These Highlights Do Not Include All The Information Needed To Use Firdapse®
f015fe60-3128-4a43-8c31-19fc6b5def3f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 ) 5/2024
Indications and Usage
FIRDAPSE ® is indicated for the treatment of Lambert-Eaton myasthenic syndrome (LEMS) in adults and pediatric patients 6 years of age and older.
Dosage and Administration
Administer orally in divided doses (3 to 5 times daily). ( 2.1 ) The recommended starting dosage for adults (any weight) and pediatric patients weighing 45 kg or more is 15 mg to 30 mg daily, in divided doses. ( 2.1 ) Dosage can be increased by 5 mg daily every 3 to 4 days. ( 2.1 ) The maximum single dose is 20 mg. ( 2.1 ) Dosage is not to exceed a maximum of 100 mg daily. ( 2.1 ) The recommended starting dosage for pediatric patients weighing less than 45 kg is 5 mg to 15 mg daily, in divided doses. ( 2.1 ) Dosage can be increased by 2.5 mg daily every 3 to 4 days. ( 2.1 ) The maximum single dose is 10 mg. ( 2.1 ) Dosage is not to exceed a maximum of 50 mg daily. ( 2.1 ) The recommended starting dosage for adult and pediatric patients with renal impairment, hepatic impairment, and in known N-acetyltransferase 2 (NAT2) poor metabolizers is the lowest recommended initial daily dosage ( 2.1 , 2.2 , 2.3 , 2.4 ) For patients with a dosage adjustment of less than 5 mg increments, or who have difficulty swallowing, or require feeding tube, a l mg/mL suspension can be prepared. ( 2.5 )
Warnings and Precautions
Seizures: FIRDAPSE can cause seizures. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment. ( 5.1 ) Hypersensitivity reactions: If a hypersensitivity reaction such as anaphylaxis occurs, FIRDAPSE should be discontinued and appropriate therapy initiated. ( 5.2 )
Contraindications
FIRDAPSE is contraindicated in patients with: A history of seizures [see Warnings and Precautions ( 5.1 )] Hypersensitivity to amifampridine phosphate or another aminopyridine [see Warnings and Precautions ( 5.2 )]
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Seizures [see Warnings and Precautions ( 5.1 )] Hypersensitivity [see Warnings and Precautions ( 5.2 )]
Drug Interactions
Drugs that lower seizure threshold: Concomitant use may lead to an increased risk of seizures. ( 7.1 ) Drugs with cholinergic effects (e.g., direct or indirect cholinesterase inhibitors): Concomitant use may increase the cholinergic effects of FIRDAPSE and of those drugs and increase the risk of adverse reactions. ( 7.2 )
Storage and Handling
Store FIRDAPSE tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP controlled room temperature]. See Dosage and Administration ( 2.5 ) for storage instructions for FIRDAPSE prepared suspension.
How Supplied
FIRDAPSE 10 mg tablets are white to off white-round, and functionally scored. Each tablet is debossed on the non-scored side with “CATALYST” and on the scored side with “211” above the score and “10” below the score. Tablets can be divided in half at the score. FIRDAPSE is supplied as follows:
Medication Information
Warnings and Precautions
Seizures: FIRDAPSE can cause seizures. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment. ( 5.1 ) Hypersensitivity reactions: If a hypersensitivity reaction such as anaphylaxis occurs, FIRDAPSE should be discontinued and appropriate therapy initiated. ( 5.2 )
Indications and Usage
FIRDAPSE ® is indicated for the treatment of Lambert-Eaton myasthenic syndrome (LEMS) in adults and pediatric patients 6 years of age and older.
Dosage and Administration
Administer orally in divided doses (3 to 5 times daily). ( 2.1 ) The recommended starting dosage for adults (any weight) and pediatric patients weighing 45 kg or more is 15 mg to 30 mg daily, in divided doses. ( 2.1 ) Dosage can be increased by 5 mg daily every 3 to 4 days. ( 2.1 ) The maximum single dose is 20 mg. ( 2.1 ) Dosage is not to exceed a maximum of 100 mg daily. ( 2.1 ) The recommended starting dosage for pediatric patients weighing less than 45 kg is 5 mg to 15 mg daily, in divided doses. ( 2.1 ) Dosage can be increased by 2.5 mg daily every 3 to 4 days. ( 2.1 ) The maximum single dose is 10 mg. ( 2.1 ) Dosage is not to exceed a maximum of 50 mg daily. ( 2.1 ) The recommended starting dosage for adult and pediatric patients with renal impairment, hepatic impairment, and in known N-acetyltransferase 2 (NAT2) poor metabolizers is the lowest recommended initial daily dosage ( 2.1 , 2.2 , 2.3 , 2.4 ) For patients with a dosage adjustment of less than 5 mg increments, or who have difficulty swallowing, or require feeding tube, a l mg/mL suspension can be prepared. ( 2.5 )
Contraindications
FIRDAPSE is contraindicated in patients with: A history of seizures [see Warnings and Precautions ( 5.1 )] Hypersensitivity to amifampridine phosphate or another aminopyridine [see Warnings and Precautions ( 5.2 )]
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Seizures [see Warnings and Precautions ( 5.1 )] Hypersensitivity [see Warnings and Precautions ( 5.2 )]
Drug Interactions
Drugs that lower seizure threshold: Concomitant use may lead to an increased risk of seizures. ( 7.1 ) Drugs with cholinergic effects (e.g., direct or indirect cholinesterase inhibitors): Concomitant use may increase the cholinergic effects of FIRDAPSE and of those drugs and increase the risk of adverse reactions. ( 7.2 )
Storage and Handling
Store FIRDAPSE tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP controlled room temperature]. See Dosage and Administration ( 2.5 ) for storage instructions for FIRDAPSE prepared suspension.
