spironolactone tablets, usp
f230f1fa-878e-4753-a16c-bf059b59cdef
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Spironolactone tablets are indicated in the management of: Primary hyperaldosteronism for: Establishing the diagnosis of primary hyperaldosteronism by therapeutic trial. Short-term preoperative treatment of patients with primary hyperaldosteronism. Long-term maintenance therapy for patients with discrete aldosterone-producing adrenal adenomas who are judged to be poor operative risks or who decline surgery. Long-term maintenance therapy for patients with bilateral micro or macronodular adrenal hyperplasia (idiopathic hyperaldosteronism). Edematous conditions for patients with:
Contraindications
Spironolactone is contraindicated for patients with anuria, acute renal insufficiency, significant impairment of renal excretory function, hyperkalemia, Addison’s disease, and with concomitant use of eplerenone.
Adverse Reactions
The following adverse reactions have been reported and, within each category (body system), are listed in order of decreasing severity. Digestive: Gastric bleeding, ulceration, gastritis, diarrhea and cramping, nausea, vomiting. Reproductive: Gynecomastia (see Precautions ), inability to achieve or maintain erection, irregular menses or amenorrhea, postmenopausal bleeding, breast pain. Carcinoma of the breast has been reported in patients taking spironolactone but a cause and effect relationship has not been established. Hematologic: Leukopenia (including agranulocytosis), thrombocytopenia. Hypersensitivity: Fever, urticaria, maculopapular or erythematous cutaneous eruptions, anaphylactic reactions, vasculitis. Metabolism: Hyperkalemia, electrolyte disturbances (see Warnings and Precautions ). Musculoskeletal: Leg cramps. Nervous system /psychiatric: Lethargy, mental confusion, ataxia, dizziness, headache, drowsiness. Liver / biliary: A very few cases of mixed cholestatic/hepatocellular toxicity, with one reported fatality, have been reported with spironolactone administration. Renal: Renal dysfunction (including renal failure). Skin: Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug rash with eosinophilia and systemic symptoms (DRESS), alopecia, pruritis.
How Supplied
Spironolactone Tablets USP 25 mg tablets are Light yellow to yellow colored, round, biconvex, film coated tablets with inscription “AD” on one side and plain on the other side having faint odour of peppermint, supplied as: NDC Number Size 16729-225-01 bottle of 100 16729-225-16 bottle of 500 16729-225-17 bottle of 1,000 16729-225-19 bottle of 2,500 Spironolactone Tablets USP 50 mg tablets are Light orange to orange colored, oval, biconvex, film coated tablets with inscription “AE” on one side and breakline on the other side having faint odour of peppermint, supplied as: NDC Number Size 16729-226-01 bottle of 100 16729-226-16 bottle of 500 Spironolactone Tablets USP 100 mg tablets are Light peach to peach colored, round, biconvex, film coated tablets with inscription “AF” on one side and breakline on the other side having faint odour of peppermint, supplied as: NDC Number Size 16729-227-01 bottle of 100 16729-227-16 bottle of 500 Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] Protect from light. Dispense in tight, Light-resistant containers. Manufactured For: Accord Healthcare, Inc., 1009 Slater Road, Suite 210-B, Durham, NC 27703, USA. Manufactured By: Intas Pharmaceuticals Limited, Plot No 5 to 12, Pharmez, Sarkhej-Bavla, National Highway No 8-A, Near Village Matoda, Tal Sanand, Ahmedabad - 382 213, Gujarat, India 51 2267 0 700753 Issued November 2016.
Description
Spironolactone has been shown to be a tumorigen in chronic toxicity studies in rats (see Precautions ). Spironolactone should be used only in those conditions described under Indications and Usage . Unnecessary use of this drug should be avoided.
Medication Information
Indications and Usage
Spironolactone tablets are indicated in the management of:
Primary hyperaldosteronism for:
Establishing the diagnosis of primary hyperaldosteronism by therapeutic trial.
Short-term preoperative treatment of patients with primary hyperaldosteronism.
Long-term maintenance therapy for patients with discrete aldosterone-producing adrenal adenomas who are judged to be poor operative risks or who decline surgery.
Long-term maintenance therapy for patients with bilateral micro or macronodular adrenal hyperplasia (idiopathic hyperaldosteronism).
Edematous conditions for patients with:
Contraindications
Spironolactone is contraindicated for patients with anuria, acute renal insufficiency, significant impairment of renal excretory function, hyperkalemia, Addison’s disease, and with concomitant use of eplerenone.
Adverse Reactions
The following adverse reactions have been reported and, within each category (body system), are listed in order of decreasing severity.
Digestive: Gastric bleeding, ulceration, gastritis, diarrhea and cramping, nausea, vomiting.
