Candida +
f5171818-902d-8ee3-da28-5048a97aa2a6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ammoniacum Gummi 6-20X 200K, Aspergillus Niger 12X 200K, Caffeic acid 12X 200K, Candida Albicans 30X 12-200K, Candida Parapsilosis 200K, Ipecacuanha 6X 30-200K, Lycopodium Clavatum 20X 20-200K, Mannan 6X 13-30-80-200K, Vaccinium Vitis Idaea 12X 200K, Vaccinium Vitis Idaea 2X
Purpose
Ammoniacum Gummi Anti-age Aspergillus Niger Urogenital drainage Caffeic acid Urogenital drainage Candida Albicans Dysbiosis Candida Parapsilosis Dysbiosis Ipecacuanha Drainage Lycopodium Clavatum Drainage Mannan Drainage Vaccinium Vitis Idaea Intestinal Vaccinium Vitis Idaea Intestinal
Medication Information
Purpose
Ammoniacum Gummi Anti-age Aspergillus Niger Urogenital drainage Caffeic acid Urogenital drainage Candida Albicans Dysbiosis Candida Parapsilosis Dysbiosis Ipecacuanha Drainage Lycopodium Clavatum Drainage Mannan Drainage Vaccinium Vitis Idaea Intestinal Vaccinium Vitis Idaea Intestinal
Description
Ammoniacum Gummi 6-20X 200K, Aspergillus Niger 12X 200K, Caffeic acid 12X 200K, Candida Albicans 30X 12-200K, Candida Parapsilosis 200K, Ipecacuanha 6X 30-200K, Lycopodium Clavatum 20X 20-200K, Mannan 6X 13-30-80-200K, Vaccinium Vitis Idaea 12X 200K, Vaccinium Vitis Idaea 2X
Keep this and all medicines out of reach of children.
Uses
For the relief of symptoms due to Candida, including precursors and byproducts
Dosage
1-5 spray(s); 1-3 time(s) per day or as recommended by your health care practitioner.
Purpose:
| Ammoniacum Gummi | Anti-age |
| Aspergillus Niger | Urogenital drainage |
| Caffeic acid | Urogenital drainage |
| Candida Albicans | Dysbiosis |
| Candida Parapsilosis | Dysbiosis |
| Ipecacuanha | Drainage |
| Lycopodium Clavatum | Drainage |
| Mannan | Drainage |
| Vaccinium Vitis Idaea | Intestinal |
| Vaccinium Vitis Idaea | Intestinal |
Warnings
Stop use and ask a health care practitioner if symptoms persist for more than five days or worsen. If pregnant or breastfeeding, ask a health care practitioner before use.
Questions
Other Information
Normal storage use.
Do not use if tamper-evident seal is broken or removed.
Other Ingredients
Alcohol and Water
Active Ingredients
Ammoniacum Gummi 6-20X 200K, Aspergillus Niger 12X 200K, Caffeic acid 12X 200K, Candida Albicans 30X 12-200K, Candida Parapsilosis 200K, Ipecacuanha 6X 30-200K, Lycopodium Clavatum 20X 20-200K, Mannan 6X 13-30-80-200K, Vaccinium Vitis Idaea 12X 200K, Vaccinium Vitis Idaea 2X
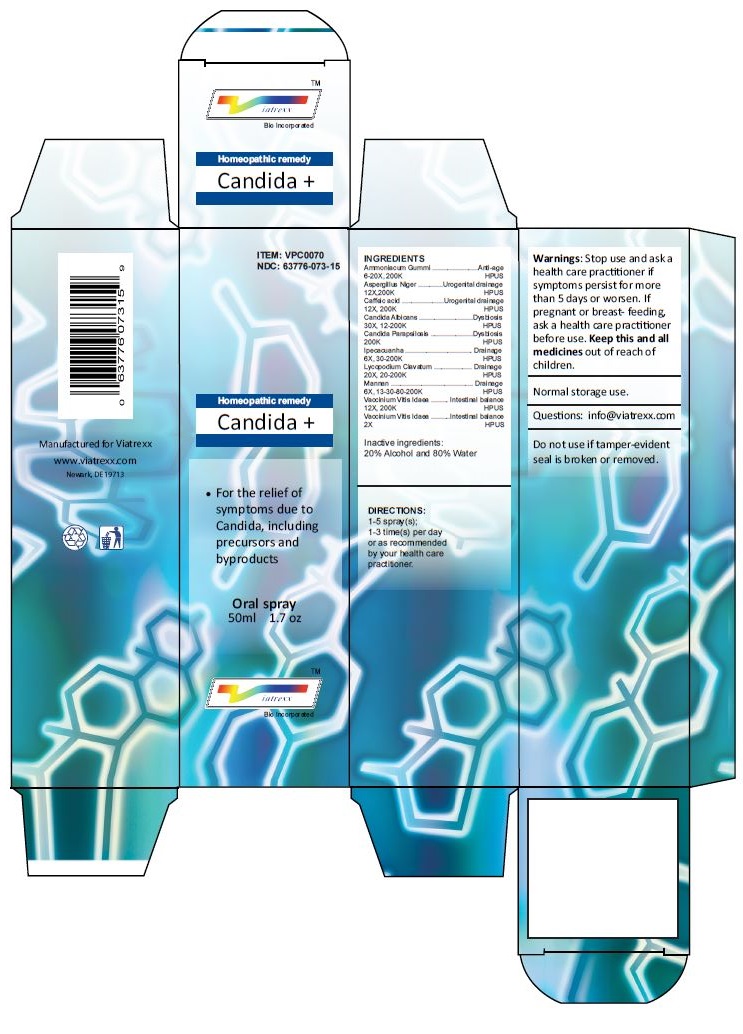
Principal Display Panel
