These Highlights Do Not Include All The Information Needed To Use Cervidil Safely And Effectively. See Full Prescribing Information For Cervidil.
f3bc9031-0620-4a50-b440-f661cadfeb1d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
CERVIDIL is indicated for the initiation and/or continuation of cervical ripening in pregnant women at or near term in whom there is a medical or obstetrical indication for the induction of labor.
Indications and Usage
CERVIDIL is indicated for the initiation and/or continuation of cervical ripening in pregnant women at or near term in whom there is a medical or obstetrical indication for the induction of labor.
Dosage and Administration
Administer a single vaginal insert (10 mg) for up to 12 hours of use (approximately 0.3 mg of dinoprostone is released per hour) ( 2.1 ) Remove upon onset of active labor or 12 hours after insertion. ( 2.1 ) Carefully monitor for uterine activity, fetal status and the progression of cervical dilatation and effacement. ( 2.1 ) CERVIDIL should be administered only by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities ( 2.2 ). See full prescribing information for complete preparation, administration, and removal instructions. ( 2.2 , 2.3 )
Warnings and Precautions
Disseminated Intravascular Coagulation : Assess for evolving fibrinolysis in the immediate post-partum period. ( 5.2 ) Amniotic Fluid Embolism Syndrome : Carefully monitor patients for clinical signs of hypotension, hypoxemia and respiratory failure, DIC, coma or seizures. Provide supportive care. ( 5.3 ) Uterine Tachysystole and Uterine Hypersystole/Hypertonicity : Monitor uterine activity; remove vaginal insert. ( 5.4 ) History of Glaucoma : Consider non-prostaglandin cervical ripening procedures. ( 5.5 )
Contraindications
CERVIDIL is contraindicated in patients with: Known hypersensitivity to prostaglandins [see Adverse Reactions (6.2) ] Evidence or clinical suspicion of fetal distress where delivery is not imminent Unexplained vaginal bleeding in the current pregnancy Evidence or clinical suspicion of marked cephalopelvic disproportion Conditions for which induction of labor is contraindicated Conditions for which oxytocic drugs are contraindicated Previous cesarean section or other uterine surgery expected to affect uterine integrity (such as myomectomy) Conditions under which prolonged contraction of the uterus may be detrimental to fetal safety Concurrent use with intravenous oxytocic agents [see Warnings and Precautions (5.4) and Drug Interactions (7) ] Six or more previous term pregnancies
Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections: Disseminated Intravascular Coagulation [see Warnings and Precautions (5.2) ] Amniotic Fluid Embolism [see Warnings and Precautions (5.3) ] Uterine Tachysytole and Uterine Hypersystole/Hypertonicity [see Warnings and Precautions (5.4) ]
Drug Interactions
Oxytocic Agents : May augment the activity of oxytocic agents; concomitant use of intravenous oxytocic agents is contraindicated ( 4 , 7.1 ). Remove vaginal insert at least 30 minutes before administering an oxytocic agent. ( 7.1 )
Medication Information
Warnings and Precautions
Disseminated Intravascular Coagulation : Assess for evolving fibrinolysis in the immediate post-partum period. ( 5.2 ) Amniotic Fluid Embolism Syndrome : Carefully monitor patients for clinical signs of hypotension, hypoxemia and respiratory failure, DIC, coma or seizures. Provide supportive care. ( 5.3 ) Uterine Tachysystole and Uterine Hypersystole/Hypertonicity : Monitor uterine activity; remove vaginal insert. ( 5.4 ) History of Glaucoma : Consider non-prostaglandin cervical ripening procedures. ( 5.5 )
Indications and Usage
CERVIDIL is indicated for the initiation and/or continuation of cervical ripening in pregnant women at or near term in whom there is a medical or obstetrical indication for the induction of labor.
Dosage and Administration
Administer a single vaginal insert (10 mg) for up to 12 hours of use (approximately 0.3 mg of dinoprostone is released per hour) ( 2.1 ) Remove upon onset of active labor or 12 hours after insertion. ( 2.1 ) Carefully monitor for uterine activity, fetal status and the progression of cervical dilatation and effacement. ( 2.1 ) CERVIDIL should be administered only by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities ( 2.2 ). See full prescribing information for complete preparation, administration, and removal instructions. ( 2.2 , 2.3 )
Contraindications
CERVIDIL is contraindicated in patients with: Known hypersensitivity to prostaglandins [see Adverse Reactions (6.2) ] Evidence or clinical suspicion of fetal distress where delivery is not imminent Unexplained vaginal bleeding in the current pregnancy Evidence or clinical suspicion of marked cephalopelvic disproportion Conditions for which induction of labor is contraindicated Conditions for which oxytocic drugs are contraindicated Previous cesarean section or other uterine surgery expected to affect uterine integrity (such as myomectomy) Conditions under which prolonged contraction of the uterus may be detrimental to fetal safety Concurrent use with intravenous oxytocic agents [see Warnings and Precautions (5.4) and Drug Interactions (7) ] Six or more previous term pregnancies
Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections: Disseminated Intravascular Coagulation [see Warnings and Precautions (5.2) ] Amniotic Fluid Embolism [see Warnings and Precautions (5.3) ] Uterine Tachysytole and Uterine Hypersystole/Hypertonicity [see Warnings and Precautions (5.4) ]
Drug Interactions
Oxytocic Agents : May augment the activity of oxytocic agents; concomitant use of intravenous oxytocic agents is contraindicated ( 4 , 7.1 ). Remove vaginal insert at least 30 minutes before administering an oxytocic agent. ( 7.1 )
Description
CERVIDIL is indicated for the initiation and/or continuation of cervical ripening in pregnant women at or near term in whom there is a medical or obstetrical indication for the induction of labor.
