These Highlights Do Not Include All The Information Needed To Use Nelarabine Injection Safely And Effectively. See Full Prescribing Information For Nelarabine Injection.
f60da70f-2f9e-421d-a277-95b1edb57a71
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Severe neurologic adverse reactions have been reported with the use of nelarabine. These adverse reactions have included altered mental states including severe somnolence, central nervous system effects including convulsions, and peripheral neuropathy ranging from numbness and paresthesias to motor weakness and paralysis. There have also been reports of adverse reactions associated with demyelination, and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome [see Warnings and Precautions ( 5.1 )]. Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Monitor frequently for signs and symptoms of neurologic toxicity during treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI Common Toxicity Criteria for Adverse Events (CTCAE) Grade 2 or greater [see Warnings and Precautions ( 5.1 )].
Indications and Usage
Nelarabine is indicated for the treatment of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) in adult and pediatric patients age 1 year and older whose disease has not responded to or has relapsed following treatment with at least two chemotherapy regimens.
Dosage and Administration
Adult Dose : 1,500 mg/m² administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. ( 2.1 ) Pediatric Dose : 650 mg/m² administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days. ( 2.1 ) Discontinue treatment for neurologic reactions greater than or equal to Grade 2. ( 2.2 ) Dosage may be delayed for hematologic reactions. ( 2.2 ) Take measures to prevent hyperuricemia. ( 2.4 )
Warnings and Precautions
Neurologic Adverse Reactions : Severe neurologic reactions have been reported. Monitor for signs and symptoms of neurologic toxicity. ( 5.1 ) Hematologic Reactions : Complete blood counts including platelets should be monitored regularly. ( 5.2 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception; and advise males to use condoms. ( 5.3 , 8.1 , 8.3 ) Effects on Ability to Drive and Use Machines : Somnolence may occur. Advise patients to refrain from these activities until somnolence has resolved. ( 5.6 )
Contraindications
None.
Adverse Reactions
Nervous system adverse reactions of any grade were reported for 223 (76%) adult patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 55 (19%) patients following initiation of nelarabine therapy [see Adverse Reactions ( 6.1 )]. Based on patients with complete data, the median time to onset of first event is 5 days from start of first infusion (range: 1-166), and the median duration is 6 days (range: 1-393 days). Nervous system adverse reactions of any grade were reported for 69 (42%) pediatric patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 25 (15%) patients following initiation of nelarabine therapy [see Adverse Reactions ( 6.1 )]. Based on patients with complete data, the median time to onset of first event is 8 days from start of first infusion (range: 1-269), and the median duration is 2 days (range: 1-82 days). Common signs and symptoms of nelarabine-related neurotoxicity include somnolence, headache, paresthesia and dysesthesia, dizziness, neuropathy (sensory and motor), cerebellar disturbances and tremor. Severe neurologic toxicity can manifest as coma, status epilepticus, craniospinal demyelination, or ascending neuropathy similar in presentation to Guillain-Barré syndrome. Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Patients treated previously or concurrently with intrathecal chemotherapy or previously with craniospinal irradiation may be at increased risk for neurologic adverse events. Monitor patients frequently for signs and symptoms of neurologic toxicity during and for at least 24 hours after completion of treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI CTCAE Grade 2 or greater and provide supportive care [see Dosage and Administration ( 2.2 ), Adverse Reactions ( 6.1 )] .
Drug Interactions
Administration of nelarabine in combination with adenosine deaminase inhibitors, such as pentostatin, is not recommended [see Clinical Pharmacology ( 12.3 )].
Storage and Handling
Nelarabine Injection is a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper and an aluminum seal with a red snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. It is supplied as follows: NDC Nelarabine Injection (5 mg per mL) Package Factor 25021-259-50 250 mg per 50 mL Single-Dose Vial 1 vial per carton 25021-259-51 250 mg per 50 mL Single-Dose Vial 6 vials per carton
How Supplied
Nelarabine Injection is a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper and an aluminum seal with a red snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. It is supplied as follows: NDC Nelarabine Injection (5 mg per mL) Package Factor 25021-259-50 250 mg per 50 mL Single-Dose Vial 1 vial per carton 25021-259-51 250 mg per 50 mL Single-Dose Vial 6 vials per carton
Medication Information
Warnings and Precautions
Neurologic Adverse Reactions : Severe neurologic reactions have been reported. Monitor for signs and symptoms of neurologic toxicity. ( 5.1 ) Hematologic Reactions : Complete blood counts including platelets should be monitored regularly. ( 5.2 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception; and advise males to use condoms. ( 5.3 , 8.1 , 8.3 ) Effects on Ability to Drive and Use Machines : Somnolence may occur. Advise patients to refrain from these activities until somnolence has resolved. ( 5.6 )
Indications and Usage
Nelarabine is indicated for the treatment of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) in adult and pediatric patients age 1 year and older whose disease has not responded to or has relapsed following treatment with at least two chemotherapy regimens.
Dosage and Administration
Adult Dose : 1,500 mg/m² administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. ( 2.1 ) Pediatric Dose : 650 mg/m² administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days. ( 2.1 ) Discontinue treatment for neurologic reactions greater than or equal to Grade 2. ( 2.2 ) Dosage may be delayed for hematologic reactions. ( 2.2 ) Take measures to prevent hyperuricemia. ( 2.4 )
Contraindications
None.
