 years of age) with cSSSI, based on age (
2.3):
Age group Dosage* Duration of therapy 12 to 17 years 5 mg/kg once every 24 hours infused over 30 minutes Up to 14 days 7 to 11 years 7 mg/kg once every 24 hours infused over 30 minutes 2 to 6 years 9 mg/kg once every 24 hours infused over 60 minutes 1 to less than 2 years 10 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
- Recommended dosage regimen for pediatric patients (1 to 17 years of age) with S. aureus bacteremia, based on age (
2.5):
Age group Dosage* Duration of therapy 12 to 17 years 7 mg/kg once every 24 hours infused over 30 minutes Up to 42 days 7 to 11 years 9 mg/kg once every 24 hours infused over 30 minutes 1 to 6 years 12 mg/kg once every 24 hours infused over 60 minutes *Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For Injection: 350 mg daptomycin as a sterile, pale yellow to light brown lyophilized powder for reconstitution in a single-dose vial.
5.8 Clostridioides Difficile (5.8 Clostridioides difficile)
Clostridioides difficile–associated diarrhea (CDAD) has been reported with the use of nearly all systemic antibacterial agents, including daptomycin, and may range in severity from mild diarrhea to fatal colitis [see Adverse Reactions ( 6.2)] . Treatment with antibacterial agents alters the normal flora of the colon, leading to overgrowth of C. difficile.
C. difficileproduces toxins A and B, which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficilecause increased morbidity and mortality, since these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficilemay need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post-approval use of daptomycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders:anemia, thrombocytopenia
General and Administration Site Conditions:pyrexia
Immune System Disorders:anaphylaxis; hypersensitivity reactions, including angioedema , pruritus, hives, shortness of breath, difficulty swallowing, truncal erythema, and pulmonary eosinophilia [see Contraindications ( 4) and Warnings and Precautions ( 5.1)]
Infections and Infestations: Clostridioides difficile–associated diarrhea [see Warnings and Precautions ( 5.8)]
Laboratory Investigations:platelet count decreased
Musculoskeletal Disorders:myoglobin increased; rhabdomyolysis (some reports involved patients treated concurrently with daptomycin and HMG-CoA reductase inhibitors) [see Warnings and Precautions ( 5.2), Drug Interactions ( 7.1), and Clinical Pharmacology ( 12.3)]
Respiratory, Thoracic, and Mediastinal Disorders:cough, eosinophilic pneumonia, organizing pneumonia [see Warnings and Precautions ( 5.3)]
Nervous System Disorders:peripheral neuropathy [see Warnings and Precautions ( 5.6)]
Skin and Subcutaneous Tissue Disorders:serious skin reactions, including drug reaction with eosinophilia and systemic symptoms (DRESS), vesiculobullous rash (with or without mucous membrane involvement, including Stevens-Johnson syndrome [SJS] and toxic epidermal necrolysis [TEN]), and acute generalized exanthematous pustulosis [see Warnings and Precautions ( 5.4)]
Gastrointestinal Disorders:nausea, vomiting
Metabolic and Nutritional Disorders:hyperkalemia
Renal and Urinary Disorders:acute kidney injury, renal insufficiency, renal failure, and tubulointerstitial nephritis (TIN) [see Warnings and Precautions ( 5.5)]
Special Senses:visual disturbances
2.2 Dosage in Adults for Csssi (2.2 Dosage in Adults for cSSSI)
Administer Daptomycin for Injection 4 mg/kg to adult patients intravenously in 0.9% sodium chloride injection once every 24 hours for 7 to 14 days.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.2 Myopathy and Rhabdomyolysis
Myopathy, defined as muscle aching or muscle weakness in conjunction with increases in creatine phosphokinase (CPK) values to greater than 10 times the upper limit of normal (ULN), has been reported with the use of daptomycin. Rhabdomyolysis, with or without acute renal failure, has been reported [see Adverse Reactions ( 6.2)] .
Patients receiving Daptomycin for Injection should be monitored for the development of muscle pain or weakness, particularly of the distal extremities. In patients who receive Daptomycin for Injection, CPK levels should be monitored weekly, and more frequently in patients who received recent prior or concomitant therapy with an HMG-CoA reductase inhibitor or in whom elevations in CPK occur during treatment with Daptomycin for Injection.
In adult patients with renal impairment, both renal function and CPK should be monitored more frequently than once weekly [see Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3)] .
