Lumify ®
ef5974b9-946c-4983-80e3-32b3aa28c207
34390-5
HUMAN OTC DRUG LABEL
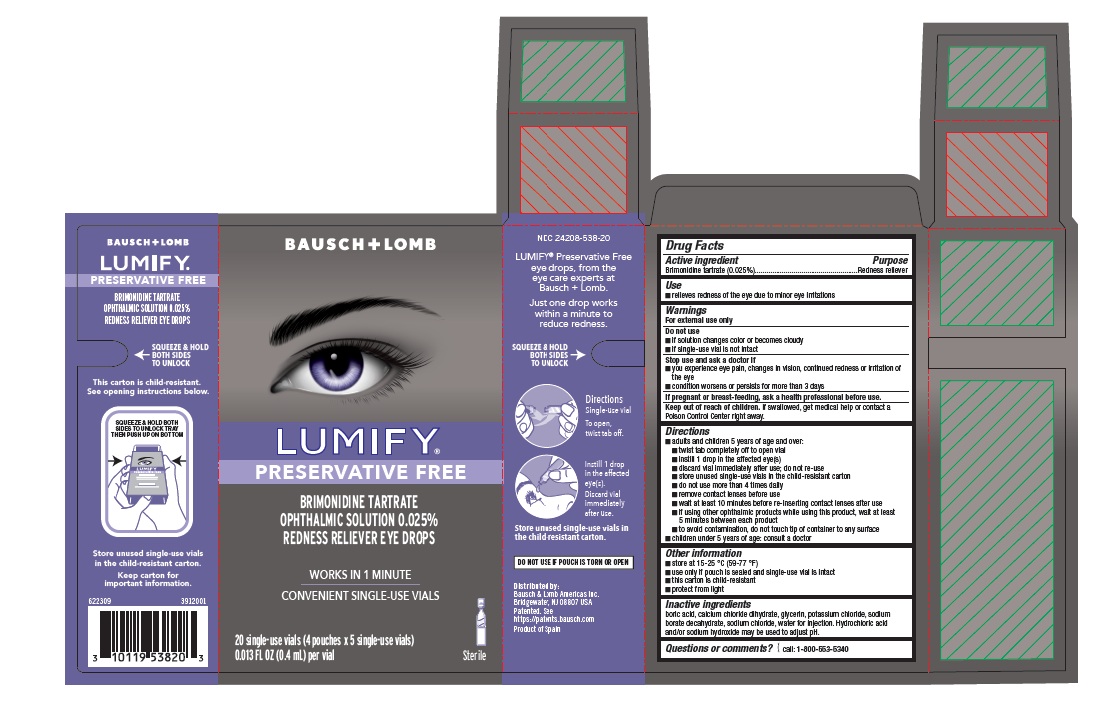
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Brimonidine tartrate (0.025%)
Purpose
Redness reliever
Medication Information
Purpose
Redness reliever
Description
Brimonidine tartrate (0.025%)
Use
- relieves redness of the eye due to minor eye irritations
Warnings
For external use only
Directions
- adults and children 5 years of age and over:
- twist tab completely off to open vial
- instill 1 drop in the affected eye(s)
- discard vial immediately after use; do not re-use
- store unused single-use vials in the child-resistant carton
- do not use more than 4 times daily
- remove contact lenses before use
- wait at least 10 minutes before re-inserting contact lenses after use
- if using other ophthalmic products while using this product, wait at least 5 minutes between each product
- to avoid contamination, do not touch tip of container to any surface
- children under 5 years of age: consult a doctor
Do Not Use
- if solution changes color or becomes cloudy
- if single-use vial is not intact
Active Ingredient
Brimonidine tartrate (0.025%)
Other Information
- store at 15-25 °C (59-77 °F)
- use only if pouch is sealed and single-use vial is intact
- this carton is child-resistant
- protect from light
Inactive Ingredients
boric acid, calcium chloride dihydrate, glycerin, potassium chloride, sodium borate decahydrate, sodium chloride, water for injection. Hydrochloric acid and/or sodium hydroxide may be used to adjust pH.
Stop Use and Ask A Doctor If
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 3 days
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel 20 Ct Carton
BAUSCH + LOMB
[Eye Image]
LUMIFY ®
PRESERVATIVE FREE
BRIMONIDINE TARTRATE
OPHTHALMIC SOLUTION 0.025%
REDNESS RELIEVER EYE DROPS
WORKS IN 1 MINUTE
CONVENIENT SINGLE-USE VIALS
20 single-use vials (4 pouches x 5 single-use vials)
0.013 FL OZ (0.4 mL) per vial
[vial icon]
Sterile
622309
3912001
Structured Label Content
Use
- relieves redness of the eye due to minor eye irritations
Purpose
Redness reliever
Warnings
For external use only
Directions
- adults and children 5 years of age and over:
- twist tab completely off to open vial
- instill 1 drop in the affected eye(s)
- discard vial immediately after use; do not re-use
- store unused single-use vials in the child-resistant carton
- do not use more than 4 times daily
- remove contact lenses before use
- wait at least 10 minutes before re-inserting contact lenses after use
- if using other ophthalmic products while using this product, wait at least 5 minutes between each product
- to avoid contamination, do not touch tip of container to any surface
- children under 5 years of age: consult a doctor
Do Not Use (Do not use)
- if solution changes color or becomes cloudy
- if single-use vial is not intact
Active Ingredient (Active ingredient)
Brimonidine tartrate (0.025%)
Other Information (Other information)
- store at 15-25 °C (59-77 °F)
- use only if pouch is sealed and single-use vial is intact
- this carton is child-resistant
- protect from light
Inactive Ingredients
boric acid, calcium chloride dihydrate, glycerin, potassium chloride, sodium borate decahydrate, sodium chloride, water for injection. Hydrochloric acid and/or sodium hydroxide may be used to adjust pH.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 3 days
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel 20 Ct Carton (Package/Label Principal Display Panel- 20 ct Carton)
BAUSCH + LOMB
[Eye Image]
LUMIFY ®
PRESERVATIVE FREE
BRIMONIDINE TARTRATE
OPHTHALMIC SOLUTION 0.025%
REDNESS RELIEVER EYE DROPS
WORKS IN 1 MINUTE
CONVENIENT SINGLE-USE VIALS
20 single-use vials (4 pouches x 5 single-use vials)
0.013 FL OZ (0.4 mL) per vial
[vial icon]
Sterile
622309
3912001
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:35.439274 · Updated: 2026-03-14T23:12:51.512687