Adverse Reactions
Nervous system adverse reactions of any grade were reported for 223 (76%) adult patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 55 (19%) patients following initiation of nelarabine therapy [see Adverse Reactions ( 6.1 )]. Based on patients with complete data, the median time to onset of first event is 5 days from start of first infusion (range: 1-166), and the median duration is 6 days (range: 1-393 days). Nervous system adverse reactions of any grade were reported for 69 (42%) pediatric patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 25 (15%) patients following initiation of nelarabine therapy [see Adverse Reactions ( 6.1 )]. Based on patients with complete data, the median time to onset of first event is 8 days from start of first infusion (range: 1-269), and the median duration is 2 days (range: 1-82 days). Common signs and symptoms of nelarabine-related neurotoxicity include somnolence, headache, paresthesia and dysesthesia, dizziness, neuropathy (sensory and motor), cerebellar disturbances and tremor. Severe neurologic toxicity can manifest as coma, status epilepticus, craniospinal demyelination, or ascending neuropathy similar in presentation to Guillain-Barré syndrome. Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Patients treated previously or concurrently with intrathecal chemotherapy or previously with craniospinal irradiation may be at increased risk for neurologic adverse events. Monitor patients frequently for signs and symptoms of neurologic toxicity during and for at least 24 hours after completion of treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI CTCAE Grade 2 or greater and provide supportive care [see Dosage and Administration ( 2.2 ), Adverse Reactions ( 6.1 )] .
Drug Interactions
Administration of nelarabine in combination with adenosine deaminase inhibitors, such as pentostatin, is not recommended [see Clinical Pharmacology ( 12.3 )].
Storage and Handling
Nelarabine Injection is a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper and an aluminum seal with a red snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. It is supplied as follows: NDC Nelarabine Injection (5 mg per mL) Package Factor 25021-259-50 250 mg per 50 mL Single-Dose Vial 1 vial per carton 25021-259-51 250 mg per 50 mL Single-Dose Vial 6 vials per carton
How Supplied
Nelarabine Injection is a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper and an aluminum seal with a red snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. It is supplied as follows: NDC Nelarabine Injection (5 mg per mL) Package Factor 25021-259-50 250 mg per 50 mL Single-Dose Vial 1 vial per carton 25021-259-51 250 mg per 50 mL Single-Dose Vial 6 vials per carton
Description
Severe neurologic adverse reactions have been reported with the use of nelarabine. These adverse reactions have included altered mental states including severe somnolence, central nervous system effects including convulsions, and peripheral neuropathy ranging from numbness and paresthesias to motor weakness and paralysis. There have also been reports of adverse reactions associated with demyelination, and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome [see Warnings and Precautions ( 5.1 )]. Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Monitor frequently for signs and symptoms of neurologic toxicity during treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI Common Toxicity Criteria for Adverse Events (CTCAE) Grade 2 or greater [see Warnings and Precautions ( 5.1 )].
Section 42229-5
Adult Dosage: The recommended adult dose of nelarabine is 1,500 mg/m² administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. Administer nelarabine undiluted.
Section 42230-3
|
PATIENT INFORMATION
Nelarabine Injection |
| Read the Patient Information that comes with nelarabine before you or your child starts treatment with nelarabine. Read the information you get each time before each treatment with nelarabine. There may be new information. This information does not take the place of talking with the doctor about your or your child's medical condition or treatment. Talk to your or your child's doctor, if you have any questions. |
|
What is the most important information I should know about nelarabine?
Nelarabine may cause blood related adverse reactions. Your doctor will do blood tests regularly during treatment to monitor blood counts, including platelets. Nelarabine may cause serious nervous system problems including:
|
Call the doctor right away if you or your child has the following symptoms:
|
| These symptoms may not go away even when treatment with nelarabine is stopped. |
|
What is nelarabine?
Nelarabine is an anti-cancer medicine used to treat adults and children who have:
|
|
What should you tell the doctor before you or your child starts nelarabine?
Tell the doctor about all health conditions you or your child have, including if you or your child:
|
| Tell the doctor about all the medicines you or your child take, including prescription and nonprescription medicines, vitamins, and herbal supplements. |
|
How is nelarabine given?
Nelarabine is an intravenous medicine. This means it is given through a tube in your vein. |
What should you or your child avoid during treatment with nelarabine?
|
|
What are the possible side effects of nelarabine?
Nelarabine may cause serious nervous system problems. See “What is the most important information I should know about nelarabine?” Nelarabine may also cause:
|
| Call your doctor right away if you experience unexplained muscle pain, tenderness, or weakness while taking nelarabine. This is because on rare occasions, muscle problems can be serious. These are not all the side effects associated with nelarabine. Ask your doctor or pharmacist for more information. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
General Advice about nelarabine
This leaflet summarizes important information about nelarabine. If you have questions or problems, talk with your or your child's doctor. You can ask your doctor or pharmacist for information about nelarabine that is written for healthcare providers. SAGENT® Mfd. for SAGENT Pharmaceuticals Schaumburg, IL 60195 (USA) Made in India ©2024 Sagent Pharmaceuticals May 2024 |
| SAGENT Pharamceucitals® |
Section 44425-7
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-259-50
Rx only
Nelarabine Injection
250 mg per 50 mL
(5 mg per mL)
For Intravenous Infusion Only
50 mL Single-Dose Vial
Caution: Cytotoxic Agent
10 Overdosage
There is no known antidote for overdoses of nelarabine. It is anticipated that overdosage would result in severe neurotoxicity (possibly including paralysis, coma), myelosuppression, and potentially death. In the event of overdose, supportive care consistent with good clinical practice should be provided.
At a dose of 2,200 mg/m2 given on Days 1, 3, and 5 every 21 days, 2 patients developed a significant Grade 3 ascending sensory neuropathy. MRI evaluations of the 2 patients demonstrated findings consistent with a demyelinating process in the cervical spine.