In Phase 1 studies and Phase 2 clinical trials in adults, CPK elevations appeared to be more frequent when daptomycin was dosed more than once daily. Therefore, daptomycin should not be dosed more frequently than once a day.
Daptomycin should be discontinued in patients with unexplained signs and symptoms of myopathy in conjunction with CPK elevations to levels >1,000 U/L (~5× ULN), and in patients without reported symptoms who have marked elevations in CPK, with levels >2,000 U/L (≥10× ULN). In addition, consideration should be given to suspending agents associated with rhabdomyolysis, such as HMG-CoA reductase inhibitors, temporarily in patients receiving daptomycin [see Drug Interactions ( 7.1)] .
7.1 Hmg Coa Reductase Inhibitors (7.1 HMG-CoA Reductase Inhibitors)
In healthy adult subjects, concomitant administration of daptomycin and simvastatin had no effect on plasma trough concentrations of simvastatin, and there were no reports of skeletal myopathy [see Clinical Pharmacology ( 12.3)] .
However, inhibitors of HMG-CoA reductase may cause myopathy, which is manifested as muscle pain or weakness associated with elevated levels of creatine phosphokinase (CPK). In the adult Phase 3 S. aureusbacteremia/endocarditis trial, some patients who received prior or concomitant treatment with an HMG-CoA reductase inhibitor developed elevated CPK [see Adverse Reactions ( 6.1)] . Experience with the coadministration of HMG-CoA reductase inhibitors and daptomycin in patients is limited; therefore, consideration should be given to suspending use of HMG-CoA reductase inhibitors temporarily in patients receiving daptomycin.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Allergic Reactions
Advise patients that allergic reactions, including serious skin, kidney, lung, or other organ reactions, could occur and that these serious reactions require immediate treatment. Patients should report any previous allergic reactions to daptomycin [see Warnings and Precautions ( 5.1, 5.4, 5.5)].
Muscle Pain or Weakness (Myopathy and Rhabdomyolysis, Peripheral Neuropathy
Advise patients to report muscle pain or weakness, especially in the forearms and lower legs, as well as tingling or numbness. [see Warnings and Precautions ( 5.2, 5.6)].
Cough, Breathlessness or Fever (Eosinophilic Pneumonia)
Advise patients to report any symptoms of cough, breathlessness, or fever [see Warnings and Precautions ( 5.3)].
C. difficile-Associated Diarrhea (CDAD)
Advise patients that diarrhea is a common problem caused by antibacterials that usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterials, including daptomycin, patients can develop watery and bloody stools (with or without stomach cramps and fever), even as late as 2 or more months after having received the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible [see Warnings and Precautions ( 5.8)].
Antibacterial Resistance
Patients should be counseled that antibacterial drugs, including Daptomycin for Injection, should be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Daptomycin for Injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be administered exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Daptomycin for Injection or other antibacterial drugs in the future.
Manufactured For:
Accord Healthcare, Inc.,
8041 Arco Corporate Drive,
Suite 200,
Raleigh, NC 27617,
USA.
Manufactured By:
Intas Pharmaceuticals Limited,
Ahmedabad-380 054, India.
10 2865 6 6035362
Issued: June 2025
8.6 Patients With Renal Impairment (8.6 Patients with Renal Impairment)
Daptomycin is eliminated primarily by the kidneys; therefore, a modification of daptomycin dosage interval is recommended for adult patients with CL CR<30 mL/min, including patients receiving hemodialysis or continuous ambulatory peritoneal dialysis (CAPD). In adult patients with renal impairment, both renal function and creatine phosphokinase (CPK) should be monitored more frequently than once weekly [see Dosage and Administration ( 2.6), Warnings and Precautions ( 5.2, 5.10), and Clinical Pharmacology ( 12.3)] .
The dosage regimen for daptomycin in pediatric patients with renal impairment has not been established.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
Daptomycin for Injection is supplied as a sterile, nonpyrogenic, preservative-free, pale yellow to light brown lyophilized cake in single-dose vials as follows:
| NDC | Daptomycin for Injection | Package Factor |
| 16729-434-05 | 350 mg Single-Dose Vial | 1 vial per carton |
| 16729-434-45 | 350 mg Single-Dose Vial | 10 vials per carton |
The container closure is not made with natural rubber latex.
2.8 Compatible Intravenous Solutions
Daptomycin for Injection is compatible with 0.9% sodium chloride injection and Lactated Ringer's injection.
