These Highlights Do Not Include All The Information Needed To Use Isturisa®
f3a5ec24-63c3-4d83-b1c0-6c550fbe7ae2
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 04/2025
Indications and Usage
ISTURISA is indicated for the treatment of endogenous hypercortisolemia in adults with Cushing's syndrome for whom surgery is not an option or has not been curative.
Dosage and Administration
Correct hypokalemia and hypomagnesemia, and obtain baseline electrocardiogram prior to starting ISTURISA ( 2.1 , 5.2 , 5.3 ) Initiate dosage at 2 mg orally twice daily, with or without food ( 2.2 ) Titrate dosage by 1 mg to 2 mg twice daily, no more frequently than every 2 weeks based on rate of cortisol changes, individual tolerability and improvement in signs and symptoms ( 2.2 ) Maximum recommended dosage is 30 mg twice daily ( 2.2 ) See Full Prescribing Information for complete titration, laboratory, and dosage modification recommendations ( 2.1 , 2.2 , 2.3 ) Patients with Hepatic Impairment: Child-Pugh B: Recommended starting dose is 1 mg twice daily ( 2.5 , 8.7 ) Child-Pugh C : Recommended starting dose is 1 mg once daily in the evening ( 2.5 , 8.7 )
Warnings and Precautions
Hypocortisolism : Monitor patients closely for hypocortisolism and potentially life-threatening adrenal insufficiency. Dosage reduction or interruption may be necessary. After interruption or discontinuation of ISTURISA, cortisol suppression may persist and patients should be regularly monitored ( 5.1 ) QTc Prolongation : Perform electrocardiogram in all patients Use with caution in patients with risk factors for QTc prolongation ( 5.2 ) Elevations in Adrenal Hormone Precursors and Androgens: Monitor for hypokalemia, worsening of hypertension, edema, and hirsutism ( 5.3 )
Contraindications
None.
Adverse Reactions
Clinically significant adverse reactions that appear in other sections of the labeling include: Hypocortisolism [see Warnings and Precautions (5.1) ] QT Prolongation [see Warnings and Precautions (5.2) ] Elevations in Adrenal Hormone Precursors and Androgens [see Warnings and Precautions (5.3) ]
Drug Interactions
CYP3A4 Inhibitor: Reduce the dose of ISTURISA by half with concomitant use of a strong CYP3A4 inhibitor ( 7.1 ) CYP3A4 and CYP2B6 Inducers: An increase of ISTURISA dosage may be needed if ISTURISA is used concomitantly with strong CYP3A4 and CYP2B6 inducers. A reduction in ISTURISA dosage may be needed if strong CYP3A4 and CYP2B6 inducers are discontinued while using ISTURISA ( 7.1 )
Medication Information
Warnings and Precautions
Hypocortisolism : Monitor patients closely for hypocortisolism and potentially life-threatening adrenal insufficiency. Dosage reduction or interruption may be necessary. After interruption or discontinuation of ISTURISA, cortisol suppression may persist and patients should be regularly monitored ( 5.1 ) QTc Prolongation : Perform electrocardiogram in all patients Use with caution in patients with risk factors for QTc prolongation ( 5.2 ) Elevations in Adrenal Hormone Precursors and Androgens: Monitor for hypokalemia, worsening of hypertension, edema, and hirsutism ( 5.3 )
Indications and Usage
ISTURISA is indicated for the treatment of endogenous hypercortisolemia in adults with Cushing's syndrome for whom surgery is not an option or has not been curative.
Dosage and Administration
Correct hypokalemia and hypomagnesemia, and obtain baseline electrocardiogram prior to starting ISTURISA ( 2.1 , 5.2 , 5.3 ) Initiate dosage at 2 mg orally twice daily, with or without food ( 2.2 ) Titrate dosage by 1 mg to 2 mg twice daily, no more frequently than every 2 weeks based on rate of cortisol changes, individual tolerability and improvement in signs and symptoms ( 2.2 ) Maximum recommended dosage is 30 mg twice daily ( 2.2 ) See Full Prescribing Information for complete titration, laboratory, and dosage modification recommendations ( 2.1 , 2.2 , 2.3 ) Patients with Hepatic Impairment: Child-Pugh B: Recommended starting dose is 1 mg twice daily ( 2.5 , 8.7 ) Child-Pugh C : Recommended starting dose is 1 mg once daily in the evening ( 2.5 , 8.7 )
Contraindications
None.
Adverse Reactions
Clinically significant adverse reactions that appear in other sections of the labeling include: Hypocortisolism [see Warnings and Precautions (5.1) ] QT Prolongation [see Warnings and Precautions (5.2) ] Elevations in Adrenal Hormone Precursors and Androgens [see Warnings and Precautions (5.3) ]
Drug Interactions
CYP3A4 Inhibitor: Reduce the dose of ISTURISA by half with concomitant use of a strong CYP3A4 inhibitor ( 7.1 ) CYP3A4 and CYP2B6 Inducers: An increase of ISTURISA dosage may be needed if ISTURISA is used concomitantly with strong CYP3A4 and CYP2B6 inducers. A reduction in ISTURISA dosage may be needed if strong CYP3A4 and CYP2B6 inducers are discontinued while using ISTURISA ( 7.1 )
Description
Indications and Usage ( 1 ) 04/2025
Section 42229-5
Study 1
The adverse reactions that occurred with frequency higher than 10% during the core 48-week period are shown in Table 1.
| Adverse Reaction Type | (N = 137) % |
|---|---|
| Adrenal insufficiency Adrenal insufficiency includes glucocorticoid deficiency, adrenocortical insufficiency acute, steroid withdrawal syndrome, cortisol free urine decreased, cortisol decreased. One-third of the subjects with this event had low cortisol levels indicative of Adrenal Insufficiency. The majority of subjects had normal cortisol levels suggesting a cortisol withdrawal syndrome.
|
43.1 |
| Fatigue Fatigue includes lethargy, asthenia.
|
38.7 |
| Nausea | 37.2 |
| Headache Headache includes head discomfort.
|
30.7 |
| Edema Edema includes edema peripheral, generalized edema, localized edema.
|
21.2 |
| Nasopharyngitis | 19.7 |
| Vomiting | 19 |
| Arthralgia | 17.5 |
| Back pain | 15.3 |
| Rash Rash includes rash erythematous, rash generalized, rash maculopapular, rash papular.
