f4264944-56c5-4f95-a8d7-8ded02953043
34390-5
HUMAN OTC DRUG LABEL
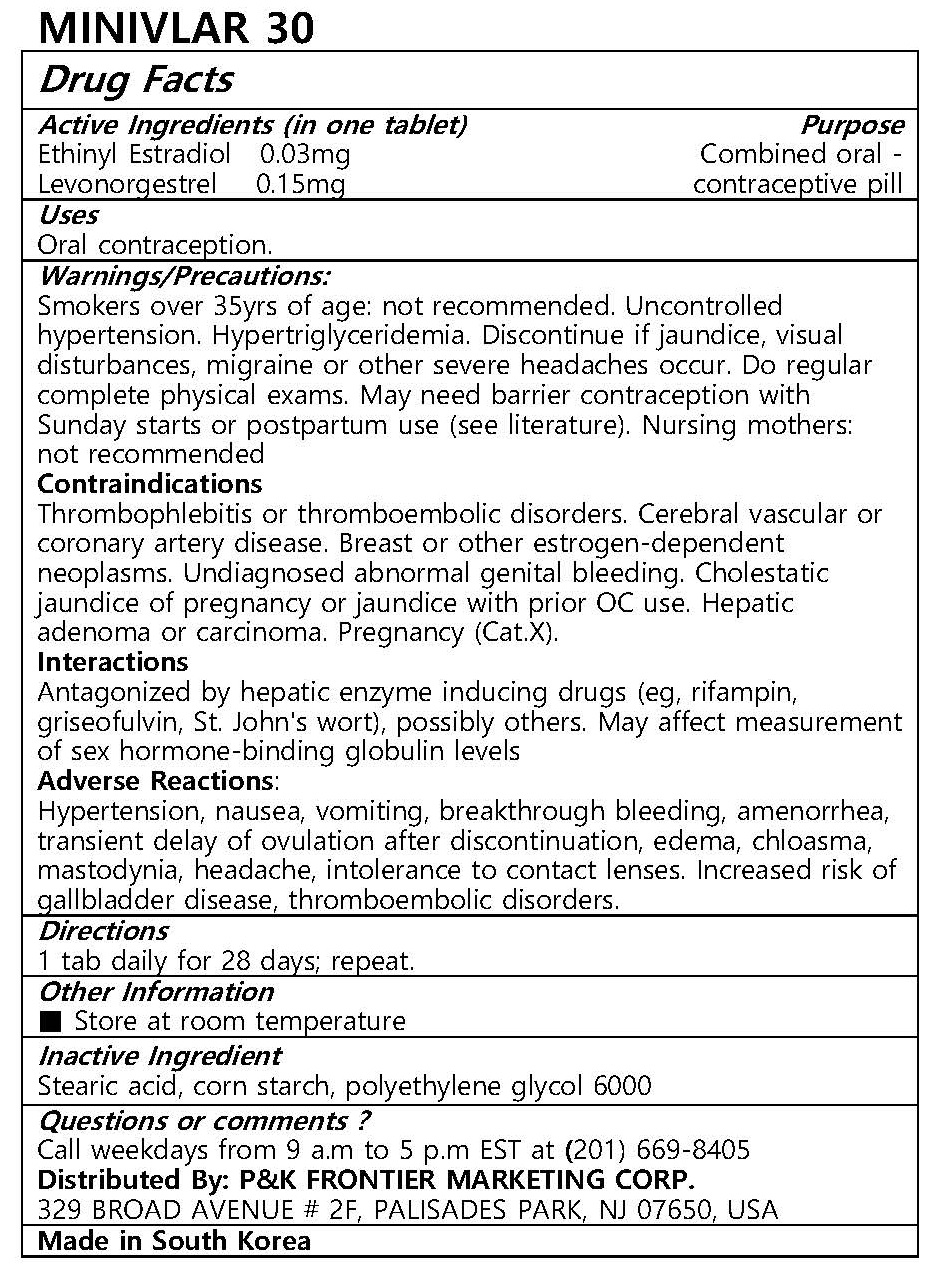
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethinyl Estradiol, Levonorgestrel
Medication Information
Warnings and Precautions
Warnings/Precautions:
Smokers over 35yrs of age: not recommended. Uncontrolled hypertension. Hypertriglyceridemia. Discontinue if jaundice, visual disturbances, migraine or other severe headaches occur. Do regular complete physical exams. May need barrier contraception with Sunday starts or postpartum use (see literature). Nursing mothers: not recommended
Contraindications
Thrombophlebitis or thromboembolic disorders. Cerebral vascular or coronary artery disease. Breast or other estrogen-dependent neoplasms. Undiagnosed abnormal genital bleeding. Cholestatic jaundice of pregnancy or jaundice with prior OC use. Hepatic adenoma or carcinoma. Pregnancy (Cat.X).
Interactions
Antagonized by hepatic enzyme inducing drugs (eg, rifampin, griseofulvin, St. John's wort), possibly others. May affect measurement of sex hormone-binding globulin levels
Adverse Reactions:
Hypertension, nausea, vomiting, breakthrough bleeding, amenorrhea, transient delay of ovulation after discontinuation, edema, chloasma, mastodynia, headache, intolerance to contact lenses. Increased risk of gallbladder disease, thromboembolic disorders.
Indications and Usage
1 tab daily for 21 days; repeat.
Dosage and Administration
For oral use only
Description
Ethinyl Estradiol, Levonorgestrel
Section 50565-1
Keep out of reach of children
Section 51727-6
Stearic acid, corn starch, polyethylene glycol 6000
Section 51945-4
Section 55105-1
Oral contraception
Section 55106-9
Ethinyl Estradiol, Levonorgestrel
Structured Label Content
Indications and Usage (34067-9)
1 tab daily for 21 days; repeat.
Dosage and Administration (34068-7)
For oral use only
Warnings and Precautions (34071-1)
Warnings/Precautions:
Smokers over 35yrs of age: not recommended. Uncontrolled hypertension. Hypertriglyceridemia. Discontinue if jaundice, visual disturbances, migraine or other severe headaches occur. Do regular complete physical exams. May need barrier contraception with Sunday starts or postpartum use (see literature). Nursing mothers: not recommended
Contraindications
Thrombophlebitis or thromboembolic disorders. Cerebral vascular or coronary artery disease. Breast or other estrogen-dependent neoplasms. Undiagnosed abnormal genital bleeding. Cholestatic jaundice of pregnancy or jaundice with prior OC use. Hepatic adenoma or carcinoma. Pregnancy (Cat.X).
Interactions
Antagonized by hepatic enzyme inducing drugs (eg, rifampin, griseofulvin, St. John's wort), possibly others. May affect measurement of sex hormone-binding globulin levels
Adverse Reactions:
Hypertension, nausea, vomiting, breakthrough bleeding, amenorrhea, transient delay of ovulation after discontinuation, edema, chloasma, mastodynia, headache, intolerance to contact lenses. Increased risk of gallbladder disease, thromboembolic disorders.
Section 50565-1 (50565-1)
Keep out of reach of children
Section 51727-6 (51727-6)
Stearic acid, corn starch, polyethylene glycol 6000
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Oral contraception
Section 55106-9 (55106-9)
Ethinyl Estradiol, Levonorgestrel
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:40.998218 · Updated: 2026-03-14T22:55:19.604592