Section 42229-5
Preparation and Administration Instructions
- Keep CERVIDIL frozen until ready for use. Do not warm CERVIDIL prior to vaginal insertion.
- Tear open the individually-wrapped foil package containing CERVIDIL along the tear mark. Never open the package using scissors or other sharp objects because this may damage the knitted polyester retrieval system. Do not cut the retrieval system and do not use CERVIDIL unless its retrieval system is intact.
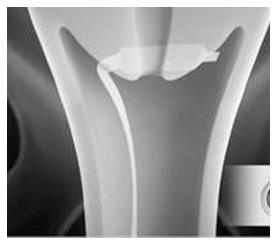
- Immediately after opening the package, insert CERVIDIL transversely, in the posterior fornix of the vagina (see Figure 1). If necessary, use a minimal amount of water-miscible lubricant to assist vaginal insertion. Do not permit excess contact or coating with the lubricant, as this could prevent release of dinoprostone from the vaginal insert. Insertion does not require sterile conditions.
Figure 1: Placement of CERVIDIL in the Posterior Vaginal Fornix - Tuck some of the excess retrieval system into the vagina to avoid movement of CERVIDIL away from the proper position; however, leave a small amount of the retrieval system outside the vagina to aid in retrieval.
- Instruct women to remain in a recumbent position during insertion of CERVIDIL and for 2 hours afterward. Women may be ambulatory 2 hours after insertion; however, ensure that the insert remains in place.
Section 44425-7
Storage and Handling
Store in a freezer between -20°C and -10°C (-4°F and 14°F). CERVIDIL, enclosed in its aluminum/polyethylene pack, is stable when stored in a freezer for a period of three years. Vaginal inserts exposed to high humidity will absorb moisture from the air and thereby alter the release characteristics of dinoprostone.
5.5 Glaucoma
Prostaglandins, including CERVIDIL, can lead to raised intraocular pressure and constriction of pupils. Consider non-prostaglandin cervical ripening procedures in patients with Glaucoma.
11 Description
CERVIDIL (dinoprostone) vaginal insert contains dinoprostone, a prostaglandin analog. Each vaginal insert contains 10 mg of dinoprostone in 241 mg of a cross-linked polyethylene oxide/urethane polymer (hydrogel polymer) that is semi-opaque, beige colored, flat, rectangular in shape with rounded corners and measuring 29 mm by 9.5 mm by 0.8 mm. The vaginal insert is contained within a pouch of an off-white knitted polyester yarn retrieval system. When placed in a moist environment, the yarn absorbs water, swells, and releases the enclosed dinoprostone. The knitted polyester retrieval system has a long tape-like end that is designed to aid retrieval of CERVIDIL at the end of the dosing interval or earlier if clinically indicated. The finished product is a controlled-release formulation that has been found to release dinoprostone in vivo at a rate of approximately 0.3 mg per hour.
The chemical name for dinoprostone (known as prostaglandin E2 or PGE2) is 11α,15S-dihydroxy-9-oxo-prosta-5Z,13E-dien-1-oic acid and the structural formula is represented below:
The molecular formula is C20H32O5 and its molecular weight is 352.47. Dinoprostone occurs as a white to off-white crystalline powder. It has a melting point within the range of 65° to 69°C. Dinoprostone is soluble in ethanol and in 25% ethanol in water.
8.4 Pediatric Use
The safety and effectiveness of CERVIDIL have not been established in pregnant girls.