15 References
- “OSHA Hazardous Drugs.” OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
Nelarabine is a prodrug of the cytotoxic deoxyguanosine analogue, 9-β-D-arabinofuranosylguanine (ara-G).
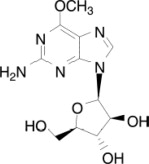
The chemical name for nelarabine is 2-amino-9-β-D-arabinofuranosyl-6-methoxy-9H-purine. It has the molecular formula C11H15N5O5 and a molecular weight of 297.27. Nelarabine has the following structural formula:
Nelarabine is slightly soluble to soluble in water and melts with decomposition between 209°C and 217°C.
Nelarabine injection is supplied as a clear, colorless, sterile solution in glass single-dose vials. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. Nelarabine is intended for intravenous infusion.
Hydrochloric acid and sodium hydroxide may have been used to adjust the pH. The solution pH ranges from 5.0 to 7.0.
5.5 Vaccinations
Avoid the administration of live vaccines to immunocompromised patients.
8.4 Pediatric Use
The safety and effectiveness of nelarabine for relapsed or refractory T-ALL and T-LBL has been established in pediatric patients age 1 year and older. The effectiveness of nelarabine in pediatric patients is supported by one single-arm clinical trial, and safety has been assessed in 165 pediatric patients age 1 year and older across multiple Phase I and Phase II trials. The trial establishing efficacy included 84 patients age 21 years and younger, who had relapsed or refractory T-ALL or T-LBL. The most frequent adverse reactions of any grade occurring on treatment in this study were hematologic laboratory abnormalities. Hematologic toxicity observed in the pediatric population was higher than that seen in the adult population [see Dosage and Administration (2.1), Adverse Reactions (6.1), Clinical Studies (14.2)].
Nervous system adverse reactions have been reported for 42% of pediatric patients across the Phase I and Phase II trials. The incidence of nervous system adverse reactions was less in the pediatric population than that seen in adult patients with relapsed/refractory T-ALL/T-LBL [see Adverse Reactions (6.1)].
In a phase III study of nelarabine in combination with multi-agent chemotherapy as first-line therapy, there were 411 patients with T-ALL or T-LBL treated with nelarabine. The safety profile in the 357 patients age 1 to 16 years was consistent with that seen in older patients in the study [see Adverse Reactions (6.1)].
Due to lack of long-term follow up data, a determination of the impact of nelarabine on the growth and pubertal development of pediatric patients cannot be made.
8.5 Geriatric Use
Clinical studies of nelarabine did not include sufficient numbers of patients age 65 and over to determine whether they respond differently from younger patients. In an exploratory analysis, increasing age, especially age 65 years and older, appeared to be associated with increased rates of neurologic adverse reactions. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection [see Use in Specific Populations (8.6)].
4 Contraindications
None.
6 Adverse Reactions
The following clinically-significant adverse reactions are discussed in greater detail in other sections of the label:
- Neurologic [see Boxed Warning, Warnings and Precautions (5.1)]
- Hematologic [see Warnings and Precautions (5.2)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.4)]
- Effects on Ability to Drive and Use Machines [see Warnings and Precautions (5.6)]
7 Drug Interactions
Administration of nelarabine in combination with adenosine deaminase inhibitors, such as pentostatin, is not recommended [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Ara-G clearance decreased as renal function decreased [see Clinical Pharmacology (12.3)]. Because the risk of adverse reactions to this drug may be greater in patients with moderate (CLCr 30 to 50 mL/min) or severe (CLCr less than 30 mL/min) renal impairment, these patients should be closely monitored for toxicities when treated with nelarabine [see Dosage and Administration (2.3)].
2.1 Recommended Dosage
This product is for intravenous use only.
8.7 Hepatic Impairment
The influence of hepatic impairment on the pharmacokinetics of nelarabine has not been evaluated. Because the risk of adverse reactions to this drug may be greater in patients with severe hepatic impairment (total bilirubin greater than 3 times upper limit of normal), these patients should be closely monitored for toxicities when treated with nelarabine.
1 Indications and Usage
Nelarabine is indicated for the treatment of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) in adult and pediatric patients age 1 year and older whose disease has not responded to or has relapsed following treatment with at least two chemotherapy regimens.
2.2 Dosage Modification
Discontinue nelarabine if the patient develops a neurologic adverse reaction of NCI CTCAE Grade 2 or greater. Dosage may be delayed for other toxicity, including hematologic toxicity [see Boxed Warning, Warnings and Precautions (5.1, 5.2)].
12.1 Mechanism of Action
Nelarabine is a prodrug of the deoxyguanosine analogue 9-β-D-arabinofuranosylguanine (ara-G), a nucleoside metabolic inhibitor. Nelarabine is demethylated by adenosine deaminase (ADA) to ara-G, mono-phosphorylated by deoxyguanosine kinase and deoxycytidine kinase, and subsequently converted to the active 5'-triphosphate, ara-GTP. Accumulation of ara-GTP in leukemic blasts allows for incorporation into deoxyribonucleic acid (DNA), leading to inhibition of DNA synthesis and cell death. Other mechanisms may contribute to the cytotoxic and systemic toxicity of nelarabine.
5.4 Tumor Lysis Syndrome
Patients receiving nelarabine should receive intravenous hydration according to standard medical practice for the management of hyperuricemia in patients at risk for tumor lysis syndrome. Consideration should be given to the use of allopurinol in patients at risk of hyperuricemia [see Dosage and Administration (2.4)].
