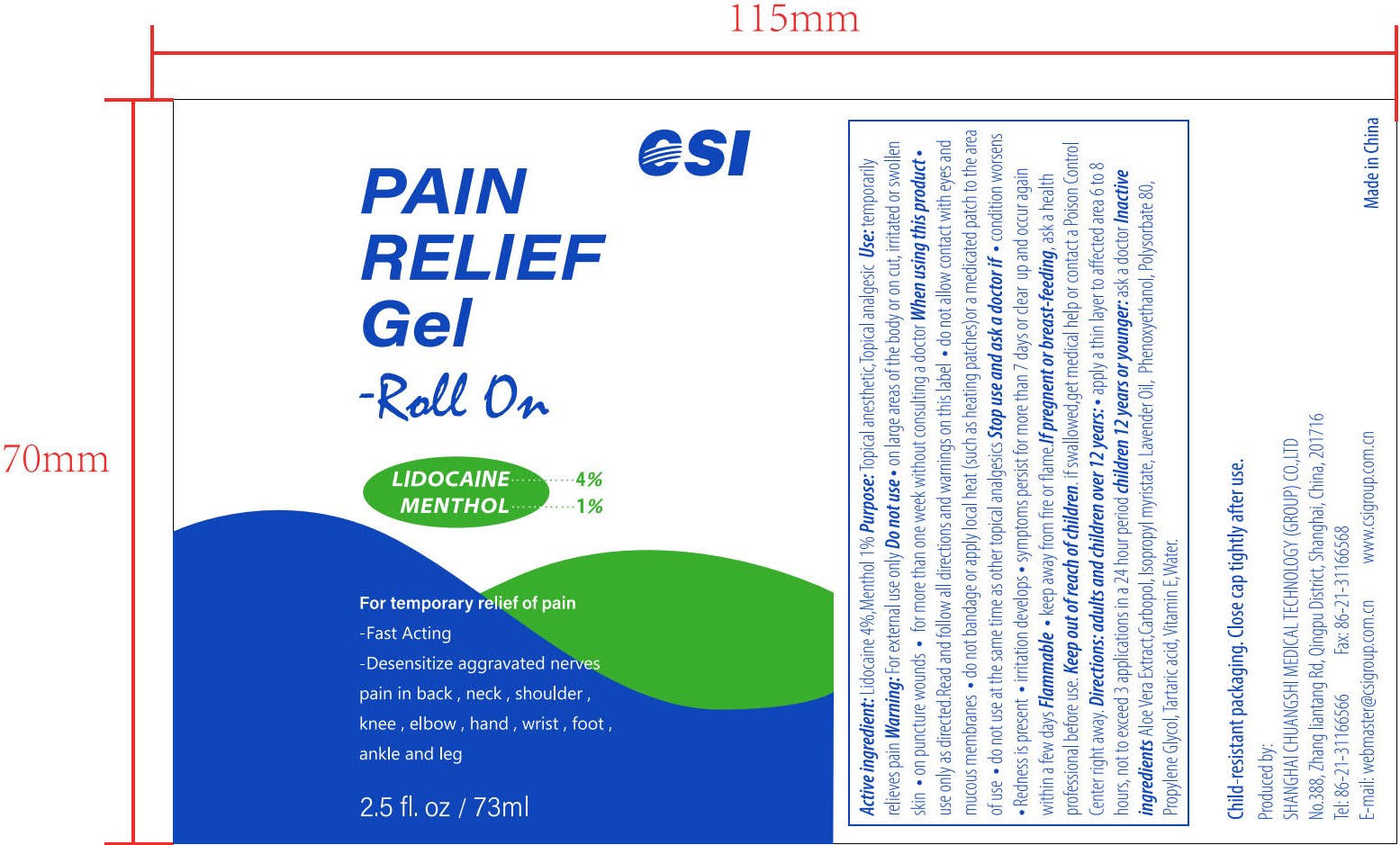
csi, lidocaine and menthol, pain relief gel-roll on, 2.5 fl. oz / 73 ml, gel, bottle
f02fa9c0-b8af-f362-e053-2a95a90ae516
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
(Lidocaine) Topical Anesthetic (Menthol) Topical Analgesic
Description
Lidocaine 4.0% (w/w ...... Purpose: Topical Anesthetic) Menthol 1.0% (w/w ...... Purpose: Topical Analgesic)
Medication Information
Warnings
For external use only
Purpose
(Lidocaine) Topical Anesthetic
(Menthol) Topical Analgesic
Directions
Adults and children 12 over years :
• apply a thin layer to affected area 6 to 8 hours, not to exceed 3 application in a 24 hour period
Children under 12 years of age : Consult a doctor
Inactive Ingredients
Aloe Vera Extract, Carbopol, Isopropyl myristate, Lavender Oil, Phenoxyethanol, Polysorbate 80, Propylene Glycol, Tartaric acid, Vitamin E, Water
Description
Lidocaine 4.0% (w/w ...... Purpose: Topical Anesthetic) Menthol 1.0% (w/w ...... Purpose: Topical Analgesic)
Use
temporary relief of pain
Section 34072-9
Child-resistant packaging. Close cap tightly after use.
