Hydralazine Hydrochloride Injection, Usp
f3c806a4-b971-421c-bcb9-9979c6d7aaec
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
(For Intramuscular or Intravenous Use) meitheal® Rx only
Indications and Usage
Severe essential hypertension when the drug cannot be given orally or when there is an urgent need to lower blood pressure.
Dosage and Administration
When there is urgent need, therapy in the hospitalized patient may be initiated intramuscularly or as a rapid intravenous bolus injection directly into the vein. Hydralazine hydrochloride injection should be used only when the drug cannot be given orally. The usual dose is 20 to 40 mg, repeated as necessary. Certain patients (especially those with marked renal damage) may require a lower dose. Blood pressure should be checked frequently. It may begin to fall within a few minutes after injection, with the average maximal decrease occurring in 10 to 80 minutes. In cases where there has been increased intracranial pressure, lowering the blood pressure may increase cerebral ischemia. Most patients can be transferred to oral hydralazine hydrochloride within 24 to 48 hours. The product should be used immediately after the vial is opened. It should not be added to infusion solutions. Hydralazine hydrochloride injection may discolor upon contact with metal; discolored solutions should be discarded. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard unused portion.
Contraindications
Hypersensitivity to hydralazine; coronary artery disease; mitral valvular rheumatic heart disease.
How Supplied
Hydralazine Hydrochloride Injection, USP is a colorless solution and is supplied as follows: NDC Hydralazine Hydrochloride for Injection, USP (20 mg per mL) Package Factor 71288- 205 -02 20 mg per mL Single-Dose Vial 25 vials per carton
Medication Information
Indications and Usage
Severe essential hypertension when the drug cannot be given orally or when there is an urgent need to lower blood pressure.
Dosage and Administration
When there is urgent need, therapy in the hospitalized patient may be initiated intramuscularly or as a rapid intravenous bolus injection directly into the vein. Hydralazine hydrochloride injection should be used only when the drug cannot be given orally. The usual dose is 20 to 40 mg, repeated as necessary.
Certain patients (especially those with marked renal damage) may require a lower dose. Blood pressure should be checked frequently. It may begin to fall within a few minutes after injection, with the average maximal decrease occurring in 10 to 80 minutes. In cases where there has been increased intracranial pressure, lowering the blood pressure may increase cerebral ischemia. Most patients can be transferred to oral hydralazine hydrochloride within 24 to 48 hours.
The product should be used immediately after the vial is opened. It should not be added to infusion solutions. Hydralazine hydrochloride injection may discolor upon contact with metal; discolored solutions should be discarded.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Discard unused portion.
Contraindications
Hypersensitivity to hydralazine; coronary artery disease; mitral valvular rheumatic heart disease.
How Supplied
Hydralazine Hydrochloride Injection, USP is a colorless solution and is supplied as follows:
| NDC | Hydralazine Hydrochloride for Injection, USP (20 mg per mL) | Package Factor |
| 71288-205-02 | 20 mg per mL Single-Dose Vial | 25 vials per carton |
Description
Hydralazine Hydrochloride Injection, USP is an antihypertensive available in a 1 mL vial for intravenous and
intramuscular administration. Hydralazine Hydrochloride Injection, USP is a sterile, nonpyrogenic colorless solution.
Each mL contains:
Active: Hydralazine Hydrochloride, USP, 20 mg
Preservatives: Methylparaben, NF, 0.65 mg; Propylparaben, NF, 0.35 mg
Inactives: Propylene Glycol, USP, 103.6 mg; Sodium Hydroxide and/or Hydrochloric Acid to adjust pH (3.4 to 4.4)
and Water for Injection.
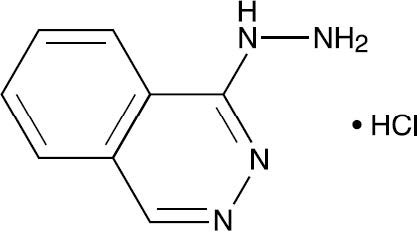
Hydralazine Hydrochloride, USP is 1-hydrazinophthalazine monohydrochloride, and its structural formula is:
Molecular Formula C8H8N4•HCl
Hydralazine Hydrochloride, USP is a white to off-white, odorless crystalline powder. It is soluble in water, slightly
soluble in alcohol, and very slightly soluble in ether. It melts at about 275°C, with decomposition, and has a molecular
weight of 196.64.