How Supplied
FIRDAPSE 10 mg tablets are white to off white-round, and functionally scored. Each tablet is debossed on the non-scored side with “CATALYST” and on the scored side with “211” above the score and “10” below the score. Tablets can be divided in half at the score. FIRDAPSE is supplied as follows:
Description
Dosage and Administration ( 2.1 ) 5/2024
Section 42229-5
Missed Dose
If a dose is missed, patients should not take double or extra doses.
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised: 5/2024 |
|
MEDICATION GUIDE
FIRDAPSE ® (FIR-dapse) (amifampridine) tablets, for oral use |
|
| Read this Medication Guide before you start taking FIRDAPSE and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. | |
|
What is the most important information I should know about FIRDAPSE?
|
|
| Stop taking FIRDAPSE and call your doctor right away if you have a seizure while taking FIRDAPSE. | |
|
What is FIRDAPSE?
FIRDAPSE is a prescription medicine used to treat Lambert-Eaton myasthenic syndrome (LEMS) in people 6 years of age and older. It is not known if FIRDAPSE is safe or effective in children less than 6 years of age. |
|
Do not take FIRDAPSE if you:
|
|
Before you take FIRDAPSE, tell your doctor about all of your medical conditions. including if you:
|
|
| Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. | |
How should I take FIRDAPSE?
|
|
|
What are the possible side effects of FIRDAPSE?
FIRDAPSE may cause serious side effects, including:
|
|
The most common side effects of FIRDAPSE include:
|
|
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of FIRDAPSE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store FIRDAPSE?
|
|
| Keep FIRDAPSE and all medicines out of the reach of children. | |
|
General Information about the safe and effective use of FIRDAPSE
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use FIRDAPSE for a condition for which it was not prescribed. Do not give FIRDAPSE to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk to your doctor or pharmacist. You can ask your pharmacist or doctor for information about FIRDAPSE that is written for health professionals. |
|
|
What are the ingredients in FIRDAPSE?
Active ingredient: amifampridine Inactive ingredients: calcium stearate, colloidal silicon dioxide, and microcrystalline cellulose. Distributed by Catalyst Pharmaceuticals, Inc., Coral Gables, FL 33134 For more information, go to www.YourCatalystPathways.com or call 1-833-422-8259 |
Section 43683-2
| Dosage and Administration (2.1) | 5/2024 |
Section 59845-8
Instructions for Use
FIRDAPSE (FIR-dapse)
(amifampridine) tablets
for oral use
This Instructions for Use contains information on how to mix and use FIRDAPSE prepared suspension. The FIRDAPSE prepared suspension can be used for people who are prescribed a dosage that cannot be obtained with whole or half tablets, who have trouble swallowing tablets, or who have a feeding tube.
Prepare FIRDAPSE oral suspension each day.
Supplies you will need:
You can get these supplies at your local pharmacy.
| your FIRDAPSE tablets | empty bottle with cap (50-100 mL recommended) | sterile water | oral syringe with catheter tip (10mL, may require smaller syringe for dosing) |
Instructions to make a 1 mg/mL suspension:
Important:
- Do not use any foods or liquids other than sterile water to mix FIRDAPSE.
- You can prepare each dose separately or all of your doses for the day at one time.
| Step 1 | Step 2 | Step 3 |
|
Place the number of FIRDAPSE tablets you need for your dose or doses in a bottle. |
For each tablet, add 10 mL of water to the bottle. Measure the water with an oral syringe, and inject the water into the bottle. Note: Depending on the number of tablets and the size of the syringe, you may need to measure and inject the water more than one time. |
Secure the cap back on the bottle. Wait for 5 minutes. Shake well for 30 seconds. |
Instructions to administer the prepared suspension:
Important:
- Prepare fresh suspensions daily.
- If you prepare all of your doses for the day at one time, refrigerate the suspension between doses. Shake well before drawing up each dose.
OPTION 1 To administer by mouth:
| Step 4 | Step 5 |
| Remove the bottle cap and use an oral syringe with a catheter tip to measure the prescribed dose. |
Push syringe plunger to administer by mouth. Note: Depending on the dose and size of syringe, you may need to repeat steps 4 and 5 until the prescribed dose is given. |
OPTION 2 To administer by feeding tube:
Important:
- Do not use any foods or liquids other than sterile water to mix FIRDAPSE.
- Use only an oral syringe with a catheter tip to give FIRDAPSE through the feeding tube. Talk to your healthcare provider about the size catheter tipped syringe you should use.
| Step 4 | Step 5 |
| Remove the bottle cap and use an oral syringe with a catheter tip to measure the prescribed dose. |
Inject the medicine using the oral syringe with a catheter tip into the feeding tube right away. Note: Depending on the dose and size of syringe, you may need to repeat steps 4 and 5 until the prescribed dose is given. |
| Step 6 | Step 7 |
| To flush the feeding tube: Refill the syringe with 10 mL of sterile water. | Shake the syringe, insert the catheter tip into the feeding tube to flush any remaining medicine from the feeding tube into the stomach. |
How should I store FIRDAPSE?
Firdapse tablets:
- Store FIRDAPSE tablets at room temperature between 68ºF to 77ºF (20ºC to 25ºC).
- Safely throw away FIRDAPSE tablets that are out of date or no longer needed.
Firdapse prepared suspension:
- Store FIRDAPSE prepared oral suspension in the refrigerator between 36°F to 46°F (2°C to 8°C) between doses for up to 24 hours.
- Safely throw away unused FIRDAPSE oral suspension after 24 hours.