|
15.3 |
| Diarrhea | 14.6 |
| Blood corticotrophin increased | 13.9 |
| Dizziness Dizziness includes dizziness postural.
|
13.9 |
| Abdominal pain Abdominal pain includes abdominal pain upper, abdominal discomfort
|
13.1 |
| Hypokalemia Hypokalemia includes blood potassium decreased.
|
12.4 |
| Myalgia | 12.4 |
| Decreased appetite | 11.7 |
| Hormone level abnormal | 11.7 |
| Hypotension Hypotension includes orthostatic hypotension, blood pressure decreased, blood pressure diastolic decreased, blood pressure systolic decreased.
|
11.7 |
| Urinary tract infection | 11.7 |
| Blood testosterone increased | 10.9 |
| Pyrexia | 10.9 |
| Anemia | 10.2 |
| Cough | 10.2 |
| Hypertension | 10.2 |
| Influenza | 10.2 |
Other notable adverse reactions which occurred with a frequency less than 10% were: hirsutism (9.5%), acne (8.8%), dyspepsia (8%), insomnia (8%), anxiety (7.3%), depression (7.3%), gastroenteritis (7.3%), malaise (6.6%), tachycardia (6.6%), alopecia (5.8%), transaminases increased (4.4%), electrocardiogram QT prolongation (3.6%), and syncope (1.5%).
Section 42230-3
| PATIENT INFORMATION ISTURISA® (is tur ee' sah) (osilodrostat) tablets, for oral use |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2025 |
|
What is the most important information I should know about ISTURISA?
|
|
|
|
| If you get symptoms of hypocortisolism while taking ISTURISA, your healthcare provider may change your dose or ask you to stop taking it. Heart problem or a heart rhythm problem, such as an irregular heartbeat which could be a sign of a heart problem called QT prolongation. Call your healthcare provider right away if you have irregular heartbeats. See "What are the possible side effects of ISTURISA?" for more information about side effects. |
|
|
What is ISTURISA?
ISTURISA is a prescription medicine that is used to treat elevated levels of cortisol in the blood (endogenous hypercortisolemia) in adults with Cushing's syndrome:
|
|
Before you take ISTURISA, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take medicines used to treat certain heart problems. Ask your healthcare provider if you are not sure whether your medicine is used to treat heart problems. |
|
How should I take ISTURISA?
|
|
|
What are the possible side effects of ISTURISA?
The most common side effects of ISTURISA include: |
|
|
|
| These are not all of the possible side effects of ISTURISA. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store ISTURISA?
|
|
|
General information about the safe and effective use of ISTURISA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ISTURISA for a condition for which it was not prescribed. Do not give ISTURISA to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for more information about ISTURISA that is written for healthcare professionals. |
|
|
What are the ingredients in ISTURISA? Active ingredient: osilodrostat phosphate Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, and magnesium stearate. Tablet film-coating: hypromellose, titanium dioxide, ferric oxide (yellow), ferric oxide (red) (1 mg only), polyethylene glycol 4000, and talc. |
|
| For more information, go to www.ISTURISA.com or call 1-888-575-8344. Manufactured for: Recordati Rare Diseases Inc., Bridgewater, NJ 08807 USA © Recordati |
Section 43683-2
| Indications and Usage (1) | 04/2025 |
Section 44425-7
Storage and Handling
Store at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted 15°C to 30°C (59°F to 86°F); protect from moisture.
14.1 Study 1
The safety and efficacy of ISTURISA was assessed in a 48-week, multicenter study (called the Core Period) that consisted of four study periods as follows:
- Period 1: 12-week, open-label, dose titration period
- Period 2: 12-week, open-label, maintenance treatment period
- Period 3: 8-week, double-blind, placebo-controlled, randomized withdrawal treatment period which provided the data for the primary efficacy endpoint
- Period 4: open-label treatment period of 14 to 24 weeks duration
The mean age at enrollment was 41 years; 77% of patients were female. There were 65% Caucasian, 28% Asian, 3% black, and 4% other race. Overall, 96% patients had received previous treatments for Cushing's disease prior to entering the study, of which 88% had undergone surgery. Persistence or recurrence of Cushing's disease was evidenced by the mean of three 24-hour UFC (mUFC) > 1.5× upper limit of normal (ULN). The mean mUFC (SD) at baseline was 1006 nmol/24 hr (1589) (365 mcg/24 hr), which corresponds to approximately 7 × ULN. The median mUFC at baseline was 476 nmol/24 hr (173 mcg/24 hr), which corresponds to approximately 3.5 × ULN.
14.2 Study 2
The safety and efficacy of ISTURISA was assessed in a 48-week, multicenter study that consisted of two core study periods as follows:
- Period 1: 12-week, double-blind, placebo-controlled, randomized treatment period with ISTURISA or placebo.
- Period 2: 36-week, open-label, treatment period with ISTURISA.
Study 2 enrolled 74 patients with Cushing's disease, of whom 73 were treated. The mean (range) age at enrollment was 41 (19 to 67) years; 84% were female. There was 67% Caucasian, 23% Asian, 3% black, and 7% other race. Overall, 96% of patients had persistent/recurring Cushing's disease prior to entering the study, of which 88% had undergone previous surgery and 62% of patients had prior medical treatment for Cushing's disease. Persistence or recurrence of Cushing's disease was evidenced by the mean of three 24-hour UFC (mUFC) > 1.3× upper limit of normal (ULN). The mean mUFC (SD) at baseline was 432 nmol/24hr (389) (157 mcg/24 hr), which corresponds to approximately 3.1 × ULN. The median mUFC at baseline was 340 nmol/24hr (123 mcg/24 hr), which corresponds to approximately 2.5 × ULN.
10 Overdosage
Overdosage may result in severe hypocortisolism. Signs and symptoms suggestive of hypocortisolism may include nausea, vomiting, fatigue, low blood pressure, abdominal pain, loss of appetite, dizziness, and syncope.
In case of suspected overdosage, ISTURISA should be temporarily discontinued, cortisol levels should be checked, and if necessary, corticosteroid supplementation should be initiated. Close surveillance may be necessary, including monitoring of the QT interval, blood pressure, glucose, fluid, and electrolyte until the patient's condition is stable.
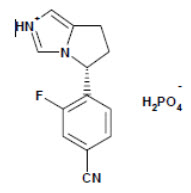
11 Description
ISTURISA (osilodrostat) is a cortisol synthesis inhibitor.