14 Clinical Studies
The effectiveness and safety of CERVIDIL for the induction of cervical ripening was evaluated in 658 pregnant women (320 CERVIDIL-treated women and 338 placebo-treated women) at or near term in three randomized, double-blind, placebo-controlled trials (Trials 1, 2, and 3). Efficacy outcomes included percentage with treatment success (defined as vaginal delivery within 12 hours, Bishop Score ≥ 6 in the 12-hour observation period, or ≥ 3 increase in the Bishop score in the 12-hour observation period), time to delivery, and time to onset of labor. Table 2 presents efficacy outcomes from Trials 1, 2, and 3.
| Primipara/Nullipara | Multipara | ||||
|---|---|---|---|---|---|
| Study # | CERVIDIL | Placebo | CERVIDIL | Placebo | P-Value |
|
Treatment Success Treatment success was defined as vaginal delivery within 12 hours, Bishop score ≥ 6 in the 12-hour observation period, or ≥ 3 increase in the Bishop score in the 12-hour observation period.
|
|||||
| Trial 1 (N=81) |
65% (n=26) |
28% (n=32) |
87% (n=16) |
29% (n=7) |
<0.001 |
| Trial 2 (N=371) |
68% (n=111) |
24% (n=123) |
77% (n=65) |
24% (n=72) |
<0.001 |
| Trial 3 (N=206) |
72% (n=60) |
48% (n=63) |
55% (n=42) |
41% (n=41) |
0.003 |
| Median Time to Delivery (hours) | |||||
| Trial 1 (N=81) |
25.7 (n=26) |
34.5 (n=32) |
12.3 (n=16) |
24.6 (n=7) |
0.001 |
| Trial 3 (N=206) |
25.5 (n=60) |
37.2 (n=63) |
20.8 (n=42) |
27.4 (n=41) |
<0.001 |
| Median Time to Onset of Labor (hours) | |||||
| Trial 1 (N=81) |
12 (n=26) |
19.2 (n=32) |
6.9 (n=16) |
18.3 (n=7) |
<0.001 |
4 Contraindications
CERVIDIL is contraindicated in patients with:
- Known hypersensitivity to prostaglandins [see Adverse Reactions (6.2)]
- Evidence or clinical suspicion of fetal distress where delivery is not imminent
- Unexplained vaginal bleeding in the current pregnancy
- Evidence or clinical suspicion of marked cephalopelvic disproportion
- Conditions for which induction of labor is contraindicated
- Conditions for which oxytocic drugs are contraindicated
- Previous cesarean section or other uterine surgery expected to affect uterine integrity (such as myomectomy)
- Conditions under which prolonged contraction of the uterus may be detrimental to fetal safety
- Concurrent use with intravenous oxytocic agents [see Warnings and Precautions (5.4) and Drug Interactions (7)]
- Six or more previous term pregnancies
6 Adverse Reactions
The following adverse reactions are described, or described in greater detail, in other sections:
- Disseminated Intravascular Coagulation [see Warnings and Precautions (5.2)]
- Amniotic Fluid Embolism [see Warnings and Precautions (5.3)]
- Uterine Tachysytole and Uterine Hypersystole/Hypertonicity [see Warnings and Precautions (5.4)]
7 Drug Interactions
7.1 Oxytocic Agents
CERVIDIL is contraindicated in patients receiving intravenous oxytocic agents because CERVIDIL may augment the activity of oxytocic agents. A dosing interval of at least 30 minutes is recommended for the sequential use of an oxytocic agent following the removal of CERVIDIL.
12.2 Pharmacodynamics
No specific pharmacodynamic studies were conducted with CERVIDIL.
12.3 Pharmacokinetics
The delivery rate of dinoprostone from CERVIDIL in vivo is approximately 0.3 mg/per hour over a period of 12 hours. Dinoprostone is metabolized in the tissues of synthesis with the half-life estimated to be 2.5 to 5 minutes. The rate limiting step for inactivation is regulated by the enzyme 15-hydroxyprostaglandin dehydrogenase (PGDH). Any dinoprostone that escapes local inactivation is cleared to the extent of 95% on the first pass through the pulmonary circulation.
No correlation could be established between the release of dinoprostone from CERVIDIL and plasma concentrations of metabolite of dinoprostone (PGEm). The relative contributions of endogenously and exogenously released dinoprostone to the plasma levels of the metabolite PGEm is not known.
1 Indications and Usage
CERVIDIL is indicated for the initiation and/or continuation of cervical ripening in pregnant women at or near term in whom there is a medical or obstetrical indication for the induction of labor.
2.1 Dosage Instructions
Administer one CERVIDIL insert (10 mg) intravaginally for use up to 12 hours (approximately 0.3 mg of dinoprostone is released per hour) [see Dosage and Administration (2.2)].
Monitor uterine activity, fetal status, and the progression of cervical dilatation and effacement with the use of CERVIDIL. Remove CERVIDIL 12 hours after insertion with the onset of active labor, prior to an amniotomy, occurrence of uterine tachysystole, uterine hypersystole/hypertonicity, or fetal distress [see Warnings and Precautions (5.4)]. Remove CERVIDIL at least 30 minutes prior to administering an oxytocic agent [see Warnings and Precautions (5.4) and Drug Interactions (7)].
