f2eb9444-333d-4b05-8cf3-2e6690a2bf99
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Levofloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Gram-positive bacteria Corynebacterium species * Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumonia Streptococcus (Groups C/F) Streptococcus (Group G) Viridans group streptococci* Gram-negative bacteria Acinetobacter lwoffii * Haemophilus influenzae Serratia marcescens* *Efficacy for this organism was studied in fewer than 10 infections
Indications and Usage
Levofloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Gram-positive bacteria Corynebacterium species * Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumonia Streptococcus (Groups C/F) Streptococcus (Group G) Viridans group streptococci* Gram-negative bacteria Acinetobacter lwoffii * Haemophilus influenzae Serratia marcescens* *Efficacy for this organism was studied in fewer than 10 infections
Dosage and Administration
Days 1 and 2 Instill one to two drops in the affected eye(s) every 2 hours while awake, up to 8 times per day. Days 3 through 7 Instill one to two drops in the affected eye(s) every 4 hours while awake, up to 4 times per day.
Warnings and Precautions
Hypersensitivity and anaphylaxis have been reported with systemic use of levofloxacin as well as topical use of other fluoroquinolones, including levofloxacin ophthalmic solution. ( 5.1 ) Prolonged use may result in the overgrowth of non- susceptible organisms, including fungi. ( 5.2 ) Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3 )
Contraindications
Levofloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to levofloxacin, to other quinolones, or to any of the components in this medication.
Adverse Reactions
The most frequently reported adverse reactions in the overall study population were transient decreased vision, fever, foreign body sensation, headache, transient ocular burning, ocular pain or discomfort, pharyngitis and photophobia. These reactions occurred in approximately 1-3% of patients. Other reported reactions occurring in less than 1% of patients included allergic reactions, lid edema, ocular dryness, and ocular itching.
How Supplied
Levofloxacin Ophthalmic Solution, 0.5% is a clear light yellow solution supplied in a white LDPE bottle with translucent LDPE nozzle and tan coloured HDPE cap in the following size: 5 mL filled in 5 mL bottle – NDC 72888-142-22 Storage: Store at 20º to 25°C (68º to 77°F) [See USP Controlled Room Temperature].
Medication Information
Warnings and Precautions
Hypersensitivity and anaphylaxis have been reported with systemic use of levofloxacin as well as topical use of other fluoroquinolones, including levofloxacin ophthalmic solution. ( 5.1 ) Prolonged use may result in the overgrowth of non- susceptible organisms, including fungi. ( 5.2 ) Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3 )
Indications and Usage
Levofloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Gram-positive bacteria Corynebacterium species * Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumonia Streptococcus (Groups C/F) Streptococcus (Group G) Viridans group streptococci* Gram-negative bacteria Acinetobacter lwoffii * Haemophilus influenzae Serratia marcescens* *Efficacy for this organism was studied in fewer than 10 infections
Dosage and Administration
Days 1 and 2 Instill one to two drops in the affected eye(s) every 2 hours while awake, up to 8 times per day. Days 3 through 7 Instill one to two drops in the affected eye(s) every 4 hours while awake, up to 4 times per day.
Contraindications
Levofloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to levofloxacin, to other quinolones, or to any of the components in this medication.
Adverse Reactions
The most frequently reported adverse reactions in the overall study population were transient decreased vision, fever, foreign body sensation, headache, transient ocular burning, ocular pain or discomfort, pharyngitis and photophobia. These reactions occurred in approximately 1-3% of patients. Other reported reactions occurring in less than 1% of patients included allergic reactions, lid edema, ocular dryness, and ocular itching.
How Supplied
Levofloxacin Ophthalmic Solution, 0.5% is a clear light yellow solution supplied in a white LDPE bottle with translucent LDPE nozzle and tan coloured HDPE cap in the following size: 5 mL filled in 5 mL bottle – NDC 72888-142-22 Storage: Store at 20º to 25°C (68º to 77°F) [See USP Controlled Room Temperature].