The chemical name of osilodrostat is 4-[(5R)-6,7-Dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl]-3-fluorobenzonitrile dihydrogen phosphate.
Molecular formula of osilodrostat salt (phosphate) form on anhydrous basis is: (C13H11FN3) (H2PO4). Relative molecular mass of osilodrostat phosphate salt form is 325.24 g/mol.
ISTURISA tablets for oral administration contains 1 mg or 5 mg of osilodrostat equivalent to 1.4 mg or 7.2 mg of osilodrostat phosphate respectively, and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, and magnesium stearate. The film coat is composed of hypromellose, titanium dioxide, ferric oxide (yellow), ferric oxide (red) (1 mg only), polyethylene glycol 4000, and talc.
2.6 Missed Dose
If a dose of ISTURISA is missed, the patient should take their next dose at the regularly scheduled time.
8.4 Pediatric Use
The safety and effectiveness of ISTURISA in pediatric patients have not been established.
8.5 Geriatric Use
Of the 167 patients in clinical trials with ISTURISA, 10 (6%) were 65 years and older. There were no patients above 75 years of age. Based on the available data on the use of ISTURISA in patients older than 65 years, no dosage adjustment is required [see Clinical Pharmacology (12.3)].
14 Clinical Studies
The safety and efficacy of ISTURISA was evaluated in 2 multicenter clinical studies, Study 1 (NCT02180217) and Study 2 (NCT02697734), in adults with persistent or recurrent Cushing's disease despite pituitary surgery or de novo patients for whom surgery was not indicated or who had refused surgery. Study 1 was a 48-week study that included an 8-week, double-blind, randomized withdrawal period at Weeks 26 to 34 in the study. After Week 48, patients who maintained clinical benefit on ISTURISA could continue in a long-term extension period until the last patient reached Week 72. Study 2 was a 48-week study in which patients were randomized 2:1 to ISTURISA or placebo and treated for 12 weeks followed by a 36-week open-label treatment. After completion of week 48, patients who maintained clinical benefit on ISTURISA could continue on ISTURISA in an optional 48-week extension period.
4 Contraindications
None.
5.1 Hypocortisolism
ISTURISA lowers cortisol levels and can lead to hypocortisolism and sometimes life-threatening adrenal insufficiency. Lowering of cortisol can cause nausea, vomiting, fatigue, abdominal pain, loss of appetite, dizziness. Significant lowering of serum cortisol may result in hypotension, abnormal electrolyte levels, and hypoglycemia [see Adverse Reactions (6)].
Hypocortisolism can occur at any time during ISTURISA treatment. Evaluate patients for precipitating causes of hypocortisolism (infection, physical stress, etc.). Monitor 24-hour urine free cortisol, serum or plasma cortisol, and patient's signs and symptoms periodically during ISTURISA treatment.
Decrease or temporarily discontinue ISTURISA if urine free cortisol levels fall below the target range, there is a rapid decrease in cortisol levels, and/or patients report symptoms of hypocortisolism. Stop ISTURISA and administer exogenous glucocorticoid replacement therapy if serum or plasma cortisol levels are below target range and patients have symptoms of adrenal insufficiency. After interruption or discontinuation of ISTURISA, cortisol suppression may persist beyond the 4 hour half-life. Monitor patients regularly and re-initiate ISTURISA at a lower dose when urine free cortisol, serum or plasma cortisol levels are within target range, and/or patient symptoms have resolved.
Educate patients on the symptoms associated with hypocortisolism and advise them to contact a healthcare provider if they occur.
6 Adverse Reactions
Clinically significant adverse reactions that appear in other sections of the labeling include:
- Hypocortisolism [see Warnings and Precautions (5.1)]
- QT Prolongation [see Warnings and Precautions (5.2)]
- Elevations in Adrenal Hormone Precursors and Androgens [see Warnings and Precautions (5.3)]
7 Drug Interactions
- CYP3A4 Inhibitor: Reduce the dose of ISTURISA by half with concomitant use of a strong CYP3A4 inhibitor (7.1)
- CYP3A4 and CYP2B6 Inducers: An increase of ISTURISA dosage may be needed if ISTURISA is used concomitantly with strong CYP3A4 and CYP2B6 inducers. A reduction in ISTURISA dosage may be needed if strong CYP3A4 and CYP2B6 inducers are discontinued while using ISTURISA (7.1)
5.2 Qtc Prolongation
ISTURISA is associated with a dose-dependent QT interval prolongation (maximum mean estimated QTcF increase of up to 5.3 ms at 30 mg), which may cause cardiac arrhythmias [see Adverse Reactions (6) , Clinical Pharmacology (12.2) ].
Perform an ECG to obtain a baseline QTc interval measurement prior to initiating therapy with ISTURISA and monitor for an effect on the QTc interval thereafter. Correct hypokalemia and/or hypomagnesemia prior to ISTURISA initiation and monitor periodically during treatment with ISTURISA. Correct electrolyte abnormalities if indicated. Consider temporary discontinuation of ISTURISA in the case of an increase in QTc interval > 480 ms.
Use caution in patients with risk factors for QT prolongation, (such as congenital long QT syndrome, congestive heart failure, bradyarrhythmias, uncorrected electrolyte abnormalities, and concomitant medications known to prolong the QT interval) and consider more frequent ECG monitoring.
8.6 Renal Impairment
No dosage adjustment of ISTURISA in patients with impaired renal function is required [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)]. In patients with moderate to severe renal impairment, UFC levels should be interpreted with caution due to reduced UFC excretion.
12.2 Pharmacodynamics
A dose dependent increase was observed in 11-deoxycortisol, the cortisol precursor, and ACTH levels in patients with Cushing's disease.
8.7 Hepatic Impairment
Dosage adjustment is not required in patients with mild hepatic impairment (Child-Pugh A) but is required for patients with moderately impaired hepatic function (Child-Pugh B) and for patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)]. More frequent monitoring of adrenal function may be required during dose titration in all patients with hepatic impairment.
1 Indications and Usage
ISTURISA is indicated for the treatment of endogenous hypercortisolemia in adults with Cushing's syndrome for whom surgery is not an option or has not been curative.
12.1 Mechanism of Action
Osilodrostat is a cortisol synthesis inhibitor. It inhibits 11beta-hydroxylase (CYP11B1), the enzyme responsible for the final step of cortisol biosynthesis in the adrenal gland. In a Chinese hamster lung cell line V79-4 that overexpresses human CYP11B1, adrenodoxin and adrenodoxin reductase, osilodrostat inhibited the activity of human CYP11B1 dose-dependently with IC50 values of 2.5 ± 0.1 nM (n = 4).
