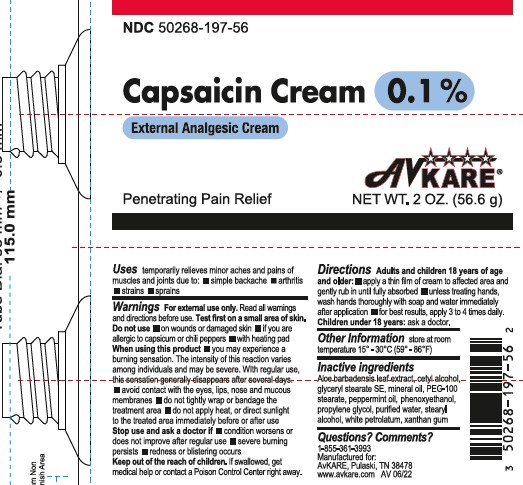
Capsaicin Cream 0.1%
ef5b0db6-8f0a-4f8e-e053-2995a90a22b7
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient: Capsaicin 0.1%
Medication Information
Warnings and Precautions
Warnings: For external use only.
Do not use:
- on wounds or damaged skin
- if you are allergic to capsicum or chili peppers
- with heating pad
When using this produce:
- you may experience a burning sensation The intensity of this reaction varies among individuals and may be severe. With regular use, the sensation generally disappears after several days.
- avoid contact with the eyes, lips, nose and mucous membranes
- do not tightly wrap or bandage the treatement area
- do not apply heat, or direct sunlight to the treated area immediately before or after use
Stop use and ask a doctor if:
- condition worsens or does not improve after regular use
- severe burning persists
- redness or blistering occurs
Indications and Usage
Uses: Provides penetrating pain relief. Temporary relief of minor aches and pains of muscles and joints associated with simple backache, arthritis, strains, bruises, and sprains.
Dosage and Administration
Directions – Adults and children 18 years of age and older
- apply a thin film of cream to affected area and gently rub in until fully absorbed
- unless treating hands, wash hands thoroughly with soap and water immediately after application
- for best results, apply 3 to 4 times daily
Children under 18 years: ask a doctor.
Description
Active Ingredient: Capsaicin 0.1%
Section 42229-5
Questions? Comments?
1-855-361-3993
Manufactured for:
AvKARE, Pulaski, TN 38478
www.avkare.com
AV 06/22
Section 44425-7
Other Information: store at room temperature 15° - 30°C (59° - 86°F)
Section 50565-1
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive Ingredients: Aloe barbadensis leaf extract, cetyl alcohol, glyceryl stearate SE, mineral oil, PEG-100 stearate, peppermint oil, phenoxyethanol, propylene glycol, purified water, stearyl alcohol, white petrolatum, xanthan gum
Section 55105-1
Purpose: Topical Analgesic
Section 55106-9
Active Ingredient: Capsaicin 0.1%
Packaging
Structured Label Content
Indications and Usage (34067-9)
Uses: Provides penetrating pain relief. Temporary relief of minor aches and pains of muscles and joints associated with simple backache, arthritis, strains, bruises, and sprains.
Dosage and Administration (34068-7)
Directions – Adults and children 18 years of age and older
- apply a thin film of cream to affected area and gently rub in until fully absorbed
- unless treating hands, wash hands thoroughly with soap and water immediately after application
- for best results, apply 3 to 4 times daily
Children under 18 years: ask a doctor.
Warnings and Precautions (34071-1)
Warnings: For external use only.
Do not use:
- on wounds or damaged skin
- if you are allergic to capsicum or chili peppers
- with heating pad
When using this produce:
- you may experience a burning sensation The intensity of this reaction varies among individuals and may be severe. With regular use, the sensation generally disappears after several days.
- avoid contact with the eyes, lips, nose and mucous membranes
- do not tightly wrap or bandage the treatement area
- do not apply heat, or direct sunlight to the treated area immediately before or after use
Stop use and ask a doctor if:
- condition worsens or does not improve after regular use
- severe burning persists
- redness or blistering occurs
Section 42229-5 (42229-5)
Questions? Comments?
1-855-361-3993
Manufactured for:
AvKARE, Pulaski, TN 38478
www.avkare.com
AV 06/22
Section 44425-7 (44425-7)
Other Information: store at room temperature 15° - 30°C (59° - 86°F)
Section 50565-1 (50565-1)
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive Ingredients: Aloe barbadensis leaf extract, cetyl alcohol, glyceryl stearate SE, mineral oil, PEG-100 stearate, peppermint oil, phenoxyethanol, propylene glycol, purified water, stearyl alcohol, white petrolatum, xanthan gum
Section 55105-1 (55105-1)
Purpose: Topical Analgesic
Section 55106-9 (55106-9)
Active Ingredient: Capsaicin 0.1%
Packaging
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:09.992548 · Updated: 2026-03-14T23:12:19.459812