These Highlights Do Not Include All The Information Needed To Use Vancomycin Hydrochloride For Injection Safely And Effectively. See Full Prescribing Information For Vancomycin Hydrochloride For Injection.
f623889c-a47a-4b3a-b3a9-e04051f5c2df
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Vancomycin Hydrochloride for Injection is a glycopeptide antibacterial indicated in adult and pediatric patients (neonates and older) for the treatment of: Septicemia ( 1.1 ) Infective Endocarditis ( 1.2 ) Skin and Skin Structure Infections ( 1.3 ) Bone Infections ( 1.4 ) Lower Respiratory Tract Infections ( 1.5 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.6 )
Indications and Usage
Vancomycin Hydrochloride for Injection is a glycopeptide antibacterial indicated in adult and pediatric patients (neonates and older) for the treatment of: Septicemia ( 1.1 ) Infective Endocarditis ( 1.2 ) Skin and Skin Structure Infections ( 1.3 ) Bone Infections ( 1.4 ) Lower Respiratory Tract Infections ( 1.5 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.6 )
Dosage and Administration
Administer Vancomycin Hydrochloride for Injection in a diluted solution over 60 minutes or greater to reduce the risk of infusion reactions. See full prescribing information for further important administration and preparation instructions ( 2.1 , 2.5 ) Adult Patients: 2 grams divided either as 0.5 grams (g) every 6 hours or 1 gram every 12 hours ( 2.2 ) Pediatric Patients (1 month and older): 10 mg/kg per dose given every 6 hours ( 2.3 ) Neonates: See full prescribing information for recommended doses in neonates ( 2.3 ) Patients with renal impairment: See full prescribing information for recommended doses in patients with renal impairment ( 2.4 )
Warnings and Precautions
Infusion Reactions: Hypotension, including shock and cardiac arrest, wheezing, dyspnea, urticaria, muscular, chest pain and “vancomycin infusion reaction” which manifests as pruritus and erythema that involves the face, neck and upper torso may occur with rapid intravenous administration. To reduce the risk of infusion reactions, administer Vancomycin Hydrochloride for Injection in a diluted solution over a period of 60 minutes or greater and also prior to intravenous anesthetic agents. ( 2.1 , 5.1 ) Nephrotoxicity: Systemic vancomycin exposure may result in acute kidney injury (AKI) including acute renal failure, mainly due to interstitial nephritis or less commonly acute tubular necrosis. Monitor serum vancomycin concentrations and renal function. ( 5.2 ) Ototoxicity: Ototoxicity has occurred in patients receiving vancomycin hydrochloride. Monitor for signs and symptoms of ototoxicity during therapy. Monitor serum vancomycin concentrations and renal function. Assessment of auditory function may be appropriate in some instances. ( 5.3 ) Severe Dermatologic Reactions: Discontinue Vancomycin Hydrochloride for Injection at the first appearance of skin rashes, mucosal lesions, or blisters ( 5.4 ). Clostridioides difficile- Associated Diarrhea: Evaluate patients if diarrhea occurs. ( 5.5 ) Neutropenia: Periodically monitor leukocyte count. ( 5.7 ) Phlebitis: To reduce the risk of local irritation and phlebitis administer Vancomycin Hydrochloride for Injection by a secure intravenous route of administration. ( 5.8 ) Development of Drug-Resistant Bacteria: Prescribing Vancomycin Hydrochloride for Injection in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug resistant bacteria. ( 5.9 )
Contraindications
Vancomycin Hydrochloride for Injection is contraindicated in patients with known hypersensitivity to vancomycin.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Infusion Reactions [see Warnings and Precautions ( 5.1 )] Nephrotoxicity [see Warnings and Precautions ( 5.2 )] Ototoxicity [see Warnings and Precautions ( 5.3 )] Clostridioides difficile -Associated Diarrhea [see Warnings and Precautions ( 5.5 )] Hemorrhagic Occlusive Retinal Vasculitis [see Warnings and Precautions ( 5.6 )] Neutropenia [see Warnings and Precautions ( 5.7 )]
Drug Interactions
Anesthetic Agents: Concomitant administration of vancomycin and anesthetic agents has been associated with erythema and histamine-like flushing. ( 2.1 , 7.1 ) Piperacillin/Tazobactam: Increased incidence of acute kidney injury in patients receiving concomitant piperacillin/tazobactam and vancomycin as compared to vancomycin alone. Monitor kidney function in patients ( 7.2 )
Storage and Handling
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored powder or plug in single-dose flip top vials that contain vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. They are available as follows: NDC Vancomycin Hydrochloride for Injection, USP Package Factor 25021-147-20 750 mg per Single-Dose vial (equivalent to 750 mg vancomycin in a 20 mL USP Type-1 clear glass tubular lyo vials with a flip off Pink color button (Matte top) seal.) 10 vials per carton 25021-148-30 1.25 grams per Single-Dose vial (equivalent to 1.25 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Purple color button (Matte top) seal.) 10 vials per carton 25021-149-30 1.5 grams per Single-Dose vial (equivalent to 1.5 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Light Green color button (Matte top) seal.) 10 vials per carton
How Supplied
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored powder or plug in single-dose flip top vials that contain vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. They are available as follows: NDC Vancomycin Hydrochloride for Injection, USP Package Factor 25021-147-20 750 mg per Single-Dose vial (equivalent to 750 mg vancomycin in a 20 mL USP Type-1 clear glass tubular lyo vials with a flip off Pink color button (Matte top) seal.) 10 vials per carton 25021-148-30 1.25 grams per Single-Dose vial (equivalent to 1.25 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Purple color button (Matte top) seal.) 10 vials per carton 25021-149-30 1.5 grams per Single-Dose vial (equivalent to 1.5 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Light Green color button (Matte top) seal.) 10 vials per carton
Medication Information
Warnings and Precautions
Infusion Reactions: Hypotension, including shock and cardiac arrest, wheezing, dyspnea, urticaria, muscular, chest pain and “vancomycin infusion reaction” which manifests as pruritus and erythema that involves the face, neck and upper torso may occur with rapid intravenous administration. To reduce the risk of infusion reactions, administer Vancomycin Hydrochloride for Injection in a diluted solution over a period of 60 minutes or greater and also prior to intravenous anesthetic agents. ( 2.1 , 5.1 ) Nephrotoxicity: Systemic vancomycin exposure may result in acute kidney injury (AKI) including acute renal failure, mainly due to interstitial nephritis or less commonly acute tubular necrosis. Monitor serum vancomycin concentrations and renal function. ( 5.2 ) Ototoxicity: Ototoxicity has occurred in patients receiving vancomycin hydrochloride. Monitor for signs and symptoms of ototoxicity during therapy. Monitor serum vancomycin concentrations and renal function. Assessment of auditory function may be appropriate in some instances. ( 5.3 ) Severe Dermatologic Reactions: Discontinue Vancomycin Hydrochloride for Injection at the first appearance of skin rashes, mucosal lesions, or blisters ( 5.4 ). Clostridioides difficile- Associated Diarrhea: Evaluate patients if diarrhea occurs. ( 5.5 ) Neutropenia: Periodically monitor leukocyte count. ( 5.7 ) Phlebitis: To reduce the risk of local irritation and phlebitis administer Vancomycin Hydrochloride for Injection by a secure intravenous route of administration. ( 5.8 ) Development of Drug-Resistant Bacteria: Prescribing Vancomycin Hydrochloride for Injection in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug resistant bacteria. ( 5.9 )