5.3 Embryo Fetal Toxicity
Based on its mechanism of action and findings in animal studies, nelarabine can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. In animal reproduction studies, intravenous administration of nelarabine to pregnant rabbits during the period of organogenesis resulted in teratogenicity at maternal doses below the recommended human adult dose of 1,500 mg/m2/day (see Data).
Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with nelarabine. Advise males with female partners of reproductive potential to use condoms during treatment with nelarabine and for 3 months after the last dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
5 Warnings and Precautions
- Neurologic Adverse Reactions: Severe neurologic reactions have been reported. Monitor for signs and symptoms of neurologic toxicity. (5.1)
- Hematologic Reactions: Complete blood counts including platelets should be monitored regularly. (5.2)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception; and advise males to use condoms. (5.3, 8.1, 8.3)
- Effects on Ability to Drive and Use Machines: Somnolence may occur. Advise patients to refrain from these activities until somnolence has resolved. (5.6)
2 Dosage and Administration
- Adult Dose: 1,500 mg/m² administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. (2.1)
- Pediatric Dose: 650 mg/m² administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days. (2.1)
- Discontinue treatment for neurologic reactions greater than or equal to Grade 2. (2.2)
- Dosage may be delayed for hematologic reactions. (2.2)
- Take measures to prevent hyperuricemia. (2.4)
3 Dosage Forms and Strengths
Injection: 250 mg per 50 mL (5 mg per mL) single-dose vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of nelarabine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and Infestations: Fatal opportunistic infections.
Metabolism and Nutrition Disorders: Tumor lysis syndrome.
Nervous System Disorders: Demyelination and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome.
Musculoskeletal and Connective Disorders: Rhabdomyolysis, blood creatine phosphokinase increased.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.4 Prevention of Hyperuricemia
Take precautions against hyperuricemia (e.g., hydration, urine alkalinization, and prophylaxis with allopurinol) [see Warnings and Precautions (5.4)].
5.1 Neurologic Adverse Reactions
Nervous system adverse reactions of any grade were reported for 223 (76%) adult patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 55 (19%) patients following initiation of nelarabine therapy [see Adverse Reactions (6.1)]. Based on patients with complete data, the median time to onset of first event is 5 days from start of first infusion (range: 1-166), and the median duration is 6 days (range: 1-393 days).
Nervous system adverse reactions of any grade were reported for 69 (42%) pediatric patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 25 (15%) patients following initiation of nelarabine therapy [see Adverse Reactions (6.1)]. Based on patients with complete data, the median time to onset of first event is 8 days from start of first infusion (range: 1-269), and the median duration is 2 days (range: 1-82 days).
Common signs and symptoms of nelarabine-related neurotoxicity include somnolence, headache, paresthesia and dysesthesia, dizziness, neuropathy (sensory and motor), cerebellar disturbances and tremor. Severe neurologic toxicity can manifest as coma, status epilepticus, craniospinal demyelination, or ascending neuropathy similar in presentation to Guillain-Barré syndrome.
Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Patients treated previously or concurrently with intrathecal chemotherapy or previously with craniospinal irradiation may be at increased risk for neurologic adverse events.
Monitor patients frequently for signs and symptoms of neurologic toxicity during and for at least 24 hours after completion of treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI CTCAE Grade 2 or greater and provide supportive care [see Dosage and Administration (2.2), Adverse Reactions (6.1)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.3 Dosage in Special Populations
Nelarabine has not been studied in patients with renal or hepatic dysfunction [see Use in Specific Populations (8.6, 8.7)]. No dose adjustment is recommended for patients with a creatinine clearance (CLCr) greater than or equal to 50 mL/min [see Clinical Pharmacology (12.3)]. There are insufficient data to support a dose recommendation for patients with a CLCr less than 50 mL/min.
5.2 Hematologic Adverse Reactions
16 How Supplied/storage and Handling
Nelarabine Injection is a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper and an aluminum seal with a red snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. It is supplied as follows:
| NDC | Nelarabine Injection (5 mg per mL) | Package Factor |
| 25021-259-50 | 250 mg per 50 mL Single-Dose Vial | 1 vial per carton |
| 25021-259-51 | 250 mg per 50 mL Single-Dose Vial | 6 vials per carton |
Warning: Neurologic Adverse Reactions
Severe neurologic adverse reactions have been reported with the use of nelarabine. These adverse reactions have included altered mental states including severe somnolence, central nervous system effects including convulsions, and peripheral neuropathy ranging from numbness and paresthesias to motor weakness and paralysis. There have also been reports of adverse reactions associated with demyelination, and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome [see Warnings and Precautions (5.1)].
Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Monitor frequently for signs and symptoms of neurologic toxicity during treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI Common Toxicity Criteria for Adverse Events (CTCAE) Grade 2 or greater [see Warnings and Precautions (5.1)].
5.6 Effects On Ability to Drive and Use Machines
Patients treated with nelarabine may experience somnolence during and for several days after treatment [see Adverse Reactions (6.1)]. Advise patients to refrain from driving or engaging in hazardous occupations or activities until somnolence has resolved.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity testing of nelarabine has not been done. However, nelarabine was mutagenic when tested in vitro in L5178Y/TK mouse lymphoma cells with and without metabolic activation. No studies have been conducted in animals to assess genotoxic potential or effects on fertility. The effect on human fertility is unknown.