Reproductive: Gynecomastia (see Precautions ), inability to achieve or maintain erection, irregular menses or amenorrhea, postmenopausal bleeding, breast pain. Carcinoma of the breast has been reported in patients taking spironolactone but a cause and effect relationship has not been established.
Hematologic: Leukopenia (including agranulocytosis), thrombocytopenia.
Hypersensitivity: Fever, urticaria, maculopapular or erythematous cutaneous eruptions, anaphylactic reactions, vasculitis.
Metabolism: Hyperkalemia, electrolyte disturbances (see Warnings and Precautions).
Musculoskeletal: Leg cramps.
Nervous system /psychiatric: Lethargy, mental confusion, ataxia, dizziness, headache, drowsiness.
Liver / biliary: A very few cases of mixed cholestatic/hepatocellular toxicity, with one reported fatality, have been reported with spironolactone administration.
Renal: Renal dysfunction (including renal failure).
Skin: Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug rash with eosinophilia and systemic symptoms (DRESS), alopecia, pruritis.
How Supplied
Spironolactone Tablets USP 25 mg tablets are Light yellow to yellow colored, round, biconvex, film coated tablets with inscription “AD” on one side and plain on the other side having faint odour of peppermint, supplied as:
| NDC Number | Size |
|---|---|
| 16729-225-01 | bottle of 100 |
| 16729-225-16 | bottle of 500 |
| 16729-225-17 | bottle of 1,000 |
| 16729-225-19 | bottle of 2,500 |
Spironolactone Tablets USP 50 mg tablets are Light orange to orange colored, oval, biconvex, film coated tablets with inscription “AE” on one side and breakline on the other side having faint odour of peppermint, supplied as:
| NDC Number | Size |
|---|---|
| 16729-226-01 | bottle of 100 |
| 16729-226-16 | bottle of 500 |
Spironolactone Tablets USP 100 mg tablets are Light peach to peach colored, round, biconvex, film coated tablets with inscription “AF” on one side and breakline on the other side having faint odour of peppermint, supplied as:
| NDC Number | Size |
|---|---|
| 16729-227-01 | bottle of 100 |
| 16729-227-16 | bottle of 500 |
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
Protect from light. Dispense in tight, Light-resistant containers.
Manufactured For:
Accord Healthcare, Inc.,
1009 Slater Road,
Suite 210-B,
Durham, NC 27703,
USA.
Manufactured By:
Intas Pharmaceuticals Limited,
Plot No 5 to 12, Pharmez,
Sarkhej-Bavla, National Highway No 8-A,
Near Village Matoda, Tal Sanand,
Ahmedabad - 382 213, Gujarat, India
51 2267 0 700753
Issued November 2016.
Description
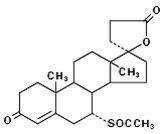
Spironolactone tablets USP, for oral administration contain 25 mg, 50 mg, or 100 mg of the aldosterone antagonist spironolactone, 17-hydroxy-7α-mercapto-3-oxo-17α-pregn-4-ene-21-carboxylic acid γ-lactone acetate, which has the following structural formula:
Spironolactone is practically insoluble in water, soluble in alcohol, and freely soluble in benzene and in chloroform.
Inactive ingredients include lactose monohydrate, dibasic calcium phosphate, povidone, peppermint oil, purified talc, pregelatinised starch, colloidal anhydrous silica, magnesium stearate, hypromellose, polyethylene glycol 400, titanium dioxide and iron oxide yellow. In addition, iron oxide red (50 mg and 100 mg tablets) is included in the film coating of specific strengths.
Section 34072-9
General: All patients receiving diuretic therapy should be observed for evidence of fluid or electrolyte imbalance, e.g., hypomagnesemia, hyponatremia, hypochloremic alkalosis, and hyperkalemia.
Serum and urine electrolyte determinations are particularly important when the patient is vomiting excessively or receiving parenteral fluids. Warning signs or symptoms of fluid and electrolyte imbalance, irrespective of cause, include dryness of the mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances such as nausea and vomiting. Hyperkalemia may occur in patients with impaired renal function or excessive potassium intake and can cause cardiac irregularities, which may be fatal. Consequently, no potassium supplement should ordinarily be given with spironolactone.
If hyperkalemia is suspected (warning signs include paresthesia, muscle weakness, fatigue, flaccid paralysis of the extremities, bradycardia and shock), an electrocardiogram (ECG) should be obtained. However, it is important to monitor serum potassium levels because mild hyperkalemia may not be associated with ECG changes.