5 Warnings and Precautions
- Hypocortisolism: Monitor patients closely for hypocortisolism and potentially life-threatening adrenal insufficiency. Dosage reduction or interruption may be necessary. After interruption or discontinuation of ISTURISA, cortisol suppression may persist and patients should be regularly monitored (5.1)
- QTc Prolongation: Perform electrocardiogram in all patients Use with caution in patients with risk factors for QTc prolongation (5.2)
- Elevations in Adrenal Hormone Precursors and Androgens: Monitor for hypokalemia, worsening of hypertension, edema, and hirsutism (5.3)
2 Dosage and Administration
- Correct hypokalemia and hypomagnesemia, and obtain baseline electrocardiogram prior to starting ISTURISA (2.1, 5.2, 5.3)
- Initiate dosage at 2 mg orally twice daily, with or without food (2.2)
- Titrate dosage by 1 mg to 2 mg twice daily, no more frequently than every 2 weeks based on rate of cortisol changes, individual tolerability and improvement in signs and symptoms (2.2)
- Maximum recommended dosage is 30 mg twice daily (2.2)
- See Full Prescribing Information for complete titration, laboratory, and dosage modification recommendations (2.1, 2.2, 2.3)
- Patients with Hepatic Impairment:
3 Dosage Forms and Strengths
ISTURISA is available as:
- 1 mg tablets: Pale yellow, unscored, round, biconvex with beveled edge tablet, debossed "1" on one side.
- 5 mg tablets: Yellow, unscored, round, biconvex with beveled edge tablet, debossed "5" on one side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of ISTURISA. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Neutropenia associated with fever and infection
8 Use in Specific Populations
- Lactation: Breastfeeding is not recommended during treatment with ISTURISA and for at least one week after treatment (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ISTURISA was evaluated in two clinical trials in adults with Cushings disease. Study 1 (NCT02180217) was a 4-period, multicenter study with a 12-week open-label titration period, 12-week open-label maintenance period, 8-week double-blind, placebo-controlled period, and 14 to 24-week open label treatment period in 137 patients with Cushing's disease. Study 2 (NCT02697734) was a 2-period, multicenter study with a 12-week randomized, double-blind, placebo-controlled period and a 36-week open-label treatment period in 74 patients with Cushing's disease [see Clinical Studies (14)].
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Patient Information).
7.1 Effect of Other Drugs On Isturisa
The effect of other drugs on ISTURISA can be found in Table 3.
| CYP3A4 Inhibitors | |
|---|---|
| Clinical Impact: | Concomitant use of ISTURISA with a strong CYP3A4 inhibitor (e.g., itraconazole, clarithromycin) may cause an increase in osilodrostat concentration and may increase the risk of ISTURISA-related adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | Reduce the dose of ISTURISA by half with concomitant use of a strong CYP3A4 inhibitor. |
| CYP3A4 and CYP2B6 Inducers | |
| Clinical Impact: | Concomitant use of ISTURISA with strong CYP3A4 and/or CYP2B6 inducers (e.g., carbamazepine, rifampin, phenobarbital) may cause a decrease in osilodrostat concentration and may reduce the efficacy of ISTURISA [see Clinical Pharmacology (12.3)]. Discontinuation of strong CYP3A4 and/or CYP2B6 inducers while using ISTURISA may cause an increase in osilodrostat concentration and may increase the risk of ISTURISA-related adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | During concomitant use of ISTURISA with strong CYP3A4 and CYP2B6 inducers, monitor cortisol concentration and patient's signs and symptoms. An increase in ISTURISA dosage may be needed. Upon discontinuation of strong CYP3A4 and CYP2B6 inducers during ISTURISA treatment, monitor cortisol concentration and patient's signs and symptoms. A reduction in ISTURISA dosage may be needed. |
7.2 Effect of Isturisa On Other Drugs
ISTURISA should be used with caution when coadministered with CYP1A2 and CYP2C19 substrates with a narrow therapeutic index, such as theophylline, tizanidine, and S-mephenytoin [see Clinical Pharmacology (12.3)].
2.3 Dosage Interruptions and Modifications
- Decrease or temporarily discontinue ISTURISA if urine free cortisol levels fall below the target range, there is a rapid decrease in cortisol levels, and/or patients report symptoms of hypocortisolism. If necessary, glucocorticoid replacement therapy should be initiated.
- Stop ISTURISA and administer exogenous glucocorticoid replacement therapy if serum or plasma cortisol levels are below target range and patients have symptoms of adrenal insufficiency [see Warnings and Precautions (5.1)].
- If treatment is interrupted, re-initiate ISTURISA at a lower dose when cortisol levels are within target ranges and patient symptoms have been resolved.
2.2 Recommended Dosage, Titration, and Monitoring
- Initiate dosing at 2 mg orally twice daily, with or without food.
- Initially, titrate the dosage by 1 mg to 2 mg twice daily, no more frequently than every 2 weeks based on the rate of cortisol changes, individual tolerability and improvement in signs and symptoms of Cushing's syndrome. If a patient tolerates ISTURISA dosage of 10 mg twice daily and continues to have elevated 24-hour urine free cortisol (UFC) levels above upper normal limit, the dosage can be titrated further by 5 mg twice daily every 2 weeks. Monitor cortisol levels from at least two 24-hour urine free cortisol collections every 1 to 2 weeks until adequate clinical response is maintained.
- The maintenance dosage of ISTURISA is individualized and determined by titration based on cortisol levels and patient's signs and symptoms.
- The maintenance dosage varied between 2 mg and 7 mg twice daily in clinical trials. The maximum recommended maintenance dosage of ISTURISA is 30 mg twice daily [see Clinical Studies (14)].
- Once the maintenance dosage is achieved, monitor cortisol levels at least every 1 to 2 months or as indicated.
Principal Display Panel 1 Mg Blister Pack Carton
NDC 55292-330-60
Isturisa® 1 mg
(osilodrostat)
tablets
Rx only
Each tablet contains 1 mg osilodrostat (as osilodrostat phosphate).
60 film-coated tablets
RECORDATI
RARE DISEASES
Principal Display Panel 5 Mg Blister Pack Carton
NDC 55292-331-60
Isturisa® 5 mg
(osilodrostat)
tablets
Rx only
Each tablet contains 5 mg osilodrostat (as osilodrostat phosphate).