12.1 Mechanism of Action
Dinoprostone is found in low concentrations in most tissues of the body and functions as a local hormone. In pregnancy, dinoprostone is secreted continuously by the fetal membranes and placenta and plays an important role in the final events leading to the initiation of labor including cervical ripening. Dinoprostone stimulates the production of prostaglandin F2α (PGF2α), which sensitizes the myometrium to endogenous or exogenously administered oxytocin. Available evidence indicates that dinoprostone, in the concentrations found during the early part of labor, plays an important role in cervical ripening without affecting uterine contractions.
In most patients, local effects of CERVIDIL on the cervix include changes in the tissue consistency, dilatation and effacement. Some women experience systemic effects, including uterine tachysystole, and uterine hypersystole/hypertonicity, as a result dinoprostone or PGF2α mediated sensitization of the myometrium to oxytocin [see Warnings and Precautions (5.4)].
2.3 Removal Instructions
- To retrieve, locate the retrieval system and pull it gently until CERVIDIL is fully removed.
- Upon removal of CERVIDIL, it is essential to perform a visual inspection to ensure that the slab has been removed, as it will continue delivering the active ingredient.
5.1 for Hospital Use Only
CERVIDIL should be administered in a hospital setting with an obstetrical care facility.
5 Warnings and Precautions
- Disseminated Intravascular Coagulation: Assess for evolving fibrinolysis in the immediate post-partum period. (5.2)
- Amniotic Fluid Embolism Syndrome: Carefully monitor patients for clinical signs of hypotension, hypoxemia and respiratory failure, DIC, coma or seizures. Provide supportive care. (5.3)
- Uterine Tachysystole and Uterine Hypersystole/Hypertonicity: Monitor uterine activity; remove vaginal insert. (5.4)
- History of Glaucoma: Consider non-prostaglandin cervical ripening procedures. (5.5)
2 Dosage and Administration
- Administer a single vaginal insert (10 mg) for up to 12 hours of use (approximately 0.3 mg of dinoprostone is released per hour) (2.1)
- Remove upon onset of active labor or 12 hours after insertion. (2.1)
- Carefully monitor for uterine activity, fetal status and the progression of cervical dilatation and effacement. (2.1)
- CERVIDIL should be administered only by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities (2.2).
- See full prescribing information for complete preparation, administration, and removal instructions. (2.2, 2.3)
3 Dosage Forms and Strengths
Vaginal Insert: 10 mg of dinoprostone (release rate approximately 0.3 mg/hour up to 12 hours) in a hydrogel polymer. The insert is beige-colored, semi-opaque, thin, flat, and rectangular in shape with rounded corners, measuring 29 mm by 9.5 mm by 0.8 mm, contained within an off-white knitted polyester pouch retrieval system (see Figure 2).
| Figure 2: CERVIDIL Components |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of CERVIDIL or other dinoprostone products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Disseminated Intravascular Coagulation
Cardiovascular disorders: Myocardial Infarction in women with a history of myocardial infarction
Immune system disorders: Hypersensitivity
Nervous system disorders: Headache
Pregnancy, puerperium and perinatal conditions: Amniotic fluid embolism
Reproductive system: reports of uterine rupture have been reported in association with use of CERVIDIL. Some required a hysterectomy and others resulted in subsequent fetal or neonatal death. Uterine hypertonus
Vascular disorders: Hypotension
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In placebo-controlled trials of 658 pregnant women (320 CERVIDIL-treated women and 338 placebo-treated women), the following treatment related adverse reactions (see Table 1) occurred at an incidence greater than 2% (and greater than that reported in the placebo group) in the CERVIDIL group [see Clinical Studies (14)].
| Trials 1and 2 | ||
| CERVIDIL (N=320) | Placebo (N=338) | |
| Uterine tachysystole with fetal distress | 2.8% | 0.3% |
| Uterine tachysystole without fetal distress | 4.7% | 0% |
| Fetal distress without uterine tachysystole - | 3.8% | 1.2% |
| Trial 3 | ||
| CERVIDIL (N=102) | Placebo (N=104) | |
| Uterine tachysystole with fetal distress | 2.9% | 0% |
| Uterine tachysystole -without fetal distress | 2% | 0% |
| Fetal distress without uterine tachysystole | 2.9% | 1% |
Drug related fever, nausea, vomiting, diarrhea, and abdominal pain occurred in less than 1% of CERVIDIL-treated patients.
In Trial 3 (with the retrieval system) cases of tachysystole uterine hyperstimulation reversed within 2 to 13 minutes of removal of CERVIDIL. Tocolytics were required in one of the five cases.
5.3 Amniotic Fluid Embolism Syndrome
The use of dinoprostone-containing products, including CERVIDIL, can result in inadvertent disruption and subsequent embolization of antigenic tissue causing the development of Amniotic Fluid Embolism Syndrome, a rare and often fatal obstetric condition.