If hyperkalemia is present, spironolactone should be discontinued immediately. With severe hyperkalemia, the clinical situation dictates the procedures to be employed. These may include the intravenous administration of calcium chloride solution, sodium bicarbonate solution and/or the oral or parenteral administration of glucose with a rapid-acting insulin preparation. These are temporary measures to be repeated as required. Cationic exchange resins such as sodium polystyrene sulfonate may be orally or rectally administered. Persistent hyperkalemia may require dialysis.
Reversible hyperchloremic metabolic acidosis, usually in association with hyperkalemia, has been reported to occur in some patients with decompensated hepatic cirrhosis, even in the presence of normal renal function.
Dilutional hyponatremia, manifested by dryness of the mouth, thirst, lethargy, and drowsiness, and confirmed by a low serum sodium level, may be caused or aggravated, especially when spironolactone is administered in combination with other diuretics, and dilutional hyponatremia may occur in edematous patients in hot weather; appropriate therapy is water restriction rather than administration of sodium, except in rare instances when the hyponatremia is life-threatening.
Spironolactone therapy may cause a transient elevation of BUN, especially in patients with pre-existing renal impairment. Spironolactone tablets may cause mild acidosis.
Gynecomastia may develop in association with the use of spironolactone; physicians should be alert to its possible onset. The development of gynecomastia appears to be related to both dosage level and duration of therapy and is normally reversible when spironolactone is discontinued. In rare instances, some breast enlargement may persist when spironolactone is discontinued.
Somnolence and dizziness have been reported to occur in some patients. Caution is advised when driving or operating machinery until the response to initial treatment has been determined.
Section 34074-5
Drug/Laboratory test interactions: Several reports of possible interference with digoxin radioimmunoassay by spironolactone, or its metabolites, have appeared in the literature. Neither the extent nor the potential clinical significance of its interference (which may be assay-specific) has been fully established.
Section 34075-2
Laboratory tests: Periodic determination of serum electrolytes to detect possible electrolyte imbalance should be done at appropriate intervals, particularly in the elderly and those with significant renal or hepatic impairments.
Section 34076-0
Information for patients: Patients who receive spironolactone should be advised to avoid potassium supplements and foods containing high levels of potassium, including salt substitutes.
Section 34080-2
Nursing mothers: Canrenone, a major (and active) metabolite of spironolactone, appears in human breast milk. Because spironolactone has been found to be tumorigenic in rats, a decision should be made whether to discontinue the drug, taking into account the importance of the drug to the mother. If use of the drug is deemed essential, an alternative method of infant feeding should be instituted.
Section 34081-0
Pediatric use: Safety and effectiveness in pediatric patients have not been established.
Section 34083-6
Carcinogenesis, mutagenesis, impairment of fertility: Orally administered spironolactone has been shown to be a tumorigen in dietary administration studies performed in rats, with its proliferative effects manifested on endocrine organs and the liver. In an 18-month study using doses of about 50, 150, and 500 mg/kg/day, there were statistically significant increases in benign adenomas of the thyroid and testes and, in male rats, a dose-related increase in proliferative changes in the liver (including hepatocytomegaly and hyperplastic nodules). In a 24-month study in which the same strain of rat was administered doses of about 10, 30, 100, and 150 mg spironolactone/kg/day, the range of proliferative effects included significant increases in hepatocellular adenomas and testicular interstitial cell tumors in males, and significant increases in thyroid follicular cell adenomas and carcinomas in both sexes. There was also a statistically significant, but not dose-related, increase in benign uterine endometrial stromal polyps in females.
A dose-related (above 20 mg/kg/day) incidence of myelocytic leukemia was observed in rats fed daily doses of potassium canrenoate (a compound chemically similar to spironolactone and whose primary metabolite, canrenone, is also a major product of spironolactone in man) for a period of one year. In two-year studies in the rat, oral administration of potassium canrenoate was associated with myelocytic leukemia and hepatic, thyroid, testicular, and mammary tumors.
Neither spironolactone nor potassium canrenoate produced mutagenic effects in tests using bacteria or yeast. In the absence of metabolic activation, neither spironolactone nor potassium canrenoate has been shown to be mutagenic in mammalian tests in vitro. In the presence of metabolic activation, spironolactone has been reported to be negative in some mammalian mutagenicity tests in vitro and inconclusive (but slightly positive) for mutagenicity in other mammalian tests in vitro. In the presence of metabolic activation, potassium canrenoate has been reported to test positive for mutagenicity in some mammalian tests in vitro, inconclusive in others, and negative in still others.