Distributed by Catalyst Pharmaceuticals, Inc., Coral Gables, FL 33134
For more information, go to www.YourCatalystPathways.com or call 1-833-422-8259
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Approved: 5/2024
5.1 Seizures
FIRDAPSE can cause seizures. Seizures have been observed in patients without a history of seizures taking FIRDAPSE at the recommended doses, at various times after initiation of treatment, at an incidence of approximately 2%. Many of the patients were taking medications or had comorbid medical conditions that may have lowered the seizure threshold [see Drug Interactions (7.1)]. Seizures may be dose-dependent. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment. FIRDAPSE is contraindicated in patients with a history of seizures.
10 Overdosage
Overdose with FIRDAPSE was not reported during clinical studies.
In a case report, a 65-year-old patient with LEMS inadvertently received a total daily amifampridine dose of 360 mg/day (approximately 4 times the maximum recommended total daily dose) and was hospitalized for general weakness, paresthesia, nausea, vomiting, and palpitations. The patient developed convulsions and paroxysmal supraventricular tachycardia, and four days after admission, experienced cardiac arrest. The patient was resuscitated and ultimately recovered following withdrawal of amifampridine.
Patients with suspected overdose with FIRDAPSE should be monitored for signs or symptoms of exaggerated FIRDAPSE adverse reactions or effects, and appropriate symptomatic treatment instituted immediately.
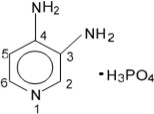
11 Description
The active ingredient of FIRDAPSE is amifampridine phosphate, which is a voltage-gated potassium channel blocker. Amifampridine phosphate is described chemically as 3,4-diaminopyridine phosphate with a molecular weight of 207.1 and a molecular formula of C5H7N3 • H3PO4. The structural formula is:
Amifampridine phosphate is a white, crystalline powder that is freely soluble in water, and slightly soluble in solvents ethanol, methanol and acetic acid. A 1% aqueous solution of amifampridine phosphate has a pH of 4.4 at ambient conditions.
Each FIRDAPSE tablet contains 10 mg amifampridine (equivalent to 18.98 mg amifampridine phosphate). The tablet formulation includes the following inactive ingredients: calcium stearate, colloidal silicon dioxide, and microcrystalline cellulose.
FIRDAPSE tablets are intended for oral administration only.
16.1 How Supplied
FIRDAPSE 10 mg tablets are white to off white-round, and functionally scored. Each tablet is debossed on the non-scored side with “CATALYST” and on the scored side with “211” above the score and “10” below the score. Tablets can be divided in half at the score. FIRDAPSE is supplied as follows:
8.4 Pediatric Use
Safety and effectiveness of FIRDAPSE for the treatment of LEMS have been established in pediatric patients 6 years of age and older. Use of FIRDAPSE for this indication is supported by evidence from adequate and well-controlled studies of FIRDAPSE in adults with LEMS, pharmacokinetic data in adult patients, pharmacokinetic modeling and simulation to identify the dosing regimen in pediatric patients, and safety data from pediatric patients aged 6 years and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients below the age of 6 years have not been established.
8.5 Geriatric Use
Clinical studies of FIRDAPSE did not include a sufficient number of subjects aged 65 and over (19 of 63 patients in Studies 1 and 2) to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Dosage and Administration (2.2, 2.3) and Drug Interactions (7.1, 7.2)].
14 Clinical Studies
The efficacy of FIRDAPSE for the treatment of LEMS was demonstrated in two randomized, double-blind, placebo-controlled discontinuation studies. A total of 64 adults (age 21 to 88 years) with LEMS were enrolled (Study 1 and Study 2). The studies enrolled patients with a confirmed diagnosis of LEMS based on either neurophysiology studies or a positive anti-P/Q type voltage-gated calcium channel antibody test. Patients were required to be on an adequate and stable dosage (30 to 80 mg daily) of amifampridine phosphate prior to entering the randomized discontinuation phases of both studies.
The two co-primary measures of efficacy in both studies were the change from baseline to the end of the discontinuation period in the Quantitative Myasthenia Gravis (QMG) score and in the Subject Global Impression (SGI) score.
The QMG is a 13-item physician-rated categorical scale assessing muscle weakness. Each item is assessed on a 4-point scale, where a score of 0 represents no weakness, and a score of 3 represents severe weakness (total score 0-39). Higher scores represent greater impairment.
The SGI is a 7-point scale on which patients rated their global impression of the effects of the study treatment on their physical well- being. Lower scores on the SGI represent lower perceived benefit with the study treatment.
A key secondary efficacy endpoint was the clinical global impression improvement (CGI-I) score, a 7-point scale on which the treating physician rated the global impression of change in clinical symptoms. A higher CGI-I score indicates a perceived worsening of clinical symptoms.
4 Contraindications
6 Adverse Reactions
7 Drug Interactions
- Drugs that lower seizure threshold: Concomitant use may lead to an increased risk of seizures. (7.1)
- Drugs with cholinergic effects (e.g., direct or indirect cholinesterase inhibitors): Concomitant use may increase the cholinergic effects of FIRDAPSE and of those drugs and increase the risk of adverse reactions. (7.2)
5.2 Hypersensitivity
In clinical trials, hypersensitivity reactions and anaphylaxis associated with FIRDAPSE administration have not been reported. Anaphylaxis has been reported in patients taking another aminopyridine; therefore, it may occur with FIRDAPSE. If anaphylaxis occurs, administration of FIRDAPSE should be discontinued and appropriate therapy initiated.
8.6 Renal Impairment
Renal clearance is an elimination pathway for amifampridine and the inactive metabolite, 3-N-acetyl-amifampridine, and exposure of amifampridine is higher in subjects with renal impairment [see Clinical Pharmacology (12.3)]. Therefore, in patients with renal impairment, FIRDAPSE should be initiated at the lowest recommended initial daily dosage, and patients should be closely monitored for adverse reactions [see Dosage and Administration (2.2)]. Consider dosage modification or discontinuation of FIRDAPSE for patients with renal impairment as needed based on clinical effect and tolerability. The safety, efficacy, and pharmacokinetics of amifampridine have not been studied in patients with end-stage renal disease (CLcr <15 mL/min or patients requiring dialysis). No dosage recommendation for FIRDAPSE can be made for patients with end-stage renal disease.