Description
Levofloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Gram-positive bacteria Corynebacterium species * Staphylococcus aureus Staphylococcus epidermidis Streptococcus pneumonia Streptococcus (Groups C/F) Streptococcus (Group G) Viridans group streptococci* Gram-negative bacteria Acinetobacter lwoffii * Haemophilus influenzae Serratia marcescens* *Efficacy for this organism was studied in fewer than 10 infections
8.1 Pregnancy
Teratogenic Effects
Levofloxacin at oral doses of 810 mg/kg/day in rats caused decreased fetal body weight and increased fetal mortality. No teratogenic effect was observed when rabbits were dosed orally as high as 50 mg/kg/day, at which systemic exposure was approximately 2,250 times that observed at the maximum recommended human ophthalmic dose, or when dosed intravenously as high as 25 mg/kg/day, at which systemic exposure was approximately 1000 times that observed at the maximum recommended human ophthalmic dose.
There are, however, no adequate and well-controlled studies in pregnant women. Levofloxacin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
11 Description
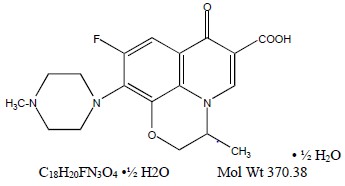
Levofloxacin Ophthalmic Solution, 0.5% is a sterile topical ophthalmic solution. Levofloxacin is a fluoroquinolone antibacterial active against a broad spectrum of Gram-positive and Gram-negative ocular pathogens. Levofloxacin is a fluorinated 4-quinolone containing a six-member (pyridobenzoxazine) ring from positions 1 to 8 of the basic ring structure. Levofloxacin is the pure (-)-(S)-enantiomer of the racemic drug substance, ofloxacin. It is more soluble in water at neutral pH than ofloxacin.
Chemical Name: (-)-(S)-9-fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido [1,2,3-de]-1,4 benzoxazine-6-carboxylic acid hemihydrate.
Levofloxacin (hemihydrate) is a light yellow powder.
Each mL of levofloxacin ophthalmic solution, 0.5% contains 5.12 mg of levofloxacin hemihydrate equivalent to 5 mg levofloxacin.
Contains
Levofloxacin 5 mg equivalent to 5.12 mg levofloxacin hemihydrate with benzalkonium chloride 0.005%, sodium chloride, water for injection, hydrochloric acid and/or sodium hydroxide (to adjust pH).
Levofloxacin ophthalmic solution, 0.5% is isotonic and formulated at pH 6.0-6.8 with an osmolality between 285-315 mOsm/kg.
12.3 Microbiology
Levofloxacin is the L-isomer of the racemate, ofloxacin, a quinolone antimicrobial agent. The antibacterial activity of ofloxacin resides primarily in the L-isomer. The mechanism of action of levofloxacin and other fluoroquinolone antimicrobials involves the inhibition of bacterial topoisomerase IV and DNA gyrase (both of which are type II topoisomerases), enzymes required for DNA replication, transcription, repair, and recombination.
Levofloxacin has in vitroactivity against a wide range of Gram-negative and Gram-positive microorganisms and is often bactericidal at concentrations equal to or slightly greater than inhibitory concentrations.
Fluoroquinolones, including levofloxacin, differ in chemical structure and mode of action from β- lactam antibiotics and aminoglycosides, and therefore may be active against bacteria resistant to β- lactam antibiotics and aminoglycosides. Additionally, β-lactam antibiotics and aminoglycosides may be active against bacteria resistant to levofloxacin.
Resistance to levofloxacin due to spontaneous mutation i n vitrois a rare occurrence (range: 10 -9to 10 -10)
Levofloxacin has been shown to be active against most strains of the following microorganisms, both in vitroand in clinical infections as described in the INDICATIONS AND USAGE section:
Aerobic gram-positive microorganisms
Corynebacterium species*
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumonia
Streptococcus(Groups C/F)
Streptococcus(Group G)
Viridans group streptococci*
Aerobic gram-negative microorganisms
Acinetobacter lwoffii*
Haemophilus influenzae
Serratia marcescens*
*Efficacy for this organism was studied in fewer than 10 infections.