Indications and Usage
Vancomycin Hydrochloride for Injection is a glycopeptide antibacterial indicated in adult and pediatric patients (neonates and older) for the treatment of: Septicemia ( 1.1 ) Infective Endocarditis ( 1.2 ) Skin and Skin Structure Infections ( 1.3 ) Bone Infections ( 1.4 ) Lower Respiratory Tract Infections ( 1.5 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.6 )
Dosage and Administration
Administer Vancomycin Hydrochloride for Injection in a diluted solution over 60 minutes or greater to reduce the risk of infusion reactions. See full prescribing information for further important administration and preparation instructions ( 2.1 , 2.5 ) Adult Patients: 2 grams divided either as 0.5 grams (g) every 6 hours or 1 gram every 12 hours ( 2.2 ) Pediatric Patients (1 month and older): 10 mg/kg per dose given every 6 hours ( 2.3 ) Neonates: See full prescribing information for recommended doses in neonates ( 2.3 ) Patients with renal impairment: See full prescribing information for recommended doses in patients with renal impairment ( 2.4 )
Contraindications
Vancomycin Hydrochloride for Injection is contraindicated in patients with known hypersensitivity to vancomycin.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Infusion Reactions [see Warnings and Precautions ( 5.1 )] Nephrotoxicity [see Warnings and Precautions ( 5.2 )] Ototoxicity [see Warnings and Precautions ( 5.3 )] Clostridioides difficile -Associated Diarrhea [see Warnings and Precautions ( 5.5 )] Hemorrhagic Occlusive Retinal Vasculitis [see Warnings and Precautions ( 5.6 )] Neutropenia [see Warnings and Precautions ( 5.7 )]
Drug Interactions
Anesthetic Agents: Concomitant administration of vancomycin and anesthetic agents has been associated with erythema and histamine-like flushing. ( 2.1 , 7.1 ) Piperacillin/Tazobactam: Increased incidence of acute kidney injury in patients receiving concomitant piperacillin/tazobactam and vancomycin as compared to vancomycin alone. Monitor kidney function in patients ( 7.2 )
Storage and Handling
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored powder or plug in single-dose flip top vials that contain vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. They are available as follows: NDC Vancomycin Hydrochloride for Injection, USP Package Factor 25021-147-20 750 mg per Single-Dose vial (equivalent to 750 mg vancomycin in a 20 mL USP Type-1 clear glass tubular lyo vials with a flip off Pink color button (Matte top) seal.) 10 vials per carton 25021-148-30 1.25 grams per Single-Dose vial (equivalent to 1.25 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Purple color button (Matte top) seal.) 10 vials per carton 25021-149-30 1.5 grams per Single-Dose vial (equivalent to 1.5 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Light Green color button (Matte top) seal.) 10 vials per carton
How Supplied
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored powder or plug in single-dose flip top vials that contain vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. They are available as follows: NDC Vancomycin Hydrochloride for Injection, USP Package Factor 25021-147-20 750 mg per Single-Dose vial (equivalent to 750 mg vancomycin in a 20 mL USP Type-1 clear glass tubular lyo vials with a flip off Pink color button (Matte top) seal.) 10 vials per carton 25021-148-30 1.25 grams per Single-Dose vial (equivalent to 1.25 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Purple color button (Matte top) seal.) 10 vials per carton 25021-149-30 1.5 grams per Single-Dose vial (equivalent to 1.5 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Light Green color button (Matte top) seal.) 10 vials per carton
Description
Vancomycin Hydrochloride for Injection is a glycopeptide antibacterial indicated in adult and pediatric patients (neonates and older) for the treatment of: Septicemia ( 1.1 ) Infective Endocarditis ( 1.2 ) Skin and Skin Structure Infections ( 1.3 ) Bone Infections ( 1.4 ) Lower Respiratory Tract Infections ( 1.5 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.6 )
Section 42229-5
Pediatric Patients (Aged 1 month and older)
The usual intravenous dosage of vancomycin is 10 mg/kg per dose given every 6 hours. Each dose should be administered over a period of at least 60 minutes. Close monitoring of serum concentrations of vancomycin may be warranted in these patients.
Section 44425-7
Storage Conditions
Prior to reconstitution, store at 20° to 25°C (68° to 77°F); excursions permitted to 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Lyophilized.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Discard unused portion.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-147-20
Rx only
Vancomycin Hydrochloride for Injection, USP
750 mg per vial
For Intravenous Use
AFTER RECONSTITUTION MUST BE FURTHER
DILUTED. SEE PACKAGE INSERT
Sterile Powder
Single-Dose Vial
1.6 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
Supportive care is advised, with maintenance of glomerular filtration. Vancomycin is poorly removed by dialysis. Hemofiltration and hemoperfusion with polysulfone resin have been reported to result in increased vancomycin clearance.
For current information on the management of overdosage, contact the National Poison Control Center at 1-800-222-1222 or www.poison.org.
15 References
- Byrd RA., Gries CL, Buening M.: Developmental Toxicology Studies of Vancomycin Hydrochloride Administered Intravenously to Rats and Rabbits. Fundam Appl Toxicol 1994; 23: 590-597.
1.1 Septicemia
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of septicemia due to:
- Susceptible isolates of methicillin-resistant Staphylococcus aureus (MRSA) and coagulase negative staphylococci.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
11 Description
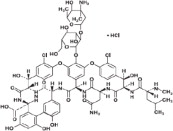
Vancomycin Hydrochloride for Injection, USP, contains the hydrochloride salt of vancomycin, a tricyclic glycopeptide antibacterial derived from Amycolatopsis orientalis (formerly Nocardia orientalis). The chemical name for vancomycin hydrochloride, USP is (Sa)-(3S,6R,7R,22R,23S,26S,36R,38aR)-44-[[2-O-(3-Amino-2,3,6-trideoxy-3-C-methyl-α-L-lyxo-hexopyranosyl)-β-D-glucopyranosyl]oxy]-3-(carbamoylmethyl)-10,19-dichloro-2,3,4,5,6,7,23,24,25,26,36,37,38,38a-tetradecahydro-7,22,28,30,32-pentahydroxy-6-[(2R)-4-methyl-2-(methylamino)valeramido]-2,5,24,38,39-pentaoxo-22H-8,11:18,21-dietheno-23,36-(iminomethano)-13,16:31,35-dimetheno-1H,16H-[l,6,9]oxadiazacyclohexadecino[4,5-m][10,2,16]-benzoxadiazacyclotetracosine-26-carboxylic acid, monohydrochloride. The molecular formula is C66H75Cl2N9O24 • HCl and the molecular weight is 1,485.71. Vancomycin hydrochloride, USP has the following structural formula:
Vancomycin Hydrochloride for Injection, USP is a sterile off-white to light tan colored lyophilized plug or powder for injection. Vancomycin Hydrochloride for Injection, USP is supplied in single-dose vials, containing 769 mg, 1.28 grams, or 1.54 grams of vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. The lyophilized powder is reconstituted with Sterile Water for Injection, USP which forms a clear, colorless or light to dark tan solution and subsequently diluted prior to intravenous administration [see Dosage and Administration (2.5)].