12.2 Pharmacodynamics
The effect of FIRDAPSE on QTc interval prolongation was studied in a double blind, randomized, placebo and positive controlled study in 52 healthy individuals who are slow acetylators. At an exposure 2-fold the expected maximum therapeutic exposure of amifampridine, FIRDAPSE did not prolong QTc to any clinically relevant extent.
12.3 Pharmacokinetics
The pharmacokinetics of amifampridine are similar between healthy individuals and LEMS patients. Following single and multiple doses, AUC, Cmax and Cmin were highly variable between individuals. FIRDAPSE exposure increased proportionally with dose across the range of 20 mg to 80 mg single oral doses.
12.5 Pharmacogenomics
Genetic variants in the N-acetyltransferase gene 2 (NAT2) affect the rate and extent of FIRDAPSE metabolism. Poor metabolizers, also referred to as “slow acetylators” (i.e., carriers of two reduced function alleles), have 3.5- to 4.5-fold higher Cmax, and 5.6- to 9- fold higher AUC than normal metabolizers, also referred to as “fast/rapid acetylators” (i.e., carriers of two normal function alleles). Therefore, FIRDAPSE should be initiated at the lowest recommended initial daily dosage in known NAT2 poor metabolizers, and such patients should be closely monitored for adverse reactions [see Dosage and Administration (2.4) and Use in Specific Populations (8.8)]. In the general population, the NAT2 poor metabolizer phenotype prevalence is 40–60% in the White and African American populations, and in 10–30% in Asian ethnic populations (individuals of Japanese, Chinese, or Korean descent).
2.1 Dosage Information
The recommended oral dosage of FIRDAPSE for adults and pediatric patients 6 years of age and older is included in Table 1. For pediatric patients, the recommended dosing regimen is dependent on body weight. Dosage should be increased every 3 to 4 days based on clinical response and tolerability. Titration increments should not exceed those shown in Table 1.
|
*See Dosage and Administration (2.5) for a method to achieve these doses |
||||
| Age and Body Weight | Initial Daily Dosage * | Titration Regimen |
Maximum Single
Dose |
Maximum Total Daily
Maintenance Dosage |
|
15 mg to 30 mg daily, in 3 to 5 divided doses |
Increase total daily dosage by 5 mg every 3 or 4 days |
20 mg | 100 mg Given in divided doses |
|
5 mg to 15 mg daily, in 3 to 5 divided doses |
Increase total daily dosage by 2.5 mg every 3 or 4 days | 10 mg | 50 mg Given in divided doses |
8.7 Hepatic Impairment
In patients with any degree of hepatic impairment, FIRDAPSE should be initiated at the lowest recommended initial daily dosage, and patients should be monitored for adverse reactions [see Dosage and Administration (2.3)]. Consider dosage modification or discontinuation of FIRDAPSE for patients with hepatic impairment as needed based on clinical effect and tolerability.
1 Indications and Usage
FIRDAPSE® is indicated for the treatment of Lambert-Eaton myasthenic syndrome (LEMS) in adults and pediatric patients 6 years of age and older.
12.1 Mechanism of Action
The mechanism by which amifampridine exerts its therapeutic effect in LEMS patients has not been fully elucidated. Amifampridine is a broad-spectrum potassium channel blocker.
16.2 Storage and Handling
Store FIRDAPSE tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP controlled room temperature]. See Dosage and Administration (2.5) for storage instructions for FIRDAPSE prepared suspension.
5 Warnings and Precautions
- Seizures: FIRDAPSE can cause seizures. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment. (5.1)
- Hypersensitivity reactions: If a hypersensitivity reaction such as anaphylaxis occurs, FIRDAPSE should be discontinued and appropriate therapy initiated. (5.2)
8.8 Nat2 Poor Metabolizers
Exposure of FIRDAPSE is increased in patients who are N-acetyltransferase 2 (NAT2) poor metabolizers [see Clinical Pharmacology (12.5)]. Therefore, initiate FIRDAPSE in patients who are known NAT2 poor metabolizers at the lowest recommended initial daily dosage and monitor for adverse reactions [see Dosage and Administration (2.4)]. Consider dosage modification of FIRDAPSE for patients who are known NAT2 poor metabolizers as needed based on clinical effect and tolerability.
2 Dosage and Administration
- Administer orally in divided doses (3 to 5 times daily). (2.1)
- The recommended starting dosage for adults (any weight) and pediatric patients weighing 45 kg or more is 15 mg to 30 mg daily, in divided doses. (2.1)
- The recommended starting dosage for pediatric patients weighing less than 45 kg is 5 mg to 15 mg daily, in divided doses. (2.1)
- The recommended starting dosage for adult and pediatric patients with renal impairment, hepatic impairment, and in known N-acetyltransferase 2 (NAT2) poor metabolizers is the lowest recommended initial daily dosage (2.1, 2.2, 2.3, 2.4)
- For patients with a dosage adjustment of less than 5 mg increments, or who have difficulty swallowing, or require feeding tube, a l mg/mL suspension can be prepared. (2.5)
3 Dosage Forms and Strengths
FIRDAPSE tablets contain 10 mg amifampridine and are white to off-white, round, and functionally scored. Each tablet is debossed on the non-scored side with “CATALYST” and on the scored side with “211” above the score and “10” below the score.
8 Use in Specific Populations
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
2.5 Administration Instructions
FIRDAPSE can be taken without regard to food.
Preparation of a 1mg/mL Suspension (see the Instructions for Use for full instructions on how to prepare the 1mg/mL suspension)
When patients require a dosage in less than 5 mg increments, have difficulty swallowing tablets, or require feeding tubes, a 1 mg/mL suspension can be prepared (e.g., by placing the required number of tablets in a 50 to 100 mL container, adding 10 mL of sterile water for each tablet, waiting for 5 minutes, and shaking well for 30 seconds).