In a three-litter reproduction study in which female rats received dietary doses of 15 and 50 mg spironolactone/kg/day, there were no effects on mating and fertility, but there was a small increase in incidence of stillborn pups at 50 mg/kg/day. When injected into female rats (100 mg/kg/day for 7 days, i.p.), spironolactone was found to increase the length of the estrous cycle by prolonging diestrus during treatment and inducing constant diestrus during a two-week post-treatment observation period. These effects were associated with retarded ovarian follicle development and a reduction in circulating estrogen levels, which would be expected to impair mating, fertility and fecundity. Spironolactone (100 mg/kg/day), administered i.p. to female mice during a two-week cohabitation period with untreated males, decreased the number of mated mice that conceived (effect shown to be caused by an inhibition of ovulation) and decreased the number of implanted embryos in those that became pregnant (effect shown to be caused by an inhibition of implantation), and at 200 mg/kg, also increased the latency period to mating.
Section 42229-5
Aldosterone antagonist activity: Increased levels of the mineralocorticoid, aldosterone, are present in primary and secondary hyperaldosteronism. Edematous states in which secondary aldosteronism is usually involved include congestive heart failure, hepatic cirrhosis, and nephrotic syndrome. By competing with aldosterone for receptor sites, spironolactone provides effective therapy for the edema and ascites in those conditions. Spironolactone counteracts secondary aldosteronism induced by the volume depletion and associated sodium loss caused by active diuretic therapy.
Spironolactone is effective in lowering the systolic and diastolic blood pressure in patients with primary hyperaldosteronism. It is also effective in most cases of essential hypertension, despite the fact that aldosterone secretion may be within normal limits in benign essential hypertension.
Through its action in antagonizing the effect of aldosterone, spironolactone inhibits the exchange of sodium for potassium in the distal renal tubule and helps to prevent potassium loss.
Spironolactone has not been demonstrated to elevate serum uric acid, to precipitate gout, or to alter carbohydrate metabolism.
Section 43679-0
Mechanism of action: Spironolactone is a specific pharmacologic antagonist of aldosterone, acting primarily through competitive binding of receptors at the aldosterone-dependent sodium-potassium exchange site in the distal convoluted renal tubule. Spironolactone causes increased amounts of sodium and water to be excreted, while potassium is retained. Spironolactone acts both as a diuretic and as an antihypertensive drug by this mechanism. It may be given alone or with other diuretic agents that act more proximally in the renal tubule.
Section 43682-4
Pharmacokinetics: Spironolactone is rapidly and extensively metabolized. Sulfur-containing products are the predominant metabolites and are thought to be primarily responsible, together with spironolactone, for the therapeutic effects of the drug. The following pharmacokinetic data were obtained from 12 healthy volunteers following the administration of 100 mg of spironolactone (film-coated tablets) daily for 15 days. On the 15th day, spironolactone was given immediately after a low-fat breakfast and blood was drawn thereafter.
| Accumulation Factor:
AUC (0–24 hr, day 15)/AUC (0–24 hr, day 1) |
Mean Peak Serum Concentration | Mean (SD) Post Steady- State Half-Life | |
|---|---|---|---|
| 7-α-(thiomethyl) spirolactone (TMS) | 1.25 | 391 ng/mL at 3.2 hr | 13.8 hr (6.4) (terminal) |
| 6-β-hydroxy-7-α-(thiomethyl) spirolactone (HTMS) | 1.50 | 125 ng/mL at 5.1 hr | 15.0 hr (4.0) (terminal) |
| Canrenone (C) | 1.41 | 181 ng/mL at 4.3 hr | 16.5 hr (6.3) (terminal) |
| Spironolactone | 1.30 | 80 ng/mL at 2.6 hr | Approximately 1.4 hr (0.5) (β half-life) |
The pharmacological activity of spironolactone metabolites in man is not known. However, in the adrenalectomized rat the antimineralocorticoid activities of the metabolites C, TMS, and HTMS, relative to spironolactone were 1.10, 1.28, and 0.32, respectively. Relative to spironolactone, their binding affinities to the aldosterone receptors in rat kidney slices were 0.19, 0.86, and 0.06, respectively.
In humans, the potencies of TMS and 7-α-thiospirolactone in reversing the effects of the synthetic mineralocorticoid, fludrocortisone, on urinary electrolyte composition were 0.33 and 0.26, respectively, relative to spironolactone. However, since the serum concentrations of these steroids were not determined, their incomplete absorption and/or first-pass metabolism could not be ruled out as a reason for their reduced in vivo activities.
Spironolactone and its metabolites are more than 90% bound to plasma proteins. The metabolites are excreted primarily in the urine and secondarily in bile.
The effect of food on spironolactone absorption (two 100 mg spironolactone tablets) was assessed in a single-dose study of 9 healthy, drug-free volunteers. Food increased the bioavailability of unmetabolized spironolactone by almost 100%. The clinical importance of this finding is not known.
Warning
Spironolactone has been shown to be a tumorigen in chronic toxicity studies in rats (see Precautions ). Spironolactone should be used only in those conditions described under Indications and Usage . Unnecessary use of this drug should be avoided.