Carefully monitor patients for clinical signs of Amniotic Fluid Embolism Syndrome including hypotension, hypoxemia and respiratory failure, DIC, coma or seizures and provide supportive care as needed.
5.2 Disseminated Intravascular Coagulation
CERVIDIL should be used with caution in women at high risk of postpartum disseminated intravascular coagulation (DIC). Physiologic or pharmacologic induction of labor, including the use of CERVIDIL, is associated with an increased risk of DIC during the postpartum period. Women aged 30 years or older, those with complications during pregnancy and those with a gestational age over 40 weeks have an increased risk of DIC during the postpartum period. As soon as possible, assess for an evolving fibrinolysis in the immediate post-partum period. Therapy consisting of prompt removal of the source of procoagulant material, replacement of depleted clotting factors and, in some cases, anti-coagulation with heparin should be instituted promptly.
Principal Display Panel 10 Mg Pouch Carton
NDC 55566-2800-1
Cervidil®
DINOPROSTONE 10mg
VAGINAL INSERT
Contains: One Cervidil® Vaginal Insert containing
10 mg Dinoprostone in 241 mg hydrogel polymer
(cross-linked polyethylene oxide/urethane)
with polyester retrieval system.
Store in a freezer:
between -20°C and -10°C (-4°F and 14°F)
FERRING
PHARMACEUTICALS
2.2 Preparation and Administration Instructions
CERVIDIL should be administered only by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity and fertility studies have not been conducted with dinoprostone. No evidence of mutagenicity has been observed with dinoprostone in the Unscheduled DNA Synthesis Assay, the Micronucleus Test, or the bacteria reverse mutation (Ames) test.
5.4 Uterine Tachysystole and Uterine Hypersystole/hypertonicity
The use of CERVIDIL may cause uterine tachysystole with or without fetal heart rate changes (see Table 1). While using CERVIDIL, carefully monitor uterine activity, fetal status and the progression of cervical dilatation and effacement. Remove CERVIDIL with any evidence of uterine tachysystole, uterine hypersystole/hypertonicity, fetal distress, or if labor commences. CERVIDIL is contraindicated when prolonged contraction of the uterus is detrimental to fetal safety or uterine integrity, such as previous cesarean section or major uterine surgery, because of the risk of uterine rupture and obstetrical complications (e.g., need for hysterectomy and the occurrence of fetal or neonatal death). Prostaglandins, including CERVIDIL, may potentiate the effect of oxytocin [see Drug Interactions (7)]. Remove CERVIDIL at least 30 minutes before administration of an oxytocic agent is initiated and continue to carefully monitor uterine activity. Remove CERVIDIL prior to amniotomy or following rupture of membranes because the higher vaginal pH that occurs with rupture of membranes may result in higher release rate of dinoprostone.
Structured Label Content
Section 42229-5 (42229-5)
Preparation and Administration Instructions
- Keep CERVIDIL frozen until ready for use. Do not warm CERVIDIL prior to vaginal insertion.
- Tear open the individually-wrapped foil package containing CERVIDIL along the tear mark. Never open the package using scissors or other sharp objects because this may damage the knitted polyester retrieval system. Do not cut the retrieval system and do not use CERVIDIL unless its retrieval system is intact.
- Immediately after opening the package, insert CERVIDIL transversely, in the posterior fornix of the vagina (see Figure 1). If necessary, use a minimal amount of water-miscible lubricant to assist vaginal insertion. Do not permit excess contact or coating with the lubricant, as this could prevent release of dinoprostone from the vaginal insert. Insertion does not require sterile conditions.
Figure 1: Placement of CERVIDIL in the Posterior Vaginal Fornix - Tuck some of the excess retrieval system into the vagina to avoid movement of CERVIDIL away from the proper position; however, leave a small amount of the retrieval system outside the vagina to aid in retrieval.
- Instruct women to remain in a recumbent position during insertion of CERVIDIL and for 2 hours afterward. Women may be ambulatory 2 hours after insertion; however, ensure that the insert remains in place.
Section 44425-7 (44425-7)
Storage and Handling
Store in a freezer between -20°C and -10°C (-4°F and 14°F). CERVIDIL, enclosed in its aluminum/polyethylene pack, is stable when stored in a freezer for a period of three years. Vaginal inserts exposed to high humidity will absorb moisture from the air and thereby alter the release characteristics of dinoprostone.
5.5 Glaucoma
Prostaglandins, including CERVIDIL, can lead to raised intraocular pressure and constriction of pupils. Consider non-prostaglandin cervical ripening procedures in patients with Glaucoma.