Crushing the tablets prior to making the suspension is not necessary. After preparation of the suspension, an oral syringe can be used to draw up and administer the correct dose by mouth or by feeding tube.
17 Patient Counseling Information
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
2.2 Patients With Renal Impairment
The recommended starting dosage of FIRDAPSE in patients with renal impairment [creatinine clearance (CLcr) 15 to 90 mL/min] is the lowest recommended initial daily dosage (i.e., 15 mg daily for pediatric patients weighing 45 kg or more and for adults, and 5 mg daily for pediatric patients weighing less than 45 kg) taken orally in divided doses. No dosage recommendation for FIRDAPSE can be made for patients with end-stage renal disease [see Dosage and Administration (2.1), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
7.2 Drugs With Cholinergic Effects
The concomitant use of FIRDAPSE and drugs with cholinergic effects (e.g., direct or indirect cholinesterase inhibitors) may increase the cholinergic effects of FIRDAPSE and of those drugs and increase the risk of adverse reactions.
2.3 Patients With Hepatic Impairment
The recommended starting dosage of FIRDAPSE in patients with any degree of hepatic impairment is the lowest recommended initial daily dosage (i.e., 15 mg daily for pediatric patients weighing 45 kg or more and for adults, and 5 mg daily for pediatric patients weighting less than 45 kg) taken orally in divided doses [see Dosage and Administration (2.1), Use in Specific Populations (8.7), and Clinical Pharmacology (12.3)].
7.1 Drugs That Lower Seizure Threshold
The concomitant use of FIRDAPSE and drugs that lower seizure threshold may lead to an increased risk of seizures [see Warnings and Precautions (5.1)]. The decision to administer FIRDAPSE concomitantly with drugs that lower the seizure threshold should be carefully considered in light of the severity of the associated risks.
2.4 Known N Acetyltransferase 2 (nat2) Poor Metabolizers
The recommended starting dosage of FIRDAPSE in known N-acetyltransferase 2 (NAT2) poor metabolizers is the lowest recommended initial daily dosage (i.e., 15 mg daily for pediatric patients weighing 45 kg or more and for adults, and 5 mg daily for pediatric patients weighing less than 45 kg) taken orally in divided doses [see Dosage and Administration (2.1), Use in Specific Populations (8.8), and Clinical Pharmacology (12.5)].
Principal Display Panel 10 Mg Tablet Blister Pack Carton
Rx Only
NDC 69616-211-06
120 Tablets Total
Provided in 12 Child-Resistant Blister Cards
Of 10 Tablets Each
FIRDAPSE®
(amifampridine) Tablets
10 mg per Tablet
Manufactured for:
Catalyst Pharmaceuticals, Inc.
Coral Gables, FL 33134
Structured Label Content
Section 42229-5 (42229-5)
Missed Dose
If a dose is missed, patients should not take double or extra doses.
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised: 5/2024 |
|
MEDICATION GUIDE
FIRDAPSE ® (FIR-dapse) (amifampridine) tablets, for oral use |
|
| Read this Medication Guide before you start taking FIRDAPSE and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. | |
|
What is the most important information I should know about FIRDAPSE?
|
|
| Stop taking FIRDAPSE and call your doctor right away if you have a seizure while taking FIRDAPSE. | |
|
What is FIRDAPSE?
FIRDAPSE is a prescription medicine used to treat Lambert-Eaton myasthenic syndrome (LEMS) in people 6 years of age and older. It is not known if FIRDAPSE is safe or effective in children less than 6 years of age. |
|
Do not take FIRDAPSE if you:
|
|
Before you take FIRDAPSE, tell your doctor about all of your medical conditions. including if you:
|
|
| Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. | |
How should I take FIRDAPSE?
|
|
|
What are the possible side effects of FIRDAPSE?
FIRDAPSE may cause serious side effects, including:
|
|
The most common side effects of FIRDAPSE include:
|
|
| Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of FIRDAPSE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store FIRDAPSE?
|
|
| Keep FIRDAPSE and all medicines out of the reach of children. | |
|
General Information about the safe and effective use of FIRDAPSE
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use FIRDAPSE for a condition for which it was not prescribed. Do not give FIRDAPSE to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk to your doctor or pharmacist. You can ask your pharmacist or doctor for information about FIRDAPSE that is written for health professionals. |
|
|
What are the ingredients in FIRDAPSE?
Active ingredient: amifampridine Inactive ingredients: calcium stearate, colloidal silicon dioxide, and microcrystalline cellulose. Distributed by Catalyst Pharmaceuticals, Inc., Coral Gables, FL 33134 For more information, go to www.YourCatalystPathways.com or call 1-833-422-8259 |
Section 43683-2 (43683-2)
| Dosage and Administration (2.1) | 5/2024 |
Section 59845-8 (59845-8)
Instructions for Use
FIRDAPSE (FIR-dapse)
(amifampridine) tablets
for oral use
This Instructions for Use contains information on how to mix and use FIRDAPSE prepared suspension. The FIRDAPSE prepared suspension can be used for people who are prescribed a dosage that cannot be obtained with whole or half tablets, who have trouble swallowing tablets, or who have a feeding tube.
Prepare FIRDAPSE oral suspension each day.
Supplies you will need:
You can get these supplies at your local pharmacy.
| your FIRDAPSE tablets | empty bottle with cap (50-100 mL recommended) | sterile water | oral syringe with catheter tip (10mL, may require smaller syringe for dosing) |
Instructions to make a 1 mg/mL suspension:
Important:
- Do not use any foods or liquids other than sterile water to mix FIRDAPSE.