Section 34072-9
General
Myocardial stimulation produced by hydralazine can cause anginal attacks and ECG changes of myocardial ischemia. The drug has been implicated in the production of myocardial infarction. It must, therefore, be used with caution in patients with suspected coronary artery disease.
The “hyperdynamic” circulation caused by hydralazine may accentuate specific cardiovascular inadequacies. For example, hydralazine may increase pulmonary artery pressure in patients with mitral valvular disease. The drug may reduce the pressor responses to epinephrine. Postural hypotension may result from hydralazine but is less common than with ganglionic blocking agents. It should be used with caution in patients with cerebral vascular accidents.
In hypertensive patients with normal kidneys who are treated with hydralazine, there is evidence of increased renal blood flow and a maintenance of glomerular filtration rate. In some instances where control values were below normal, improved renal function has been noted after administration of hydralazine. However, as with any antihypertensive agent, hydralazine should be used with caution in patients with advanced renal damage.
Peripheral neuritis, evidenced by paresthesia, numbness, and tingling, has been observed. Published evidence suggests an antipyridoxine effect, and that pyridoxine should be added to the regimen if symptoms develop.
Section 42229-5
(For Intramuscular or Intravenous Use)
meitheal®
Rx only
Warnings
In a few patients hydralazine may produce a clinical picture simulating systemic lupus erythematosus including glomerulonephritis. In such patients hydralazine should be discontinued unless the benefit-to-risk determination requires continued antihypertensive therapy with this drug. Symptoms and signs usually regress when the drug is discontinued but residua have been detected many years later. Long-term treatment with steroids may be necessary (see PRECAUTIONS, Laboratory Tests ).
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Sterile, Nonpyrogenic.
The container closure is not made with natural rubber latex.
meitheal®
Mfd. for Meitheal Pharmaceuticals
Chicago, IL 60631 (USA)
©2023 Meitheal Pharmaceuticals Inc.
Mfd. by Kindos Pharmaceuticals Co., Ltd.
Chengdu, China 611731
November 2023
Overdosage
Acute Toxicity: No deaths due to acute poisoning have been reported.
Highest known dose survived: adults, 10 g orally.
Oral LD50 in rats: 173 and 187 mg/kg.
Principal Display Panel – Hydralazine Hydrochloride Injection, Usp 20 Mg Per Ml Vial
NDC 71288-205-01
Rx Only
hydrALAZINE Hydrochloride Injection, USP
20 mg per mL
For Intramuscular or Intravenous Use
1 mL Single-Dose Vial
Principal Display Panel – Hydralazine Hydrochloride Injection, Usp 20 Mg Per Ml Carton
NDC 71288-205-02
Rx Only
hydrALAZINE Hydrochloride Injection, USP
20 mg per mL
For Intramuscular or Intravenous Use
25 x 1 mL Single-Dose Vials
Structured Label Content
Section 34072-9 (34072-9)
General
Myocardial stimulation produced by hydralazine can cause anginal attacks and ECG changes of myocardial ischemia. The drug has been implicated in the production of myocardial infarction. It must, therefore, be used with caution in patients with suspected coronary artery disease.
The “hyperdynamic” circulation caused by hydralazine may accentuate specific cardiovascular inadequacies. For example, hydralazine may increase pulmonary artery pressure in patients with mitral valvular disease. The drug may reduce the pressor responses to epinephrine. Postural hypotension may result from hydralazine but is less common than with ganglionic blocking agents. It should be used with caution in patients with cerebral vascular accidents.
In hypertensive patients with normal kidneys who are treated with hydralazine, there is evidence of increased renal blood flow and a maintenance of glomerular filtration rate. In some instances where control values were below normal, improved renal function has been noted after administration of hydralazine. However, as with any antihypertensive agent, hydralazine should be used with caution in patients with advanced renal damage.
Peripheral neuritis, evidenced by paresthesia, numbness, and tingling, has been observed. Published evidence suggests an antipyridoxine effect, and that pyridoxine should be added to the regimen if symptoms develop.