5.3 Ototoxicity
Ototoxicity has occurred in patients receiving Vancomycin Hydrochloride for Injection. It may be transient or permanent. Ototoxicity manifests as tinnitus, hearing loss, dizziness or vertigo. The risk is higher in older patients, patients who are receiving higher doses, who have an underlying hearing loss, who are receiving concomitant therapy with another ototoxic agent, such as an aminoglycoside or who have underlying renal impairment. Monitor for signs and symptoms of ototoxicity during therapy. Monitor serum vancomycin concentrations and renal function in all patients receiving parenteral vancomycin. Discontinue Vancomycin Hydrochloride for Injection if ototoxicity occurs. Dosage of Vancomycin Hydrochloride for Injection must be adjusted for patients with renal impairment [see Dosage and Administration (2.3)]. Serial tests of auditory function may be helpful in order to minimize the risk of ototoxicity.
5.7 Neutropenia
Reversible neutropenia has been reported in patients receiving Vancomycin Hydrochloride for Injection [see Adverse Reactions (6.1)]. Patients who will undergo prolonged therapy with Vancomycin Hydrochloride for Injection or those who are receiving concomitant drugs which may cause neutropenia should have periodic monitoring of the leukocyte count.
8.4 Pediatric Use
Vancomycin Hydrochloride for Injection is indicated in pediatric patients (neonates and older). In pediatric patients, monitor vancomycin serum concentration and renal function when administering Vancomycin Hydrochloride for Injection [see Dosage and Administration (2.2, 2.3) and Warnings and Precautions (5.2)]. More severe infusion related reactions related to vancomycin administration may occur in pediatric patients. Concomitant administration of vancomycin and intravenous anesthetic agents has been associated with erythema and histamine-like flushing in all patients including pediatric patients [see Warnings and Precautions (5.1)].
8.5 Geriatric Use
Vancomycin Hydrochloride for Injection is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection [see Dosage and Administration (2.2)], and it may be useful to monitor renal function [see Warnings and Precautions (5.2)].
5.2 Nephrotoxicity
Vancomycin Hydrochloride for Injection can result in acute kidney injury (AKI), including acute renal failure, mainly due to interstitial nephritis or less commonly acute tubular necrosis. AKI is manifested by increasing blood urea nitrogen (BUN) and serum creatinine (Cr). The risk of AKI increases with higher vancomycin serum levels, prolonged exposure, concomitant administration of other nephrotoxic drugs, concomitant administration of piperacillin-tazobactam [see Drug Interactions (7.2)], volume depletion, pre-existing renal impairment and in critically ill patients and patients with co-morbid conditions that predispose to renal impairment.
Monitor serum vancomycin concentrations and renal function in all patients receiving Vancomycin Hydrochloride for Injection. More frequent monitoring is recommended in patients with comorbidities that predispose to impairment in renal function or are concomitantly receiving other nephrotoxic drugs, in critically ill patients, in patients with changing renal function, and in patients requiring higher therapeutic vancomycin levels. If acute kidney injury occurs, discontinue Vancomycin Hydrochloride for Injection or reduce the dose.
1.4 Bone Infections
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of bone infections due to:
- Susceptible isolates of MRSA and coagulase negative staphylococci.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
4 Contraindications
Vancomycin Hydrochloride for Injection is contraindicated in patients with known hypersensitivity to vancomycin.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infusion Reactions [see Warnings and Precautions (5.1)]
- Nephrotoxicity [see Warnings and Precautions (5.2)]
- Ototoxicity [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.5)]
- Hemorrhagic Occlusive Retinal Vasculitis [see Warnings and Precautions (5.6)]
- Neutropenia [see Warnings and Precautions (5.7)]
7 Drug Interactions
- Anesthetic Agents: Concomitant administration of vancomycin and anesthetic agents has been associated with erythema and histamine-like flushing. (2.1, 7.1)
- Piperacillin/Tazobactam: Increased incidence of acute kidney injury in patients receiving concomitant piperacillin/tazobactam and vancomycin as compared to vancomycin alone. Monitor kidney function in patients (7.2)
12.2 Pharmacodynamics
The pharmacodynamics of vancomycin is unknown.
12.3 Pharmacokinetics
In subjects with normal kidney function, multiple intravenous dosing of 1 g of vancomycin (15 mg/kg) infused over 60 minutes produces mean plasma concentrations of approximately 63 mcg/mL immediately after the completion of infusion, mean plasma concentrations of approximately 23 mcg/mL 2 hours after infusion, and mean plasma concentrations of approximately 8 mcg/mL 11 hours after the end of the infusion. Multiple dosing of 500 mg infused over 30 minutes produces mean plasma concentrations of about 49 mcg/mL at the completion of infusion, mean plasma concentrations of about 19 mcg/mL 2 hours after infusion, and mean plasma concentrations of about 10 mcg/mL 6 hours after infusion. The plasma concentrations during multiple dosing are like those after a single dose.
7.1 Anesthetic Agents
5.1 Infusion Reactions
Hypotension, including shock and cardiac arrest, wheezing, dyspnea, urticaria, muscular and chest pain may occur with rapid Vancomycin Hydrochloride for Injection administration. The reactions may be more severe in younger patients, particularly children, and in patients receiving concomitant muscle relaxant anesthetics.
Rapid intravenous administration of Vancomycin Hydrochloride for Injection may also be associated with “vancomycin infusion reaction”, which manifests as pruritus and erythema that involves the face, neck and upper torso.
Infusion-related adverse reactions are related to both the concentration and the rate of administration of vancomycin. Infusion-related adverse reactions may occur, however, at any rate or concentration.
Administer Vancomycin Hydrochloride for Injection in a diluted solution over a period of 60 minutes or greater to reduce the risk of infusion-related adverse reactions. In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used; use of such higher concentrations may increase the risk of infusion-related adverse reactions. Administer prior to intravenous anesthetic agents when feasible. Stop the infusion if a reaction occurs.
1 Indications and Usage
Vancomycin Hydrochloride for Injection is a glycopeptide antibacterial indicated in adult and pediatric patients (neonates and older) for the treatment of:
- Septicemia (1.1)
- Infective Endocarditis (1.2)
- Skin and Skin Structure Infections (1.3)
- Bone Infections (1.4)
- Lower Respiratory Tract Infections (1.5)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.6)
12.1 Mechanism of Action
Vancomycin is an antibacterial drug [see Microbiology (12.4)].
1.2 Infective Endocarditis
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of infective endocarditis due to:
- Susceptible isolates of MRSA.