- You can prepare each dose separately or all of your doses for the day at one time.
| Step 1 | Step 2 | Step 3 |
|
Place the number of FIRDAPSE tablets you need for your dose or doses in a bottle. |
For each tablet, add 10 mL of water to the bottle. Measure the water with an oral syringe, and inject the water into the bottle. Note: Depending on the number of tablets and the size of the syringe, you may need to measure and inject the water more than one time. |
Secure the cap back on the bottle. Wait for 5 minutes. Shake well for 30 seconds. |
Instructions to administer the prepared suspension:
Important:
- Prepare fresh suspensions daily.
- If you prepare all of your doses for the day at one time, refrigerate the suspension between doses. Shake well before drawing up each dose.
OPTION 1 To administer by mouth:
| Step 4 | Step 5 |
| Remove the bottle cap and use an oral syringe with a catheter tip to measure the prescribed dose. |
Push syringe plunger to administer by mouth. Note: Depending on the dose and size of syringe, you may need to repeat steps 4 and 5 until the prescribed dose is given. |
OPTION 2 To administer by feeding tube:
Important:
- Do not use any foods or liquids other than sterile water to mix FIRDAPSE.
- Use only an oral syringe with a catheter tip to give FIRDAPSE through the feeding tube. Talk to your healthcare provider about the size catheter tipped syringe you should use.
| Step 4 | Step 5 |
| Remove the bottle cap and use an oral syringe with a catheter tip to measure the prescribed dose. |
Inject the medicine using the oral syringe with a catheter tip into the feeding tube right away. Note: Depending on the dose and size of syringe, you may need to repeat steps 4 and 5 until the prescribed dose is given. |
| Step 6 | Step 7 |
| To flush the feeding tube: Refill the syringe with 10 mL of sterile water. | Shake the syringe, insert the catheter tip into the feeding tube to flush any remaining medicine from the feeding tube into the stomach. |
How should I store FIRDAPSE?
Firdapse tablets:
- Store FIRDAPSE tablets at room temperature between 68ºF to 77ºF (20ºC to 25ºC).
- Safely throw away FIRDAPSE tablets that are out of date or no longer needed.
Firdapse prepared suspension:
- Store FIRDAPSE prepared oral suspension in the refrigerator between 36°F to 46°F (2°C to 8°C) between doses for up to 24 hours.
- Safely throw away unused FIRDAPSE oral suspension after 24 hours.
Distributed by Catalyst Pharmaceuticals, Inc., Coral Gables, FL 33134
For more information, go to www.YourCatalystPathways.com or call 1-833-422-8259
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Approved: 5/2024
5.1 Seizures
FIRDAPSE can cause seizures. Seizures have been observed in patients without a history of seizures taking FIRDAPSE at the recommended doses, at various times after initiation of treatment, at an incidence of approximately 2%. Many of the patients were taking medications or had comorbid medical conditions that may have lowered the seizure threshold [see Drug Interactions (7.1)]. Seizures may be dose-dependent. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment. FIRDAPSE is contraindicated in patients with a history of seizures.
10 Overdosage (10 OVERDOSAGE)
Overdose with FIRDAPSE was not reported during clinical studies.
In a case report, a 65-year-old patient with LEMS inadvertently received a total daily amifampridine dose of 360 mg/day (approximately 4 times the maximum recommended total daily dose) and was hospitalized for general weakness, paresthesia, nausea, vomiting, and palpitations. The patient developed convulsions and paroxysmal supraventricular tachycardia, and four days after admission, experienced cardiac arrest. The patient was resuscitated and ultimately recovered following withdrawal of amifampridine.
Patients with suspected overdose with FIRDAPSE should be monitored for signs or symptoms of exaggerated FIRDAPSE adverse reactions or effects, and appropriate symptomatic treatment instituted immediately.
11 Description (11 DESCRIPTION)
The active ingredient of FIRDAPSE is amifampridine phosphate, which is a voltage-gated potassium channel blocker. Amifampridine phosphate is described chemically as 3,4-diaminopyridine phosphate with a molecular weight of 207.1 and a molecular formula of C5H7N3 • H3PO4. The structural formula is:
Amifampridine phosphate is a white, crystalline powder that is freely soluble in water, and slightly soluble in solvents ethanol, methanol and acetic acid. A 1% aqueous solution of amifampridine phosphate has a pH of 4.4 at ambient conditions.
Each FIRDAPSE tablet contains 10 mg amifampridine (equivalent to 18.98 mg amifampridine phosphate). The tablet formulation includes the following inactive ingredients: calcium stearate, colloidal silicon dioxide, and microcrystalline cellulose.
FIRDAPSE tablets are intended for oral administration only.
16.1 How Supplied
FIRDAPSE 10 mg tablets are white to off white-round, and functionally scored. Each tablet is debossed on the non-scored side with “CATALYST” and on the scored side with “211” above the score and “10” below the score. Tablets can be divided in half at the score. FIRDAPSE is supplied as follows:
8.4 Pediatric Use
Safety and effectiveness of FIRDAPSE for the treatment of LEMS have been established in pediatric patients 6 years of age and older. Use of FIRDAPSE for this indication is supported by evidence from adequate and well-controlled studies of FIRDAPSE in adults with LEMS, pharmacokinetic data in adult patients, pharmacokinetic modeling and simulation to identify the dosing regimen in pediatric patients, and safety data from pediatric patients aged 6 years and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Safety and effectiveness in pediatric patients below the age of 6 years have not been established.
8.5 Geriatric Use
Clinical studies of FIRDAPSE did not include a sufficient number of subjects aged 65 and over (19 of 63 patients in Studies 1 and 2) to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Dosage and Administration (2.2, 2.3) and Drug Interactions (7.1, 7.2)].