The following i n vitrodata are also available, but their clinical significance in ophthalmic infections is unknown. The safety and effectiveness of levofloxacin in treating ophthalmological infections due to these microorganisms have not been established in adequate and well- controlled trials.
These organisms are considered susceptible when evaluated using systemic breakpoints. However, a correlation between the in vitrosystemic breakpoint and ophthalmological efficacy has not been established. The list of organisms is provided as guidance only in assessing the potential treatment of conjunctival infections. Levofloxacin exhibits in vitrominimal inhibitory concentrations (MICs) of 2 mcg/mL or less (systemic susceptible breakpoint) against most (≥90%) strains of the following ocular pathogens:
Aerobicgram-positive microorganisms
Enterococcus faecalis
Staphylococcus saprophyticus
Streptococcus agalactiae
Streptococcus pyogenes
Aerobic gram-negative microorganisms
Acinetobacter anitratus
Acinetobacter baumannii
Citrobacter koseri
Citrobacter freundii
Enterobacter aerogenes
Enterobacter agglomerans
Enterobacter cloacae
Escherichia coli
Haemophilus parainfluenzae
Klebsiella oxytoca
Klebsiella pneumonia
Legionella pneumophila
Moraxella catarrhalis
Morganella morganii
Neisseria gonorrhoeae
Proteus mirabilis
Proteus vulgaris
Providencia rettgeri
Providencia stuartii
Pseudomonas aeruginosa
Pseudomonas fluorescens
8.4 Pediatric Use
Safety and effectiveness in children below the age of six years have not been established. Oral administration of systemic quinolones has been shown to cause arthropathy in immature animals. There is no evidence that the ophthalmic administration of levofloxacin has any effect on weight bearing joints.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and other adult patients.
14 Clinical Studies
In randomized, double-masked, multicenter controlled clinical trials where patients were dosed for 5 days, Levofloxacin ophthalmic® demonstrated clinical cures in 79% of patients treated for bacterial conjunctivitis on the final study visit day (day 6 to10). Microbial outcomes for the same clinical trials demonstrated an eradication rate for presumed pathogens of 90%.
4 Contraindications
Levofloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to levofloxacin, to other quinolones, or to any of the components in this medication.
6 Adverse Reactions
The most frequently reported adverse reactions in the overall study population were transient decreased vision, fever, foreign body sensation, headache, transient ocular burning, ocular pain or discomfort, pharyngitis and photophobia. These reactions occurred in approximately 1-3% of patients. Other reported reactions occurring in less than 1% of patients included allergic reactions, lid edema, ocular dryness, and ocular itching.
8.3 Nursing Mothers
Levofloxacin has not been measured in human milk. Based upon data from ofloxacin, it can be presumed that levofloxacin is excreted in human milk. Caution should be exercised when levofloxacin ophthalmic solution is administered to a nursing mother.
12.2 Pharmacokinetics
Levofloxacin concentration in plasma was measured in 15 healthy adult volunteers at various time points during a 15-day course of treatment with levofloxacin ophthalmic solution. The mean levofloxacin concentration in plasma 1 hour postdose, ranged from 0.86 ng/mL on Day 1 to 2.05 ng/mL on Day 15. The highest maximum mean levofloxacin concentration of 2.25 ng/mL was measured on Day 4 following 2 days of dosing every 2 hours for a total of 8 doses per day. Maximum mean levofloxacin concentrations increased from 0.94 ng/mL on Day 1 to 2.15 ng/mL on Day 15, which is more than 1,000 times lower than those reported after standard oral doses of levofloxacin.
Levofloxacin concentration in tears was measured in 30 healthy adult volunteers at various time points following instillation of a single drop of levofloxacin ophthalmic solution. Mean levofloxacin concentrations in tears ranged from 34.9 to 221.1 mcg/mL during the 60-minute period following the single dose. The mean tear concentrations measured 4 and 6 hours postdose were 17.0 and 6.6 mcg/mL. The clinical significance of these concentrations is unknown.
