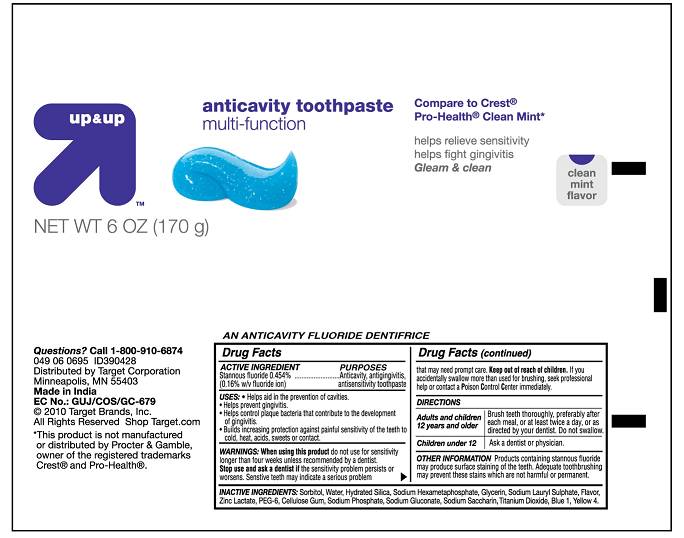
Up And Up Anticavity Toothpaste Multi-function
eef7c016-0b43-41cc-8dc5-2e95393f2544
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
STANNOUS FLUORIDE 0.454%...................... ANTICAVITY, ANTIGINGIVITIS, (0.16% W/V FLUORIDE ION) ANTISENSITIVITY TOOTHPASTE
Medication Information
Description
STANNOUS FLUORIDE 0.454%...................... ANTICAVITY, ANTIGINGIVITIS, (0.16% W/V FLUORIDE ION) ANTISENSITIVITY TOOTHPASTE
Uses
- HELPS AID IN THE PREVENTION OF CAVITIES
- HELPS PREVENT GINGIVITIS
- HELPS CONTROL PLAQUE BACTERIA THAT CONTRIBUTE TO THE DEVELOPMENT OF GINGIVITIS
- BUILD INCREASING PROTECTION AGAINST PAINFUL SENSITIVITY OF THE TEETH TO COLD, HEAT, ACIDS, SWEETS OR CONTACT.
Section 50565-1
KEEP OUT OF REACH OF CHILDREN
Section 55105-1
ANTICAVITY, ANTIGINGIVITIS, ANTISENSITIVITY TOOTHPASTE
Warnings
Directions
Adults and children 12 years and older Brush teeth thoroughly, PREFERABLY AFTER EACH MEAL,or at least twice a day, or as directed by your dentist. DO NOT SWALLOW
Children under 12 ASK A dentist or PHYSICIAN
Other information PRODUCTS CONTAINING STANNOUS FLUORIDE MAY PRODUCE SURFACE STAINING OF THE TEETH. ADEQUATE TOOTHBRUSHING MAY PREVENT THESE STAIN WHICH ARE NOT HARMFUL OR PERMANENT.
Active Ingredients
STANNOUS FLUORIDE 0.454%...................... ANTICAVITY, ANTIGINGIVITIS,
(0.16% W/V FLUORIDE ION) ANTISENSITIVITY TOOTHPASTE
Inactive Ingredients:
Principal Display Panel
Structured Label Content
Uses (USES)
- HELPS AID IN THE PREVENTION OF CAVITIES
- HELPS PREVENT GINGIVITIS
- HELPS CONTROL PLAQUE BACTERIA THAT CONTRIBUTE TO THE DEVELOPMENT OF GINGIVITIS
- BUILD INCREASING PROTECTION AGAINST PAINFUL SENSITIVITY OF THE TEETH TO COLD, HEAT, ACIDS, SWEETS OR CONTACT.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN
Section 55105-1 (55105-1)
ANTICAVITY, ANTIGINGIVITIS, ANTISENSITIVITY TOOTHPASTE
Warnings (WARNINGS)
Directions
Adults and children 12 years and older Brush teeth thoroughly, PREFERABLY AFTER EACH MEAL,or at least twice a day, or as directed by your dentist. DO NOT SWALLOW
Children under 12 ASK A dentist or PHYSICIAN
Other information PRODUCTS CONTAINING STANNOUS FLUORIDE MAY PRODUCE SURFACE STAINING OF THE TEETH. ADEQUATE TOOTHBRUSHING MAY PREVENT THESE STAIN WHICH ARE NOT HARMFUL OR PERMANENT.
Active Ingredients
STANNOUS FLUORIDE 0.454%...................... ANTICAVITY, ANTIGINGIVITIS,
(0.16% W/V FLUORIDE ION) ANTISENSITIVITY TOOTHPASTE
Inactive Ingredients: (Inactive ingredients:)
Principal Display Panel
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:07.238444 · Updated: 2026-03-14T22:52:27.184723