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of FIRDAPSE for the treatment of LEMS was demonstrated in two randomized, double-blind, placebo-controlled discontinuation studies. A total of 64 adults (age 21 to 88 years) with LEMS were enrolled (Study 1 and Study 2). The studies enrolled patients with a confirmed diagnosis of LEMS based on either neurophysiology studies or a positive anti-P/Q type voltage-gated calcium channel antibody test. Patients were required to be on an adequate and stable dosage (30 to 80 mg daily) of amifampridine phosphate prior to entering the randomized discontinuation phases of both studies.
The two co-primary measures of efficacy in both studies were the change from baseline to the end of the discontinuation period in the Quantitative Myasthenia Gravis (QMG) score and in the Subject Global Impression (SGI) score.
The QMG is a 13-item physician-rated categorical scale assessing muscle weakness. Each item is assessed on a 4-point scale, where a score of 0 represents no weakness, and a score of 3 represents severe weakness (total score 0-39). Higher scores represent greater impairment.
The SGI is a 7-point scale on which patients rated their global impression of the effects of the study treatment on their physical well- being. Lower scores on the SGI represent lower perceived benefit with the study treatment.
A key secondary efficacy endpoint was the clinical global impression improvement (CGI-I) score, a 7-point scale on which the treating physician rated the global impression of change in clinical symptoms. A higher CGI-I score indicates a perceived worsening of clinical symptoms.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
7 Drug Interactions (7 DRUG INTERACTIONS)
- Drugs that lower seizure threshold: Concomitant use may lead to an increased risk of seizures. (7.1)
- Drugs with cholinergic effects (e.g., direct or indirect cholinesterase inhibitors): Concomitant use may increase the cholinergic effects of FIRDAPSE and of those drugs and increase the risk of adverse reactions. (7.2)
5.2 Hypersensitivity
In clinical trials, hypersensitivity reactions and anaphylaxis associated with FIRDAPSE administration have not been reported. Anaphylaxis has been reported in patients taking another aminopyridine; therefore, it may occur with FIRDAPSE. If anaphylaxis occurs, administration of FIRDAPSE should be discontinued and appropriate therapy initiated.
8.6 Renal Impairment
Renal clearance is an elimination pathway for amifampridine and the inactive metabolite, 3-N-acetyl-amifampridine, and exposure of amifampridine is higher in subjects with renal impairment [see Clinical Pharmacology (12.3)]. Therefore, in patients with renal impairment, FIRDAPSE should be initiated at the lowest recommended initial daily dosage, and patients should be closely monitored for adverse reactions [see Dosage and Administration (2.2)]. Consider dosage modification or discontinuation of FIRDAPSE for patients with renal impairment as needed based on clinical effect and tolerability. The safety, efficacy, and pharmacokinetics of amifampridine have not been studied in patients with end-stage renal disease (CLcr <15 mL/min or patients requiring dialysis). No dosage recommendation for FIRDAPSE can be made for patients with end-stage renal disease.
12.2 Pharmacodynamics
The effect of FIRDAPSE on QTc interval prolongation was studied in a double blind, randomized, placebo and positive controlled study in 52 healthy individuals who are slow acetylators. At an exposure 2-fold the expected maximum therapeutic exposure of amifampridine, FIRDAPSE did not prolong QTc to any clinically relevant extent.
12.3 Pharmacokinetics
The pharmacokinetics of amifampridine are similar between healthy individuals and LEMS patients. Following single and multiple doses, AUC, Cmax and Cmin were highly variable between individuals. FIRDAPSE exposure increased proportionally with dose across the range of 20 mg to 80 mg single oral doses.
12.5 Pharmacogenomics
Genetic variants in the N-acetyltransferase gene 2 (NAT2) affect the rate and extent of FIRDAPSE metabolism. Poor metabolizers, also referred to as “slow acetylators” (i.e., carriers of two reduced function alleles), have 3.5- to 4.5-fold higher Cmax, and 5.6- to 9- fold higher AUC than normal metabolizers, also referred to as “fast/rapid acetylators” (i.e., carriers of two normal function alleles). Therefore, FIRDAPSE should be initiated at the lowest recommended initial daily dosage in known NAT2 poor metabolizers, and such patients should be closely monitored for adverse reactions [see Dosage and Administration (2.4) and Use in Specific Populations (8.8)]. In the general population, the NAT2 poor metabolizer phenotype prevalence is 40–60% in the White and African American populations, and in 10–30% in Asian ethnic populations (individuals of Japanese, Chinese, or Korean descent).
2.1 Dosage Information
The recommended oral dosage of FIRDAPSE for adults and pediatric patients 6 years of age and older is included in Table 1. For pediatric patients, the recommended dosing regimen is dependent on body weight. Dosage should be increased every 3 to 4 days based on clinical response and tolerability. Titration increments should not exceed those shown in Table 1.
|
*See Dosage and Administration (2.5) for a method to achieve these doses |
||||
| Age and Body Weight | Initial Daily Dosage * | Titration Regimen |
Maximum Single
Dose |
Maximum Total Daily
Maintenance Dosage |
|
15 mg to 30 mg daily, in 3 to 5 divided doses |
Increase total daily dosage by 5 mg every 3 or 4 days |
20 mg | 100 mg Given in divided doses |
|
5 mg to 15 mg daily, in 3 to 5 divided doses |
Increase total daily dosage by 2.5 mg every 3 or 4 days | 10 mg | 50 mg Given in divided doses |
8.7 Hepatic Impairment
In patients with any degree of hepatic impairment, FIRDAPSE should be initiated at the lowest recommended initial daily dosage, and patients should be monitored for adverse reactions [see Dosage and Administration (2.3)]. Consider dosage modification or discontinuation of FIRDAPSE for patients with hepatic impairment as needed based on clinical effect and tolerability.
1 Indications and Usage (1 INDICATIONS AND USAGE)
FIRDAPSE® is indicated for the treatment of Lambert-Eaton myasthenic syndrome (LEMS) in adults and pediatric patients 6 years of age and older.