5.9 Persisting Or Relapsing S. Aureus (5.9 Persisting or Relapsing S. aureus)
Patients with persisting or relapsing S. aureusbacteremia/endocarditis or poor clinical response should have repeat blood cultures. If a blood culture is positive for S. aureus, minimum inhibitory concentration (MIC) susceptibility testing of the isolate should be performed using a standardized procedure, and diagnostic evaluation of the patient should be performed to rule out sequestered foci of infection. Appropriate surgical intervention (e.g., debridement, removal of prosthetic devices, valve replacement surgery) and/or consideration of a change in antibacterial regimen may be required.
Failure of treatment due to persisting or relapsing S. aureusbacteremia/endocarditis may be due to reduced daptomycin susceptibility (as evidenced by increasing MIC of the S. aureusisolate) [see Clinical Studies ( 14.2)] .
7.2 Drug Laboratory Test Interactions (7.2 Drug-Laboratory Test Interactions)
Clinically relevant plasma concentrations of daptomycin have been observed to cause a significant concentration-dependent false prolongation of prothrombin time (PT) and elevation of International Normalized Ratio (INR) when certain recombinant thromboplastin reagents are utilized for the assay. The possibility of an erroneously elevated PT/INR result due to interaction with a recombinant thromboplastin reagent may be minimized by drawing specimens for PT or INR testing near the time of trough plasma concentrations of daptomycin. However, sufficient daptomycin concentrations may be present at trough to cause interaction.
If confronted with an abnormally high PT/INR result in a patient being treated with daptomycin, it is recommended that clinicians:
- Repeat the assessment of PT/INR, requesting that the specimen be drawn just prior to the next daptomycin dose (i.e., at trough concentration). If the PT/INR value obtained at trough remains substantially elevated above what would otherwise be expected, consider evaluating PT/INR utilizing an alternative method.
- Evaluate for other causes of abnormally elevated PT/INR results.
5.5 Tubulointerstitial Nephritis (tin) (5.5 Tubulointerstitial Nephritis (TIN))
TIN has been reported in post-marketing experience with daptomycin [see Adverse Reactions ( 6.2)]. Patients who develop new or worsening renal impairment while receiving daptomycin should undergo medical evaluation. If TIN is suspected, discontinue daptomycin promptly and institute appropriate treatment.
5.1 Anaphylaxis/hypersensitivity Reactions (5.1 Anaphylaxis/Hypersensitivity Reactions)
Anaphylaxis/hypersensitivity reactions have been reported with the use of antibacterial agents, including daptomycin for injection, and may be life-threatening. If an allergic reaction to Daptomycin for Injection occurs, discontinue the drug and institute appropriate therapy [see Adverse Reactions ( 6.2)].
5.12 Development of Drug Resistant Bacteria (5.12 Development of Drug-Resistant Bacteria)
Prescribing Daptomycin for Injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.6 Dosage in Patients With Renal Impairment (2.6 Dosage in Patients with Renal Impairment)
Adult Patients
No dosage adjustment is required in adult patients with creatinine clearance (CL CR) greater than or equal to 30 mL/min. The recommended dosage regimen for Daptomycin for Injection in adult patients with CL CRless than 30 mL/min, including adult patients on hemodialysis or continuous ambulatory peritoneal dialysis (CAPD), is 4 mg/kg (cSSSI) or 6 mg/kg ( S. aureusbloodstream infections) once every 48 hours ( Table 3). When possible, Daptomycin for Injection should be administered following the completion of hemodialysis on hemodialysis days [see Warnings and Precautions ( 5.2, 5.10), Use in Specific Populations ( 8.6), and Clinical Pharmacology ( 12.3)] .
|
* When possible, administer Daptomycin for Injection following the completion of hemodialysis on hemodialysis days. |
||
|
Creatinine Clearance
(CL CR ) |
Dosage Regimen in Adults | |
| cSSSI | S. aureus Bloodstream Infections | |
| Greater than or equal to 30 mL/min | 4 mg/kg once every 24 hours | 6 mg/kg once every 24 hours |
| Less than 30 mL/min, including hemodialysis and CAPD | 4 mg/kg once every 48 hours* | 6 mg/kg once every 48 hours* |
Pediatric Patients
The dosage regimen for Daptomycin for Injection in pediatric patients with renal impairment has not been established.
2.1 Important Administration Duration Instructions
Adults
Administer the appropriate volume of the reconstituted Daptomycin for Injection (concentration of 50 mg per mL) to adult patientsintravenously either by injection over a two (2) minute period or by intravenous infusion over a thirty (30) minute period [see Dosage and Administration ( 2.2, 2.4, 2.7)].