1 Indications and Usage
Levofloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms:
Gram-positive bacteria
Corynebacterium species*
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumonia
Streptococcus(Groups C/F)
Streptococcus(Group G)
Viridans group streptococci*
Gram-negative bacteria
Acinetobacter lwoffii *
Haemophilus influenzae
Serratia marcescens*
*Efficacy for this organism was studied in fewer than 10 infections
12.1 Mechanism of Action
Levofloxacin is a member of the fluoroquinolone class of anti-infective drugs. (See 12.4 Microbiology)
5 Warnings and Precautions
- Hypersensitivity and anaphylaxis have been reported with systemic use of levofloxacin as well as topical use of other fluoroquinolones, including levofloxacin ophthalmic solution. ( 5.1)
- Prolonged use may result in the overgrowth of non- susceptible organisms, including fungi. ( 5.2)
- Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3)
2 Dosage and Administration
Days 1 and 2
Instill one to two drops in the affected eye(s) every 2 hours while awake, up to 8 times per day.
Days 3 through 7
Instill one to two drops in the affected eye(s) every 4 hours while awake, up to 4 times per day.
17.2 Avoid Contact Lens Wear
Advise patients not to wear contact lenses if they have signs and symptoms of bacterial conjunctivitis.
3 Dosage Forms and Strengths
5 mL white LDPE bottle filled with 5 mL sterile ophthalmic solution of levofloxacin, 0.5%.
5.1 Hypersensitivity Reactions
In patients receiving systemically administered quinolones, including levofloxacin, serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported, some following the first dose. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema, (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria and itching. If allergic reaction to levofloxacin occurs, discontinue the drug. Serious acute hypersensitivity reactions may require immediate emergency treatment. Oxygen and airway management should be administered as clinically indicated.
17.3 Hypersensitivity Reactions
Systemically administered quinolones, including levofloxacin, have been associated with hypersensitivity reactions, even following a single dose. Advise patients to discontinue use immediately and contact their physician at the first sign of a rash or allergic reaction.
Distributed by:
Advagen Pharma Ltd.,
East Windsor, NJ 08520, USA
Rev. 02/2025
16 How Supplied/storage Handling
Levofloxacin Ophthalmic Solution, 0.5% is a clear light yellow solution supplied in a white LDPE bottle with translucent LDPE nozzle and tan coloured HDPE cap in the following size:
5 mL filled in 5 mL bottle – NDC 72888-142-22
Storage:
Store at 20º to 25°C (68º to 77°F) [See USP Controlled Room Temperature].
5.3 Avoidance of Contact Lens Wear
Patients should be advised not to wear contact lenses if they have signs and symptoms of bacterial conjunctivitis.
Package Label.principal Display Panel
Levofloxacin Ophthalmic Solution, 0.5% Sterile Ophthalmic Solution - NDC 72888-142-22 - 5 mL Carton Label
Levofloxacin Ophthalmic Solution, 0.5% Sterile Ophthalmic Solution - NDC 72888-142-22 - 5 mL Container Label
17.1 Avoid Contamination of the Product
Advise patients to avoid contaminating the applicator tip with material from the eye, finger, or other source.
5.2 Growth of Resistant Organisms With Prolonged Use
As with other anti-infectives, prolonged use may result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. Whenever clinical judgment dictates, the patient should be examined with the aid of magnification, such as slit-lamp biomicroscopy, and where appropriate, fluorescein staining.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a long term carcinogenicity study in rats, levofloxacin exhibited no carcinogenic or tumorigenic potential following daily dietary administration for 2 years at doses up to 100 mg/kg/day, corresponding to plasma levels that were 1235 times the maximum clinical exposure, based on C max.
Levofloxacin was not mutagenic in the following assays: Ames bacterial mutation assay ( S. typhimuriumand E . coli), CHO/HGPRT forward mutation assay, mouse micronucleus test, mouse dominant lethal test, rat unscheduled DNA synthesis assay, and the in vivomouse sister chromatid exchange assay. It was positive in the in vitrochromosomal aberration (CHL cell line) and in vitrosister chromatid exchange (CHL/IU cell line) assays. Levofloxacin caused no impairment of fertility or reproduction in rats at oral doses as high as 360 mg/kg/day, at which systemic exposure was estimated to be 23,000 times that at the maximum recommended human ophthalmic dose.