12.1 Mechanism of Action
The mechanism by which amifampridine exerts its therapeutic effect in LEMS patients has not been fully elucidated. Amifampridine is a broad-spectrum potassium channel blocker.
16.2 Storage and Handling
Store FIRDAPSE tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP controlled room temperature]. See Dosage and Administration (2.5) for storage instructions for FIRDAPSE prepared suspension.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Seizures: FIRDAPSE can cause seizures. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment. (5.1)
- Hypersensitivity reactions: If a hypersensitivity reaction such as anaphylaxis occurs, FIRDAPSE should be discontinued and appropriate therapy initiated. (5.2)
8.8 Nat2 Poor Metabolizers (8.8 NAT2 Poor Metabolizers)
Exposure of FIRDAPSE is increased in patients who are N-acetyltransferase 2 (NAT2) poor metabolizers [see Clinical Pharmacology (12.5)]. Therefore, initiate FIRDAPSE in patients who are known NAT2 poor metabolizers at the lowest recommended initial daily dosage and monitor for adverse reactions [see Dosage and Administration (2.4)]. Consider dosage modification of FIRDAPSE for patients who are known NAT2 poor metabolizers as needed based on clinical effect and tolerability.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administer orally in divided doses (3 to 5 times daily). (2.1)
- The recommended starting dosage for adults (any weight) and pediatric patients weighing 45 kg or more is 15 mg to 30 mg daily, in divided doses. (2.1)
- The recommended starting dosage for pediatric patients weighing less than 45 kg is 5 mg to 15 mg daily, in divided doses. (2.1)
- The recommended starting dosage for adult and pediatric patients with renal impairment, hepatic impairment, and in known N-acetyltransferase 2 (NAT2) poor metabolizers is the lowest recommended initial daily dosage (2.1, 2.2, 2.3, 2.4)
- For patients with a dosage adjustment of less than 5 mg increments, or who have difficulty swallowing, or require feeding tube, a l mg/mL suspension can be prepared. (2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
FIRDAPSE tablets contain 10 mg amifampridine and are white to off-white, round, and functionally scored. Each tablet is debossed on the non-scored side with “CATALYST” and on the scored side with “211” above the score and “10” below the score.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
2.5 Administration Instructions
FIRDAPSE can be taken without regard to food.
Preparation of a 1mg/mL Suspension (see the Instructions for Use for full instructions on how to prepare the 1mg/mL suspension)
When patients require a dosage in less than 5 mg increments, have difficulty swallowing tablets, or require feeding tubes, a 1 mg/mL suspension can be prepared (e.g., by placing the required number of tablets in a 50 to 100 mL container, adding 10 mL of sterile water for each tablet, waiting for 5 minutes, and shaking well for 30 seconds).
Crushing the tablets prior to making the suspension is not necessary. After preparation of the suspension, an oral syringe can be used to draw up and administer the correct dose by mouth or by feeding tube.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
2.2 Patients With Renal Impairment (2.2 Patients with Renal Impairment)
The recommended starting dosage of FIRDAPSE in patients with renal impairment [creatinine clearance (CLcr) 15 to 90 mL/min] is the lowest recommended initial daily dosage (i.e., 15 mg daily for pediatric patients weighing 45 kg or more and for adults, and 5 mg daily for pediatric patients weighing less than 45 kg) taken orally in divided doses. No dosage recommendation for FIRDAPSE can be made for patients with end-stage renal disease [see Dosage and Administration (2.1), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
7.2 Drugs With Cholinergic Effects (7.2 Drugs with Cholinergic Effects)
The concomitant use of FIRDAPSE and drugs with cholinergic effects (e.g., direct or indirect cholinesterase inhibitors) may increase the cholinergic effects of FIRDAPSE and of those drugs and increase the risk of adverse reactions.
2.3 Patients With Hepatic Impairment (2.3 Patients with Hepatic Impairment)
The recommended starting dosage of FIRDAPSE in patients with any degree of hepatic impairment is the lowest recommended initial daily dosage (i.e., 15 mg daily for pediatric patients weighing 45 kg or more and for adults, and 5 mg daily for pediatric patients weighting less than 45 kg) taken orally in divided doses [see Dosage and Administration (2.1), Use in Specific Populations (8.7), and Clinical Pharmacology (12.3)].
7.1 Drugs That Lower Seizure Threshold (7.1 Drugs that Lower Seizure Threshold)
The concomitant use of FIRDAPSE and drugs that lower seizure threshold may lead to an increased risk of seizures [see Warnings and Precautions (5.1)]. The decision to administer FIRDAPSE concomitantly with drugs that lower the seizure threshold should be carefully considered in light of the severity of the associated risks.
2.4 Known N Acetyltransferase 2 (nat2) Poor Metabolizers (2.4 Known N-acetyltransferase 2 (NAT2) Poor Metabolizers)
The recommended starting dosage of FIRDAPSE in known N-acetyltransferase 2 (NAT2) poor metabolizers is the lowest recommended initial daily dosage (i.e., 15 mg daily for pediatric patients weighing 45 kg or more and for adults, and 5 mg daily for pediatric patients weighing less than 45 kg) taken orally in divided doses [see Dosage and Administration (2.1), Use in Specific Populations (8.8), and Clinical Pharmacology (12.5)].
Principal Display Panel 10 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Blister Pack Carton)
Rx Only
NDC 69616-211-06
120 Tablets Total
Provided in 12 Child-Resistant Blister Cards
Of 10 Tablets Each
FIRDAPSE®
(amifampridine) Tablets
10 mg per Tablet
Manufactured for:
Catalyst Pharmaceuticals, Inc.
Coral Gables, FL 33134
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:40.429179 · Updated: 2026-03-14T22:06:28.970908