Pseudoephedrine Hydrocloride
f398ceab-e3d2-421d-813e-b66369f65472
34390-5
HUMAN OTC DRUG LABEL
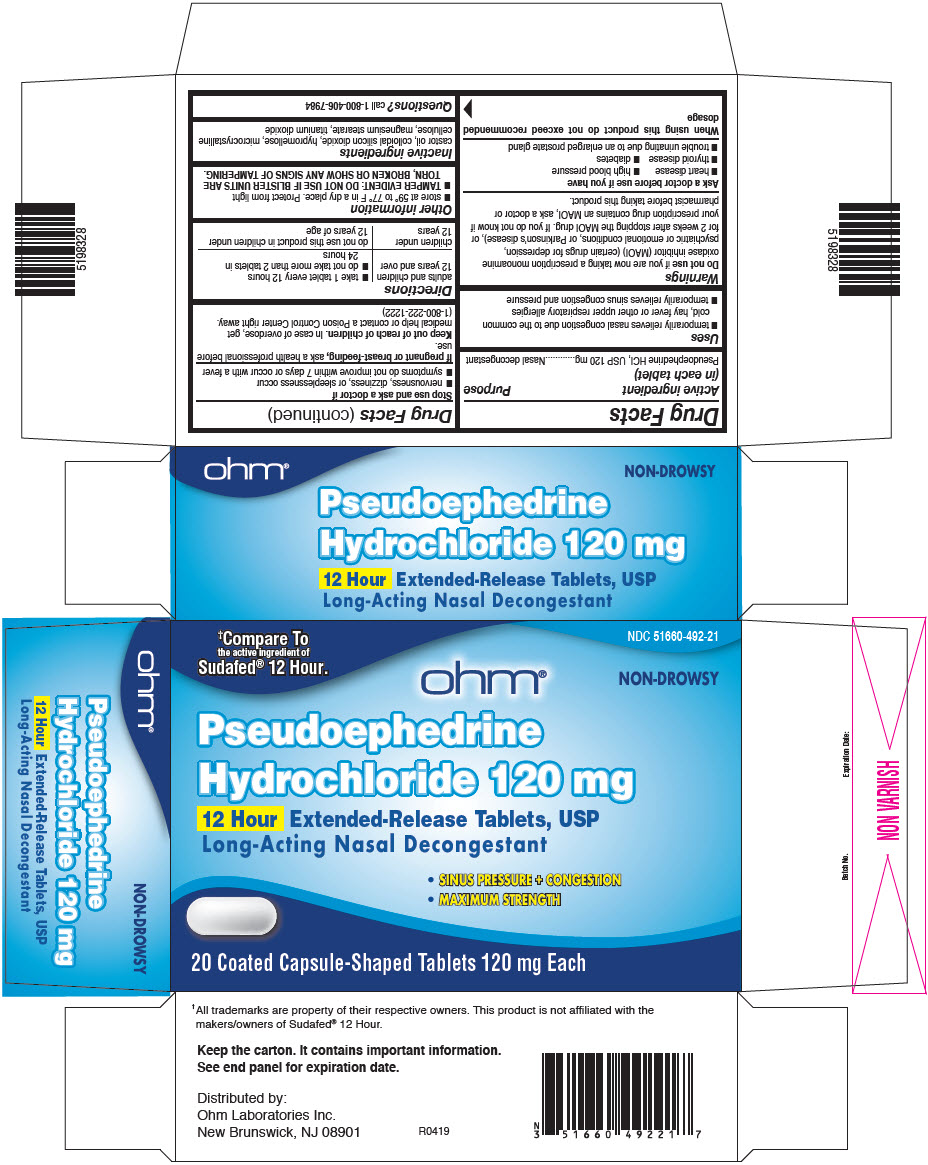
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Nasal decongestant
Medication Information
Purpose
Nasal decongestant
Description
Drug Facts
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Section 50567-7
When using this product do not exceed recommended dosage
Section 50570-1
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
| adults and children 12 years and over |
|
| children under 12 years | do not use this product in children under 12 years of age |
Questions?
call 1-800-406-7984
Other Information
- store at 59° to 77° F in a dry place. Protect from light
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.
Inactive Ingredients
castor oil, colloidal silicon dioxide, hypromellose, microcrystalline cellulose, magnesium stearate, titanium dioxide
Stop Use and Ask A Doctor If
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
Active Ingredient (in Each Tablet)
Pseudoephedrine HCl, USP 120 mg
Ask A Doctor Before Use If You Have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
Principal Display Panel 20 Tablet Blister Pack
†Compare To
the active ingredient of
Sudafed® 12 Hour.
NDC 51660-492-21
ohm®
NON-DROWSY
Pseudoephedrine
Hydrochloride 120 mg
12 Hour Extended-Release Tablets, USP
Long-Acting Nasal Decongestant
- SINUS PRESSURE + CONGESTION
- MAXIMUM STRENGTH
20 Coated Capsule-Shaped Tablets 120 mg Each
Structured Label Content
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Section 50567-7 (50567-7)
When using this product do not exceed recommended dosage
Section 50570-1 (50570-1)
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nasal decongestant
Directions
| adults and children 12 years and over |
|
| children under 12 years | do not use this product in children under 12 years of age |
Questions?
call 1-800-406-7984
Other Information (Other information)
- store at 59° to 77° F in a dry place. Protect from light
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.
Inactive Ingredients (Inactive ingredients)
castor oil, colloidal silicon dioxide, hypromellose, microcrystalline cellulose, magnesium stearate, titanium dioxide
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Pseudoephedrine HCl, USP 120 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
Principal Display Panel 20 Tablet Blister Pack (PRINCIPAL DISPLAY PANEL - 20 Tablet Blister Pack)
†Compare To
the active ingredient of
Sudafed® 12 Hour.
NDC 51660-492-21
ohm®
NON-DROWSY
Pseudoephedrine
Hydrochloride 120 mg
12 Hour Extended-Release Tablets, USP
Long-Acting Nasal Decongestant
- SINUS PRESSURE + CONGESTION
- MAXIMUM STRENGTH
20 Coated Capsule-Shaped Tablets 120 mg Each
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:48.667755 · Updated: 2026-03-14T22:55:33.121473