60 film-coated tablets
RECORDATI
RARE DISEASES
2.1 Laboratory Testing Prior to Isturisa Initiation
- Correct hypokalemia and hypomagnesemia prior to starting ISTURISA [see Warnings and Precautions (5.2, 5.3)].
- Obtain baseline electrocardiogram (ECG). Repeat ECG within one week after treatment initiation, and as clinically indicated thereafter [see Warnings and Precautions (5.2)].
Principal Display Panel 10 Mg Blister Pack Carton
NDC 55292-322-60
Isturisa® 10 mg
(osilodrostat)
tablets
Rx only
Each tablet contains 10 mg osilodrostat (as osilodrostat phosphate).
60 film-coated tablets
RECORDATI
RARE DISEASES
5.3 Elevations in Adrenal Hormone Precursors and Androgens
ISTURISA blocks cortisol synthesis and may increase circulating levels of cortisol and aldosterone precursors (11-deoxy cortisol and 11-deoxycorticosterone) and androgens.
Elevated 11-deoxycorticosterone levels may activate mineralocorticoid receptors and cause hypokalemia, edema and hypertension [see Adverse Reactions (6)]. Hypokalemia should be corrected prior to initiating ISTURISA. Monitor patients treated with ISTURISA for hypokalemia, worsening of hypertension and edema. ISTURISA-induced hypokalemia should be treated with intravenous or oral potassium supplementation based on event severity. If hypokalemia persists despite potassium supplementation, consider adding mineralocorticoid antagonists. ISTURISA dose reduction or discontinuation may be necessary.
Accumulation of androgens may lead to hirsutism, hypertrichosis and acne (in females). Inform patients of the symptoms associated with hyperandrogenism and advise them to contact a healthcare provider if they occur.
2.4 Recommended Dosage and Monitoring in Patients With Renal Impairment
- No dose adjustment is required for patients with renal impairment. Use caution in interpreting urine free cortisol levels in patients with moderate to severe renal impairment, due to reduced urine free cortisol excretion [see Clinical Pharmacology (12.3)].
2.5 Recommended Dosage and Monitoring in Patients With Hepatic Impairment
- For patients with moderate hepatic impairment (Child-Pugh B), the recommended starting dose is 1 mg twice daily. For patients with severe hepatic impairment (Child-Pugh C), the recommended starting dose is 1 mg once daily in the evening.
- No dose adjustment is required for patients with mild hepatic impairment (Child-Pugh A).
- More frequent monitoring of adrenal function may be required during dose titration in all patients with hepatic impairment [see Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Study 1
The adverse reactions that occurred with frequency higher than 10% during the core 48-week period are shown in Table 1.
| Adverse Reaction Type | (N = 137) % |
|---|---|
| Adrenal insufficiency Adrenal insufficiency includes glucocorticoid deficiency, adrenocortical insufficiency acute, steroid withdrawal syndrome, cortisol free urine decreased, cortisol decreased. One-third of the subjects with this event had low cortisol levels indicative of Adrenal Insufficiency. The majority of subjects had normal cortisol levels suggesting a cortisol withdrawal syndrome.
|
43.1 |
| Fatigue Fatigue includes lethargy, asthenia.
|
38.7 |
| Nausea | 37.2 |
| Headache Headache includes head discomfort.
|
30.7 |
| Edema Edema includes edema peripheral, generalized edema, localized edema.
|
21.2 |
| Nasopharyngitis | 19.7 |
| Vomiting | 19 |
| Arthralgia | 17.5 |
| Back pain | 15.3 |
| Rash Rash includes rash erythematous, rash generalized, rash maculopapular, rash papular.
|
15.3 |
| Diarrhea | 14.6 |
| Blood corticotrophin increased | 13.9 |
| Dizziness Dizziness includes dizziness postural.
|
13.9 |
| Abdominal pain Abdominal pain includes abdominal pain upper, abdominal discomfort
|
13.1 |
| Hypokalemia Hypokalemia includes blood potassium decreased.
|
12.4 |
| Myalgia | 12.4 |
| Decreased appetite | 11.7 |
| Hormone level abnormal | 11.7 |
| Hypotension Hypotension includes orthostatic hypotension, blood pressure decreased, blood pressure diastolic decreased, blood pressure systolic decreased.
|
11.7 |
| Urinary tract infection | 11.7 |
| Blood testosterone increased | 10.9 |
| Pyrexia | 10.9 |
| Anemia | 10.2 |
| Cough | 10.2 |
| Hypertension | 10.2 |
| Influenza | 10.2 |
Other notable adverse reactions which occurred with a frequency less than 10% were: hirsutism (9.5%), acne (8.8%), dyspepsia (8%), insomnia (8%), anxiety (7.3%), depression (7.3%), gastroenteritis (7.3%), malaise (6.6%), tachycardia (6.6%), alopecia (5.8%), transaminases increased (4.4%), electrocardiogram QT prolongation (3.6%), and syncope (1.5%).
Section 42230-3 (42230-3)
| PATIENT INFORMATION ISTURISA® (is tur ee' sah) (osilodrostat) tablets, for oral use |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2025 |
|
What is the most important information I should know about ISTURISA?
|
|
|
|
| If you get symptoms of hypocortisolism while taking ISTURISA, your healthcare provider may change your dose or ask you to stop taking it. Heart problem or a heart rhythm problem, such as an irregular heartbeat which could be a sign of a heart problem called QT prolongation. Call your healthcare provider right away if you have irregular heartbeats. See "What are the possible side effects of ISTURISA?" for more information about side effects. |
|
|
What is ISTURISA?
ISTURISA is a prescription medicine that is used to treat elevated levels of cortisol in the blood (endogenous hypercortisolemia) in adults with Cushing's syndrome:
|
|
Before you take ISTURISA, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take medicines used to treat certain heart problems. Ask your healthcare provider if you are not sure whether your medicine is used to treat heart problems. |
|
How should I take ISTURISA?
|
|
|
What are the possible side effects of ISTURISA?