- Viridans group streptococci Streptococcus gallolyticus (previously known as Streptococcus bovis), Enterococcus species and Corynebacterium species. For enterococcal endocarditis, use Vancomycin Hydrochloride for Injection in combination with an aminoglycoside.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of early-onset prosthetic valve endocarditis caused by Staphylococcus epidermidis in combination with rifampin and an aminoglycoside.
5 Warnings and Precautions
- Infusion Reactions: Hypotension, including shock and cardiac arrest, wheezing, dyspnea, urticaria, muscular, chest pain and “vancomycin infusion reaction” which manifests as pruritus and erythema that involves the face, neck and upper torso may occur with rapid intravenous administration. To reduce the risk of infusion reactions, administer Vancomycin Hydrochloride for Injection in a diluted solution over a period of 60 minutes or greater and also prior to intravenous anesthetic agents. (2.1, 5.1)
- Nephrotoxicity: Systemic vancomycin exposure may result in acute kidney injury (AKI) including acute renal failure, mainly due to interstitial nephritis or less commonly acute tubular necrosis. Monitor serum vancomycin concentrations and renal function. (5.2)
- Ototoxicity: Ototoxicity has occurred in patients receiving vancomycin hydrochloride. Monitor for signs and symptoms of ototoxicity during therapy. Monitor serum vancomycin concentrations and renal function. Assessment of auditory function may be appropriate in some instances. (5.3)
- Severe Dermatologic Reactions: Discontinue Vancomycin Hydrochloride for Injection at the first appearance of skin rashes, mucosal lesions, or blisters (5.4).
- Clostridioides difficile- Associated Diarrhea: Evaluate patients if diarrhea occurs. (5.5)
- Neutropenia: Periodically monitor leukocyte count. (5.7)
- Phlebitis: To reduce the risk of local irritation and phlebitis administer Vancomycin Hydrochloride for Injection by a secure intravenous route of administration. (5.8)
- Development of Drug-Resistant Bacteria: Prescribing Vancomycin Hydrochloride for Injection in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug resistant bacteria. (5.9)
2 Dosage and Administration
- Administer Vancomycin Hydrochloride for Injection in a diluted solution over 60 minutes or greater to reduce the risk of infusion reactions.
- See full prescribing information for further important administration and preparation instructions (2.1, 2.5)
- Adult Patients: 2 grams divided either as 0.5 grams (g) every 6 hours or 1 gram every 12 hours (2.2)
- Pediatric Patients (1 month and older): 10 mg/kg per dose given every 6 hours (2.3)
- Neonates: See full prescribing information for recommended doses in neonates (2.3)
- Patients with renal impairment: See full prescribing information for recommended doses in patients with renal impairment (2.4)
7.2 Piperacillin Tazobactam
Studies have detected an increased incidence of acute kidney injury in patients administered concomitant piperacillin/tazobactam and vancomycin as compared to vancomycin alone. Monitor kidney function in patients receiving concomitant piperacillin/tazobactam and vancomycin. No pharmacokinetic interactions have been noted between piperacillin/tazobactam and vancomycin.
3 Dosage Forms and Strengths
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored plug or powder in single-dose vials containing vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base.
5.5 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Vancomycin Hydrochloride for Injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Clinically significant serum concentrations have been reported in some patients being treated for active C. difficile-induced pseudomembranous colitis after multiple oral doses of vancomycin.
Prolonged use of Vancomycin Hydrochloride for Injection may result in the overgrowth of nonsusceptible microorganisms. Careful observation of the patient is essential. If superinfection occurs during therapy, appropriate measures should be taken. In rare instances, there have been reports of pseudomembranous colitis due to C. difficile developing in patients who received intravenous Vancomycin Hydrochloride for Injection.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of vancomycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders: Drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP) [see Warnings and Precautions (5.4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following adverse reactions associated with the use of Vancomycin Hydrochloride for Injection were identified in clinical trials:
Immune system disorders: Hypersensitivity reactions including anaphylaxis and “vancomycin infusion reaction” [see Warnings and Precautions (5.1)]
Skin and subcutaneous tissue disorders: Erythema (especially of the face, neck and upper torso) and pruritus which are manifestations of rashes including exfoliative dermatitis, toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), Linear IgA bullous dermatosis (LABD) [see Warnings and Precautions (5.4)].
Renal and urinary disorders: Acute kidney injury and interstitial nephritis
Ear and Labyrinth Disorders: Tinnitus, hearing loss, vertigo
Blood and Lymphatic System Disorders: Agranulocytosis, neutropenia, pancytopenia, leukopenia, thrombocytopenia, eosinophilia
Gastrointestinal Disorders: Pseudomembranous colitis [see Warnings and Precautions (5.5)]
Cardiac Disorders: Cardiac arrest, chest pain
General Disorders and Administration Site Conditions: General discomfort, fever, chills, phlebitis, injection site irritation, injection site pain and necrosis following intramuscular injection, chemical peritonitis following intraperitoneal administration
(Vancomycin Hydrochloride for Injection is not approved for intramuscular and intraperitoneal administration) [see Warnings and Precautions (5.7)]
Laboratory Abnormalities: Elevated blood urea nitrogen, elevated serum creatinine
Musculoskeletal and connective tissue disorders: Muscle pain
Nervous system disorders: Dizziness
Respiratory, thoracic and mediastinal disorders: Wheezing, dyspnea
Vascular disorders: Hypotension, shock, vasculitis
5.4 Severe Dermatologic Reactions
Severe dermatologic reactions such as toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and linear IgA bullous dermatosis (LABD) have been reported in association with the use of vancomycin. Cutaneous signs or symptoms reported include skin rashes, mucosal lesions, and blisters.
Discontinue Vancomycin Hydrochloride for Injection at the first appearance of signs and symptoms of TEN, SJS, DRESS, AGEP, or LABD.
16 How Supplied/storage and Handling
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored powder or plug in single-dose flip top vials that contain vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. They are available as follows:
| NDC | Vancomycin Hydrochloride for Injection, USP | Package Factor |
| 25021-147-20 | 750 mg per Single-Dose vial (equivalent to 750 mg vancomycin in a 20 mL USP Type-1 clear glass tubular lyo vials with a flip off Pink color button (Matte top) seal.) |
10 vials per carton |
| 25021-148-30 | 1.25 grams per Single-Dose vial (equivalent to 1.25 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Purple color button (Matte top) seal.) |
10 vials per carton |
| 25021-149-30 | 1.5 grams per Single-Dose vial (equivalent to 1.5 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Light Green color button (Matte top) seal.) |
10 vials per carton |
7.3 Ototoxic And/or Nephrotoxic Drugs
Concurrent and/or sequential systemic or topical use of other potentially, neurotoxic and/or nephrotoxic drugs requires more frequent monitoring of renal function.