Section 51945-4
Flammable
• keep away from fire or flame
Do Not Use
• on large areas of the body or on cut, irritated or swollen skin not apply to wounds or damaged skin • on puncture wounds • for more than one week without consulting a doctor
Stop Use If
• condition worsens • redness is present • irritation develops • symptoms persist for more than 7 days or clear up and occur within a few days
Ask A Doctor If
• condition worsens • redness is present • irritation develops • symptoms persist for more than 7 days or clear up and occur within a few days
Active Ingredient
Lidocaine 4.0% (w/w ...... Purpose: Topical Anesthetic)
Menthol 1.0% (w/w ...... Purpose: Topical Analgesic)
Questions Or Comments
86-21-31166566
When Using This Product
• use only as directed. Read and follow all directions and warnings on this label • do not allow contact with eyes and mucous membranes • do not bandage or apply local heat (such as heating patches) or a medicated patch to the area of use • do not use at the same time as other topical analgesics
If Pregnant Or Breast Feeding
Ask a health professional before use
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away
Structured Label Content
Warnings
For external use only
Purpose
(Lidocaine) Topical Anesthetic
(Menthol) Topical Analgesic
Directions
Adults and children 12 over years :
• apply a thin layer to affected area 6 to 8 hours, not to exceed 3 application in a 24 hour period
Children under 12 years of age : Consult a doctor
Inactive Ingredients
Aloe Vera Extract, Carbopol, Isopropyl myristate, Lavender Oil, Phenoxyethanol, Polysorbate 80, Propylene Glycol, Tartaric acid, Vitamin E, Water
Use
temporary relief of pain
Section 34072-9 (34072-9)
Child-resistant packaging. Close cap tightly after use.
Section 51945-4 (51945-4)
Flammable
• keep away from fire or flame
Do Not Use (Do not use)
• on large areas of the body or on cut, irritated or swollen skin not apply to wounds or damaged skin • on puncture wounds • for more than one week without consulting a doctor
Stop Use If (Stop use if)
• condition worsens • redness is present • irritation develops • symptoms persist for more than 7 days or clear up and occur within a few days
Ask A Doctor If (Ask a doctor if)
• condition worsens • redness is present • irritation develops • symptoms persist for more than 7 days or clear up and occur within a few days
Active Ingredient
Lidocaine 4.0% (w/w ...... Purpose: Topical Anesthetic)
Menthol 1.0% (w/w ...... Purpose: Topical Analgesic)
Questions Or Comments (Questions or Comments)
86-21-31166566
When Using This Product (When using this product)
• use only as directed. Read and follow all directions and warnings on this label • do not allow contact with eyes and mucous membranes • do not bandage or apply local heat (such as heating patches) or a medicated patch to the area of use • do not use at the same time as other topical analgesics
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
Ask a health professional before use
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:46.534106 · Updated: 2026-03-14T23:04:01.123954