14.1 Adult Clinical Trial in Relapsed Or Refractory T All and T Lbl
The safety and efficacy of nelarabine in adult patients were studied in a clinical trial which included 39 treated patients, 28 who had T-ALL or T-LBL that had relapsed following or was refractory to at least two prior induction regimens. A 1,500-mg/m2 dose of nelarabine was administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. Patients who experienced signs or symptoms of Grade 2 or greater neurologic toxicity on therapy were to be discontinued from further therapy with nelarabine. Seventeen patients had a diagnosis of T-ALL and 11 had a diagnosis of T-LBL. For patients with ≥ 2 prior inductions, the age range was 16 to 65 years (mean: 34 years) and most patients were male (82%) and Caucasian (61%). Patients with central nervous system (CNS) disease were not eligible.
Complete response (CR) in this trial was defined as bone marrow blast counts ≤ 5%, no other evidence of disease, and full recovery of peripheral blood counts. Complete response without complete hematologic recovery (CR*) was also assessed. The results of the trial for patients who had received ≥ 2 prior inductions are shown in Table 5.
|
Abbreviations: CR, complete response; CI, confidence interval; CR*, complete response without hematologic recovery. |
|
|
a Does not include 1 patient who was transplanted (duration of response was 156+ weeks). |
|
| N = 28 | |
| CR plus CR* % (n) [95% CI] | 21% (6) [8%, 41%] |
| CR % (n) [95% CI] | 18% (5) [6%, 37%] |
| CR* % (n) [95% CI] | 4% (1) [0%, 18%] |
| Duration of CR plus CR* (range in weeks)a | 4 to 195+ |
| Median overall survival (weeks) [95% CI] | 20.6 weeks [10.4, 36.4] |
The mean number of days on therapy was 56 days (range of 10 to 136 days). Time to CR plus CR* ranged from 2.9 to 11.7 weeks.
14.2 Pediatric Clinical Trial in Relapsed Or Refractory T All and T Lbl
The safety and efficacy of nelarabine in pediatric patients were studied in a clinical trial which included patients age 21 years and younger, who had relapsed or refractory T-ALL or T-LBL. Eighty-four (84) patients, 39 of whom had received two or more prior induction regimens, were treated with 650 mg/m2/day of nelarabine administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days (see Table 6). Patients who experienced signs or symptoms of Grade 2 or greater neurologic toxicity on therapy were to be discontinued from further therapy with nelarabine.
|
Abbreviations: T-ALL, T-cell acute lymphoblastic leukemia; T-LBL, T-cell lymphoblastic lymphoma. |
|
| Patient Population | N |
| Patients treated at 650 mg/m2/day x 5 days, every 21 days. | 84 |
| Patients with T-ALL or T-LBL with two or more prior induction treated at 650 mg/m2/day x 5 days, every 21 days. | 39 |
| Patients with T-ALL or T-LBL with one prior induction treated at 650 mg/m2/day x 5 days, every 21 days. | 31 |
The 84 patients ranged in age from 2.5 to 21.7 years (overall mean: 11.9 years), 52% were 3 to 12 years of age and most were male (74%) and Caucasian (62%). The majority (77%) of patients had a diagnosis of T-ALL.
Complete response (CR) in this trial was defined as bone marrow blast counts ≤ 5%, no other evidence of disease, and full recovery of peripheral blood counts. Complete response without full hematologic recovery (CR*) was also assessed as a meaningful outcome in this trial. Duration of response is reported from date of response to date of relapse, and may include subsequent stem cell transplant. Efficacy results are presented in Table 7.
|
Abbreviations: CR, complete response; CI, confidence interval; CR*, complete response without hematologic recovery. |
|
|
a Does not include 5 patients who were transplanted or had subsequent systemic chemotherapy (duration of response in these 5 patients was 4.7 to 42.1 weeks). |
|
| N = 39 | |
| CR plus CR* % (n) [95% CI] | 23% (9) [11%, 39%] |
| CR % (n) [95% CI] | 13% (5) [4%, 27%] |
| CR* % (n) [95% CI] | 10% (4) [3%, 24%] |
| Duration of CR plus CR* (range in weeks)a | 3.3 to 9.3 |
| Median overall survival (weeks) [95% CI] | 13.1 [8.7, 17.4] |
The mean number of days on therapy was 46 days (range: 7 to 129 days). Median time to CR plus CR* was 3.4 weeks (95% CI: 3.0, 3.7).
Structured Label Content
Section 42229-5 (42229-5)
Adult Dosage: The recommended adult dose of nelarabine is 1,500 mg/m² administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. Administer nelarabine undiluted.
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
Nelarabine Injection |
| Read the Patient Information that comes with nelarabine before you or your child starts treatment with nelarabine. Read the information you get each time before each treatment with nelarabine. There may be new information. This information does not take the place of talking with the doctor about your or your child's medical condition or treatment. Talk to your or your child's doctor, if you have any questions. |
|
What is the most important information I should know about nelarabine?
Nelarabine may cause blood related adverse reactions. Your doctor will do blood tests regularly during treatment to monitor blood counts, including platelets. Nelarabine may cause serious nervous system problems including:
|
Call the doctor right away if you or your child has the following symptoms:
|
| These symptoms may not go away even when treatment with nelarabine is stopped. |
|
What is nelarabine?
Nelarabine is an anti-cancer medicine used to treat adults and children who have:
|
|
What should you tell the doctor before you or your child starts nelarabine?
Tell the doctor about all health conditions you or your child have, including if you or your child:
|
| Tell the doctor about all the medicines you or your child take, including prescription and nonprescription medicines, vitamins, and herbal supplements. |
|
How is nelarabine given?
Nelarabine is an intravenous medicine. This means it is given through a tube in your vein. |
What should you or your child avoid during treatment with nelarabine?
|
|
What are the possible side effects of nelarabine?