1.3 Skin and Skin Structure Infections
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of skin and skin structure infections due to:
- Susceptible isolates of MRSA and coagulase negative staphylococci.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
1.5 Lower Respiratory Tract Infections
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of lower respiratory tract infections due to:
- Susceptible isolates of MRSA
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
2.1 Important Administration Instructions
To reduce the risk of infusion related adverse reactions, administer Vancomycin Hydrochloride for Injection in a diluted solution over 60 minutes or greater [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. Vancomycin Hydrochloride for Injection concentrations of no more than 5 mg/mL are recommended in adults [see Dosage and Administration (2.2)]. See also age-specific recommendations [see Dosage and Administration (2.3)]. In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used [see Warnings and Precautions (5.1)].
Administer Vancomycin Hydrochloride for Injection prior to intravenous anesthetic agents to reduce the risk of infusion related adverse reactions [see Warnings and Precautions (5.1)].
Administer Vancomycin Hydrochloride by a secure intravenous route of administration to avoid local irritation and phlebitis reactions [see Warnings and Precautions (5.8)].
The supplied lyophilized powder must be reconstituted and subsequently diluted prior to intravenous use [see Dosage and Administration (2.5)].
2.6 Compatibility With Intravenous Fluids
The following diluents are physically and chemically compatible with 5 g/L vancomycin hydrochloride:
5% Dextrose Injection, USP
5% Dextrose Injection and 0.9% Sodium Chloride Injection, USP
Lactated Ringer's Injection, USP
Lactated Ringer's and 5% Dextrose Injection, USP
0.9% Sodium Chloride Injection, USP
2.7 Incompatibilities for Intravenous Use
Vancomycin solution has a low pH and may cause chemical or physical instability when it is mixed with other compounds.
Mixtures of solutions of vancomycin and beta-lactam antibacterial drugs have been shown to be physically incompatible. The likelihood of precipitation increases with higher concentrations of vancomycin. It is recommended to adequately flush the intravenous lines between the administration of these antibacterial drugs. It is also recommended to dilute solutions of vancomycin to 5 mg/mL or less.
13.2 Animal Toxicology And/or Pharmacology
In animal studies, hypotension and bradycardia occurred in dogs receiving an intravenous infusion of vancomycin hydrochloride 25 mg/kg, at a concentration of 25 mg/mL and an infusion rate of 13.3 mL/min.
5.9 Development of Drug Resistant Bacteria
Prescribing Vancomycin Hydrochloride for Injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.4 Dosage in Patients With Renal Impairment
Dosage adjustment must be made in patients with renal impairment. The initial dose should be no less than 15 mg/kg, in patients with any degree of renal impairment.
In premature infants and the elderly, greater dosage reductions than expected may be necessary because of decreased renal function. Measure trough vancomycin serum concentrations to guide therapy, especially in seriously ill patients with changing renal function.
For functionally anephric patients, an initial dose of 15 mg/kg of body weight should be given to achieve prompt therapeutic serum concentration. A dose of 1.9 mg/kg/24 hr should be given after the initial dose of 15 mg/kg.
5.6 Hemorrhagic Occlusive Retinal Vasculitis (horv)
Hemorrhagic occlusive retinal vasculitis, including permanent loss of vision, occurred in patients receiving intracameral or intravitreal administration of vancomycin during or after cataract surgery. The safety and efficacy of vancomycin administered by the intracameral or the intravitreal route have not been established by adequate and well-controlled trials. Vancomycin is not indicated for the prophylaxis of endophthalmitis.
5.8 Phlebitis and Other Administration Site Reactions
Inflammation at the site of injection of Vancomycin Hydrochloride for Injection has been reported. Vancomycin Hydrochloride for Injection is irritating to tissue and must be given by a secure intravenous route of administration to reduce the risk of local irritation and phlebitis.
Administration of Vancomycin Hydrochloride for Injection by intramuscular (IM), intraperitoneal, intrathecal (intralumbar or intraventricular), or intravitreal routes has not been approved and is not recommended. The safety and efficacy of vancomycin administered by the intrathecal (intralumbar or intraventricular) route or by the intraperitoneal route have not been established by adequate and well controlled trials.
Pain, tenderness, and necrosis occur with IM injection of Vancomycin Hydrochloride for Injection or with inadvertent extravasation. Thrombophlebitis may occur, the frequency and severity of which can be minimized by administering the drug slowly as a dilute solution (2.5 to 5 g/L) and by rotation of venous access sites.
Intraperitoneal administration during continuous ambulatory peritoneal dialysis (CAPD) can result in chemical peritonitis. Manifestations range from cloudy dialysate alone to a cloudy dialysate accompanied by variable degrees of abdominal pain and fever. This syndrome appears to be resolve after discontinuation of intraperitoneal vancomycin.
2.2 Dosage in Adult Patients With Normal Renal Function
The usual daily intravenous dose is 2 grams divided either as 500 mg every 6 hours or 1 g every 12 hours. Administer each dose over a period of 60 minutes or greater. Other patient factors, such as age or obesity, may call for modification of the usual intravenous daily dose.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Although no long-term studies in animals have been performed to evaluate carcinogenic potential, no mutagenic potential of Vancomycin Hydrochloride for Injection was found in standard laboratory tests. No definitive fertility studies have been performed.
2.5 Preparation of Vancomycin Hydrochloride for Injection for Intravenous Administration and Storage Instructions
Vancomycin Hydrochloride for Injection must be reconstituted and further diluted.
Structured Label Content
Section 42229-5 (42229-5)
Pediatric Patients (Aged 1 month and older)
The usual intravenous dosage of vancomycin is 10 mg/kg per dose given every 6 hours. Each dose should be administered over a period of at least 60 minutes. Close monitoring of serum concentrations of vancomycin may be warranted in these patients.
Section 44425-7 (44425-7)
Storage Conditions
Prior to reconstitution, store at 20° to 25°C (68° to 77°F); excursions permitted to 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Lyophilized.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Discard unused portion.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-147-20
Rx only
Vancomycin Hydrochloride for Injection, USP
750 mg per vial
For Intravenous Use
AFTER RECONSTITUTION MUST BE FURTHER
DILUTED. SEE PACKAGE INSERT
Sterile Powder
Single-Dose Vial
1.6 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
Supportive care is advised, with maintenance of glomerular filtration. Vancomycin is poorly removed by dialysis. Hemofiltration and hemoperfusion with polysulfone resin have been reported to result in increased vancomycin clearance.
For current information on the management of overdosage, contact the National Poison Control Center at 1-800-222-1222 or www.poison.org.
15 References (15 REFERENCES)
- Byrd RA., Gries CL, Buening M.: Developmental Toxicology Studies of Vancomycin Hydrochloride Administered Intravenously to Rats and Rabbits. Fundam Appl Toxicol 1994; 23: 590-597.