The most common side effects of ISTURISA include: |
|
|
|
| These are not all of the possible side effects of ISTURISA. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store ISTURISA?
|
|
|
General information about the safe and effective use of ISTURISA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ISTURISA for a condition for which it was not prescribed. Do not give ISTURISA to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for more information about ISTURISA that is written for healthcare professionals. |
|
|
What are the ingredients in ISTURISA? Active ingredient: osilodrostat phosphate Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, and magnesium stearate. Tablet film-coating: hypromellose, titanium dioxide, ferric oxide (yellow), ferric oxide (red) (1 mg only), polyethylene glycol 4000, and talc. |
|
| For more information, go to www.ISTURISA.com or call 1-888-575-8344. Manufactured for: Recordati Rare Diseases Inc., Bridgewater, NJ 08807 USA © Recordati |
Section 43683-2 (43683-2)
| Indications and Usage (1) | 04/2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted 15°C to 30°C (59°F to 86°F); protect from moisture.
14.1 Study 1
The safety and efficacy of ISTURISA was assessed in a 48-week, multicenter study (called the Core Period) that consisted of four study periods as follows:
- Period 1: 12-week, open-label, dose titration period
- Period 2: 12-week, open-label, maintenance treatment period
- Period 3: 8-week, double-blind, placebo-controlled, randomized withdrawal treatment period which provided the data for the primary efficacy endpoint
- Period 4: open-label treatment period of 14 to 24 weeks duration
The mean age at enrollment was 41 years; 77% of patients were female. There were 65% Caucasian, 28% Asian, 3% black, and 4% other race. Overall, 96% patients had received previous treatments for Cushing's disease prior to entering the study, of which 88% had undergone surgery. Persistence or recurrence of Cushing's disease was evidenced by the mean of three 24-hour UFC (mUFC) > 1.5× upper limit of normal (ULN). The mean mUFC (SD) at baseline was 1006 nmol/24 hr (1589) (365 mcg/24 hr), which corresponds to approximately 7 × ULN. The median mUFC at baseline was 476 nmol/24 hr (173 mcg/24 hr), which corresponds to approximately 3.5 × ULN.
14.2 Study 2
The safety and efficacy of ISTURISA was assessed in a 48-week, multicenter study that consisted of two core study periods as follows:
- Period 1: 12-week, double-blind, placebo-controlled, randomized treatment period with ISTURISA or placebo.
- Period 2: 36-week, open-label, treatment period with ISTURISA.
Study 2 enrolled 74 patients with Cushing's disease, of whom 73 were treated. The mean (range) age at enrollment was 41 (19 to 67) years; 84% were female. There was 67% Caucasian, 23% Asian, 3% black, and 7% other race. Overall, 96% of patients had persistent/recurring Cushing's disease prior to entering the study, of which 88% had undergone previous surgery and 62% of patients had prior medical treatment for Cushing's disease. Persistence or recurrence of Cushing's disease was evidenced by the mean of three 24-hour UFC (mUFC) > 1.3× upper limit of normal (ULN). The mean mUFC (SD) at baseline was 432 nmol/24hr (389) (157 mcg/24 hr), which corresponds to approximately 3.1 × ULN. The median mUFC at baseline was 340 nmol/24hr (123 mcg/24 hr), which corresponds to approximately 2.5 × ULN.
10 Overdosage (10 OVERDOSAGE)
Overdosage may result in severe hypocortisolism. Signs and symptoms suggestive of hypocortisolism may include nausea, vomiting, fatigue, low blood pressure, abdominal pain, loss of appetite, dizziness, and syncope.
In case of suspected overdosage, ISTURISA should be temporarily discontinued, cortisol levels should be checked, and if necessary, corticosteroid supplementation should be initiated. Close surveillance may be necessary, including monitoring of the QT interval, blood pressure, glucose, fluid, and electrolyte until the patient's condition is stable.
11 Description (11 DESCRIPTION)
ISTURISA (osilodrostat) is a cortisol synthesis inhibitor.
The chemical name of osilodrostat is 4-[(5R)-6,7-Dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl]-3-fluorobenzonitrile dihydrogen phosphate.
Molecular formula of osilodrostat salt (phosphate) form on anhydrous basis is: (C13H11FN3) (H2PO4). Relative molecular mass of osilodrostat phosphate salt form is 325.24 g/mol.
ISTURISA tablets for oral administration contains 1 mg or 5 mg of osilodrostat equivalent to 1.4 mg or 7.2 mg of osilodrostat phosphate respectively, and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, and magnesium stearate. The film coat is composed of hypromellose, titanium dioxide, ferric oxide (yellow), ferric oxide (red) (1 mg only), polyethylene glycol 4000, and talc.
2.6 Missed Dose
If a dose of ISTURISA is missed, the patient should take their next dose at the regularly scheduled time.
8.4 Pediatric Use
The safety and effectiveness of ISTURISA in pediatric patients have not been established.
8.5 Geriatric Use
Of the 167 patients in clinical trials with ISTURISA, 10 (6%) were 65 years and older. There were no patients above 75 years of age. Based on the available data on the use of ISTURISA in patients older than 65 years, no dosage adjustment is required [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of ISTURISA was evaluated in 2 multicenter clinical studies, Study 1 (NCT02180217) and Study 2 (NCT02697734), in adults with persistent or recurrent Cushing's disease despite pituitary surgery or de novo patients for whom surgery was not indicated or who had refused surgery. Study 1 was a 48-week study that included an 8-week, double-blind, randomized withdrawal period at Weeks 26 to 34 in the study. After Week 48, patients who maintained clinical benefit on ISTURISA could continue in a long-term extension period until the last patient reached Week 72. Study 2 was a 48-week study in which patients were randomized 2:1 to ISTURISA or placebo and treated for 12 weeks followed by a 36-week open-label treatment. After completion of week 48, patients who maintained clinical benefit on ISTURISA could continue on ISTURISA in an optional 48-week extension period.
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.1 Hypocortisolism
ISTURISA lowers cortisol levels and can lead to hypocortisolism and sometimes life-threatening adrenal insufficiency. Lowering of cortisol can cause nausea, vomiting, fatigue, abdominal pain, loss of appetite, dizziness. Significant lowering of serum cortisol may result in hypotension, abnormal electrolyte levels, and hypoglycemia [see Adverse Reactions (6)].
Hypocortisolism can occur at any time during ISTURISA treatment. Evaluate patients for precipitating causes of hypocortisolism (infection, physical stress, etc.). Monitor 24-hour urine free cortisol, serum or plasma cortisol, and patient's signs and symptoms periodically during ISTURISA treatment.