11 Description (11 DESCRIPTION)
CERVIDIL (dinoprostone) vaginal insert contains dinoprostone, a prostaglandin analog. Each vaginal insert contains 10 mg of dinoprostone in 241 mg of a cross-linked polyethylene oxide/urethane polymer (hydrogel polymer) that is semi-opaque, beige colored, flat, rectangular in shape with rounded corners and measuring 29 mm by 9.5 mm by 0.8 mm. The vaginal insert is contained within a pouch of an off-white knitted polyester yarn retrieval system. When placed in a moist environment, the yarn absorbs water, swells, and releases the enclosed dinoprostone. The knitted polyester retrieval system has a long tape-like end that is designed to aid retrieval of CERVIDIL at the end of the dosing interval or earlier if clinically indicated. The finished product is a controlled-release formulation that has been found to release dinoprostone in vivo at a rate of approximately 0.3 mg per hour.
The chemical name for dinoprostone (known as prostaglandin E2 or PGE2) is 11α,15S-dihydroxy-9-oxo-prosta-5Z,13E-dien-1-oic acid and the structural formula is represented below:
The molecular formula is C20H32O5 and its molecular weight is 352.47. Dinoprostone occurs as a white to off-white crystalline powder. It has a melting point within the range of 65° to 69°C. Dinoprostone is soluble in ethanol and in 25% ethanol in water.
8.4 Pediatric Use
The safety and effectiveness of CERVIDIL have not been established in pregnant girls.
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness and safety of CERVIDIL for the induction of cervical ripening was evaluated in 658 pregnant women (320 CERVIDIL-treated women and 338 placebo-treated women) at or near term in three randomized, double-blind, placebo-controlled trials (Trials 1, 2, and 3). Efficacy outcomes included percentage with treatment success (defined as vaginal delivery within 12 hours, Bishop Score ≥ 6 in the 12-hour observation period, or ≥ 3 increase in the Bishop score in the 12-hour observation period), time to delivery, and time to onset of labor. Table 2 presents efficacy outcomes from Trials 1, 2, and 3.
| Primipara/Nullipara | Multipara | ||||
|---|---|---|---|---|---|
| Study # | CERVIDIL | Placebo | CERVIDIL | Placebo | P-Value |
|
Treatment Success Treatment success was defined as vaginal delivery within 12 hours, Bishop score ≥ 6 in the 12-hour observation period, or ≥ 3 increase in the Bishop score in the 12-hour observation period.
|
|||||
| Trial 1 (N=81) |
65% (n=26) |
28% (n=32) |
87% (n=16) |
29% (n=7) |
<0.001 |
| Trial 2 (N=371) |
68% (n=111) |
24% (n=123) |
77% (n=65) |
24% (n=72) |
<0.001 |
| Trial 3 (N=206) |
72% (n=60) |
48% (n=63) |
55% (n=42) |
41% (n=41) |
0.003 |
| Median Time to Delivery (hours) | |||||
| Trial 1 (N=81) |
25.7 (n=26) |
34.5 (n=32) |
12.3 (n=16) |
24.6 (n=7) |
0.001 |
| Trial 3 (N=206) |
25.5 (n=60) |
37.2 (n=63) |
20.8 (n=42) |
27.4 (n=41) |
<0.001 |
| Median Time to Onset of Labor (hours) | |||||
| Trial 1 (N=81) |
12 (n=26) |
19.2 (n=32) |
6.9 (n=16) |
18.3 (n=7) |
<0.001 |
4 Contraindications (4 CONTRAINDICATIONS)
CERVIDIL is contraindicated in patients with:
- Known hypersensitivity to prostaglandins [see Adverse Reactions (6.2)]
- Evidence or clinical suspicion of fetal distress where delivery is not imminent
- Unexplained vaginal bleeding in the current pregnancy
- Evidence or clinical suspicion of marked cephalopelvic disproportion
- Conditions for which induction of labor is contraindicated
- Conditions for which oxytocic drugs are contraindicated
- Previous cesarean section or other uterine surgery expected to affect uterine integrity (such as myomectomy)
- Conditions under which prolonged contraction of the uterus may be detrimental to fetal safety
- Concurrent use with intravenous oxytocic agents [see Warnings and Precautions (5.4) and Drug Interactions (7)]
- Six or more previous term pregnancies
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described, or described in greater detail, in other sections:
- Disseminated Intravascular Coagulation [see Warnings and Precautions (5.2)]
- Amniotic Fluid Embolism [see Warnings and Precautions (5.3)]
- Uterine Tachysytole and Uterine Hypersystole/Hypertonicity [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
7.1 Oxytocic Agents
CERVIDIL is contraindicated in patients receiving intravenous oxytocic agents because CERVIDIL may augment the activity of oxytocic agents. A dosing interval of at least 30 minutes is recommended for the sequential use of an oxytocic agent following the removal of CERVIDIL.
12.2 Pharmacodynamics
No specific pharmacodynamic studies were conducted with CERVIDIL.