Pediatric Patients (1 to 17 Years of Age)
Unlike in adults, do NOT administer Daptomycin for Injection by injection over a two (2) minute period to pediatric patients.
- Pediatric Patients 7 to 17 years of Age:Administer Daptomycin for Injection intravenously by infusion over a 30-minute period [see Dosage and Administration ( 2.3, 2.5, 2.7)].
- Pediatric Patients 1 to 6 years of Age:Administer Daptomycin for Injection intravenously by infusion over a 60-minute period [see Dosage and Administration ( 2.3, 2.5, 2.7)].
14.1 Complicated Skin and Skin Structure Infections
Adults with cSSSI
Adult patients with clinically documented complicated skin and skin structure infections (cSSSI) ( Table 15) were enrolled in two randomized, multinational, multicenter, investigator-blinded trials comparing daptomycin (4 mg/kg IV every 24h) with either vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g IV per day). Patients could switch to oral therapy after a minimum of 4 days of IV treatment if clinical improvement was demonstrated. Patients known to have bacteremia at baseline were excluded. Patients with creatinine clearance (CL CR) between 30 and 70 mL/min were to receive a lower dose of daptomycin as specified in the protocol; however, the majority of patients in this subpopulation did not have the dose of daptomycin adjusted.
|
* Comparator: vancomycin (1 g IV q12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses). |
|||
|
†The majority of cases were subsequently categorized as complicated cellulitis, major abscesses, or traumatic wound infections. |
|||
| Primary Diagnosis |
Adult Patients
(Daptomycin / Comparator*) |
||
|
Study 9801
N=264 / N=266 |
Study 9901
N=270 / N=292 |
Pooled
N=534 / N=558 |
|
| Wound Infection | 99 (38%) / 116 (44%) | 102 (38%) / 108 (37%) | 201 (38%) / 224 (40%) |
| Major Abscess | 55 (21%) / 43 (16%) | 59 (22%) / 65 (22%) | 114 (21%) / 108 (19%) |
| Ulcer Infection | 71 (27%) / 75 (28%) | 53 (20%) / 68 (23%) | 124 (23%) / 143 (26%) |
| Other Infection † | 39 (15%) / 32 (12%) | 56 (21%) / 51 (18%) | 95 (18%) / 83 (15%) |
One trial was conducted primarily in the United States and South Africa (study 9801), and the second was conducted at non-US sites only (study 9901). The two trials were similar in design but differed in patient characteristics, including history of diabetes and peripheral vascular disease. There were a total of 534 adult patients treated with daptomycin and 558 treated with comparator in the two trials. The majority (89.7%) of patients received IV medication exclusively.
The efficacy endpoints in both trials were the clinical success rates in the intent-to-treat (ITT) population and in the clinically evaluable (CE) population. In study 9801, clinical success rates in the ITT population were 62.5% (165/264) in patients treated with daptomycin and 60.9% (162/266) in patients treated with comparator drugs. Clinical success rates in the CE population were 76.0% (158/208) in patients treated with daptomycin and 76.7% (158/206) in patients treated with comparator drugs. In study 9901, clinical success rates in the ITT population were 80.4% (217/270) in patients treated with daptomycin and 80.5% (235/292) in patients treated with comparator drugs. Clinical success rates in the CE population were 89.9% (214/238) in patients treated with daptomycin and 90.4% (226/250) in patients treated with comparator drugs.
The success rates by pathogen for microbiologically evaluable patients are presented in Table 16.
|
* Comparator: vancomycin (1 g IV q12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses). |
||
|
† As determined by the central laboratory. |
||
| Pathogen |
Success Rate
n/N (%) |
|
| Daptomycin | Comparator* | |
| Methicillin-susceptible Staphylococcus aureus(MSSA) † | 170/198 (86%) | 180/207 (87%) |
| Methicillin-resistant Staphylococcus aureus(MRSA) † | 21/28 (75%) | 25/36 (69%) |
| Streptococcus pyogenes | 79/84 (94%) | 80/88 (91%) |
| Streptococcus agalactiae | 23/27 (85%) | 22/29 (76%) |
| Streptococcus dysgalactiaesubsp. equisimilis | 8/8 (100%) | 9/11 (82%) |
| Enterococcus faecalis(vancomycin-susceptible only) | 27/37 (73%) | 40/53 (76%) |
Pediatric Patients (1 to 17 Years of Age) with cSSSI
The cSSSI pediatric trial was a single prospective multi-center, randomized, comparative trial. A total of 396 pediatric patients aged 1 to 17 years with cSSSI caused by Gram positive pathogens were enrolled into the study. Patients known to have bacteremia, osteomyelitis, endocarditis, and pneumonia at baseline were excluded. Patients were enrolled in a stepwise approach into four age groups and given age-dependent doses of daptomycin once daily for up to 14 days. The different age groups and doses evaluated were as follows: Adolescents (12 to 17 years) treated with 5 mg/kg of daptomycin (n=113), Children (7 to 11 years) treated with 7 mg/kg of daptomycin (n=113), Children (2 to 6 years) treated with 9 mg/kg of daptomycin (n=125) and Infants (1 to <2 years) treated with 10 mg/kg (n= 45).