Structured Label Content
8.1 Pregnancy
Teratogenic Effects
Levofloxacin at oral doses of 810 mg/kg/day in rats caused decreased fetal body weight and increased fetal mortality. No teratogenic effect was observed when rabbits were dosed orally as high as 50 mg/kg/day, at which systemic exposure was approximately 2,250 times that observed at the maximum recommended human ophthalmic dose, or when dosed intravenously as high as 25 mg/kg/day, at which systemic exposure was approximately 1000 times that observed at the maximum recommended human ophthalmic dose.
There are, however, no adequate and well-controlled studies in pregnant women. Levofloxacin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
11 Description (11 DESCRIPTION)
Levofloxacin Ophthalmic Solution, 0.5% is a sterile topical ophthalmic solution. Levofloxacin is a fluoroquinolone antibacterial active against a broad spectrum of Gram-positive and Gram-negative ocular pathogens. Levofloxacin is a fluorinated 4-quinolone containing a six-member (pyridobenzoxazine) ring from positions 1 to 8 of the basic ring structure. Levofloxacin is the pure (-)-(S)-enantiomer of the racemic drug substance, ofloxacin. It is more soluble in water at neutral pH than ofloxacin.
Chemical Name: (-)-(S)-9-fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido [1,2,3-de]-1,4 benzoxazine-6-carboxylic acid hemihydrate.
Levofloxacin (hemihydrate) is a light yellow powder.
Each mL of levofloxacin ophthalmic solution, 0.5% contains 5.12 mg of levofloxacin hemihydrate equivalent to 5 mg levofloxacin.
Contains
Levofloxacin 5 mg equivalent to 5.12 mg levofloxacin hemihydrate with benzalkonium chloride 0.005%, sodium chloride, water for injection, hydrochloric acid and/or sodium hydroxide (to adjust pH).
Levofloxacin ophthalmic solution, 0.5% is isotonic and formulated at pH 6.0-6.8 with an osmolality between 285-315 mOsm/kg.
12.3 Microbiology
Levofloxacin is the L-isomer of the racemate, ofloxacin, a quinolone antimicrobial agent. The antibacterial activity of ofloxacin resides primarily in the L-isomer. The mechanism of action of levofloxacin and other fluoroquinolone antimicrobials involves the inhibition of bacterial topoisomerase IV and DNA gyrase (both of which are type II topoisomerases), enzymes required for DNA replication, transcription, repair, and recombination.
Levofloxacin has in vitroactivity against a wide range of Gram-negative and Gram-positive microorganisms and is often bactericidal at concentrations equal to or slightly greater than inhibitory concentrations.
Fluoroquinolones, including levofloxacin, differ in chemical structure and mode of action from β- lactam antibiotics and aminoglycosides, and therefore may be active against bacteria resistant to β- lactam antibiotics and aminoglycosides. Additionally, β-lactam antibiotics and aminoglycosides may be active against bacteria resistant to levofloxacin.
Resistance to levofloxacin due to spontaneous mutation i n vitrois a rare occurrence (range: 10 -9to 10 -10)
Levofloxacin has been shown to be active against most strains of the following microorganisms, both in vitroand in clinical infections as described in the INDICATIONS AND USAGE section:
Aerobic gram-positive microorganisms
Corynebacterium species*
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumonia
Streptococcus(Groups C/F)
Streptococcus(Group G)
Viridans group streptococci*
Aerobic gram-negative microorganisms
Acinetobacter lwoffii*
Haemophilus influenzae
Serratia marcescens*
*Efficacy for this organism was studied in fewer than 10 infections.
The following i n vitrodata are also available, but their clinical significance in ophthalmic infections is unknown. The safety and effectiveness of levofloxacin in treating ophthalmological infections due to these microorganisms have not been established in adequate and well- controlled trials.