Overdosage
The oral LD 50 of spironolactone is greater than 1000 mg/kg in mice, rats, and rabbits.
Acute overdosage of spironolactone may be manifested by drowsiness, mental confusion, maculopapular or erythematous rash, nausea, vomiting, dizziness, or diarrhea. Rarely, instances of hyponatremia, hyperkalemia, or hepatic coma may occur in patients with severe liver disease, but these are unlikely due to acute overdosage. Hyperkalemia may occur, especially in patients with impaired renal function.
Hypokalemia
For the treatment of patients with hypokalemia when other measures are considered inappropriate or inadequate. Spironolactone tablets are also indicated for the prophylaxis of hypokalemia in patients taking digitalis when other measures are considered inadequate or inappropriate.
Essential Hypertension
Spironolactone tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Usually in combination with other drugs, spironolactone tablets are indicated for patients who cannot be treated adequately with other agents or for whom other agents are considered inappropriate.
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 50 mg | 100 mg |
| 30 | 71610-287-30 | 71610-288-30 |
| 45 | 71610-287-45 | 71610-288-45 |
| 90 | - | 71610-288-60 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20190603JH
Principal Display Panel 50 Mg
NDC 71610-287 - Spironolactone, USP 50 mg Tablets - Rx Only
Principal Display Panel 100 Mg
NDC 71610-288 - Spironolactone, USP 100 mg Tablets - Rx Only
Severe Heart Failure (nyha Class Iii – Iv)
To increase survival, and to reduce the need for hospitalization for heart failure when used in addition to standard therapy.
Structured Label Content
Indications and Usage (INDICATIONS AND USAGE)
Spironolactone tablets are indicated in the management of:
Primary hyperaldosteronism for:
Establishing the diagnosis of primary hyperaldosteronism by therapeutic trial.
Short-term preoperative treatment of patients with primary hyperaldosteronism.
Long-term maintenance therapy for patients with discrete aldosterone-producing adrenal adenomas who are judged to be poor operative risks or who decline surgery.
Long-term maintenance therapy for patients with bilateral micro or macronodular adrenal hyperplasia (idiopathic hyperaldosteronism).
Edematous conditions for patients with:
Contraindications (CONTRAINDICATIONS)
Spironolactone is contraindicated for patients with anuria, acute renal insufficiency, significant impairment of renal excretory function, hyperkalemia, Addison’s disease, and with concomitant use of eplerenone.
Adverse Reactions (ADVERSE REACTIONS)
The following adverse reactions have been reported and, within each category (body system), are listed in order of decreasing severity.
Digestive: Gastric bleeding, ulceration, gastritis, diarrhea and cramping, nausea, vomiting.
Reproductive: Gynecomastia (see Precautions ), inability to achieve or maintain erection, irregular menses or amenorrhea, postmenopausal bleeding, breast pain. Carcinoma of the breast has been reported in patients taking spironolactone but a cause and effect relationship has not been established.
Hematologic: Leukopenia (including agranulocytosis), thrombocytopenia.
Hypersensitivity: Fever, urticaria, maculopapular or erythematous cutaneous eruptions, anaphylactic reactions, vasculitis.
Metabolism: Hyperkalemia, electrolyte disturbances (see Warnings and Precautions).
Musculoskeletal: Leg cramps.
Nervous system /psychiatric: Lethargy, mental confusion, ataxia, dizziness, headache, drowsiness.
Liver / biliary: A very few cases of mixed cholestatic/hepatocellular toxicity, with one reported fatality, have been reported with spironolactone administration.
Renal: Renal dysfunction (including renal failure).
Skin: Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug rash with eosinophilia and systemic symptoms (DRESS), alopecia, pruritis.
How Supplied (HOW SUPPLIED)
Spironolactone Tablets USP 25 mg tablets are Light yellow to yellow colored, round, biconvex, film coated tablets with inscription “AD” on one side and plain on the other side having faint odour of peppermint, supplied as:
| NDC Number | Size |
|---|---|
| 16729-225-01 | bottle of 100 |
| 16729-225-16 | bottle of 500 |
| 16729-225-17 | bottle of 1,000 |
| 16729-225-19 | bottle of 2,500 |
Spironolactone Tablets USP 50 mg tablets are Light orange to orange colored, oval, biconvex, film coated tablets with inscription “AE” on one side and breakline on the other side having faint odour of peppermint, supplied as:
| NDC Number | Size |
|---|---|
| 16729-226-01 | bottle of 100 |
| 16729-226-16 | bottle of 500 |
Spironolactone Tablets USP 100 mg tablets are Light peach to peach colored, round, biconvex, film coated tablets with inscription “AF” on one side and breakline on the other side having faint odour of peppermint, supplied as:
| NDC Number | Size |
|---|---|
| 16729-227-01 | bottle of 100 |
| 16729-227-16 | bottle of 500 |
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
Protect from light. Dispense in tight, Light-resistant containers.