Section 42229-5 (42229-5)
(For Intramuscular or Intravenous Use)
meitheal®
Rx only
Warnings (WARNINGS)
In a few patients hydralazine may produce a clinical picture simulating systemic lupus erythematosus including glomerulonephritis. In such patients hydralazine should be discontinued unless the benefit-to-risk determination requires continued antihypertensive therapy with this drug. Symptoms and signs usually regress when the drug is discontinued but residua have been detected many years later. Long-term treatment with steroids may be necessary (see PRECAUTIONS, Laboratory Tests ).
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Sterile, Nonpyrogenic.
The container closure is not made with natural rubber latex.
meitheal®
Mfd. for Meitheal Pharmaceuticals
Chicago, IL 60631 (USA)
©2023 Meitheal Pharmaceuticals Inc.
Mfd. by Kindos Pharmaceuticals Co., Ltd.
Chengdu, China 611731
November 2023
Overdosage (OVERDOSAGE)
Acute Toxicity: No deaths due to acute poisoning have been reported.
Highest known dose survived: adults, 10 g orally.
Oral LD50 in rats: 173 and 187 mg/kg.
Description (DESCRIPTION)
Hydralazine Hydrochloride Injection, USP is an antihypertensive available in a 1 mL vial for intravenous and
intramuscular administration. Hydralazine Hydrochloride Injection, USP is a sterile, nonpyrogenic colorless solution.
Each mL contains:
Active: Hydralazine Hydrochloride, USP, 20 mg
Preservatives: Methylparaben, NF, 0.65 mg; Propylparaben, NF, 0.35 mg
Inactives: Propylene Glycol, USP, 103.6 mg; Sodium Hydroxide and/or Hydrochloric Acid to adjust pH (3.4 to 4.4)
and Water for Injection.
Hydralazine Hydrochloride, USP is 1-hydrazinophthalazine monohydrochloride, and its structural formula is:
Molecular Formula C8H8N4•HCl
Hydralazine Hydrochloride, USP is a white to off-white, odorless crystalline powder. It is soluble in water, slightly
soluble in alcohol, and very slightly soluble in ether. It melts at about 275°C, with decomposition, and has a molecular
weight of 196.64.
How Supplied (HOW SUPPLIED)
Hydralazine Hydrochloride Injection, USP is a colorless solution and is supplied as follows:
| NDC | Hydralazine Hydrochloride for Injection, USP (20 mg per mL) | Package Factor |
| 71288-205-02 | 20 mg per mL Single-Dose Vial | 25 vials per carton |
Contraindications (CONTRAINDICATIONS)
Hypersensitivity to hydralazine; coronary artery disease; mitral valvular rheumatic heart disease.
Indications and Usage (INDICATIONS AND USAGE)
Severe essential hypertension when the drug cannot be given orally or when there is an urgent need to lower blood pressure.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
When there is urgent need, therapy in the hospitalized patient may be initiated intramuscularly or as a rapid intravenous bolus injection directly into the vein. Hydralazine hydrochloride injection should be used only when the drug cannot be given orally. The usual dose is 20 to 40 mg, repeated as necessary.
Certain patients (especially those with marked renal damage) may require a lower dose. Blood pressure should be checked frequently. It may begin to fall within a few minutes after injection, with the average maximal decrease occurring in 10 to 80 minutes. In cases where there has been increased intracranial pressure, lowering the blood pressure may increase cerebral ischemia. Most patients can be transferred to oral hydralazine hydrochloride within 24 to 48 hours.
The product should be used immediately after the vial is opened. It should not be added to infusion solutions. Hydralazine hydrochloride injection may discolor upon contact with metal; discolored solutions should be discarded.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Discard unused portion.
Principal Display Panel – Hydralazine Hydrochloride Injection, Usp 20 Mg Per Ml Vial (Principal Display Panel – hydrALAZINE Hydrochloride Injection, USP 20 mg per mL Vial)
NDC 71288-205-01
Rx Only
hydrALAZINE Hydrochloride Injection, USP
20 mg per mL
For Intramuscular or Intravenous Use
1 mL Single-Dose Vial
Principal Display Panel – Hydralazine Hydrochloride Injection, Usp 20 Mg Per Ml Carton (Principal Display Panel – hydrALAZINE Hydrochloride Injection, USP 20 mg per mL Carton)
NDC 71288-205-02
Rx Only
hydrALAZINE Hydrochloride Injection, USP
20 mg per mL
For Intramuscular or Intravenous Use
25 x 1 mL Single-Dose Vials
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:34.169460 · Updated: 2026-03-14T22:27:35.794501