12.3 Pharmacokinetics
The delivery rate of dinoprostone from CERVIDIL in vivo is approximately 0.3 mg/per hour over a period of 12 hours. Dinoprostone is metabolized in the tissues of synthesis with the half-life estimated to be 2.5 to 5 minutes. The rate limiting step for inactivation is regulated by the enzyme 15-hydroxyprostaglandin dehydrogenase (PGDH). Any dinoprostone that escapes local inactivation is cleared to the extent of 95% on the first pass through the pulmonary circulation.
No correlation could be established between the release of dinoprostone from CERVIDIL and plasma concentrations of metabolite of dinoprostone (PGEm). The relative contributions of endogenously and exogenously released dinoprostone to the plasma levels of the metabolite PGEm is not known.
1 Indications and Usage (1 INDICATIONS AND USAGE)
CERVIDIL is indicated for the initiation and/or continuation of cervical ripening in pregnant women at or near term in whom there is a medical or obstetrical indication for the induction of labor.
2.1 Dosage Instructions
Administer one CERVIDIL insert (10 mg) intravaginally for use up to 12 hours (approximately 0.3 mg of dinoprostone is released per hour) [see Dosage and Administration (2.2)].
Monitor uterine activity, fetal status, and the progression of cervical dilatation and effacement with the use of CERVIDIL. Remove CERVIDIL 12 hours after insertion with the onset of active labor, prior to an amniotomy, occurrence of uterine tachysystole, uterine hypersystole/hypertonicity, or fetal distress [see Warnings and Precautions (5.4)]. Remove CERVIDIL at least 30 minutes prior to administering an oxytocic agent [see Warnings and Precautions (5.4) and Drug Interactions (7)].
12.1 Mechanism of Action
Dinoprostone is found in low concentrations in most tissues of the body and functions as a local hormone. In pregnancy, dinoprostone is secreted continuously by the fetal membranes and placenta and plays an important role in the final events leading to the initiation of labor including cervical ripening. Dinoprostone stimulates the production of prostaglandin F2α (PGF2α), which sensitizes the myometrium to endogenous or exogenously administered oxytocin. Available evidence indicates that dinoprostone, in the concentrations found during the early part of labor, plays an important role in cervical ripening without affecting uterine contractions.
In most patients, local effects of CERVIDIL on the cervix include changes in the tissue consistency, dilatation and effacement. Some women experience systemic effects, including uterine tachysystole, and uterine hypersystole/hypertonicity, as a result dinoprostone or PGF2α mediated sensitization of the myometrium to oxytocin [see Warnings and Precautions (5.4)].
2.3 Removal Instructions
- To retrieve, locate the retrieval system and pull it gently until CERVIDIL is fully removed.
- Upon removal of CERVIDIL, it is essential to perform a visual inspection to ensure that the slab has been removed, as it will continue delivering the active ingredient.
5.1 for Hospital Use Only (5.1 For Hospital Use Only)
CERVIDIL should be administered in a hospital setting with an obstetrical care facility.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Disseminated Intravascular Coagulation: Assess for evolving fibrinolysis in the immediate post-partum period. (5.2)
- Amniotic Fluid Embolism Syndrome: Carefully monitor patients for clinical signs of hypotension, hypoxemia and respiratory failure, DIC, coma or seizures. Provide supportive care. (5.3)
- Uterine Tachysystole and Uterine Hypersystole/Hypertonicity: Monitor uterine activity; remove vaginal insert. (5.4)
- History of Glaucoma: Consider non-prostaglandin cervical ripening procedures. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer a single vaginal insert (10 mg) for up to 12 hours of use (approximately 0.3 mg of dinoprostone is released per hour) (2.1)
- Remove upon onset of active labor or 12 hours after insertion. (2.1)
- Carefully monitor for uterine activity, fetal status and the progression of cervical dilatation and effacement. (2.1)
- CERVIDIL should be administered only by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities (2.2).
- See full prescribing information for complete preparation, administration, and removal instructions. (2.2, 2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Vaginal Insert: 10 mg of dinoprostone (release rate approximately 0.3 mg/hour up to 12 hours) in a hydrogel polymer. The insert is beige-colored, semi-opaque, thin, flat, and rectangular in shape with rounded corners, measuring 29 mm by 9.5 mm by 0.8 mm, contained within an off-white knitted polyester pouch retrieval system (see Figure 2).