Nelarabine may cause serious nervous system problems. See “What is the most important information I should know about nelarabine?” Nelarabine may also cause:
|
| Call your doctor right away if you experience unexplained muscle pain, tenderness, or weakness while taking nelarabine. This is because on rare occasions, muscle problems can be serious. These are not all the side effects associated with nelarabine. Ask your doctor or pharmacist for more information. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
General Advice about nelarabine
This leaflet summarizes important information about nelarabine. If you have questions or problems, talk with your or your child's doctor. You can ask your doctor or pharmacist for information about nelarabine that is written for healthcare providers. SAGENT® Mfd. for SAGENT Pharmaceuticals Schaumburg, IL 60195 (USA) Made in India ©2024 Sagent Pharmaceuticals May 2024 |
| SAGENT Pharamceucitals® |
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-259-50
Rx only
Nelarabine Injection
250 mg per 50 mL
(5 mg per mL)
For Intravenous Infusion Only
50 mL Single-Dose Vial
Caution: Cytotoxic Agent
10 Overdosage (10 OVERDOSAGE)
There is no known antidote for overdoses of nelarabine. It is anticipated that overdosage would result in severe neurotoxicity (possibly including paralysis, coma), myelosuppression, and potentially death. In the event of overdose, supportive care consistent with good clinical practice should be provided.
At a dose of 2,200 mg/m2 given on Days 1, 3, and 5 every 21 days, 2 patients developed a significant Grade 3 ascending sensory neuropathy. MRI evaluations of the 2 patients demonstrated findings consistent with a demyelinating process in the cervical spine.
15 References (15 REFERENCES)
- “OSHA Hazardous Drugs.” OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Nelarabine is a prodrug of the cytotoxic deoxyguanosine analogue, 9-β-D-arabinofuranosylguanine (ara-G).
The chemical name for nelarabine is 2-amino-9-β-D-arabinofuranosyl-6-methoxy-9H-purine. It has the molecular formula C11H15N5O5 and a molecular weight of 297.27. Nelarabine has the following structural formula:
Nelarabine is slightly soluble to soluble in water and melts with decomposition between 209°C and 217°C.
Nelarabine injection is supplied as a clear, colorless, sterile solution in glass single-dose vials. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. Nelarabine is intended for intravenous infusion.
Hydrochloric acid and sodium hydroxide may have been used to adjust the pH. The solution pH ranges from 5.0 to 7.0.
5.5 Vaccinations
Avoid the administration of live vaccines to immunocompromised patients.
8.4 Pediatric Use
The safety and effectiveness of nelarabine for relapsed or refractory T-ALL and T-LBL has been established in pediatric patients age 1 year and older. The effectiveness of nelarabine in pediatric patients is supported by one single-arm clinical trial, and safety has been assessed in 165 pediatric patients age 1 year and older across multiple Phase I and Phase II trials. The trial establishing efficacy included 84 patients age 21 years and younger, who had relapsed or refractory T-ALL or T-LBL. The most frequent adverse reactions of any grade occurring on treatment in this study were hematologic laboratory abnormalities. Hematologic toxicity observed in the pediatric population was higher than that seen in the adult population [see Dosage and Administration (2.1), Adverse Reactions (6.1), Clinical Studies (14.2)].
Nervous system adverse reactions have been reported for 42% of pediatric patients across the Phase I and Phase II trials. The incidence of nervous system adverse reactions was less in the pediatric population than that seen in adult patients with relapsed/refractory T-ALL/T-LBL [see Adverse Reactions (6.1)].
In a phase III study of nelarabine in combination with multi-agent chemotherapy as first-line therapy, there were 411 patients with T-ALL or T-LBL treated with nelarabine. The safety profile in the 357 patients age 1 to 16 years was consistent with that seen in older patients in the study [see Adverse Reactions (6.1)].
Due to lack of long-term follow up data, a determination of the impact of nelarabine on the growth and pubertal development of pediatric patients cannot be made.
8.5 Geriatric Use
Clinical studies of nelarabine did not include sufficient numbers of patients age 65 and over to determine whether they respond differently from younger patients. In an exploratory analysis, increasing age, especially age 65 years and older, appeared to be associated with increased rates of neurologic adverse reactions. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection [see Use in Specific Populations (8.6)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically-significant adverse reactions are discussed in greater detail in other sections of the label:
- Neurologic [see Boxed Warning, Warnings and Precautions (5.1)]
- Hematologic [see Warnings and Precautions (5.2)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.4)]
- Effects on Ability to Drive and Use Machines [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Administration of nelarabine in combination with adenosine deaminase inhibitors, such as pentostatin, is not recommended [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Ara-G clearance decreased as renal function decreased [see Clinical Pharmacology (12.3)]. Because the risk of adverse reactions to this drug may be greater in patients with moderate (CLCr 30 to 50 mL/min) or severe (CLCr less than 30 mL/min) renal impairment, these patients should be closely monitored for toxicities when treated with nelarabine [see Dosage and Administration (2.3)].
2.1 Recommended Dosage
This product is for intravenous use only.
8.7 Hepatic Impairment
The influence of hepatic impairment on the pharmacokinetics of nelarabine has not been evaluated. Because the risk of adverse reactions to this drug may be greater in patients with severe hepatic impairment (total bilirubin greater than 3 times upper limit of normal), these patients should be closely monitored for toxicities when treated with nelarabine.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Nelarabine is indicated for the treatment of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) in adult and pediatric patients age 1 year and older whose disease has not responded to or has relapsed following treatment with at least two chemotherapy regimens.