These organisms are considered susceptible when evaluated using systemic breakpoints. However, a correlation between the in vitrosystemic breakpoint and ophthalmological efficacy has not been established. The list of organisms is provided as guidance only in assessing the potential treatment of conjunctival infections. Levofloxacin exhibits in vitrominimal inhibitory concentrations (MICs) of 2 mcg/mL or less (systemic susceptible breakpoint) against most (≥90%) strains of the following ocular pathogens:
Aerobicgram-positive microorganisms
Enterococcus faecalis
Staphylococcus saprophyticus
Streptococcus agalactiae
Streptococcus pyogenes
Aerobic gram-negative microorganisms
Acinetobacter anitratus
Acinetobacter baumannii
Citrobacter koseri
Citrobacter freundii
Enterobacter aerogenes
Enterobacter agglomerans
Enterobacter cloacae
Escherichia coli
Haemophilus parainfluenzae
Klebsiella oxytoca
Klebsiella pneumonia
Legionella pneumophila
Moraxella catarrhalis
Morganella morganii
Neisseria gonorrhoeae
Proteus mirabilis
Proteus vulgaris
Providencia rettgeri
Providencia stuartii
Pseudomonas aeruginosa
Pseudomonas fluorescens
8.4 Pediatric Use
Safety and effectiveness in children below the age of six years have not been established. Oral administration of systemic quinolones has been shown to cause arthropathy in immature animals. There is no evidence that the ophthalmic administration of levofloxacin has any effect on weight bearing joints.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and other adult patients.
14 Clinical Studies (14 CLINICAL STUDIES)
In randomized, double-masked, multicenter controlled clinical trials where patients were dosed for 5 days, Levofloxacin ophthalmic® demonstrated clinical cures in 79% of patients treated for bacterial conjunctivitis on the final study visit day (day 6 to10). Microbial outcomes for the same clinical trials demonstrated an eradication rate for presumed pathogens of 90%.
4 Contraindications (4 CONTRAINDICATIONS)
Levofloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to levofloxacin, to other quinolones, or to any of the components in this medication.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most frequently reported adverse reactions in the overall study population were transient decreased vision, fever, foreign body sensation, headache, transient ocular burning, ocular pain or discomfort, pharyngitis and photophobia. These reactions occurred in approximately 1-3% of patients. Other reported reactions occurring in less than 1% of patients included allergic reactions, lid edema, ocular dryness, and ocular itching.
8.3 Nursing Mothers
Levofloxacin has not been measured in human milk. Based upon data from ofloxacin, it can be presumed that levofloxacin is excreted in human milk. Caution should be exercised when levofloxacin ophthalmic solution is administered to a nursing mother.
12.2 Pharmacokinetics
Levofloxacin concentration in plasma was measured in 15 healthy adult volunteers at various time points during a 15-day course of treatment with levofloxacin ophthalmic solution. The mean levofloxacin concentration in plasma 1 hour postdose, ranged from 0.86 ng/mL on Day 1 to 2.05 ng/mL on Day 15. The highest maximum mean levofloxacin concentration of 2.25 ng/mL was measured on Day 4 following 2 days of dosing every 2 hours for a total of 8 doses per day. Maximum mean levofloxacin concentrations increased from 0.94 ng/mL on Day 1 to 2.15 ng/mL on Day 15, which is more than 1,000 times lower than those reported after standard oral doses of levofloxacin.
Levofloxacin concentration in tears was measured in 30 healthy adult volunteers at various time points following instillation of a single drop of levofloxacin ophthalmic solution. Mean levofloxacin concentrations in tears ranged from 34.9 to 221.1 mcg/mL during the 60-minute period following the single dose. The mean tear concentrations measured 4 and 6 hours postdose were 17.0 and 6.6 mcg/mL. The clinical significance of these concentrations is unknown.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Levofloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms:
Gram-positive bacteria
Corynebacterium species*
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumonia
Streptococcus(Groups C/F)
Streptococcus(Group G)
Viridans group streptococci*
Gram-negative bacteria
Acinetobacter lwoffii *
Haemophilus influenzae
Serratia marcescens*
*Efficacy for this organism was studied in fewer than 10 infections
12.1 Mechanism of Action
Levofloxacin is a member of the fluoroquinolone class of anti-infective drugs. (See 12.4 Microbiology)
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity and anaphylaxis have been reported with systemic use of levofloxacin as well as topical use of other fluoroquinolones, including levofloxacin ophthalmic solution. ( 5.1)
- Prolonged use may result in the overgrowth of non- susceptible organisms, including fungi. ( 5.2)
- Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Days 1 and 2
Instill one to two drops in the affected eye(s) every 2 hours while awake, up to 8 times per day.