Manufactured For:
Accord Healthcare, Inc.,
1009 Slater Road,
Suite 210-B,
Durham, NC 27703,
USA.
Manufactured By:
Intas Pharmaceuticals Limited,
Plot No 5 to 12, Pharmez,
Sarkhej-Bavla, National Highway No 8-A,
Near Village Matoda, Tal Sanand,
Ahmedabad - 382 213, Gujarat, India
51 2267 0 700753
Issued November 2016.
Description (DESCRIPTION)
Spironolactone tablets USP, for oral administration contain 25 mg, 50 mg, or 100 mg of the aldosterone antagonist spironolactone, 17-hydroxy-7α-mercapto-3-oxo-17α-pregn-4-ene-21-carboxylic acid γ-lactone acetate, which has the following structural formula:
Spironolactone is practically insoluble in water, soluble in alcohol, and freely soluble in benzene and in chloroform.
Inactive ingredients include lactose monohydrate, dibasic calcium phosphate, povidone, peppermint oil, purified talc, pregelatinised starch, colloidal anhydrous silica, magnesium stearate, hypromellose, polyethylene glycol 400, titanium dioxide and iron oxide yellow. In addition, iron oxide red (50 mg and 100 mg tablets) is included in the film coating of specific strengths.
Section 34072-9 (34072-9)
General: All patients receiving diuretic therapy should be observed for evidence of fluid or electrolyte imbalance, e.g., hypomagnesemia, hyponatremia, hypochloremic alkalosis, and hyperkalemia.
Serum and urine electrolyte determinations are particularly important when the patient is vomiting excessively or receiving parenteral fluids. Warning signs or symptoms of fluid and electrolyte imbalance, irrespective of cause, include dryness of the mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances such as nausea and vomiting. Hyperkalemia may occur in patients with impaired renal function or excessive potassium intake and can cause cardiac irregularities, which may be fatal. Consequently, no potassium supplement should ordinarily be given with spironolactone.
If hyperkalemia is suspected (warning signs include paresthesia, muscle weakness, fatigue, flaccid paralysis of the extremities, bradycardia and shock), an electrocardiogram (ECG) should be obtained. However, it is important to monitor serum potassium levels because mild hyperkalemia may not be associated with ECG changes.
If hyperkalemia is present, spironolactone should be discontinued immediately. With severe hyperkalemia, the clinical situation dictates the procedures to be employed. These may include the intravenous administration of calcium chloride solution, sodium bicarbonate solution and/or the oral or parenteral administration of glucose with a rapid-acting insulin preparation. These are temporary measures to be repeated as required. Cationic exchange resins such as sodium polystyrene sulfonate may be orally or rectally administered. Persistent hyperkalemia may require dialysis.
Reversible hyperchloremic metabolic acidosis, usually in association with hyperkalemia, has been reported to occur in some patients with decompensated hepatic cirrhosis, even in the presence of normal renal function.
Dilutional hyponatremia, manifested by dryness of the mouth, thirst, lethargy, and drowsiness, and confirmed by a low serum sodium level, may be caused or aggravated, especially when spironolactone is administered in combination with other diuretics, and dilutional hyponatremia may occur in edematous patients in hot weather; appropriate therapy is water restriction rather than administration of sodium, except in rare instances when the hyponatremia is life-threatening.
Spironolactone therapy may cause a transient elevation of BUN, especially in patients with pre-existing renal impairment. Spironolactone tablets may cause mild acidosis.
Gynecomastia may develop in association with the use of spironolactone; physicians should be alert to its possible onset. The development of gynecomastia appears to be related to both dosage level and duration of therapy and is normally reversible when spironolactone is discontinued. In rare instances, some breast enlargement may persist when spironolactone is discontinued.
Somnolence and dizziness have been reported to occur in some patients. Caution is advised when driving or operating machinery until the response to initial treatment has been determined.
Section 34074-5 (34074-5)
Drug/Laboratory test interactions: Several reports of possible interference with digoxin radioimmunoassay by spironolactone, or its metabolites, have appeared in the literature. Neither the extent nor the potential clinical significance of its interference (which may be assay-specific) has been fully established.
Section 34075-2 (34075-2)
Laboratory tests: Periodic determination of serum electrolytes to detect possible electrolyte imbalance should be done at appropriate intervals, particularly in the elderly and those with significant renal or hepatic impairments.
Section 34076-0 (34076-0)
Information for patients: Patients who receive spironolactone should be advised to avoid potassium supplements and foods containing high levels of potassium, including salt substitutes.