2.2 Dosage Modification
Discontinue nelarabine if the patient develops a neurologic adverse reaction of NCI CTCAE Grade 2 or greater. Dosage may be delayed for other toxicity, including hematologic toxicity [see Boxed Warning, Warnings and Precautions (5.1, 5.2)].
12.1 Mechanism of Action
Nelarabine is a prodrug of the deoxyguanosine analogue 9-β-D-arabinofuranosylguanine (ara-G), a nucleoside metabolic inhibitor. Nelarabine is demethylated by adenosine deaminase (ADA) to ara-G, mono-phosphorylated by deoxyguanosine kinase and deoxycytidine kinase, and subsequently converted to the active 5'-triphosphate, ara-GTP. Accumulation of ara-GTP in leukemic blasts allows for incorporation into deoxyribonucleic acid (DNA), leading to inhibition of DNA synthesis and cell death. Other mechanisms may contribute to the cytotoxic and systemic toxicity of nelarabine.
5.4 Tumor Lysis Syndrome
Patients receiving nelarabine should receive intravenous hydration according to standard medical practice for the management of hyperuricemia in patients at risk for tumor lysis syndrome. Consideration should be given to the use of allopurinol in patients at risk of hyperuricemia [see Dosage and Administration (2.4)].
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
Based on its mechanism of action and findings in animal studies, nelarabine can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)]. In animal reproduction studies, intravenous administration of nelarabine to pregnant rabbits during the period of organogenesis resulted in teratogenicity at maternal doses below the recommended human adult dose of 1,500 mg/m2/day (see Data).
Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with nelarabine. Advise males with female partners of reproductive potential to use condoms during treatment with nelarabine and for 3 months after the last dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Neurologic Adverse Reactions: Severe neurologic reactions have been reported. Monitor for signs and symptoms of neurologic toxicity. (5.1)
- Hematologic Reactions: Complete blood counts including platelets should be monitored regularly. (5.2)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception; and advise males to use condoms. (5.3, 8.1, 8.3)
- Effects on Ability to Drive and Use Machines: Somnolence may occur. Advise patients to refrain from these activities until somnolence has resolved. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Adult Dose: 1,500 mg/m² administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. (2.1)
- Pediatric Dose: 650 mg/m² administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days. (2.1)
- Discontinue treatment for neurologic reactions greater than or equal to Grade 2. (2.2)
- Dosage may be delayed for hematologic reactions. (2.2)
- Take measures to prevent hyperuricemia. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 250 mg per 50 mL (5 mg per mL) single-dose vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of nelarabine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and Infestations: Fatal opportunistic infections.
Metabolism and Nutrition Disorders: Tumor lysis syndrome.
Nervous System Disorders: Demyelination and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome.
Musculoskeletal and Connective Disorders: Rhabdomyolysis, blood creatine phosphokinase increased.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.4 Prevention of Hyperuricemia
Take precautions against hyperuricemia (e.g., hydration, urine alkalinization, and prophylaxis with allopurinol) [see Warnings and Precautions (5.4)].
5.1 Neurologic Adverse Reactions
Nervous system adverse reactions of any grade were reported for 223 (76%) adult patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 55 (19%) patients following initiation of nelarabine therapy [see Adverse Reactions (6.1)]. Based on patients with complete data, the median time to onset of first event is 5 days from start of first infusion (range: 1-166), and the median duration is 6 days (range: 1-393 days).
Nervous system adverse reactions of any grade were reported for 69 (42%) pediatric patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 25 (15%) patients following initiation of nelarabine therapy [see Adverse Reactions (6.1)]. Based on patients with complete data, the median time to onset of first event is 8 days from start of first infusion (range: 1-269), and the median duration is 2 days (range: 1-82 days).
Common signs and symptoms of nelarabine-related neurotoxicity include somnolence, headache, paresthesia and dysesthesia, dizziness, neuropathy (sensory and motor), cerebellar disturbances and tremor. Severe neurologic toxicity can manifest as coma, status epilepticus, craniospinal demyelination, or ascending neuropathy similar in presentation to Guillain-Barré syndrome.
Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Patients treated previously or concurrently with intrathecal chemotherapy or previously with craniospinal irradiation may be at increased risk for neurologic adverse events.
Monitor patients frequently for signs and symptoms of neurologic toxicity during and for at least 24 hours after completion of treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI CTCAE Grade 2 or greater and provide supportive care [see Dosage and Administration (2.2), Adverse Reactions (6.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.3 Dosage in Special Populations
Nelarabine has not been studied in patients with renal or hepatic dysfunction [see Use in Specific Populations (8.6, 8.7)]. No dose adjustment is recommended for patients with a creatinine clearance (CLCr) greater than or equal to 50 mL/min [see Clinical Pharmacology (12.3)]. There are insufficient data to support a dose recommendation for patients with a CLCr less than 50 mL/min.
5.2 Hematologic Adverse Reactions
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Nelarabine Injection is a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper and an aluminum seal with a red snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. It is supplied as follows:
| NDC | Nelarabine Injection (5 mg per mL) | Package Factor |
| 25021-259-50 | 250 mg per 50 mL Single-Dose Vial | 1 vial per carton |
| 25021-259-51 | 250 mg per 50 mL Single-Dose Vial | 6 vials per carton |
Warning: Neurologic Adverse Reactions (WARNING: NEUROLOGIC ADVERSE REACTIONS)
Severe neurologic adverse reactions have been reported with the use of nelarabine. These adverse reactions have included altered mental states including severe somnolence, central nervous system effects including convulsions, and peripheral neuropathy ranging from numbness and paresthesias to motor weakness and paralysis. There have also been reports of adverse reactions associated with demyelination, and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome [see Warnings and Precautions (5.1)].
Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine. Monitor frequently for signs and symptoms of neurologic toxicity during treatment with nelarabine. Discontinue nelarabine for neurologic adverse reactions of NCI Common Toxicity Criteria for Adverse Events (CTCAE) Grade 2 or greater [see Warnings and Precautions (5.1)].
5.6 Effects On Ability to Drive and Use Machines (5.6 Effects on Ability to Drive and Use Machines)
Patients treated with nelarabine may experience somnolence during and for several days after treatment [see Adverse Reactions (6.1)]. Advise patients to refrain from driving or engaging in hazardous occupations or activities until somnolence has resolved.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity testing of nelarabine has not been done. However, nelarabine was mutagenic when tested in vitro in L5178Y/TK mouse lymphoma cells with and without metabolic activation. No studies have been conducted in animals to assess genotoxic potential or effects on fertility. The effect on human fertility is unknown.
14.1 Adult Clinical Trial in Relapsed Or Refractory T All and T Lbl (14.1 Adult Clinical Trial in Relapsed or Refractory T-ALL and T-LBL)
The safety and efficacy of nelarabine in adult patients were studied in a clinical trial which included 39 treated patients, 28 who had T-ALL or T-LBL that had relapsed following or was refractory to at least two prior induction regimens. A 1,500-mg/m2 dose of nelarabine was administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. Patients who experienced signs or symptoms of Grade 2 or greater neurologic toxicity on therapy were to be discontinued from further therapy with nelarabine. Seventeen patients had a diagnosis of T-ALL and 11 had a diagnosis of T-LBL. For patients with ≥ 2 prior inductions, the age range was 16 to 65 years (mean: 34 years) and most patients were male (82%) and Caucasian (61%). Patients with central nervous system (CNS) disease were not eligible.
Complete response (CR) in this trial was defined as bone marrow blast counts ≤ 5%, no other evidence of disease, and full recovery of peripheral blood counts. Complete response without complete hematologic recovery (CR*) was also assessed. The results of the trial for patients who had received ≥ 2 prior inductions are shown in Table 5.
|
Abbreviations: CR, complete response; CI, confidence interval; CR*, complete response without hematologic recovery. |
|
|
a Does not include 1 patient who was transplanted (duration of response was 156+ weeks). |
|
| N = 28 | |
| CR plus CR* % (n) [95% CI] | 21% (6) [8%, 41%] |
| CR % (n) [95% CI] | 18% (5) [6%, 37%] |
| CR* % (n) [95% CI] | 4% (1) [0%, 18%] |
| Duration of CR plus CR* (range in weeks)a | 4 to 195+ |
| Median overall survival (weeks) [95% CI] | 20.6 weeks [10.4, 36.4] |
The mean number of days on therapy was 56 days (range of 10 to 136 days). Time to CR plus CR* ranged from 2.9 to 11.7 weeks.
14.2 Pediatric Clinical Trial in Relapsed Or Refractory T All and T Lbl (14.2 Pediatric Clinical Trial in Relapsed or Refractory T-ALL and T-LBL)
The safety and efficacy of nelarabine in pediatric patients were studied in a clinical trial which included patients age 21 years and younger, who had relapsed or refractory T-ALL or T-LBL. Eighty-four (84) patients, 39 of whom had received two or more prior induction regimens, were treated with 650 mg/m2/day of nelarabine administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days (see Table 6). Patients who experienced signs or symptoms of Grade 2 or greater neurologic toxicity on therapy were to be discontinued from further therapy with nelarabine.
|
Abbreviations: T-ALL, T-cell acute lymphoblastic leukemia; T-LBL, T-cell lymphoblastic lymphoma. |
|
| Patient Population | N |
| Patients treated at 650 mg/m2/day x 5 days, every 21 days. | 84 |
| Patients with T-ALL or T-LBL with two or more prior induction treated at 650 mg/m2/day x 5 days, every 21 days. | 39 |
| Patients with T-ALL or T-LBL with one prior induction treated at 650 mg/m2/day x 5 days, every 21 days. | 31 |
The 84 patients ranged in age from 2.5 to 21.7 years (overall mean: 11.9 years), 52% were 3 to 12 years of age and most were male (74%) and Caucasian (62%). The majority (77%) of patients had a diagnosis of T-ALL.
Complete response (CR) in this trial was defined as bone marrow blast counts ≤ 5%, no other evidence of disease, and full recovery of peripheral blood counts. Complete response without full hematologic recovery (CR*) was also assessed as a meaningful outcome in this trial. Duration of response is reported from date of response to date of relapse, and may include subsequent stem cell transplant. Efficacy results are presented in Table 7.
|
Abbreviations: CR, complete response; CI, confidence interval; CR*, complete response without hematologic recovery. |
|
|
a Does not include 5 patients who were transplanted or had subsequent systemic chemotherapy (duration of response in these 5 patients was 4.7 to 42.1 weeks). |
|
| N = 39 | |
| CR plus CR* % (n) [95% CI] | 23% (9) [11%, 39%] |
| CR % (n) [95% CI] | 13% (5) [4%, 27%] |
| CR* % (n) [95% CI] | 10% (4) [3%, 24%] |
| Duration of CR plus CR* (range in weeks)a | 3.3 to 9.3 |
| Median overall survival (weeks) [95% CI] | 13.1 [8.7, 17.4] |
The mean number of days on therapy was 46 days (range: 7 to 129 days). Median time to CR plus CR* was 3.4 weeks (95% CI: 3.0, 3.7).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:13.250693 · Updated: 2026-03-14T22:08:06.283890