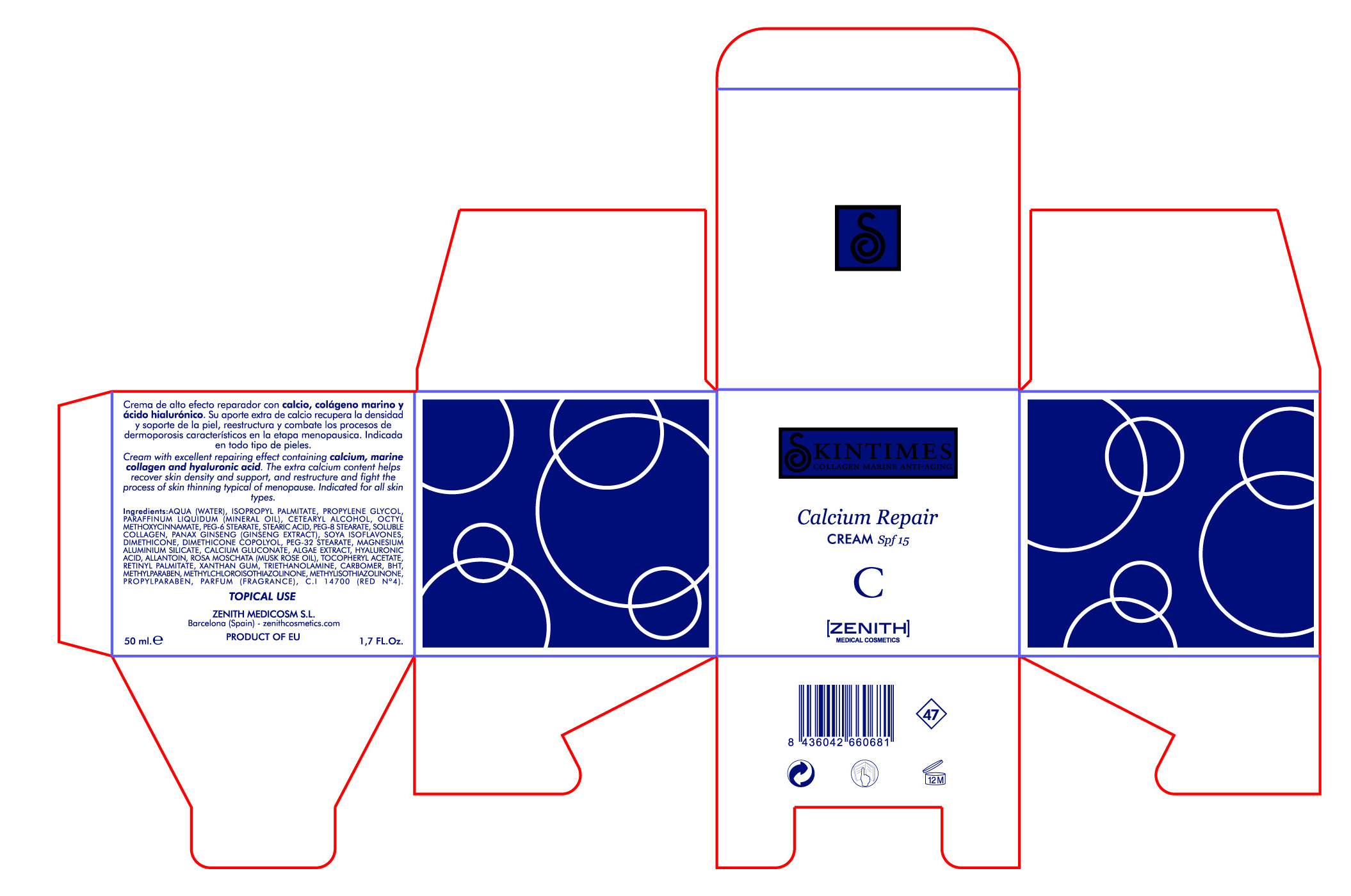
[{"name": "MAGNESIUM ALUMINUM SILICATE", "unii": "6M3P64V0NC", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLPARABEN", "unii": "Z8IX2SC1OH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALPHA-TOCOPHEROL ACETATE", "unii": "9E8X80D2L0", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIMETHICONE", "unii": "92RU3N3Y1O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "XANTHAN GUM", "unii": "TTV12P4NEE", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ROSA MOSCHATA OIL", "unii": "J99W255AWF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MINERAL OIL", "unii": "T5L8T28FGP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CETOSTEARYL ALCOHOL", "unii": "2DMT128M1S", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PEG-8 STEARATE", "unii": "2P9L47VI5E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TROLAMINE", "unii": "9O3K93S3TK", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SOY ISOFLAVONES", "unii": "71B37NR06D", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CALCIUM GLUCONATE", "unii": "SQE6VB453K", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "BUTYLATED HYDROXYTOLUENE", "unii": "1P9D0Z171K", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "HYALURONIC ACID", "unii": "S270N0TRQY", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CARBOMER 934", "unii": "Z135WT9208", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLPARABEN", "unii": "A2I8C7HI9T", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "COLLAGEN, SOLUBLE, FISH SKIN", "unii": "8JC99XGU4W", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "OCTINOXATE", "unii": "4Y5P7MUD51", "classCode": "ACTIB", "strengthNumerator": "3.75", "strengthDenominator": "50", "strengthNumeratorUnit": "mL", "strengthDenominatorUnit": "mL"}, {"name": "ISOPROPYL PALMITATE", "unii": "8CRQ2TH63M", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PEG-6 STEARATE", "unii": "8LQC57C6B0", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PEG-32 STEARATE", "unii": "33GX5WQC0M", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "STEARIC ACID", "unii": "4ELV7Z65AP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PANAX GINSENG FRUIT", "unii": "E35T4MOI3E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FUCUS VESICULOSUS", "unii": "535G2ABX9M", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALLANTOIN", "unii": "344S277G0Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VITAMIN A PALMITATE", "unii": "1D1K0N0VVC", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLENE GLYCOL", "unii": "6DC9Q167V3", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLCHLOROISOTHIAZOLINONE", "unii": "DEL7T5QRPN", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLISOTHIAZOLINONE", "unii": "229D0E1QFA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C RED NO. 4", "unii": "X3W0AM1JLX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}]