Decrease or temporarily discontinue ISTURISA if urine free cortisol levels fall below the target range, there is a rapid decrease in cortisol levels, and/or patients report symptoms of hypocortisolism. Stop ISTURISA and administer exogenous glucocorticoid replacement therapy if serum or plasma cortisol levels are below target range and patients have symptoms of adrenal insufficiency. After interruption or discontinuation of ISTURISA, cortisol suppression may persist beyond the 4 hour half-life. Monitor patients regularly and re-initiate ISTURISA at a lower dose when urine free cortisol, serum or plasma cortisol levels are within target range, and/or patient symptoms have resolved.
Educate patients on the symptoms associated with hypocortisolism and advise them to contact a healthcare provider if they occur.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Clinically significant adverse reactions that appear in other sections of the labeling include:
- Hypocortisolism [see Warnings and Precautions (5.1)]
- QT Prolongation [see Warnings and Precautions (5.2)]
- Elevations in Adrenal Hormone Precursors and Androgens [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- CYP3A4 Inhibitor: Reduce the dose of ISTURISA by half with concomitant use of a strong CYP3A4 inhibitor (7.1)
- CYP3A4 and CYP2B6 Inducers: An increase of ISTURISA dosage may be needed if ISTURISA is used concomitantly with strong CYP3A4 and CYP2B6 inducers. A reduction in ISTURISA dosage may be needed if strong CYP3A4 and CYP2B6 inducers are discontinued while using ISTURISA (7.1)
5.2 Qtc Prolongation (5.2 QTc Prolongation)
ISTURISA is associated with a dose-dependent QT interval prolongation (maximum mean estimated QTcF increase of up to 5.3 ms at 30 mg), which may cause cardiac arrhythmias [see Adverse Reactions (6) , Clinical Pharmacology (12.2) ].
Perform an ECG to obtain a baseline QTc interval measurement prior to initiating therapy with ISTURISA and monitor for an effect on the QTc interval thereafter. Correct hypokalemia and/or hypomagnesemia prior to ISTURISA initiation and monitor periodically during treatment with ISTURISA. Correct electrolyte abnormalities if indicated. Consider temporary discontinuation of ISTURISA in the case of an increase in QTc interval > 480 ms.
Use caution in patients with risk factors for QT prolongation, (such as congenital long QT syndrome, congestive heart failure, bradyarrhythmias, uncorrected electrolyte abnormalities, and concomitant medications known to prolong the QT interval) and consider more frequent ECG monitoring.
8.6 Renal Impairment
No dosage adjustment of ISTURISA in patients with impaired renal function is required [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)]. In patients with moderate to severe renal impairment, UFC levels should be interpreted with caution due to reduced UFC excretion.
12.2 Pharmacodynamics
A dose dependent increase was observed in 11-deoxycortisol, the cortisol precursor, and ACTH levels in patients with Cushing's disease.
8.7 Hepatic Impairment
Dosage adjustment is not required in patients with mild hepatic impairment (Child-Pugh A) but is required for patients with moderately impaired hepatic function (Child-Pugh B) and for patients with severe hepatic impairment (Child-Pugh C) [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)]. More frequent monitoring of adrenal function may be required during dose titration in all patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ISTURISA is indicated for the treatment of endogenous hypercortisolemia in adults with Cushing's syndrome for whom surgery is not an option or has not been curative.
12.1 Mechanism of Action
Osilodrostat is a cortisol synthesis inhibitor. It inhibits 11beta-hydroxylase (CYP11B1), the enzyme responsible for the final step of cortisol biosynthesis in the adrenal gland. In a Chinese hamster lung cell line V79-4 that overexpresses human CYP11B1, adrenodoxin and adrenodoxin reductase, osilodrostat inhibited the activity of human CYP11B1 dose-dependently with IC50 values of 2.5 ± 0.1 nM (n = 4).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypocortisolism: Monitor patients closely for hypocortisolism and potentially life-threatening adrenal insufficiency. Dosage reduction or interruption may be necessary. After interruption or discontinuation of ISTURISA, cortisol suppression may persist and patients should be regularly monitored (5.1)
- QTc Prolongation: Perform electrocardiogram in all patients Use with caution in patients with risk factors for QTc prolongation (5.2)
- Elevations in Adrenal Hormone Precursors and Androgens: Monitor for hypokalemia, worsening of hypertension, edema, and hirsutism (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Correct hypokalemia and hypomagnesemia, and obtain baseline electrocardiogram prior to starting ISTURISA (2.1, 5.2, 5.3)
- Initiate dosage at 2 mg orally twice daily, with or without food (2.2)
- Titrate dosage by 1 mg to 2 mg twice daily, no more frequently than every 2 weeks based on rate of cortisol changes, individual tolerability and improvement in signs and symptoms (2.2)
- Maximum recommended dosage is 30 mg twice daily (2.2)
- See Full Prescribing Information for complete titration, laboratory, and dosage modification recommendations (2.1, 2.2, 2.3)
- Patients with Hepatic Impairment:
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
ISTURISA is available as:
- 1 mg tablets: Pale yellow, unscored, round, biconvex with beveled edge tablet, debossed "1" on one side.
- 5 mg tablets: Yellow, unscored, round, biconvex with beveled edge tablet, debossed "5" on one side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of ISTURISA. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Neutropenia associated with fever and infection
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Breastfeeding is not recommended during treatment with ISTURISA and for at least one week after treatment (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ISTURISA was evaluated in two clinical trials in adults with Cushings disease. Study 1 (NCT02180217) was a 4-period, multicenter study with a 12-week open-label titration period, 12-week open-label maintenance period, 8-week double-blind, placebo-controlled period, and 14 to 24-week open label treatment period in 137 patients with Cushing's disease. Study 2 (NCT02697734) was a 2-period, multicenter study with a 12-week randomized, double-blind, placebo-controlled period and a 36-week open-label treatment period in 74 patients with Cushing's disease [see Clinical Studies (14)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Patient Information).