| Figure 2: CERVIDIL Components |
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of CERVIDIL or other dinoprostone products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Disseminated Intravascular Coagulation
Cardiovascular disorders: Myocardial Infarction in women with a history of myocardial infarction
Immune system disorders: Hypersensitivity
Nervous system disorders: Headache
Pregnancy, puerperium and perinatal conditions: Amniotic fluid embolism
Reproductive system: reports of uterine rupture have been reported in association with use of CERVIDIL. Some required a hysterectomy and others resulted in subsequent fetal or neonatal death. Uterine hypertonus
Vascular disorders: Hypotension
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In placebo-controlled trials of 658 pregnant women (320 CERVIDIL-treated women and 338 placebo-treated women), the following treatment related adverse reactions (see Table 1) occurred at an incidence greater than 2% (and greater than that reported in the placebo group) in the CERVIDIL group [see Clinical Studies (14)].
| Trials 1and 2 | ||
| CERVIDIL (N=320) | Placebo (N=338) | |
| Uterine tachysystole with fetal distress | 2.8% | 0.3% |
| Uterine tachysystole without fetal distress | 4.7% | 0% |
| Fetal distress without uterine tachysystole - | 3.8% | 1.2% |
| Trial 3 | ||
| CERVIDIL (N=102) | Placebo (N=104) | |
| Uterine tachysystole with fetal distress | 2.9% | 0% |
| Uterine tachysystole -without fetal distress | 2% | 0% |
| Fetal distress without uterine tachysystole | 2.9% | 1% |
Drug related fever, nausea, vomiting, diarrhea, and abdominal pain occurred in less than 1% of CERVIDIL-treated patients.
In Trial 3 (with the retrieval system) cases of tachysystole uterine hyperstimulation reversed within 2 to 13 minutes of removal of CERVIDIL. Tocolytics were required in one of the five cases.
5.3 Amniotic Fluid Embolism Syndrome
The use of dinoprostone-containing products, including CERVIDIL, can result in inadvertent disruption and subsequent embolization of antigenic tissue causing the development of Amniotic Fluid Embolism Syndrome, a rare and often fatal obstetric condition.
Carefully monitor patients for clinical signs of Amniotic Fluid Embolism Syndrome including hypotension, hypoxemia and respiratory failure, DIC, coma or seizures and provide supportive care as needed.
5.2 Disseminated Intravascular Coagulation
CERVIDIL should be used with caution in women at high risk of postpartum disseminated intravascular coagulation (DIC). Physiologic or pharmacologic induction of labor, including the use of CERVIDIL, is associated with an increased risk of DIC during the postpartum period. Women aged 30 years or older, those with complications during pregnancy and those with a gestational age over 40 weeks have an increased risk of DIC during the postpartum period. As soon as possible, assess for an evolving fibrinolysis in the immediate post-partum period. Therapy consisting of prompt removal of the source of procoagulant material, replacement of depleted clotting factors and, in some cases, anti-coagulation with heparin should be instituted promptly.
Principal Display Panel 10 Mg Pouch Carton (PRINCIPAL DISPLAY PANEL - 10 mg Pouch Carton)
NDC 55566-2800-1
Cervidil®
DINOPROSTONE 10mg
VAGINAL INSERT
Contains: One Cervidil® Vaginal Insert containing
10 mg Dinoprostone in 241 mg hydrogel polymer
(cross-linked polyethylene oxide/urethane)
with polyester retrieval system.
Store in a freezer:
between -20°C and -10°C (-4°F and 14°F)
FERRING
PHARMACEUTICALS
2.2 Preparation and Administration Instructions
CERVIDIL should be administered only by trained obstetrical personnel in a hospital setting with appropriate obstetrical care facilities.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity and fertility studies have not been conducted with dinoprostone. No evidence of mutagenicity has been observed with dinoprostone in the Unscheduled DNA Synthesis Assay, the Micronucleus Test, or the bacteria reverse mutation (Ames) test.
5.4 Uterine Tachysystole and Uterine Hypersystole/hypertonicity (5.4 Uterine Tachysystole and Uterine Hypersystole/Hypertonicity)
The use of CERVIDIL may cause uterine tachysystole with or without fetal heart rate changes (see Table 1). While using CERVIDIL, carefully monitor uterine activity, fetal status and the progression of cervical dilatation and effacement. Remove CERVIDIL with any evidence of uterine tachysystole, uterine hypersystole/hypertonicity, fetal distress, or if labor commences. CERVIDIL is contraindicated when prolonged contraction of the uterus is detrimental to fetal safety or uterine integrity, such as previous cesarean section or major uterine surgery, because of the risk of uterine rupture and obstetrical complications (e.g., need for hysterectomy and the occurrence of fetal or neonatal death). Prostaglandins, including CERVIDIL, may potentiate the effect of oxytocin [see Drug Interactions (7)]. Remove CERVIDIL at least 30 minutes before administration of an oxytocic agent is initiated and continue to carefully monitor uterine activity. Remove CERVIDIL prior to amniotomy or following rupture of membranes because the higher vaginal pH that occurs with rupture of membranes may result in higher release rate of dinoprostone.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:22.623414 · Updated: 2026-03-14T21:54:22.637255