1.1 Septicemia
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of septicemia due to:
- Susceptible isolates of methicillin-resistant Staphylococcus aureus (MRSA) and coagulase negative staphylococci.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
11 Description (11 DESCRIPTION)
Vancomycin Hydrochloride for Injection, USP, contains the hydrochloride salt of vancomycin, a tricyclic glycopeptide antibacterial derived from Amycolatopsis orientalis (formerly Nocardia orientalis). The chemical name for vancomycin hydrochloride, USP is (Sa)-(3S,6R,7R,22R,23S,26S,36R,38aR)-44-[[2-O-(3-Amino-2,3,6-trideoxy-3-C-methyl-α-L-lyxo-hexopyranosyl)-β-D-glucopyranosyl]oxy]-3-(carbamoylmethyl)-10,19-dichloro-2,3,4,5,6,7,23,24,25,26,36,37,38,38a-tetradecahydro-7,22,28,30,32-pentahydroxy-6-[(2R)-4-methyl-2-(methylamino)valeramido]-2,5,24,38,39-pentaoxo-22H-8,11:18,21-dietheno-23,36-(iminomethano)-13,16:31,35-dimetheno-1H,16H-[l,6,9]oxadiazacyclohexadecino[4,5-m][10,2,16]-benzoxadiazacyclotetracosine-26-carboxylic acid, monohydrochloride. The molecular formula is C66H75Cl2N9O24 • HCl and the molecular weight is 1,485.71. Vancomycin hydrochloride, USP has the following structural formula:
Vancomycin Hydrochloride for Injection, USP is a sterile off-white to light tan colored lyophilized plug or powder for injection. Vancomycin Hydrochloride for Injection, USP is supplied in single-dose vials, containing 769 mg, 1.28 grams, or 1.54 grams of vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. The lyophilized powder is reconstituted with Sterile Water for Injection, USP which forms a clear, colorless or light to dark tan solution and subsequently diluted prior to intravenous administration [see Dosage and Administration (2.5)].
5.3 Ototoxicity
Ototoxicity has occurred in patients receiving Vancomycin Hydrochloride for Injection. It may be transient or permanent. Ototoxicity manifests as tinnitus, hearing loss, dizziness or vertigo. The risk is higher in older patients, patients who are receiving higher doses, who have an underlying hearing loss, who are receiving concomitant therapy with another ototoxic agent, such as an aminoglycoside or who have underlying renal impairment. Monitor for signs and symptoms of ototoxicity during therapy. Monitor serum vancomycin concentrations and renal function in all patients receiving parenteral vancomycin. Discontinue Vancomycin Hydrochloride for Injection if ototoxicity occurs. Dosage of Vancomycin Hydrochloride for Injection must be adjusted for patients with renal impairment [see Dosage and Administration (2.3)]. Serial tests of auditory function may be helpful in order to minimize the risk of ototoxicity.
5.7 Neutropenia
Reversible neutropenia has been reported in patients receiving Vancomycin Hydrochloride for Injection [see Adverse Reactions (6.1)]. Patients who will undergo prolonged therapy with Vancomycin Hydrochloride for Injection or those who are receiving concomitant drugs which may cause neutropenia should have periodic monitoring of the leukocyte count.
8.4 Pediatric Use
Vancomycin Hydrochloride for Injection is indicated in pediatric patients (neonates and older). In pediatric patients, monitor vancomycin serum concentration and renal function when administering Vancomycin Hydrochloride for Injection [see Dosage and Administration (2.2, 2.3) and Warnings and Precautions (5.2)]. More severe infusion related reactions related to vancomycin administration may occur in pediatric patients. Concomitant administration of vancomycin and intravenous anesthetic agents has been associated with erythema and histamine-like flushing in all patients including pediatric patients [see Warnings and Precautions (5.1)].
8.5 Geriatric Use
Vancomycin Hydrochloride for Injection is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection [see Dosage and Administration (2.2)], and it may be useful to monitor renal function [see Warnings and Precautions (5.2)].
5.2 Nephrotoxicity
Vancomycin Hydrochloride for Injection can result in acute kidney injury (AKI), including acute renal failure, mainly due to interstitial nephritis or less commonly acute tubular necrosis. AKI is manifested by increasing blood urea nitrogen (BUN) and serum creatinine (Cr). The risk of AKI increases with higher vancomycin serum levels, prolonged exposure, concomitant administration of other nephrotoxic drugs, concomitant administration of piperacillin-tazobactam [see Drug Interactions (7.2)], volume depletion, pre-existing renal impairment and in critically ill patients and patients with co-morbid conditions that predispose to renal impairment.
Monitor serum vancomycin concentrations and renal function in all patients receiving Vancomycin Hydrochloride for Injection. More frequent monitoring is recommended in patients with comorbidities that predispose to impairment in renal function or are concomitantly receiving other nephrotoxic drugs, in critically ill patients, in patients with changing renal function, and in patients requiring higher therapeutic vancomycin levels. If acute kidney injury occurs, discontinue Vancomycin Hydrochloride for Injection or reduce the dose.
1.4 Bone Infections
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of bone infections due to:
- Susceptible isolates of MRSA and coagulase negative staphylococci.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
4 Contraindications (4 CONTRAINDICATIONS)
Vancomycin Hydrochloride for Injection is contraindicated in patients with known hypersensitivity to vancomycin.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infusion Reactions [see Warnings and Precautions (5.1)]
- Nephrotoxicity [see Warnings and Precautions (5.2)]
- Ototoxicity [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.5)]
- Hemorrhagic Occlusive Retinal Vasculitis [see Warnings and Precautions (5.6)]
- Neutropenia [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Anesthetic Agents: Concomitant administration of vancomycin and anesthetic agents has been associated with erythema and histamine-like flushing. (2.1, 7.1)
- Piperacillin/Tazobactam: Increased incidence of acute kidney injury in patients receiving concomitant piperacillin/tazobactam and vancomycin as compared to vancomycin alone. Monitor kidney function in patients (7.2)
12.2 Pharmacodynamics
The pharmacodynamics of vancomycin is unknown.
12.3 Pharmacokinetics
In subjects with normal kidney function, multiple intravenous dosing of 1 g of vancomycin (15 mg/kg) infused over 60 minutes produces mean plasma concentrations of approximately 63 mcg/mL immediately after the completion of infusion, mean plasma concentrations of approximately 23 mcg/mL 2 hours after infusion, and mean plasma concentrations of approximately 8 mcg/mL 11 hours after the end of the infusion. Multiple dosing of 500 mg infused over 30 minutes produces mean plasma concentrations of about 49 mcg/mL at the completion of infusion, mean plasma concentrations of about 19 mcg/mL 2 hours after infusion, and mean plasma concentrations of about 10 mcg/mL 6 hours after infusion. The plasma concentrations during multiple dosing are like those after a single dose.
7.1 Anesthetic Agents
5.1 Infusion Reactions
Hypotension, including shock and cardiac arrest, wheezing, dyspnea, urticaria, muscular and chest pain may occur with rapid Vancomycin Hydrochloride for Injection administration. The reactions may be more severe in younger patients, particularly children, and in patients receiving concomitant muscle relaxant anesthetics.
Rapid intravenous administration of Vancomycin Hydrochloride for Injection may also be associated with “vancomycin infusion reaction”, which manifests as pruritus and erythema that involves the face, neck and upper torso.