Section 34080-2 (34080-2)
Nursing mothers: Canrenone, a major (and active) metabolite of spironolactone, appears in human breast milk. Because spironolactone has been found to be tumorigenic in rats, a decision should be made whether to discontinue the drug, taking into account the importance of the drug to the mother. If use of the drug is deemed essential, an alternative method of infant feeding should be instituted.
Section 34081-0 (34081-0)
Pediatric use: Safety and effectiveness in pediatric patients have not been established.
Section 34083-6 (34083-6)
Carcinogenesis, mutagenesis, impairment of fertility: Orally administered spironolactone has been shown to be a tumorigen in dietary administration studies performed in rats, with its proliferative effects manifested on endocrine organs and the liver. In an 18-month study using doses of about 50, 150, and 500 mg/kg/day, there were statistically significant increases in benign adenomas of the thyroid and testes and, in male rats, a dose-related increase in proliferative changes in the liver (including hepatocytomegaly and hyperplastic nodules). In a 24-month study in which the same strain of rat was administered doses of about 10, 30, 100, and 150 mg spironolactone/kg/day, the range of proliferative effects included significant increases in hepatocellular adenomas and testicular interstitial cell tumors in males, and significant increases in thyroid follicular cell adenomas and carcinomas in both sexes. There was also a statistically significant, but not dose-related, increase in benign uterine endometrial stromal polyps in females.
A dose-related (above 20 mg/kg/day) incidence of myelocytic leukemia was observed in rats fed daily doses of potassium canrenoate (a compound chemically similar to spironolactone and whose primary metabolite, canrenone, is also a major product of spironolactone in man) for a period of one year. In two-year studies in the rat, oral administration of potassium canrenoate was associated with myelocytic leukemia and hepatic, thyroid, testicular, and mammary tumors.
Neither spironolactone nor potassium canrenoate produced mutagenic effects in tests using bacteria or yeast. In the absence of metabolic activation, neither spironolactone nor potassium canrenoate has been shown to be mutagenic in mammalian tests in vitro. In the presence of metabolic activation, spironolactone has been reported to be negative in some mammalian mutagenicity tests in vitro and inconclusive (but slightly positive) for mutagenicity in other mammalian tests in vitro. In the presence of metabolic activation, potassium canrenoate has been reported to test positive for mutagenicity in some mammalian tests in vitro, inconclusive in others, and negative in still others.
In a three-litter reproduction study in which female rats received dietary doses of 15 and 50 mg spironolactone/kg/day, there were no effects on mating and fertility, but there was a small increase in incidence of stillborn pups at 50 mg/kg/day. When injected into female rats (100 mg/kg/day for 7 days, i.p.), spironolactone was found to increase the length of the estrous cycle by prolonging diestrus during treatment and inducing constant diestrus during a two-week post-treatment observation period. These effects were associated with retarded ovarian follicle development and a reduction in circulating estrogen levels, which would be expected to impair mating, fertility and fecundity. Spironolactone (100 mg/kg/day), administered i.p. to female mice during a two-week cohabitation period with untreated males, decreased the number of mated mice that conceived (effect shown to be caused by an inhibition of ovulation) and decreased the number of implanted embryos in those that became pregnant (effect shown to be caused by an inhibition of implantation), and at 200 mg/kg, also increased the latency period to mating.
Section 42229-5 (42229-5)
Aldosterone antagonist activity: Increased levels of the mineralocorticoid, aldosterone, are present in primary and secondary hyperaldosteronism. Edematous states in which secondary aldosteronism is usually involved include congestive heart failure, hepatic cirrhosis, and nephrotic syndrome. By competing with aldosterone for receptor sites, spironolactone provides effective therapy for the edema and ascites in those conditions. Spironolactone counteracts secondary aldosteronism induced by the volume depletion and associated sodium loss caused by active diuretic therapy.
Spironolactone is effective in lowering the systolic and diastolic blood pressure in patients with primary hyperaldosteronism. It is also effective in most cases of essential hypertension, despite the fact that aldosterone secretion may be within normal limits in benign essential hypertension.
Through its action in antagonizing the effect of aldosterone, spironolactone inhibits the exchange of sodium for potassium in the distal renal tubule and helps to prevent potassium loss.
Spironolactone has not been demonstrated to elevate serum uric acid, to precipitate gout, or to alter carbohydrate metabolism.
Section 43679-0 (43679-0)
Mechanism of action: Spironolactone is a specific pharmacologic antagonist of aldosterone, acting primarily through competitive binding of receptors at the aldosterone-dependent sodium-potassium exchange site in the distal convoluted renal tubule. Spironolactone causes increased amounts of sodium and water to be excreted, while potassium is retained. Spironolactone acts both as a diuretic and as an antihypertensive drug by this mechanism. It may be given alone or with other diuretic agents that act more proximally in the renal tubule.