7.1 Effect of Other Drugs On Isturisa (7.1 Effect of Other Drugs on ISTURISA)
The effect of other drugs on ISTURISA can be found in Table 3.
| CYP3A4 Inhibitors | |
|---|---|
| Clinical Impact: | Concomitant use of ISTURISA with a strong CYP3A4 inhibitor (e.g., itraconazole, clarithromycin) may cause an increase in osilodrostat concentration and may increase the risk of ISTURISA-related adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | Reduce the dose of ISTURISA by half with concomitant use of a strong CYP3A4 inhibitor. |
| CYP3A4 and CYP2B6 Inducers | |
| Clinical Impact: | Concomitant use of ISTURISA with strong CYP3A4 and/or CYP2B6 inducers (e.g., carbamazepine, rifampin, phenobarbital) may cause a decrease in osilodrostat concentration and may reduce the efficacy of ISTURISA [see Clinical Pharmacology (12.3)]. Discontinuation of strong CYP3A4 and/or CYP2B6 inducers while using ISTURISA may cause an increase in osilodrostat concentration and may increase the risk of ISTURISA-related adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | During concomitant use of ISTURISA with strong CYP3A4 and CYP2B6 inducers, monitor cortisol concentration and patient's signs and symptoms. An increase in ISTURISA dosage may be needed. Upon discontinuation of strong CYP3A4 and CYP2B6 inducers during ISTURISA treatment, monitor cortisol concentration and patient's signs and symptoms. A reduction in ISTURISA dosage may be needed. |
7.2 Effect of Isturisa On Other Drugs (7.2 Effect of ISTURISA on Other Drugs)
ISTURISA should be used with caution when coadministered with CYP1A2 and CYP2C19 substrates with a narrow therapeutic index, such as theophylline, tizanidine, and S-mephenytoin [see Clinical Pharmacology (12.3)].
2.3 Dosage Interruptions and Modifications
- Decrease or temporarily discontinue ISTURISA if urine free cortisol levels fall below the target range, there is a rapid decrease in cortisol levels, and/or patients report symptoms of hypocortisolism. If necessary, glucocorticoid replacement therapy should be initiated.
- Stop ISTURISA and administer exogenous glucocorticoid replacement therapy if serum or plasma cortisol levels are below target range and patients have symptoms of adrenal insufficiency [see Warnings and Precautions (5.1)].
- If treatment is interrupted, re-initiate ISTURISA at a lower dose when cortisol levels are within target ranges and patient symptoms have been resolved.
2.2 Recommended Dosage, Titration, and Monitoring
- Initiate dosing at 2 mg orally twice daily, with or without food.
- Initially, titrate the dosage by 1 mg to 2 mg twice daily, no more frequently than every 2 weeks based on the rate of cortisol changes, individual tolerability and improvement in signs and symptoms of Cushing's syndrome. If a patient tolerates ISTURISA dosage of 10 mg twice daily and continues to have elevated 24-hour urine free cortisol (UFC) levels above upper normal limit, the dosage can be titrated further by 5 mg twice daily every 2 weeks. Monitor cortisol levels from at least two 24-hour urine free cortisol collections every 1 to 2 weeks until adequate clinical response is maintained.
- The maintenance dosage of ISTURISA is individualized and determined by titration based on cortisol levels and patient's signs and symptoms.
- The maintenance dosage varied between 2 mg and 7 mg twice daily in clinical trials. The maximum recommended maintenance dosage of ISTURISA is 30 mg twice daily [see Clinical Studies (14)].
- Once the maintenance dosage is achieved, monitor cortisol levels at least every 1 to 2 months or as indicated.
Principal Display Panel 1 Mg Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 1 mg Blister Pack Carton)
NDC 55292-330-60
Isturisa® 1 mg
(osilodrostat)
tablets
Rx only
Each tablet contains 1 mg osilodrostat (as osilodrostat phosphate).
60 film-coated tablets
RECORDATI
RARE DISEASES
Principal Display Panel 5 Mg Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 5 mg Blister Pack Carton)
NDC 55292-331-60
Isturisa® 5 mg
(osilodrostat)
tablets
Rx only
Each tablet contains 5 mg osilodrostat (as osilodrostat phosphate).
60 film-coated tablets
RECORDATI
RARE DISEASES
2.1 Laboratory Testing Prior to Isturisa Initiation (2.1 Laboratory Testing Prior to ISTURISA Initiation)
- Correct hypokalemia and hypomagnesemia prior to starting ISTURISA [see Warnings and Precautions (5.2, 5.3)].
- Obtain baseline electrocardiogram (ECG). Repeat ECG within one week after treatment initiation, and as clinically indicated thereafter [see Warnings and Precautions (5.2)].
Principal Display Panel 10 Mg Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 10 mg Blister Pack Carton)
NDC 55292-322-60
Isturisa® 10 mg
(osilodrostat)
tablets
Rx only
Each tablet contains 10 mg osilodrostat (as osilodrostat phosphate).
60 film-coated tablets
RECORDATI
RARE DISEASES
5.3 Elevations in Adrenal Hormone Precursors and Androgens
ISTURISA blocks cortisol synthesis and may increase circulating levels of cortisol and aldosterone precursors (11-deoxy cortisol and 11-deoxycorticosterone) and androgens.
Elevated 11-deoxycorticosterone levels may activate mineralocorticoid receptors and cause hypokalemia, edema and hypertension [see Adverse Reactions (6)]. Hypokalemia should be corrected prior to initiating ISTURISA. Monitor patients treated with ISTURISA for hypokalemia, worsening of hypertension and edema. ISTURISA-induced hypokalemia should be treated with intravenous or oral potassium supplementation based on event severity. If hypokalemia persists despite potassium supplementation, consider adding mineralocorticoid antagonists. ISTURISA dose reduction or discontinuation may be necessary.
Accumulation of androgens may lead to hirsutism, hypertrichosis and acne (in females). Inform patients of the symptoms associated with hyperandrogenism and advise them to contact a healthcare provider if they occur.
2.4 Recommended Dosage and Monitoring in Patients With Renal Impairment (2.4 Recommended Dosage and Monitoring in Patients with Renal Impairment)
- No dose adjustment is required for patients with renal impairment. Use caution in interpreting urine free cortisol levels in patients with moderate to severe renal impairment, due to reduced urine free cortisol excretion [see Clinical Pharmacology (12.3)].
2.5 Recommended Dosage and Monitoring in Patients With Hepatic Impairment (2.5 Recommended Dosage and Monitoring in Patients with Hepatic Impairment)
- For patients with moderate hepatic impairment (Child-Pugh B), the recommended starting dose is 1 mg twice daily. For patients with severe hepatic impairment (Child-Pugh C), the recommended starting dose is 1 mg once daily in the evening.
- No dose adjustment is required for patients with mild hepatic impairment (Child-Pugh A).
- More frequent monitoring of adrenal function may be required during dose titration in all patients with hepatic impairment [see Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:22.311720 · Updated: 2026-03-14T22:37:11.582303