Days 3 through 7
Instill one to two drops in the affected eye(s) every 4 hours while awake, up to 4 times per day.
17.2 Avoid Contact Lens Wear
Advise patients not to wear contact lenses if they have signs and symptoms of bacterial conjunctivitis.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
5 mL white LDPE bottle filled with 5 mL sterile ophthalmic solution of levofloxacin, 0.5%.
5.1 Hypersensitivity Reactions
In patients receiving systemically administered quinolones, including levofloxacin, serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported, some following the first dose. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema, (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria and itching. If allergic reaction to levofloxacin occurs, discontinue the drug. Serious acute hypersensitivity reactions may require immediate emergency treatment. Oxygen and airway management should be administered as clinically indicated.
17.3 Hypersensitivity Reactions
Systemically administered quinolones, including levofloxacin, have been associated with hypersensitivity reactions, even following a single dose. Advise patients to discontinue use immediately and contact their physician at the first sign of a rash or allergic reaction.
Distributed by:
Advagen Pharma Ltd.,
East Windsor, NJ 08520, USA
Rev. 02/2025
16 How Supplied/storage Handling (16 HOW SUPPLIED/STORAGE HANDLING)
Levofloxacin Ophthalmic Solution, 0.5% is a clear light yellow solution supplied in a white LDPE bottle with translucent LDPE nozzle and tan coloured HDPE cap in the following size:
5 mL filled in 5 mL bottle – NDC 72888-142-22
Storage:
Store at 20º to 25°C (68º to 77°F) [See USP Controlled Room Temperature].
5.3 Avoidance of Contact Lens Wear
Patients should be advised not to wear contact lenses if they have signs and symptoms of bacterial conjunctivitis.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Levofloxacin Ophthalmic Solution, 0.5% Sterile Ophthalmic Solution - NDC 72888-142-22 - 5 mL Carton Label
Levofloxacin Ophthalmic Solution, 0.5% Sterile Ophthalmic Solution - NDC 72888-142-22 - 5 mL Container Label
17.1 Avoid Contamination of the Product
Advise patients to avoid contaminating the applicator tip with material from the eye, finger, or other source.
5.2 Growth of Resistant Organisms With Prolonged Use (5.2 Growth of Resistant Organisms with Prolonged Use)
As with other anti-infectives, prolonged use may result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. Whenever clinical judgment dictates, the patient should be examined with the aid of magnification, such as slit-lamp biomicroscopy, and where appropriate, fluorescein staining.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a long term carcinogenicity study in rats, levofloxacin exhibited no carcinogenic or tumorigenic potential following daily dietary administration for 2 years at doses up to 100 mg/kg/day, corresponding to plasma levels that were 1235 times the maximum clinical exposure, based on C max.
Levofloxacin was not mutagenic in the following assays: Ames bacterial mutation assay ( S. typhimuriumand E . coli), CHO/HGPRT forward mutation assay, mouse micronucleus test, mouse dominant lethal test, rat unscheduled DNA synthesis assay, and the in vivomouse sister chromatid exchange assay. It was positive in the in vitrochromosomal aberration (CHL cell line) and in vitrosister chromatid exchange (CHL/IU cell line) assays. Levofloxacin caused no impairment of fertility or reproduction in rats at oral doses as high as 360 mg/kg/day, at which systemic exposure was estimated to be 23,000 times that at the maximum recommended human ophthalmic dose.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:05.565255 · Updated: 2026-03-14T22:23:58.586609