Infusion-related adverse reactions are related to both the concentration and the rate of administration of vancomycin. Infusion-related adverse reactions may occur, however, at any rate or concentration.
Administer Vancomycin Hydrochloride for Injection in a diluted solution over a period of 60 minutes or greater to reduce the risk of infusion-related adverse reactions. In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used; use of such higher concentrations may increase the risk of infusion-related adverse reactions. Administer prior to intravenous anesthetic agents when feasible. Stop the infusion if a reaction occurs.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Vancomycin Hydrochloride for Injection is a glycopeptide antibacterial indicated in adult and pediatric patients (neonates and older) for the treatment of:
- Septicemia (1.1)
- Infective Endocarditis (1.2)
- Skin and Skin Structure Infections (1.3)
- Bone Infections (1.4)
- Lower Respiratory Tract Infections (1.5)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for Injection and other antibacterial drugs, Vancomycin Hydrochloride for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.6)
12.1 Mechanism of Action
Vancomycin is an antibacterial drug [see Microbiology (12.4)].
1.2 Infective Endocarditis
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of infective endocarditis due to:
- Susceptible isolates of MRSA.
- Viridans group streptococci Streptococcus gallolyticus (previously known as Streptococcus bovis), Enterococcus species and Corynebacterium species. For enterococcal endocarditis, use Vancomycin Hydrochloride for Injection in combination with an aminoglycoside.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of early-onset prosthetic valve endocarditis caused by Staphylococcus epidermidis in combination with rifampin and an aminoglycoside.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infusion Reactions: Hypotension, including shock and cardiac arrest, wheezing, dyspnea, urticaria, muscular, chest pain and “vancomycin infusion reaction” which manifests as pruritus and erythema that involves the face, neck and upper torso may occur with rapid intravenous administration. To reduce the risk of infusion reactions, administer Vancomycin Hydrochloride for Injection in a diluted solution over a period of 60 minutes or greater and also prior to intravenous anesthetic agents. (2.1, 5.1)
- Nephrotoxicity: Systemic vancomycin exposure may result in acute kidney injury (AKI) including acute renal failure, mainly due to interstitial nephritis or less commonly acute tubular necrosis. Monitor serum vancomycin concentrations and renal function. (5.2)
- Ototoxicity: Ototoxicity has occurred in patients receiving vancomycin hydrochloride. Monitor for signs and symptoms of ototoxicity during therapy. Monitor serum vancomycin concentrations and renal function. Assessment of auditory function may be appropriate in some instances. (5.3)
- Severe Dermatologic Reactions: Discontinue Vancomycin Hydrochloride for Injection at the first appearance of skin rashes, mucosal lesions, or blisters (5.4).
- Clostridioides difficile- Associated Diarrhea: Evaluate patients if diarrhea occurs. (5.5)
- Neutropenia: Periodically monitor leukocyte count. (5.7)
- Phlebitis: To reduce the risk of local irritation and phlebitis administer Vancomycin Hydrochloride for Injection by a secure intravenous route of administration. (5.8)
- Development of Drug-Resistant Bacteria: Prescribing Vancomycin Hydrochloride for Injection in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug resistant bacteria. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer Vancomycin Hydrochloride for Injection in a diluted solution over 60 minutes or greater to reduce the risk of infusion reactions.
- See full prescribing information for further important administration and preparation instructions (2.1, 2.5)
- Adult Patients: 2 grams divided either as 0.5 grams (g) every 6 hours or 1 gram every 12 hours (2.2)
- Pediatric Patients (1 month and older): 10 mg/kg per dose given every 6 hours (2.3)
- Neonates: See full prescribing information for recommended doses in neonates (2.3)
- Patients with renal impairment: See full prescribing information for recommended doses in patients with renal impairment (2.4)
7.2 Piperacillin Tazobactam (7.2 Piperacillin-Tazobactam)
Studies have detected an increased incidence of acute kidney injury in patients administered concomitant piperacillin/tazobactam and vancomycin as compared to vancomycin alone. Monitor kidney function in patients receiving concomitant piperacillin/tazobactam and vancomycin. No pharmacokinetic interactions have been noted between piperacillin/tazobactam and vancomycin.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored plug or powder in single-dose vials containing vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base.
5.5 Clostridioides Difficile (5.5 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Vancomycin Hydrochloride for Injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Clinically significant serum concentrations have been reported in some patients being treated for active C. difficile-induced pseudomembranous colitis after multiple oral doses of vancomycin.
Prolonged use of Vancomycin Hydrochloride for Injection may result in the overgrowth of nonsusceptible microorganisms. Careful observation of the patient is essential. If superinfection occurs during therapy, appropriate measures should be taken. In rare instances, there have been reports of pseudomembranous colitis due to C. difficile developing in patients who received intravenous Vancomycin Hydrochloride for Injection.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of vancomycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders: Drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP) [see Warnings and Precautions (5.4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following adverse reactions associated with the use of Vancomycin Hydrochloride for Injection were identified in clinical trials:
Immune system disorders: Hypersensitivity reactions including anaphylaxis and “vancomycin infusion reaction” [see Warnings and Precautions (5.1)]
Skin and subcutaneous tissue disorders: Erythema (especially of the face, neck and upper torso) and pruritus which are manifestations of rashes including exfoliative dermatitis, toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), Linear IgA bullous dermatosis (LABD) [see Warnings and Precautions (5.4)].
Renal and urinary disorders: Acute kidney injury and interstitial nephritis
Ear and Labyrinth Disorders: Tinnitus, hearing loss, vertigo
Blood and Lymphatic System Disorders: Agranulocytosis, neutropenia, pancytopenia, leukopenia, thrombocytopenia, eosinophilia
Gastrointestinal Disorders: Pseudomembranous colitis [see Warnings and Precautions (5.5)]
Cardiac Disorders: Cardiac arrest, chest pain
General Disorders and Administration Site Conditions: General discomfort, fever, chills, phlebitis, injection site irritation, injection site pain and necrosis following intramuscular injection, chemical peritonitis following intraperitoneal administration
(Vancomycin Hydrochloride for Injection is not approved for intramuscular and intraperitoneal administration) [see Warnings and Precautions (5.7)]
Laboratory Abnormalities: Elevated blood urea nitrogen, elevated serum creatinine
Musculoskeletal and connective tissue disorders: Muscle pain
Nervous system disorders: Dizziness
Respiratory, thoracic and mediastinal disorders: Wheezing, dyspnea
Vascular disorders: Hypotension, shock, vasculitis
5.4 Severe Dermatologic Reactions
Severe dermatologic reactions such as toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and linear IgA bullous dermatosis (LABD) have been reported in association with the use of vancomycin. Cutaneous signs or symptoms reported include skin rashes, mucosal lesions, and blisters.