Patients were randomized 2:1 to receive daptomycin or a standard of care (SOC) comparator, which included intravenous therapy with either vancomycin, clindamycin, or an anti-staphylococcal semi-synthetic penicillin (nafcillin, oxacillin, or cloxacillin). Patients could switch to oral therapy after clinical improvement was demonstrated (no minimum IV dosing was required).
The primary objective of this study was to evaluate the safety of daptomycin. The clinical outcome was determined by resolution or improvement of symptoms at the End-of-Treatment (EOT), 3 days after the last dose, and Test-of-Cure (TOC), 7 to 14 days after the last dose. Investigator observed outcomes were verified in a blinded fashion. Of the 396 subjects randomized in the study, 389 subjects were treated with daptomycin or comparator and included in the ITT population. Of these, 257 subjects were randomized to the daptomycin group and 132 subjects were randomized to the comparator group. Approximately 95% of subjects switched to oral therapy. The mean day of switch was day 4, and ranged from day 1 to day 14. The clinical success rates determined at 7 to 14 days after last dose of therapy (IV and oral) (TOC visit) were 88% (227/257) for daptomycin and 86% (114/132) for comparator.
2.4 Dosage in Adult Patients With Staphylococcus Aureus (2.4 Dosage in Adult Patients with Staphylococcus aureus)
Administer Daptomycin for Injection 6 mg/kg to adult patients intravenously in 0.9% sodium chloride injection once every 24 hours for 2 to 6 weeks. There are limited safety data for the use of Daptomycin for Injection for more than 28 days of therapy. In the Phase 3 trial, there were a total of 14 adult patients who were treated with daptomycin for more than 28 days.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies in animals have not been conducted to evaluate the carcinogenic potential of daptomycin. However, neither mutagenic nor clastogenic potential was found in a battery of genotoxicity tests, including the Ames assay, a mammalian cell gene mutation assay, a test for chromosomal aberrations in Chinese hamster ovary cells, an in vivomicronucleus assay, an in vitroDNA repair assay, and an in vivosister chromatid exchange assay in Chinese hamsters.
Daptomycin did not affect the fertility or reproductive performance of male and female rats when administered intravenously at doses of 25, 75, or 150 mg/kg/day, which is approximately up to 9 times the estimated human exposure level based upon AUCs (or approximately up to 4 times the recommended human dose of 6 mg/kg based on body surface area comparison).
1.1 Complicated Skin and Skin Structure Infections (csssi) (1.1 Complicated Skin and Skin Structure Infections (cSSSI))
Daptomycin for Injection is indicated for the treatment of adult and pediatric patients (1 to 17 years of age) with complicated skin and skin structure infections (cSSSI) caused by susceptible isolates of the following Gram-positive bacteria: Staphylococcus aureus(including methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiaesubsp. equisimilis,and Enterococcus faecalis(vancomycin-susceptible isolates only).
2.7 Preparation and Administration of Daptomycin for Injection
There are other formulations of daptomycin that have differences concerning reconstitution and storage. Carefully follow the reconstitution and storage procedures described in this labeling.