Section 43682-4 (43682-4)
Pharmacokinetics: Spironolactone is rapidly and extensively metabolized. Sulfur-containing products are the predominant metabolites and are thought to be primarily responsible, together with spironolactone, for the therapeutic effects of the drug. The following pharmacokinetic data were obtained from 12 healthy volunteers following the administration of 100 mg of spironolactone (film-coated tablets) daily for 15 days. On the 15th day, spironolactone was given immediately after a low-fat breakfast and blood was drawn thereafter.
| Accumulation Factor:
AUC (0–24 hr, day 15)/AUC (0–24 hr, day 1) |
Mean Peak Serum Concentration | Mean (SD) Post Steady- State Half-Life | |
|---|---|---|---|
| 7-α-(thiomethyl) spirolactone (TMS) | 1.25 | 391 ng/mL at 3.2 hr | 13.8 hr (6.4) (terminal) |
| 6-β-hydroxy-7-α-(thiomethyl) spirolactone (HTMS) | 1.50 | 125 ng/mL at 5.1 hr | 15.0 hr (4.0) (terminal) |
| Canrenone (C) | 1.41 | 181 ng/mL at 4.3 hr | 16.5 hr (6.3) (terminal) |
| Spironolactone | 1.30 | 80 ng/mL at 2.6 hr | Approximately 1.4 hr (0.5) (β half-life) |
The pharmacological activity of spironolactone metabolites in man is not known. However, in the adrenalectomized rat the antimineralocorticoid activities of the metabolites C, TMS, and HTMS, relative to spironolactone were 1.10, 1.28, and 0.32, respectively. Relative to spironolactone, their binding affinities to the aldosterone receptors in rat kidney slices were 0.19, 0.86, and 0.06, respectively.
In humans, the potencies of TMS and 7-α-thiospirolactone in reversing the effects of the synthetic mineralocorticoid, fludrocortisone, on urinary electrolyte composition were 0.33 and 0.26, respectively, relative to spironolactone. However, since the serum concentrations of these steroids were not determined, their incomplete absorption and/or first-pass metabolism could not be ruled out as a reason for their reduced in vivo activities.
Spironolactone and its metabolites are more than 90% bound to plasma proteins. The metabolites are excreted primarily in the urine and secondarily in bile.
The effect of food on spironolactone absorption (two 100 mg spironolactone tablets) was assessed in a single-dose study of 9 healthy, drug-free volunteers. Food increased the bioavailability of unmetabolized spironolactone by almost 100%. The clinical importance of this finding is not known.
Warning (WARNING)
Spironolactone has been shown to be a tumorigen in chronic toxicity studies in rats (see Precautions ). Spironolactone should be used only in those conditions described under Indications and Usage . Unnecessary use of this drug should be avoided.
Overdosage (OVERDOSAGE)
The oral LD 50 of spironolactone is greater than 1000 mg/kg in mice, rats, and rabbits.
Acute overdosage of spironolactone may be manifested by drowsiness, mental confusion, maculopapular or erythematous rash, nausea, vomiting, dizziness, or diarrhea. Rarely, instances of hyponatremia, hyperkalemia, or hepatic coma may occur in patients with severe liver disease, but these are unlikely due to acute overdosage. Hyperkalemia may occur, especially in patients with impaired renal function.
Hypokalemia
For the treatment of patients with hypokalemia when other measures are considered inappropriate or inadequate. Spironolactone tablets are also indicated for the prophylaxis of hypokalemia in patients taking digitalis when other measures are considered inadequate or inappropriate.
Essential Hypertension (Essential hypertension)
Spironolactone tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Usually in combination with other drugs, spironolactone tablets are indicated for patients who cannot be treated adequately with other agents or for whom other agents are considered inappropriate.
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 50 mg | 100 mg |
| 30 | 71610-287-30 | 71610-288-30 |
| 45 | 71610-287-45 | 71610-288-45 |
| 90 | - | 71610-288-60 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20190603JH
Principal Display Panel 50 Mg (PRINCIPAL DISPLAY PANEL - 50 mg)
NDC 71610-287 - Spironolactone, USP 50 mg Tablets - Rx Only
Principal Display Panel 100 Mg (PRINCIPAL DISPLAY PANEL - 100 mg)
NDC 71610-288 - Spironolactone, USP 100 mg Tablets - Rx Only
Severe Heart Failure (nyha Class Iii – Iv) (Severe heart failure (NYHA class III – IV))
To increase survival, and to reduce the need for hospitalization for heart failure when used in addition to standard therapy.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:44.152674 · Updated: 2026-03-14T21:41:24.246163