Discontinue Vancomycin Hydrochloride for Injection at the first appearance of signs and symptoms of TEN, SJS, DRESS, AGEP, or LABD.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Vancomycin Hydrochloride for Injection, USP is a sterile lyophilized powder for injection supplied as an off-white to light tan colored powder or plug in single-dose flip top vials that contain vancomycin hydrochloride, USP equivalent to 750 mg, 1.25 grams, or 1.5 grams of vancomycin base. They are available as follows:
| NDC | Vancomycin Hydrochloride for Injection, USP | Package Factor |
| 25021-147-20 | 750 mg per Single-Dose vial (equivalent to 750 mg vancomycin in a 20 mL USP Type-1 clear glass tubular lyo vials with a flip off Pink color button (Matte top) seal.) |
10 vials per carton |
| 25021-148-30 | 1.25 grams per Single-Dose vial (equivalent to 1.25 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Purple color button (Matte top) seal.) |
10 vials per carton |
| 25021-149-30 | 1.5 grams per Single-Dose vial (equivalent to 1.5 grams vancomycin in a 30 mL USP Type-1 clear glass moulded lyo vials with a flip off Light Green color button (Matte top) seal.) |
10 vials per carton |
7.3 Ototoxic And/or Nephrotoxic Drugs (7.3 Ototoxic and/or Nephrotoxic Drugs)
Concurrent and/or sequential systemic or topical use of other potentially, neurotoxic and/or nephrotoxic drugs requires more frequent monitoring of renal function.
1.3 Skin and Skin Structure Infections
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of skin and skin structure infections due to:
- Susceptible isolates of MRSA and coagulase negative staphylococci.
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
1.5 Lower Respiratory Tract Infections
Vancomycin Hydrochloride for Injection is indicated in adults and pediatric patients (neonates and older) for the treatment of lower respiratory tract infections due to:
- Susceptible isolates of MRSA
- Methicillin-susceptible staphylococci in penicillin-allergic patients, or those patients who cannot receive or who have failed to respond to other drugs, including penicillins or cephalosporins.
2.1 Important Administration Instructions
To reduce the risk of infusion related adverse reactions, administer Vancomycin Hydrochloride for Injection in a diluted solution over 60 minutes or greater [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. Vancomycin Hydrochloride for Injection concentrations of no more than 5 mg/mL are recommended in adults [see Dosage and Administration (2.2)]. See also age-specific recommendations [see Dosage and Administration (2.3)]. In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used [see Warnings and Precautions (5.1)].
Administer Vancomycin Hydrochloride for Injection prior to intravenous anesthetic agents to reduce the risk of infusion related adverse reactions [see Warnings and Precautions (5.1)].
Administer Vancomycin Hydrochloride by a secure intravenous route of administration to avoid local irritation and phlebitis reactions [see Warnings and Precautions (5.8)].
The supplied lyophilized powder must be reconstituted and subsequently diluted prior to intravenous use [see Dosage and Administration (2.5)].
2.6 Compatibility With Intravenous Fluids (2.6 Compatibility with Intravenous Fluids)
The following diluents are physically and chemically compatible with 5 g/L vancomycin hydrochloride:
5% Dextrose Injection, USP
5% Dextrose Injection and 0.9% Sodium Chloride Injection, USP
Lactated Ringer's Injection, USP
Lactated Ringer's and 5% Dextrose Injection, USP
0.9% Sodium Chloride Injection, USP
2.7 Incompatibilities for Intravenous Use
Vancomycin solution has a low pH and may cause chemical or physical instability when it is mixed with other compounds.
Mixtures of solutions of vancomycin and beta-lactam antibacterial drugs have been shown to be physically incompatible. The likelihood of precipitation increases with higher concentrations of vancomycin. It is recommended to adequately flush the intravenous lines between the administration of these antibacterial drugs. It is also recommended to dilute solutions of vancomycin to 5 mg/mL or less.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In animal studies, hypotension and bradycardia occurred in dogs receiving an intravenous infusion of vancomycin hydrochloride 25 mg/kg, at a concentration of 25 mg/mL and an infusion rate of 13.3 mL/min.
5.9 Development of Drug Resistant Bacteria (5.9 Development of Drug-Resistant Bacteria)
Prescribing Vancomycin Hydrochloride for Injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.4 Dosage in Patients With Renal Impairment
Dosage adjustment must be made in patients with renal impairment. The initial dose should be no less than 15 mg/kg, in patients with any degree of renal impairment.
In premature infants and the elderly, greater dosage reductions than expected may be necessary because of decreased renal function. Measure trough vancomycin serum concentrations to guide therapy, especially in seriously ill patients with changing renal function.
For functionally anephric patients, an initial dose of 15 mg/kg of body weight should be given to achieve prompt therapeutic serum concentration. A dose of 1.9 mg/kg/24 hr should be given after the initial dose of 15 mg/kg.
5.6 Hemorrhagic Occlusive Retinal Vasculitis (horv) (5.6 Hemorrhagic Occlusive Retinal Vasculitis (HORV))
Hemorrhagic occlusive retinal vasculitis, including permanent loss of vision, occurred in patients receiving intracameral or intravitreal administration of vancomycin during or after cataract surgery. The safety and efficacy of vancomycin administered by the intracameral or the intravitreal route have not been established by adequate and well-controlled trials. Vancomycin is not indicated for the prophylaxis of endophthalmitis.
5.8 Phlebitis and Other Administration Site Reactions
Inflammation at the site of injection of Vancomycin Hydrochloride for Injection has been reported. Vancomycin Hydrochloride for Injection is irritating to tissue and must be given by a secure intravenous route of administration to reduce the risk of local irritation and phlebitis.
Administration of Vancomycin Hydrochloride for Injection by intramuscular (IM), intraperitoneal, intrathecal (intralumbar or intraventricular), or intravitreal routes has not been approved and is not recommended. The safety and efficacy of vancomycin administered by the intrathecal (intralumbar or intraventricular) route or by the intraperitoneal route have not been established by adequate and well controlled trials.
Pain, tenderness, and necrosis occur with IM injection of Vancomycin Hydrochloride for Injection or with inadvertent extravasation. Thrombophlebitis may occur, the frequency and severity of which can be minimized by administering the drug slowly as a dilute solution (2.5 to 5 g/L) and by rotation of venous access sites.
Intraperitoneal administration during continuous ambulatory peritoneal dialysis (CAPD) can result in chemical peritonitis. Manifestations range from cloudy dialysate alone to a cloudy dialysate accompanied by variable degrees of abdominal pain and fever. This syndrome appears to be resolve after discontinuation of intraperitoneal vancomycin.
2.2 Dosage in Adult Patients With Normal Renal Function
The usual daily intravenous dose is 2 grams divided either as 500 mg every 6 hours or 1 g every 12 hours. Administer each dose over a period of 60 minutes or greater. Other patient factors, such as age or obesity, may call for modification of the usual intravenous daily dose.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Although no long-term studies in animals have been performed to evaluate carcinogenic potential, no mutagenic potential of Vancomycin Hydrochloride for Injection was found in standard laboratory tests. No definitive fertility studies have been performed.
2.5 Preparation of Vancomycin Hydrochloride for Injection for Intravenous Administration and Storage Instructions
Vancomycin Hydrochloride for Injection must be reconstituted and further diluted.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:30.427578 · Updated: 2026-03-14T22:27:19.902515