2.3 Dosage in Pediatric Patients (1 to 17 Years of Age) for Csssi (2.3 Dosage in Pediatric Patients (1 to 17 Years of Age) for cSSSI)
The recommended dosage regimens based on age for pediatric patients with cSSSI are shown in Table 1. Administer Daptomycin for Injection intravenously in 0.9% sodium chloride injection once every 24 hours for up to 14 days.
| Age Range | Dosage Regimen* | Duration of therapy |
| 12 to 17 years | 5 mg/kg once every 24 hours infused over 30 minutes | Up to 14 days |
| 7 to 11 years | 7 mg/kg once every 24 hours infused over 30 minutes | |
| 2 to 6 years | 9 mg/kg once every 24 hours infused over 60 minutes | |
| 1 to less than 2 years | 10 mg/kg once every 24 hours infused over 60 minutes | |
|
*Recommended dosage regimen is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. |
5.4 Drug Reaction With Eosinophilia and Systemic Symptoms (dress) (5.4 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS))
DRESS has been reported in post-marketing experience with daptomycin [see Adverse Reactions ( 6.2)]. Patients who develop skin rash, fever, peripheral eosinophilia, and systemic organ (for example, hepatic, renal, pulmonary) impairment while receiving daptomycin should undergo medical evaluation. If DRESS is suspected, discontinue daptomycin promptly and institute appropriate treatment.
5.10 Decreased Efficacy in Patients With Moderate Baseline Renal Impairment (5.10 Decreased Efficacy in Patients with Moderate Baseline Renal Impairment)
Limited data are available from the two Phase 3 complicated skin and skin structure infection (cSSSI) trials regarding clinical efficacy of daptomycin treatment in adult patients with creatinine clearance (CL CR) <50 mL/min; only 31/534 (6%) patients treated with daptomycin in the intent-to-treat (ITT) population had a baseline CL CR<50 mL/min. Table 4shows the number of adult patients by renal function and treatment group who were clinical successes in the Phase 3 cSSSI trials.
| CL CR |
Success Rate
n/N (%) |
|
|
Daptomycin
4 mg/kg every 24h |
Comparator | |
| 50 to 70 mL/min | 25/38 (66%) | 30/48 (63%) |
| 30 to <50 mL/min | 7/15 (47%) | 20/35 (57%) |
In a subgroup analysis of the ITT population in the Phase 3 S. aureusbacteremia/endocarditis trial, clinical success rates, as determined by a treatment-blinded Adjudication Committee [see Clinical Studies ( 14.2)] , in the daptomycin-treated adult patients were lower in patients with baseline CL CR<50 mL/min (see Table 5). A decrease of the magnitude shown in Table 5was not observed in comparator-treated patients.
| Baseline CL CR |
Success Rate
n/N (%) |
|||
|
Daptomycin
6 mg/kg every 24h |
Comparator | |||
| Bacteremia | Right-Sided Infective Endocarditis | Bacteremia | Right-Sided Infective Endocarditis | |
| >80 mL/min | 30/50 (60%) | 7/14 (50%) | 19/42 (45%) | 5/11 (46%) |
| 50 to 80 mL/min | 12/26 (46%) | 1/4 (25%) | 13/31 (42%) | 1/2 (50%) |
| 30 to <50 mL/min | 2/14 (14%) | 0/1 (0%) | 7/17 (41%) | 1/1 (100%) |
Consider these data when selecting antibacterial therapy for use in adult patients with baseline moderate to severe renal impairment.
5.11 Increased International Normalized Ratio (inr)/prolonged Prothrombin Time (5.11 Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time)
Clinically relevant plasma concentrations of daptomycin have been observed to cause a significant concentration-dependent false prolongation of prothrombin time (PT) and elevation of International Normalized Ratio (INR) when certain recombinant thromboplastin reagents are utilized for the assay [see Drug Interactions ( 7.2)].
2.5 Dosage in Pediatric Patients (1 to 17 Years of Age) With Staphylococcus Aureus (2.5 Dosage in Pediatric Patients (1 to 17 Years of Age) with Staphylococcus aureus)
The recommended dosage regimens based on age for pediatric patients with S. aureusbloodstream infections (bacteremia) are shown in Table 2. Administer Daptomycin for Injection intravenously in 0.9% sodium chloride injection once every 24 hours for up to 42 days.
| Age group | Dosage* | Duration of therapy |
| 12 to 17 years | 7 mg/kg once every 24 hours infused over 30 minutes | Up to 42 days |
| 7 to 11 years | 9 mg/kg once every 24 hours infused over 30 minutes | |
| 1 to 6 years | 12 mg/kg once every 24 hours infused over 60 minutes | |
|
*Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established. |
5.7 Potential Nervous System And/or Muscular System Effects in Pediatric Patients Younger Than 12 Months (5.7 Potential Nervous System and/or Muscular System Effects in Pediatric Patients Younger than 12 Months)
Avoid use of daptomycin in pediatric patients younger than 12 months due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs with intravenous daptomycin [see Nonclinical Toxicology ( 13.2)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:39.175686 · Updated: 2026-03-14T22:46:59.478961