ITEM: VPC0070
NDC 63776-073-15
Homeopathic remedy
Candida +
• For the relief of symptoms due to Candida, including precursors and byproducts
Oral spray
50ml 1.7 oz
Viatrexx™ Bio Incorporated
Manufactured by Viatrexx
www.viatrexx.com
Newark, DE 19713
Candida +
50 mL
1.7 oz
Viatrexx™ Bio Incorporated
ITEM: VPC0070
NDC: 63776-073-15
INDICATIONS:
For the relief of symptoms due to Candida, including precursors and byproducts
DIRECTIONS:
1-5 spray(s); 1-3 time(s) per day or as recommended by your health care practitioner.
Mfg. for
Viatrexx Bio Incorporated.
www.viatrexx.com
Newark, DE 19713
Structured Label Content
Keep this and all medicines out of reach of children.
Uses
For the relief of symptoms due to Candida, including precursors and byproducts
Dosage
1-5 spray(s); 1-3 time(s) per day or as recommended by your health care practitioner.
Purpose:
| Ammoniacum Gummi | Anti-age |
| Aspergillus Niger | Urogenital drainage |
| Caffeic acid | Urogenital drainage |
| Candida Albicans | Dysbiosis |
| Candida Parapsilosis | Dysbiosis |
| Ipecacuanha | Drainage |
| Lycopodium Clavatum | Drainage |
| Mannan | Drainage |
| Vaccinium Vitis Idaea | Intestinal |
| Vaccinium Vitis Idaea | Intestinal |
Warnings
Stop use and ask a health care practitioner if symptoms persist for more than five days or worsen. If pregnant or breastfeeding, ask a health care practitioner before use.
Questions
Other Information
Normal storage use.
Do not use if tamper-evident seal is broken or removed.
Other Ingredients
Alcohol and Water
Active Ingredients
Ammoniacum Gummi 6-20X 200K, Aspergillus Niger 12X 200K, Caffeic acid 12X 200K, Candida Albicans 30X 12-200K, Candida Parapsilosis 200K, Ipecacuanha 6X 30-200K, Lycopodium Clavatum 20X 20-200K, Mannan 6X 13-30-80-200K, Vaccinium Vitis Idaea 12X 200K, Vaccinium Vitis Idaea 2X
Principal Display Panel
ITEM: VPC0070
NDC 63776-073-15
Homeopathic remedy
Candida +
• For the relief of symptoms due to Candida, including precursors and byproducts
Oral spray
50ml 1.7 oz
Viatrexx™ Bio Incorporated
Manufactured by Viatrexx
www.viatrexx.com
Newark, DE 19713
Candida +
50 mL
1.7 oz
Viatrexx™ Bio Incorporated
ITEM: VPC0070
NDC: 63776-073-15
INDICATIONS:
For the relief of symptoms due to Candida, including precursors and byproducts
DIRECTIONS:
1-5 spray(s); 1-3 time(s) per day or as recommended by your health care practitioner.
Mfg. for
Viatrexx Bio Incorporated.
www.viatrexx.com
Newark, DE 19713
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:10:50.897244 · Updated: 2026-03-14T23:15:47.351284