Zensa - 5% Lidocaine Cream
f56146b6-3c11-4608-a6ab-6142f27e9ac3
34390-5
HUMAN OTC DRUG LABEL
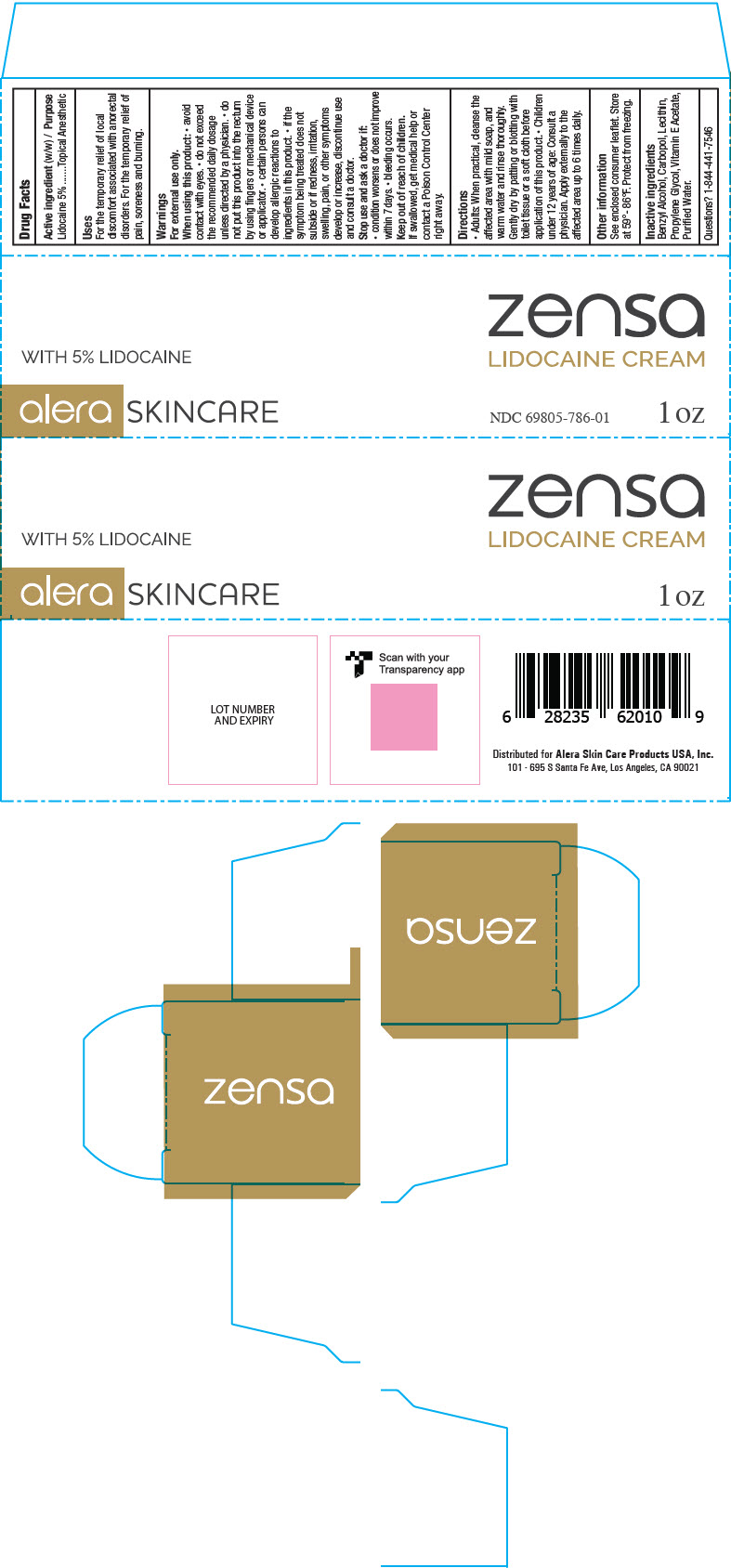
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 5%
Purpose
Topical Anaesthetic
Medication Information
Purpose
Topical Anaesthetic
Description
Lidocaine 5%
Uses
- For the temporary relief of local discomfort associated with anorectal disorders
- For the temporary relief of pain, soreness, and burning
Warnings
For external use only.
Directions
- Adults: when practical, cleanse the affected area with mild soap, and warm water and rinse thoroughly. Gently dry by patting or blottig with toilet tissue or a soft cloth before application of this product. Children under 12 years of age: consult a physician.
- Apply externally to the affected area up to 6 times daily
Active Ingredient
Lidocaine 5%
Other Information
See enclosed consumer leaflet.
Store at 15°C to 30°C. Protect from freezing
Inactive Ingredients
Benzyl Alcohol, Carbopol, Lecithin, Propylene Glycol, Vitamin E Acetate, Purified Water
When Using This Product
- avoid contact with eyes
- do not exceed the recommended daily dosage unless directed by a physician
- do not put this product into the rectum by using fingers or any mechanical device or applicator
- certain persons can develop allergic reactions to ingredients in this product. If the symptom being treated does not subside or if redness, irritation, swelling, pain, or other symptoms develop or increase, discontinue use and consult a doctor
Stop Use and Ask A Doctor If
- condition worsens or does not improve within 7 days
- bleeding occurs
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away
Principal Display Panel 1 Oz Tube Carton
zensa
LIDOCAINE CREAM
WITH 5% LIDOCAINE
alera SKINCARE
NDC 69805-786-01
1 oz
Structured Label Content
Uses
- For the temporary relief of local discomfort associated with anorectal disorders
- For the temporary relief of pain, soreness, and burning
Purpose
Topical Anaesthetic
Warnings
For external use only.
Directions
- Adults: when practical, cleanse the affected area with mild soap, and warm water and rinse thoroughly. Gently dry by patting or blottig with toilet tissue or a soft cloth before application of this product. Children under 12 years of age: consult a physician.
- Apply externally to the affected area up to 6 times daily
Active Ingredient (Active ingredient)
Lidocaine 5%
Other Information (Other information)
See enclosed consumer leaflet.
Store at 15°C to 30°C. Protect from freezing
Inactive Ingredients (Inactive ingredients)
Benzyl Alcohol, Carbopol, Lecithin, Propylene Glycol, Vitamin E Acetate, Purified Water
When Using This Product (When using this product)
- avoid contact with eyes
- do not exceed the recommended daily dosage unless directed by a physician
- do not put this product into the rectum by using fingers or any mechanical device or applicator
- certain persons can develop allergic reactions to ingredients in this product. If the symptom being treated does not subside or if redness, irritation, swelling, pain, or other symptoms develop or increase, discontinue use and consult a doctor
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens or does not improve within 7 days
- bleeding occurs
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away
Principal Display Panel 1 Oz Tube Carton (PRINCIPAL DISPLAY PANEL - 1 oz Tube Carton)
zensa
LIDOCAINE CREAM
WITH 5% LIDOCAINE
alera SKINCARE
NDC 69805-786-01
1 oz
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:33.180935 · Updated: 2026-03-14T23:05:22.628914